PARP Inhibitor Market
The PARP Inhibitor Market is Segmented by Drug Type, Indication, Distribution Channel, Mechanism of Action, End User, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
PARP Inhibitor Market Size, Market Forecast and Outlook By FMI
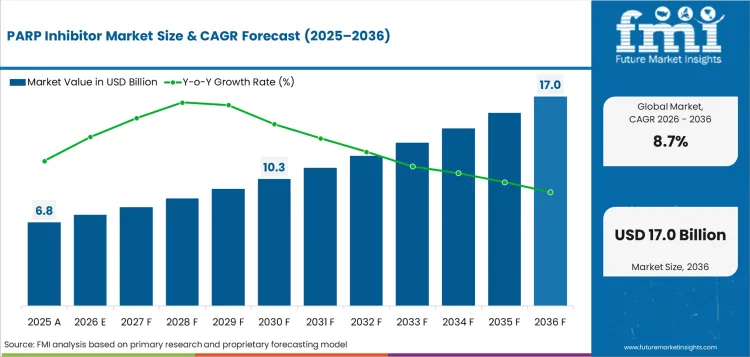
The PARP inhibitor market size stood at USD 6.8 billion in 2025 and is projected to reach USD 7.4 billion in 2026. Industry revenue is anticipated to rise to USD 17.0 billion by 2036 at 8.7% CAGR. By drug type, olaparib is estimated to account for 86.2% share in 2026. Ovarian cancer is anticipated to represent 83.9% share of indication in 2026.
Summary of the PARP Inhibitor Market
- Demand and Growth Drivers
- Oncology centers are widening maintenance treatment use after platinum response in ovarian cancer pathways.
- Genetic testing is raising eligible patient identification across BRCA-mutated breast and prostate cancer settings.
- Oral targeted therapy use is expanding as hospitals shift selected cancer care away from infusion-only pathways.
- Product and Segment View
- Olaparib is projected to account for 86.2% share in 2026 as broader label coverage supports repeat clinical adoption.
- Ovarian cancer is anticipated to capture 83.9% share in 2026 through maintenance therapy after platinum-based treatment.
- Hospital pharmacies are forecast to account for 60.0% share in 2026 due to oncology dispensing needing safety review.
- Geography and Competitive Outlook
- India is projected to record 8.0% CAGR by 2036, driven by private oncology networks expanding targeted therapy access.
- United Kingdom is expected to expand at 5.1% CAGR by 2036, reflected by National Health Service pathways influencing structured cancer medicine review.
- AstraZeneca and Merck & Co. shape competition through olaparib reach. GSK plc supports niraparib access.
- Analyst Opinion
- Sabyasachi Ghosh, , Principal Consultant for Healthcare at FMI, opines, “PARP inhibitors deliver a precision label depth opportunity in oncology. Market leaders compete by linking approved indications to genetic testing and evidence packages satisfying payers. Ovarian maintenance therapy forms the foundation of today’s revenue. Expansion into prostate and breast cancers opens the next revenue tier. Long-term commercial success depends on building and sustaining prescriber confidence through clear patient selection and reliable follow-up management.”
- PARP Inhibitor Market Value Analysis
- PARP inhibitor revenue is moving from ovarian-only maintenance use toward broader precision oncology use in selected solid tumors.
- Operator spending depends on mutation testing as hospitals need clear proof before approving targeted oral treatment.
- Value growth depends on treatment duration and approved label fit more than general cancer prevalence alone.
- Country growth varies since payer rules and genetic testing access shape how fast eligible patients reach therapy.
Treatment selection in oncology is shifting from broad use toward gene-defined and regimen-specific approaches. The FDA’s December 2025 approval of Akeega supports the use of niraparib in combination therapy for patients with BRCA2-mutated metastatic castration-sensitive prostate cancer. The AMPLITUDE trial, which enrolled 696 patients, demonstrated a 54% reduction in the risk of disease progression or death (rPFS hazard ratio of 0.46) in the BRCA2 subgroup.
Wider adoption of combination therapy in prostate cancer is estimated to depend on effective diagnostic coordination to identify eligible patients. Beyond prostate cancer, endometrial cancer is emerging as a new revenue opportunity. In August 2024, AstraZeneca received EU approval for Lynparza in combination with Imfinzi following chemotherapy in pMMR disease. This regimen demonstrated a 43% risk reduction and a median progression-free survival of 15 months, strengthening the case for expansion into gynecologic oncology beyond ovarian cancer.
Commercial success in this market is dependent on funding capacity. AstraZeneca reported Lynparza sales of USD 3,072 million in FY 2024, highlighting the ability of larger companies to support label expansion and market access efforts. As reimbursement bodies demand stronger clinical and economic evidence, smaller players face increasing pressure to target narrower patient groups and demonstrate clear value within hospital settings.
