Extended-Action Local Anaesthetic Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Extended-Action Local Anaesthetic Market Analysis – Size, Share, and Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Extended-Action Local Anaesthetic Market Forecast and Outlook 2025 to 2035
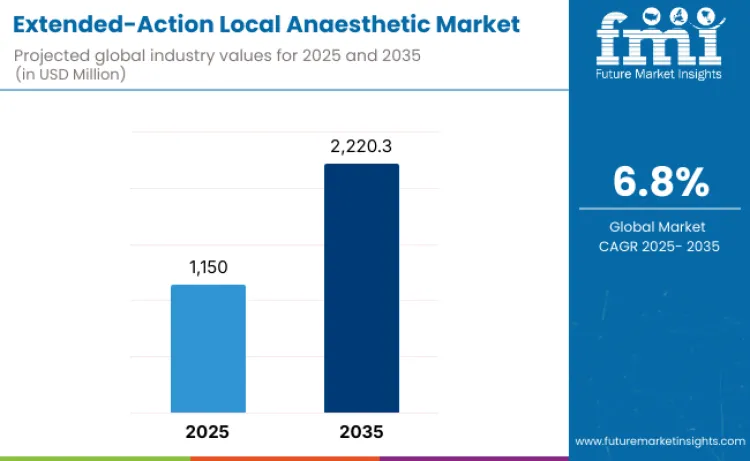
Global extended-action local anaesthetic market is expected to grow from USD 1,150.0 million in 2025 to USD 2,220.3 million by 2035, at a CAGR of 6.8%. Rising global surgical volumes, broader implementation of Enhanced Recovery After Surgery (ERAS) protocols, and continued preference for day-care surgical models are creating sustained momentum for long-acting anesthetic formulations. While adoption is accelerating across developed healthcare systems, higher product pricing and limited availability in price-sensitive regions may temper uptake, particularly where hospitals rely on traditional short-acting anesthetics due to budget constraints.
Extended-release anesthetics are transforming perioperative pain management by delivering predictable analgesia for two to three days, reducing opioid consumption and enabling faster ambulation. Surgeons and anesthesiologists increasingly favor these formulations for orthopedic, plastic, general, obstetric, and regional anesthesia procedures, where prolonged analgesic duration improves patient comfort and recovery quality. Their role in minimizing opioid exposure has strengthened clinical preference, especially in settings prioritizing early discharge and reduced readmissions. The shift toward catheter-free nerve blocks and ultrasound-guided techniques further supports the use of sustained-release agents that simplify workflows and enhance safety.
Quick Stats for the Extended-Action Local Anaesthetic Market
- Extended-Action Local Anaesthetic Market Value (2025): USD 1,150.0 million
- Extended-Action Local Anaesthetic Market Forecast Value (2035): USD 2,220.3 million
- Extended-Action Local Anaesthetic Market Forecast CAGR: 6.8%
- Leading Market Segment: Surgical analgesia (50% share)
- Key Growth Regions: North America, Europe, and Asia Pacific
- Top Players: PaciraBioSciences, Heron Therapeutics, AstraZeneca, B. Braun, Mylan / Viatris, Others
Innovation continues to accelerate as companies develop advanced polymer-based carriers, liposomal delivery platforms, biodegradable microspheres, and targeted nerve-release technologies designed to prolong effects while reducing systemic toxicity. These next-generation formulations integrate seamlessly with modern regional anesthesia practices and ERAS guidelines. Government-backed opioid-reduction initiatives, academic training programs, and expanding adoption across outpatient surgical centers reinforce long-term demand, positioning extended-action anesthetics as a core component of contemporary multimodal pain-management strategies.
Extended-Action Local Anaesthetic Market Year-Over-Year Forecast (2025 to 2035)
Between 2025 and 2030, the extended-action local anaesthetic market will expand as rising surgical volumes and widespread adoption of opioid-sparing protocols drive stronger demand for long-acting formulations. Hospitals and ambulatory centers are integrating extended-release anesthetics into ERAS pathways to lower postoperative pain and shorten recovery times. Companies are advancing competitive positioning through investments in controlled-release delivery systems, liposomal technologies, and clinical training programs for perioperative teams.