PARP Inhibitor Market Definition
PARP inhibitors cover prescription oncology medicines blocking poly ADP-ribose polymerase enzymes involved in DNA repair. Scope includes approved oral tablets and capsules used for ovarian and breast cancer. Prostate and pancreatic cancer use is included if approved drug revenue is recorded. Revenue covers branded medicines and authorized channels linked to treatment use. Unapproved research compounds are excluded unless commercial drug revenue is recorded.
PARP Inhibitor Market Inclusions
Scope includes olaparib and niraparib sold through approved oncology channels. Rucaparib and talazoparib are included as separate approved drug types. Maintenance therapy and monotherapy use are included. Combination therapy is counted if PARP inhibitor drug value is identifiable. Hospital pharmacies and specialty pharmacies are included as distribution channels.
PARP Inhibitor Market Exclusions
Scope excludes companion diagnostic test revenue and genetic sequencing service revenue. Cytotoxic drugs and injectable oncology medicines are outside the PARP inhibitor boundary unless an oral PARP product is involved. Clinical trial supply is excluded unless it converts into commercial drug revenue. Supportive care products and general oncology pharmacy fees are excluded from the core value view.
PARP Inhibitor Market Research Methodology
- Primary Research: FMI analysts reviewed oncology prescribing pathways, hospital pharmacy controls and specialist treatment access across major countries.
- Desk Research: Drug labels and cancer registry data supported indication mapping. Official cancer burden sources supported country demand review.
- Market Sizing and Forecasting: Forecast values used supplied anchors, product-level label breadth and country-level cancer care access checks.
- Data Validation: Checks compared 2026 value with 2036 value, and product dominance and country growth patterns against oncology treatment maturity.
Why is the PARP Inhibitor Market Growing?
- Ovarian maintenance therapy is widening PARP inhibitor demand after platinum response in BRCA-mutated or HRD-positive patient groups.
- BRCA and HRD testing supports adoption by matching eligible patients with approved oral targeted therapy.
- Prostate cancer combinations are opening a revenue channel beyond ovarian cancer treatment by pairing PARP inhibitors with androgen-pathway therapy.
Targeted oncology care gives PARP inhibitors a clearer role in treatment planning. U.S. Food and Drug Administration labeling for Lynparza lists uses in ovarian and breast cancers. Pancreatic and prostate cancer uses widen the clinical path for olaparib. Hospital oncology teams can connect one familiar oral drug with tumor boards across several indications. FDA-approved companion diagnostic language keeps biomarker testing central to drug access.
Broader label coverage supports repeated use when physicians need known options across mutation-defined patients. International Agency for Research on Cancer and World Health Organization released 2022 global estimates in February 2024 showing about 20.0 million new cancer cases. Only a smaller subset becomes suitable for PARP treatment after BRCA or homologous recombination repair testing. Diagnostic access has a direct effect on commercial uptake. Suppliers with clear label evidence and testing support can move identified patients into treatment.
Market Segmentation Analysis
- Olaparib is projected to capture 86.2% share in 2026 due to broad label use influencing ovarian and breast cancer treatment.
- Ovarian cancer is anticipated to hold 83.9% share in 2026, led by maintenance treatment after platinum response sustaining a high repeat therapy use.
- Hospital pharmacies are forecast to account for 60.0% share in 2026 since oncology medicines need safety review and controlled dispensing.
- DNA damage repair inhibition is projected to represent 25.0% share in 2026, driven by mechanism messaging guiding specialist education.
- Hospitals are expected to hold 65.0% share in 2026 as patient initiation and monitoring stay centered in cancer care facilities.
FMI divides market segmentation into five parent groups by how PARP inhibitors are prescribed and paid for. Drug type separates brand positions and approved molecule options. Indication shows whether therapy starts in ovarian and breast cancer. Prostate and pancreatic cancer complete indication coverage. Distribution channel explains how therapy reaches patients after specialist review. Mechanism of action captures clinical positioning in DNA repair treatment pathways. End user separates treatment centers from research and specialty clinic demand.
Insights into PARP Inhibitor Market by Drug Type
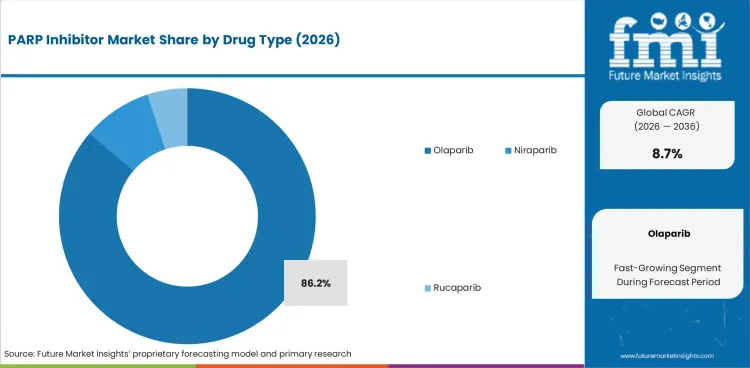
- Olaparib is projected to account for 86.2% share in 2026 as multiple tumor indications give it broad commercial reach.
- Niraparib retains a defined role in ovarian maintenance therapy as ZEJULA labeling supports use after response to platinum-based chemotherapy.
Insights into PARP Inhibitor Market by Indication
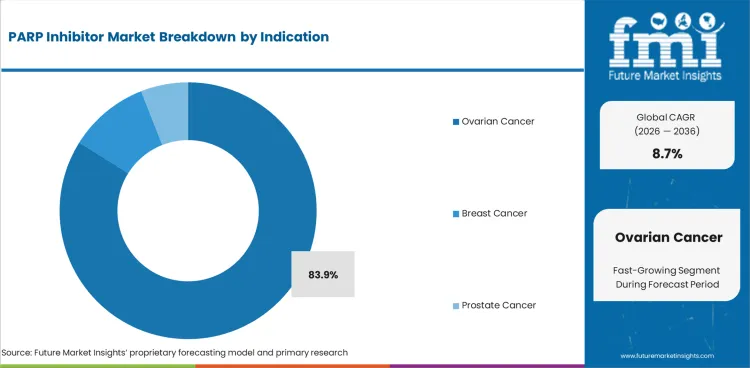
- Ovarian cancer is anticipated to account for 83.9% share in 2026 since maintenance therapy after platinum response remains the main treatment anchor.
- Breast cancer use is narrower but remains commercially important because germline BRCA mutation status defines eligible patients.
Insights into PARP Inhibitor Market by Distribution Channel
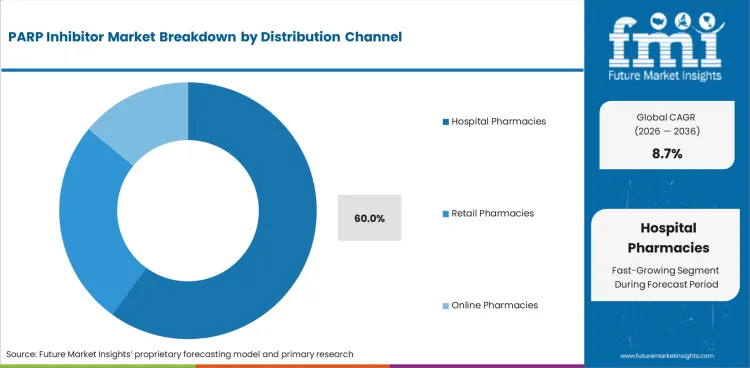
- Hospital pharmacies are forecast to account for 60.0% share in 2026 as oncology drug review stays close to specialist teams.
- Cancer treatment centers gain share as targeted medicine approvals require patient records and therapy follow-up.
Insights into PARP Inhibitor Market by Mechanism of Action
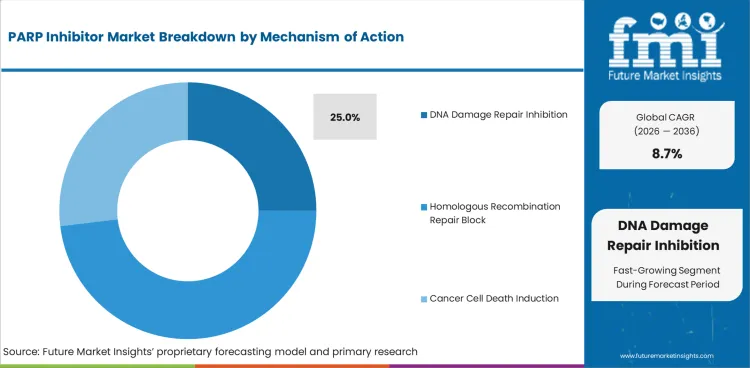
- DNA damage repair inhibition is expected to account for 25.0% share in 2026 as doctors focus on targeting weak repair pathways in treatment.
- Homologous recombination repair block remains a core clinical concept as BRCA1 and BRCA2 defects define many eligible patients.
Insights into PARP Inhibitor Market by End User
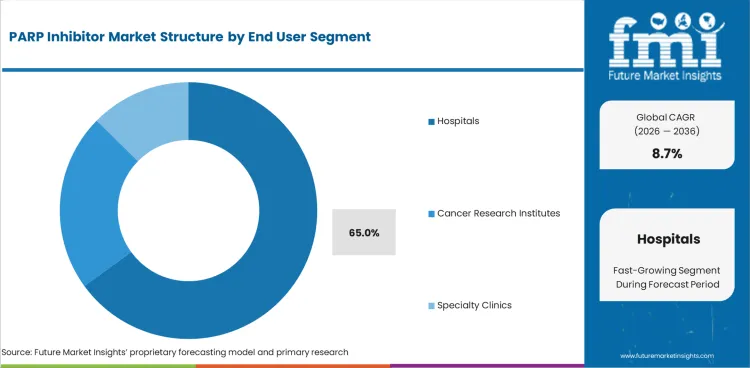
- Hospitals are expected to account for 65.0% share in 2026 as specialist cancer care remains the main initiation point.