From 2030 to 2035, growth accelerates, driven by broader adoption of orthopedic, abdominal, thoracic, and plastic procedures, supported by expanding day-care and minimally invasive surgery programs. Collaborations between pharmaceutical innovators, regional anesthesia platforms, and ERAS committees will strengthen adoption as evidence demonstrates improved recovery outcomes, reduced opioid dependence, and higher patient satisfaction.
Extended-Action Local Anaesthetic Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 1,150.0 million |
| Market Forecast Value (2035) | USD 2,220.3 million |
| Forecast CAGR (2025 to 2035) | 6.8% |
Why Is the Extended-Action Local Anaesthetic Market Growing Worldwide?
The extended-action local anaesthetic landscape grows as clinicians adopt long-acting formulations that offer 48-72 hours of pain relief, reduce opioid use by more than half, and support faster recovery across orthopedic, abdominal, and soft-tissue procedures. Rising demand for same-day surgery, multimodal analgesia, and ERAS pathways strengthens preference for solutions that deliver durable analgesia without catheters or repeated dosing.
Government-backed opioid-reduction initiatives, hospital quality programs, and payer incentives are accelerating use across hospitals, ASCs, and pain clinics. Training programs are expanding clinician proficiency in sustained-release anesthesia, while outpatient procedure growth increases the need for mobility-friendly analgesia. Cost barriers and uneven reimbursement may still constrain adoption in resource-limited settings.
How Is the Extended-Action Local Anaesthetic Market Segmented by product type, Application, end user and Region?
The extended-action local anaesthetic market is segmented based on product type, application focus, and end-user. By product type, the market includes liposomal formulations, prolonged-release injectable systems, transdermal anaesthetic patches, and device-integrated nerve-block platforms, along with other emerging delivery innovations. Based on application, the industry is categorized into surgical analgesia, post-operative pain management, chronic pain and regional nerve blocks, and dental and minor procedural use cases. End-user segmentation includes hospitals and surgical centers, ambulatory surgery centers (ASCs), dental clinics, and specialized pain management clinics. Regionally, the market is divided into Asia Pacific, Europe, North America, Latin America, and Middle East & Africa.
Liposomal Formulations Hold Dominant Share in the Extended-Action Local Anaesthetic Market
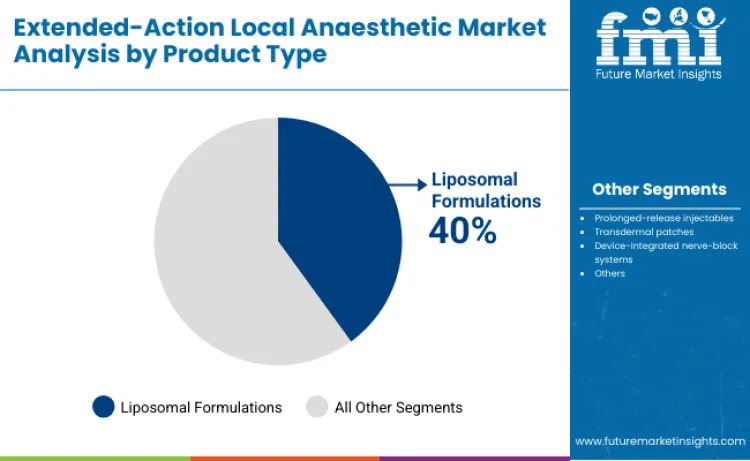
The liposomal formulation segment leads the extended-action local anaesthetic landscape with about 40% share in 2025, driven by strong clinical evidence, ERAS alignment, and broad acceptance of liposomal bupivacaine for multi-day analgesia and opioid reduction. Prolonged-release injectables hold 25%, supported by predictable dosing for postoperative and ambulatory care. Device-integrated nerve-block systems account for 13%, while transdermal patches represent 12% and emerging polymer-based depots 10%. Liposomal formulations dominate due to extended analgesia, proven opioid-sparing effects, predictable pharmacokinetics, and meaningful improvements in mobility, recovery, and overall patient experience.
Surgical Analgesia Represents the Primary Application Segment in the Extended-Action Local Anaesthetic Market

The surgical analgesia segment leads the extended-action local anaesthetic landscape with about 50% revenue in 2025, driven by widespread use in orthopedic, abdominal, gynecologic, colorectal, thoracic, and trauma surgeries where multi-day pain relief supports ERAS pathways and reduces opioid dependence. Post-operative pain management holds 30%, benefiting from growing adoption in ASCs and same-day recovery programs. Chronic pain and regional block applications contribute 12%, while dental and minor procedures account for 8%. Leadership is sustained by improved recovery timelines, standardized analgesia, reduced opioid-related complications, and lower readmission risk.
Hospitals & Surgical Centers Account for the Largest Share of Extended-Action Local Anaesthetic Utilization
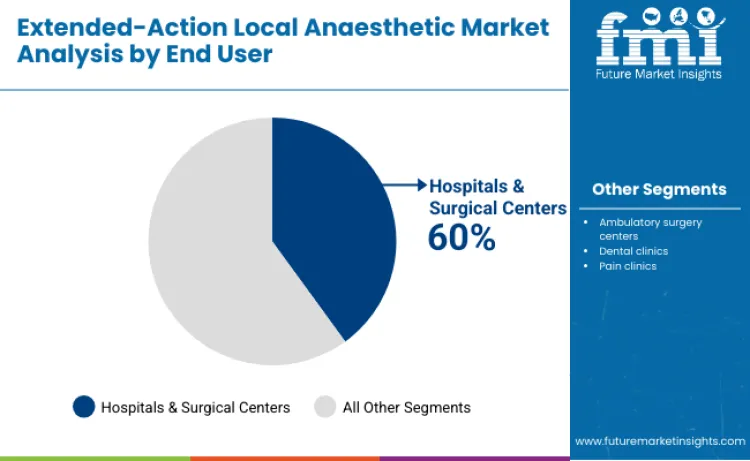
Hospitals and surgical centers form the largest end-user segment, generating about 60% of revenue in 2025, driven by high surgical volumes and the need for consistent, long-acting analgesia across orthopedic, abdominal, gynecologic, thoracic, and trauma procedures. Their structured perioperative teams, ERAS integration, and formulary oversight accelerate adoption of sustained-release anesthetics to reduce opioid use and improve recovery outcomes. Ambulatory surgery centers hold notable share as outpatient procedures expand, while pain and dental clinics adopt extended-duration solutions for chronic pain management and prolonged postoperative comfort without narcotic dependence.
What Are the Major Drivers, Restraints, and Emerging Trends Influencing the Extended-Action Local Anaesthetic Market Growing Worldwide?
The extended-action local anaesthetic market is driven by global adoption of opioid-sparing pain-management strategies and modernization of perioperative care. Hospitals increasingly implement multimodal analgesia protocols that reduce opioid exposure and postoperative complications, accelerating demand for long-acting formulations. Growth in outpatient and minimally invasive procedures, including orthopedic, plastic, gynecologic, and abdominal surgeries, further strengthens uptake as sustained-release anesthetics support early ambulation and same-day discharge. Expansion of ERAS programs across major health systems reinforces their necessity by improving postoperative comfort, reducing resource utilization, and enhancing overall patient recovery outcomes.
What Are the Major Restraints Limiting Adoption of Extended-Action Local Anaesthetics?
Market restraints include premium pricing, with extended-action formulations costing three to six times more than conventional anesthetics, creating reimbursement challenges for hospitals with constrained budgets. Variability in insurance policies limits accessibility, particularly in lower-income regions. Technical considerations, including dosing familiarity, infiltration techniques, and practitioner training, affect adoption among anesthesiologists accustomed to catheters or standard injections. Clinical hesitation persists where long-term safety and performance consistency are prioritized, especially for newer polymer-based sustained-release systems and depot platforms requiring additional evidence to support widespread perioperative integration.
What Emerging Trends Are Shaping the Extended-Action Local Anaesthetic Market Worldwide?
Key trends include rapid uptake in North America and Europe driven by opioid-reduction mandates, with Asia-Pacific adoption rising alongside expanding surgical infrastructure. Pharmaceutical innovation is shifting toward advanced encapsulation technologies, bioresorbable microspheres, and nerve-targeted depot systems extending analgesia beyond 72 hours. Integration with ultrasound-guided regional anesthesia and catheter-free nerve block techniques increases procedural efficiency and broadens clinical applicability. Market dynamics may evolve as generics, hybrid sustained-release formulations, and device-assisted regional anesthesia technologies emerge, influencing provider preferences across perioperative pain-management pathways and long-term recovery frameworks.