- Tertiary care hospitals carry higher use upon managing recurrent ovarian cancer and advanced prostate cancer cases.
PARP Inhibitor Market Drivers, Restraints and Opportunities
- Mutation-defined prescribing is making BRCA and HRR evidence central to PARP inhibitor prescribing.
- Health technology review can slow access since oral oncology drugs move through cost and safety checks.
- Combination therapy is creating opportunity as prostate cancer programs pair PARP inhibitors with androgen pathway drugs.
PARP inhibitor growth is shifting toward tested patient groups instead of broad cancer use. U.S. Food and Drug Administration labeling defines olaparib adoption by tumor type and mutation status across approved cancer types. FDA labeling for Talzenna states talazoparib is used with enzalutamide for HRR gene-mutated metastatic castration-resistant prostate cancer. NICE’s 2025 olaparib review shows access can depend on cost comparison and commercial terms. Growth outlook favors manufacturers supporting BRCA and HRR testing and clear safety monitoring. Combination use in castration-resistant prostate cancer treatment is likely to widen revenue paths as oncology teams link targeted oral drugs with defined treatment sequences.
Trade Flow and Import-Export Analysis
Trade flow analysis for PARP inhibitor supply should focus on finished oral oncology products and licensed regional channels instead of only API movement. AstraZeneca reported FY 2025 Lynparza revenue of USD 1,434 million in the United States and USD 914 million in Europe. Emerging markets generated USD 669 million with China affected by generic competition and stock compensation ahead of expected volume-based procurement in Q1 2026. Outlook depends on branded access in mature countries and generic pressure in China. Suppliers will need region-specific pricing and inventory plans as PARP inhibitor trade becomes more exposed to reimbursement timing.
Therapy-wise Demand and Prescription Pattern Analysis
Therapy-wise oral oncology drug adoption is expected to stay centered on tested patients rather than broad cancer use. FDA’s 2025 Lynparza label covers approved cancer indications. Patient selection depends on BRCA or HRR mutation evidence across several approved settings. Recommended dosage is 300 mg taken orally twice daily. Prescription patterns favor hospitals connecting genetic testing with treatment sequencing and safety monitoring. Manufacturers with clear testing support and payer-ready label evidence will be better placed to move oncology referrals into treated patient volume.
Analysis of PARP Inhibitor Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 8.0% |
| United Kingdom | 5.1% |
| France | 4.6% |
| China | 3.4% |
| South Korea | 2.8% |
| Japan | 1.6% |
| United States | 1.2% |
Source: Future Market Insights, 2026.
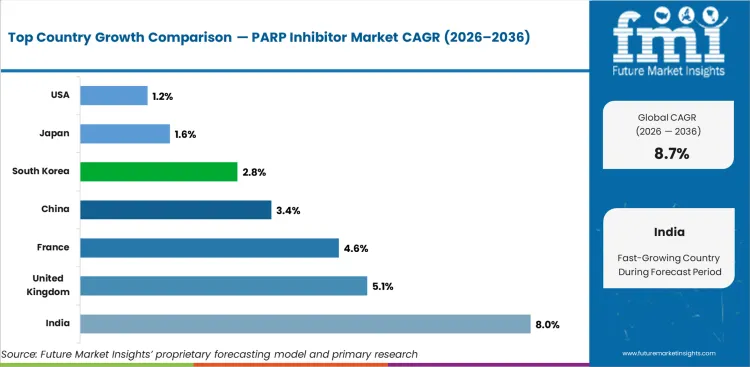
PARP Inhibitor Market CAGR Analysis by Country
- India is projected to record 8.0% CAGR by 2036, driven by private oncology access and genetic testing expanding in major care networks.
- United Kingdom is expected to expand at 5.1% CAGR by 2036 due to structured cancer medicine review influencing controlled use.
- France is forecast to grow at 4.6% CAGR by 2036, led by hospital oncology pathways retaining strong targeted therapy access.
- China is expected to advance at 3.4% CAGR by 2036, reflected by high patient volume meeting pricing pressure and local competition.
- South Korea is projected to rise at 2.8% CAGR by 2036 since mature oncology care supports selective uptake.
- Japan is forecast at 1.6% CAGR by 2036 as reimbursement review keeps adoption measured across advanced cancer care. United States is expected to post 1.2% CAGR by 2036 through mature use and payer controls narrowing incremental value expansion.