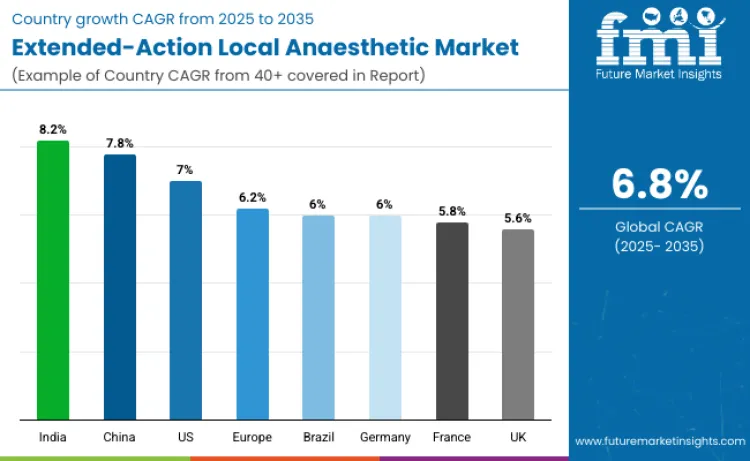
How Are Leading Countries Driving Growth in the Extended-Action Local Anaesthetic Market?
| Country | CAGR (2025 to 2035) |
|---|---|
| USA | 7.0% |
| Brazil | 6.0% |
| China | 7.8% |
| India | 8.2% |
| Europe | 6.2% |
| Germany | 6.0% |
| France | 5.8% |
| UK | 5.6% |
The extended-action local anaesthetic landscape is expanding rapidly, with the United States leading growth at a CAGR of 7.0%, supported by advanced surgical infrastructure, ERAS adherence, and strong opioid-reduction mandates. Europe follows at 6.2%, with Germany (6.0%), France (5.8%), and the UK (5.6%) driving adoption through established regional-anesthesia practices. India shows the fastest acceleration at 8.2%, driven by rising surgical volumes and growing ambulatory care capacity, while China advances at 7.8% through hospital modernization. Brazil expands at 6.0% as private hospital networks grow. Together, North America and Europe anchor premium usage, while Asia-Pacific drives high-volume adoption.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
How Are Surgical Expansion & Opioid-Reduction Policies Accelerating Market Growth in China?
China shows strong momentum in the extended-action local anaestheticmarket, expanding at a CAGR of 7.8% through 2035, supported by rapid growth in surgical infrastructure and rising adoption of multimodal analgesia across major hospitals in Beijing, Shanghai, Shenzhen, and Guangzhou. National policies promoting reduced postoperative opioid use and ERAS-aligned care are accelerating uptake of long-acting formulations. Expansion of day-care surgery, improved anesthesia training, and domestic innovation in sustained-release anesthetics are strengthening accessibility and affordability, enabling broader adoption across public and private hospitals while advancing overall postoperative recovery standards.
Why Are Perioperative Care Improvements & Clinical Training Programs Enhancing Extended-Action Local Anaesthetic Adoption in India?
India’s extended-action local anaesthetic market is expanding rapidly, advancing at a CAGR of 8.2% through 2035 as multispecialty hospitals and ambulatory surgery units in Mumbai, Delhi-NCR, Bengaluru, and Hyderabad adopt long-acting anesthetic protocols. Growth is driven by rising surgical volumes, ERAS pathway integration, and strong national focus on opioid-sparing recovery. Clinicians increasingly use sustained-release anesthetics to enhance mobility, shorten hospital stays, and support home-based recovery. Expanded anesthesia training, government modernization efforts, and partnerships with global pharma companies are further strengthening clinical capability and accelerating nationwide adoption.
How Do Advanced Surgical Standards Sustain Market Leadership in Germany?
Extended-action local anaesthetic market in Germany advanced healthcare system enables strong use of extended-action local anesthetic solutions, improving post-operative pain control, lowering opioid consumption, and accelerating mobilization across orthopedic, abdominal, and day-care surgeries. Major centers in Berlin, Munich, Hamburg, and Frankfurt integrate sustained-release anesthetics into ERAS and regional anesthesia pathways, supported by rigorous evidence-based standards. Clinicians emphasize predictable analgesic duration and long-term safety, sustaining demand for premium formulations. The segment records a 6.0% CAGR through 2035, supported by ERAS adoption, enhanced anesthesia training, stable distributor partnerships, and catheter-free recovery techniques.
Why Is Expanding Surgical Infrastructure Fueling Extended-Action Local Anaesthetic Demand in Brazil?
Extended-action local anaesthetic market in Brazil leads Latin American adoption of extended-action local anesthetic solutions, supported by rising surgical volumes, expansion of private hospitals, and stronger opioid-sparing postoperative practices. The segment grows at a 6.0% CAGR through 2035, driven by increased orthopedic, bariatric, cosmetic, and general surgeries across São Paulo, Rio de Janeiro, Brasília, and Porto Alegre. Providers are adopting long-acting formulations to improve recovery, reduce opioid use, and enable rapid discharge in day-care settings. Growing ERAS participation, expanded regional anesthesia training, and collaborations with multinational distributors are strengthening nationwide access to sustained-release anesthetics.
How Do Advanced Perioperative Practices Strengthen Extended-Action Local Anaesthetic Market Performance in the United States?
Sales of extended-action local anaestheticin United States shows mature adoption of extended-action local anesthetics, supported by advanced perioperative pain-management protocols, strong ERAS implementation, and a well-established regional anesthesia culture across major hubs in California, Texas, New York, and Massachusetts. The segment grows at a 7.0% CAGR through 2035, driven by opioid-reduction mandates, payer incentives, and expansion of outpatient surgical care. Academic centers and integrated health systems routinely use extended-release anesthetics to improve recovery and reduce opioid dependence. Pharmaceutical innovators strengthen national leadership through advanced formulations, clinician education, and real-world evidence programs.
How Is Perioperative Care Optimization Supporting Market Growth in the United Kingdom?
Demand for extended-action local anaesthetic in United Kingdom is accelerating adoption of extended-action local anesthetics as major hubs such as London, Manchester, Birmingham, and Edinburgh integrate ERAS frameworks and strengthen post-operative pain standards across NHS and private surgical centers. The segment grows at a 5.6% CAGR through 2035, supported by national opioid-reduction initiatives, day-surgery expansion, and focus on early ambulation. Clinicians incorporate long-acting formulations into multimodal pathways to improve patient experience and reduce opioid use. National guidelines, academic training programs, and NHS research collaborations further reinforce uptake across orthopedic, gynecologic, colorectal, and plastic-surgery procedures.
How Is Extended-Action Local Anaesthetic Market Expanding Across Europe?
Europe’s extended-action local anaesthetic market is set to grow from USD 345.0 million in 2025 to USD 621.7 million by 2035, at a 6.2% CAGR. Germany leads with 6.0% share in 2025, supported by advanced perioperative systems and strong ERAS integration across Berlin, Munich, and Hamburg. France follows with 23% in 2025 and 22% in 2035, driven by uptake in oncology, orthopedic, and ambulatory surgery programs in Paris and Lyon.
Italy holds 12% in 2025 and 11.5% in 2035, reflecting rising orthopedic and general surgery demand. Spain records 10% in 2025 and 9.5% in 2035 as ERAS adoption accelerates in Madrid and Barcelona. The Netherlands maintains 8.5%, while Rest of Western Europe grows from 23% to 24.5% with modernization of multimodal surgical care.
Precision-Driven Analgesia Standards Dominate Requirements in Japan
The extended-action local anaesthetic market in Japan is demonstrates a highly mature and precision-focused clinical environment, characterized by sophisticated adoption of long-acting analgesic techniques across leading university hospitals, surgical centers, and specialty orthopedic and oncology facilities. Japan’s strong focus on controlled analgesic duration, pharmacokinetic consistency, and patient-comfort optimization drives demand for premium extended-release local anesthetics that support rigorous safety and recovery standards.
The market benefits from strong collaboration between domestic pharmaceutical companies and advanced anesthesiology departments, creating tightly integrated clinical pathways that prioritize predictable anesthesia performance and minimal systemic exposure. Clinicians in Tokyo, Osaka, and other metropolitan hubs showcase advanced adoption of sustained-release anesthetic solutions, where products deliver stable analgesic effects aligned with Japan’s stringent perioperative performance metrics and postoperative recovery benchmarks.