Country performance differs by testing access and payer control. India and the United Kingdom track above the global line in the supplied country outlook due to broader access gaps leaving prospects for uptake. China and France fall in the middle of the country set as large patient pools meet structured price review. South Korea, Japan and the United States show slower value expansion as mature care pathways already include PARP therapy for eligible groups.
Demand Outlook for PARP Inhibitors in India
Demand in India is projected to record 8.0% CAGR by 2036, driven by targeted oncology integrating into wider specialist adoption. India's Ministry of Health and Family Welfare cited 1.53 million estimated cancer cases for 2024 in a Lok Sabha answer released in August 2025. Such burden gives oncologists a larger pool for genetic evaluation in breast and ovarian cancer. Prostate cancer testing can add another eligible group. Access will depend on affordability and hospital-led testing. Private oncology networks are expected to support faster uptake than public hospitals.
- Urban cancer hospitals can broaden PARP therapy use as genetic testing becomes more common before treatment selection.
- Oncology departments are likely to favor olaparib first as broad label coverage reduces formulary complexity.
- Cost and test access limit wider use leading patient assistance and local distribution to shape supplier success.
Demand Analysis of PARP Inhibitors in the United Kingdom
The market in United Kingdom is expected to expand at 5.1% CAGR by 2036 as national review systems support controlled access. National Ovarian Cancer Audit work in 2024 examined ovarian cancer care across England and Wales. Audit reporting keeps attention on diagnosis and treatment pathways for a core PARP indication. Medicine use is shaped by National Health Service review and clinical evidence. Stable reimbursement routes can support uptake once guidance accepts a defined patient group.
- Specialist cancer centers can use structured testing records to match ovarian cancer patients with maintenance therapy.
- Hospital pharmacy control supports safe use as oral oncology drugs still need specialist monitoring and repeat review.
- Budget review may slow new combinations even though clear survival evidence can improve pathway acceptance.
Sales Analysis of PARP Inhibitors in China
Sales in China are forecast to grow at 3.4% CAGR by 2036, influenced by oncology volume remaining consistently high. International Agency for Research on Cancer counted 4.82 million new cancer cases in China in its February 2024 GLOBOCAN fact sheet. Patient scale supports clinical need across breast and ovarian cancer. Prostate cancer adds a further targeted therapy pool. Central purchasing pressure and local oncology competition can cap revenue per treated patient. Domestic testing capacity should still lift identified eligible groups over the forecast period.
- Large hospital oncology departments create steady demand for mutation-tested treatment in advanced cancer pathways.
- Local price review keeps value expansion below global CAGR even as treated patient volume improves.
- Domestic oncology firms may compete in adjacent targeted classes, sharpening pricing pressure on imported brands.
Opportunity Analysis of PARP Inhibitors in South Korea
The sector in South Korea is projected to rise at 2.8% CAGR by 2036 due to a mature cancer system maintaining measured targeted therapy use. International Agency for Research on Cancer recorded 237,647 new cancer cases in South Korea in the February 2024 country sheet. High-quality diagnosis and specialist care support patient selection. Reimbursement rules remain a gatekeeper for high-value oral therapies.
- Cancer centers can use existing molecular testing workflows to identify suitable ovarian and breast cancer patients.
- Public reimbursement review will keep treatment access focused on approved indications with clear evidence.
- Specialist follow-up capacity supports safe oral therapy use as monitoring needs continue through treatment cycles.
Future Outlook for PARP Inhibitors in Japan
Demand in Japan is forecast at 1.6% CAGR by 2036, reflected by careful reimbursement review sustaining a measured value expansion. February 2024 GLOBOCAN country data recorded 104,318 new prostate cancer cases in Japan. Advanced prostate cancer continues as a relevant target for PARP combinations. Uptake relies on mutation testing and prior therapy sequence, with payer acceptance remaining critical.
- Quality-focused cancer hospitals will use PARP inhibitors mainly for clearly defined mutation-positive patients.
- Prostate cancer pathways create selective upside as combination use becomes better defined in advanced disease.
- Reimbursement and safety review are likely to maintain gradual adoption across oral oncology treatment cycles.
Opportunity Analysis of PARP Inhibitors in the United States
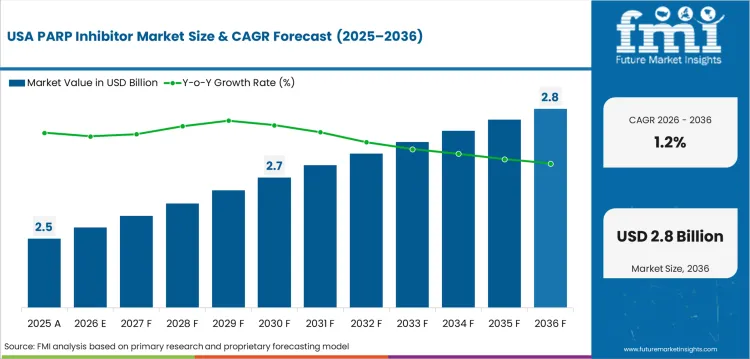
Industry in the United States is expected to post 1.2% CAGR by 2036 since PARP inhibitor use is already established in major oncology pathways. National Cancer Institute SEER summaries expect about 2.1 million new cancer diagnoses in 2026. High diagnosis rates do not lead to strong value growth as most eligible cases are already covered. Payer checks and specialty pharmacy controls slow further expansion. Incremental opportunity will be gained from label changes and combination evidence.
- Specialty oncology practices can move suitable patients into PARP therapy after tumor testing confirms eligibility.
- Prior authorization keeps access tied to label wording and companion diagnostic proof for high-value oral drugs.
- Combination updates in prostate cancer offer incremental upside. Mature uptake limits country expansion.
Demand Outlook for PARP Inhibitors in France
Demand for PARP inhibitors in France is forecast to grow at 4.6% CAGR by 2036 as hospital oncology access supports targeted treatment review. International Agency for Research on Cancer recorded 65,659 new breast cancer cases in metropolitan France in its February 2024 country sheet. Breast and ovarian cancer pathways keep genetic testing relevant for PARP use. Hospital pharmacies remain central to dispensing control.
- Cancer centers can support PARP use through established genetic counseling and oncology pharmacy workflows.
- Hospital-based dispensing keeps treatment oversight close to adverse event monitoring and dose management.
- Value assessment will favor products with clear survival evidence and manageable safety profiles.
Competitive Landscape and Strategic Positioning
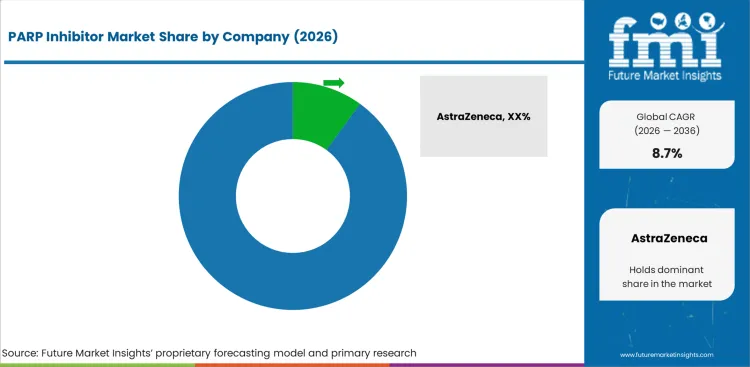
- Broad-label brands hold an advantage as hospitals prefer therapies with evidence across several tumor pathways.
- Specialty pharmacy reach has a direct role as oral oncology drugs need documentation support after physician selection.
- Pipeline combinations are expected to shift value from single-agent maintenance toward mutation-led combination treatment.
Competition in the PARP inhibitor market is influenced by approved indications and access support. Companies such as AstraZeneca and Merck & Co. hold a strong position through olaparib and its multi-indication reach. GSK plc competes based on niraparib in ovarian maintenance settings. Pfizer uses talazoparib to maintain positions in breast cancer and prostate cancer. pharma& GmbH supports rucaparib supply for specific ovarian and prostate cancer uses. Hospital accounts compare products on safety guidance and reimbursement readiness.
Smaller asset owners face a narrower path as oncology buyers prefer products with clear clinical placement. Biomarker-based treatment planning promotes PARP use once mutation evidence is available. Adjacent treatment planning shapes combination pathways across prostate and gynecologic cancer care. Companies with clear treatment sequencing and strong safety support are better placed to defend share in mature oncology systems.
Key Companies in the PARP Inhibitor Market
Competitive structure is easier to assess by grouping companies around approved therapy ownership and oncology access support.
- Approved PARP Leaders: AstraZeneca and Merck & Co. support olaparib through broad oncology label coverage and global hospital reach. GSK plc supports niraparib in ovarian maintenance therapy. Pfizer manages talazoparib across breast cancer and prostate cancer use.
- Specialty Asset Owners: pharma& GmbH supports Rubraca supply and access after the former Clovis Oncology asset changed ownership. Johnson & Johnson Innovative Medicine participates through prostate cancer combinations and broader advanced oncology pathways.
- Oncology Ecosystem Players: Diagnostic firms and specialty pharmacies influence uptake through mutation confirmation and access checks. Hospitals prefer suppliers able to reduce delays from test result to prescription approval and patient start.
Competitive Benchmarking: PARP Inhibitor Market
| Company | Approved Label Breadth | Oncology Access Depth | Mutation Selection Support | Geographic Footprint |
|---|---|---|---|---|
| AstraZeneca | High | Strong | Strong | Global |
| Merck & Co. | High | Strong | Strong | Global partnership reach |
| GSK plc | Medium | Strong | Medium | United States and Europe |
| Pfizer Inc. | Medium | Strong | Medium | Global |
| pharma& GmbH | Medium | Medium | Medium | United States and Europe |
| Johnson & Johnson Innovative Medicine | Medium | Strong | Medium | Global oncology reach |
| Janssen Biotech, Inc. | Low | Strong | Medium | Global prostate cancer channels |
| Astellas Pharma Inc. | Medium | Medium | Medium | Global prostate cancer co-commercialization |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on approved label breadth, oncology access depth and mutation selection support.
Key Developments in PARP Inhibitor Market
- In June 2025, the U.S. Food and Drug Administration approved ZEJULA labeling changes narrowing first-line ovarian maintenance use to HRD-positive tumors.
- In June 2025, Pfizer said final overall survival data were added to the existing TALZENNA label for HRR-mutated metastatic castration-resistant prostate cancer.
- In December 2025, the U.S. Food and Drug Administration granted regular approval to rucaparib for BRCA-mutated metastatic castration-resistant prostate cancer after prior androgen receptor-directed therapy.
Key Players in the PARP Inhibitor Market
Major Global Players:
- AstraZeneca
- Merck & Co.
- GSK plc
- Pfizer Inc.
- Johnson & Johnson Innovative Medicine
Specialist and Regional Players:
- Janssen Biotech, Inc.
- pharma& GmbH
- Astellas Pharma Inc.
- IMPACT Therapeutics
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 7.4 billion to USD 17.0 billion at 8.7% CAGR |
| Market Definition | Approved oral PARP inhibitor medicines used in selected cancer treatment pathways |
| Drug Type | Olaparib, niraparib, rucaparib, talazoparib |
| Indication | Ovarian cancer, breast cancer, prostate cancer, pancreatic cancer |
| Distribution Channel | Hospital pharmacies, retail pharmacies, online pharmacies |
| Mechanism of Action | DNA damage repair inhibition, homologous recombination repair block, synthetic lethality mechanism |
| End User | Hospitals, cancer research institutes, specialty clinics |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | India, United Kingdom, China, South Korea, Japan, United States, France |
| Key Companies Profiled | AstraZeneca and Merck & Co.; GSK plc; Pfizer Inc.; Johnson & Johnson Innovative Medicine; Janssen Biotech, Inc.; Astellas Pharma Inc., pharma& GmbH |
| Forecast Period | 2026 to 2036 |
| Approach | Supplied value anchors and label review. Country cancer burden checks and segment share modeling |
Source: Future Market Insights, 2026.
PARP Inhibitor Market Breakdown by Drug Type, Indication, Distribution Channel, Mechanism of Action, End User, and Region
ARP Inhibitor Market Segmented by Drug Type:
- Olaparib
- Niraparib
- Rucaparib
- Talazoparib
PARP Inhibitor Market Segmented by Indication:
- Ovarian Cancer
- Breast Cancer
- Prostate Cancer
- Pancreatic Cancer
PARP Inhibitor Market Segmented by Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
PARP Inhibitor Market Segmented by Mechanism of Action:
- DNA Damage Repair Inhibition
- Homologous Recombination Repair Block
- Synthetic Lethality Mechanism
- Cancer Cell Death Induction
- Replication Stress Induction
PARP Inhibitor Market Segmented by End User:
- Hospitals
- Cancer Research Institutes
- Specialty Clinics
PARP Inhibitor Market Segmented by Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Bibliography
- AstraZeneca. (2024, August 14). Lynparza and Imfinzi combination approved in the EU for patients with mismatch repair proficient advanced or recurrent endometrial cancer.
- AstraZeneca. (2025, February 6). Full year and Q4 2024 results: Conference call and webcast for investors and analysts.
- AstraZeneca. (2026, February 10). AstraZeneca results: FY and Q4 2025.
- GSK plc. (2025). Annual Report 2024.
- Government of India, Ministry of Health and Family Welfare, Department of Health Research. (2025, August 8). Rising cases of cancer: Lok Sabha unstarred question no. 3233.
- National Cancer Institute. (2026). Common cancer sites: Cancer Stat Facts.
- National Institute for Health and Care Excellence. (2025, February 12). Olaparib for treating BRCA mutation-positive HER2-negative advanced breast cancer after chemotherapy (Technology appraisal guidance No. TA1040).
- National Ovarian Cancer Audit. (2024, September). State of the Nation Report 2024: An audit of care received by women diagnosed with ovarian cancer in England in 2021 and in Wales in 2022. National Cancer Audit Collaborating Centre.
- Pfizer Inc. (2025, June 13). Pfizer provides update on U.S. regulatory review of TALZENNA in combination with XTANDI for broader use in metastatic castration-resistant prostate cancer.
- pharma& GmbH. (2025, December 19). U.S. FDA grants approval to Rubraca® (rucaparib) for the treatment of chemotherapy-naïve BRCA 1/2-mutated metastatic castration-resistant prostate cancer.
- USA Food and Drug Administration. (2025, June). ZEJULA® (niraparib) tablets, for oral use: Prescribing information.
- USA Food and Drug Administration. (2025, July). LYNPARZA® (olaparib) tablets, for oral use: Prescribing information.
- USA Food and Drug Administration. (2025, December). RUBRACA® (rucaparib) tablets, for oral use: Prescribing information.
- USA Food and Drug Administration. (2025, December 12). FDA approves niraparib and abiraterone acetate plus prednisone for BRCA2-mutated metastatic castration-sensitive prostate cancer.
- USA Food and Drug Administration. (2025, December 17). FDA grants regular approval to rucaparib for metastatic castration-resistant prostate cancer.
- World Health Organization. (2024, February 1). Global cancer burden growing, amidst mounting need for services.
Bibliography uses government sources, official regulatory evidence and company-issued information relevant to PARP inhibitor demand signals.
This Report Answers
- What is the current and future size of the PARP inhibitor market?
- How fast is the PARP inhibitor market projected to expand between 2026 and 2036?
- Which drug type is expected to account for the leading PARP inhibitor share in 2026?
- Which indication is projected to account for the leading PARP inhibitor share in 2026?
- Why does olaparib account for a high PARP inhibitor share?
- How does ovarian cancer support PARP inhibitor adoption?
- Which countries are projected to record faster expansion during the forecast period?
- Who are the main companies active in PARP inhibitor supply?
- How does FMI define the PARP inhibitor market scope?
- How does FMI estimate and validate the PARP inhibitor forecast?
Frequently Asked Questions
What is the global market demand for PARP inhibitors in 2026?
In 2026, PARP inhibitor market value is projected to reach USD 7.4 billion as mutation-matched oncology treatment expands across approved cancer settings.
How big will the PARP inhibitor market be in 2036?
By 2036, PARP inhibitor industry value is forecast to reach USD 17.0 billion as oral targeted therapy use widens.
How much is demand for PARP inhibitors projected to expand between 2026 and 2036?
Between 2026 and 2036, PARP inhibitor sector revenue is anticipated to expand at 8.7% CAGR as label breadth widens.
Which drug type is expected to lead the PARP inhibitor market by 2026?
Olaparib is projected to represent 86.2% share in 2026 due to multiple approved indications influencing broader clinical use.
Which indication is expected to lead PARP inhibitor demand in 2026?
Ovarian cancer is anticipated to account for 83.9% share in 2026 since maintenance therapy supports repeated use.
Which country is forecast to grow fastest in the PARP inhibitor market?
India is projected to record 8.0% CAGR by 2036 as oncology access and genetic testing improve.
What does the PARP inhibitor market include?
Scope includes approved olaparib and niraparib medicines. Rucaparib and talazoparib complete the core approved drug set.
How does FMI estimate the PARP inhibitor market forecast?
FMI uses supplied value anchors, drug label review and country cancer burden checks to validate forecast size and segment shares.
Why do hospital pharmacies matter in the PARP inhibitor market?
Hospital pharmacies are forecast to hold 60.0% share in 2026 as targeted oral oncology drugs need safety review and repeat oversight.
Who uses PARP inhibitor medicines?
Main users include hospitals and cancer centers. Research institutes and specialty clinics support trials, patient matching and treatment follow-up.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Clinical Management Systems
- Electronic Health Record Systems
- Patient Monitoring Systems
- Clinical Management Systems
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Usage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Usage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Usage, 2026 to 2036
- Clinical Homecare Systems
- Chronic Disease Management
- Post-Acute Care Monitoring
- Clinical Homecare Systems
- Y to o to Y Growth Trend Analysis By Usage, 2021 to 2025
- Absolute $ Opportunity Analysis By Usage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Cloud-Based Systems
- Software as a Service Platforms
- On-Premise Systems
- Cloud-Based Systems
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Large Hospitals
- Small and Medium Hospitals
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Sales
- Enterprise Contracts
- Resellers
- Direct Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Usage
- By Technology
- By End User
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Teladoc Health
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Homecare Homebase
- Kyruus
- HealthViewX Remote Patient Monitoring
- Loyal Health
- Teladoc Health
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Usage, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Usage
- Figure 9: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Technology
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product
- Figure 32: North America Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Usage
- Figure 35: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Technology
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Distribution Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product
- Figure 48: Latin America Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Usage
- Figure 51: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Technology
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product
- Figure 64: Western Europe Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Usage
- Figure 67: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Technology
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Usage
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product
- Figure 96: East Asia Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Usage
- Figure 99: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Technology
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Usage
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Usage, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Usage, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Usage
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