International Clinical Collaboration Drives Advanced Adoption in South Korea
Extended-action local anaestheticmarket in South Korea is characterized by strong involvement of international pharmaceutical and pain-management technology providers, who maintain significant clinical presence through advanced medical education programs, perioperative training support, and technical service partnerships across leading hospitals. The market demonstrates growing focus on enhanced regional anesthesia practices and opioid-sparing recovery pathways, as Korean surgical centers increasingly incorporate long-acting injectable anesthetics into protocols for orthopedic, cosmetic, obstetric, and general surgery cases.
Large tertiary hospitals and private surgical networks in Seoul, Busan, and Daegu increasingly collaborate with multinational drug manufacturers to gain access to sustained-release formulations, clinical workshops, and best-practice standards aligned with global ERAS and acute pain-management frameworks. The competitive landscape reflects expanding collaboration between international anesthesia companies and Korean pain-medicine societies, creating hybrid adoption models that combine global product innovation with localized clinical integration and physician-training programs.
How Competitive Is the Global Extended-Action Local Anaesthetic Market and What Defines Its Structure?
The extended-action local anaesthetic landscape comprises a moderately concentrated group of clinically active pharmaceutical developers, with leading companies holding a dominant share due to strong clinical evidence, proprietary formulation technology, and deep access to hospital formularies. Competition centers on analgesic duration, safety performance, delivery innovation, and recovery outcomes rather than pricing. PaciraBioSciences remains the foremost participant, supported by broad adoption of its sustained-release bupivacaine platform and alignment with postoperative ERAS pathways. Market leaders such as PaciraBioSciences, Heron Therapeutics, and AstraZeneca strengthen their positions through advanced delivery systems, extensive safety datasets, influential perioperative networks, and established reimbursement support.
Challenger firms including B. Braun and Viatris maintain relevance through wide anaesthesia portfolios, strong procurement relationships, and cost-aligned offerings suitable for routine and emergency surgical settings. Their strategies rely on tender participation and long-standing clinician familiarity with injectable anaesthetics. Regional manufacturers across Asia-Pacific and Latin America intensify competition with lower-cost formulations and tender-driven contracting, particularly within public systems. Overall dynamics continue to favor companies delivering proven extended-release pharmacology, dependable plasma profiles, procedure-specific clinical validation, and education programs that reinforce opioid-sparing pathways.
Key Players in the Extended-Action Local Anaesthetic market
- PaciraBioSciences
- Heron Therapeutics
- AstraZeneca
- B. Braun
- Mylan / Viatris
- Others
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 1,150.0 million |
| Product Type | Liposomal formulations, Prolonged-release injectables, Transdermal patches, Device -integrated nerve-block systems, Others |
| Application | Surgical analgesia, post-op pain management, Chronic pain / regional blocks, Dental & minor procedures |
| End User | Hospitals & surgical centers, A mbulatory surgery centers, Dental clinics, Pain clinics |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Country Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40+ countries |
| Key Companies Profiled | Pacira BioSciences, Heron Therapeutics, AstraZeneca, B. Braun, Mylan / Viatris, Others |
| Additional Attributes | Dollar sales by extended-release anaesthetic formulations and surgical applications, regional adoption across North America, Europe, and Asia-Pacific, competitive landscape of ERAS-focused innovators, performance benchmarks for analgesia duration and opioid reduction, and advances in liposomal, polymer, and catheter-based delivery driving integration into ERAS and ambulatory care models. |
Extended-Action Local Anaesthetic market by Segments
Product Type:
- Liposomal formulations
- Prolonged release-injectables
- Transdermal patches
- Device-integrated nerve-block systems
- Others
Application:
- Surgical analgesia
- Post-op pain management
- Chronic pain / regional blocks
- Dental & minor procedures
End User:
- Hospitals & surgical centers
- Ambulatory surgery centers
- Dental clinics
- Pain clinics
Region:
- Asia Pacific
- China
- India
- Japan
- South Korea
- Vietnam
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Netherlands
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the extended-action local anaesthetic market in 2025?
The global extended-action local anaesthetic market is valued at USD 1,150.0 million in 2025.
What will be the size of the extended-action local anaesthetic market in 2035?
The market is projected to reach USD 2,220.3 million by 2035.
How fast will the extended-action local anaesthetic market grow between 2025 and 2035?
The market will grow at a CAGR of 6.8% from 2025 to 2035.
Which application segment leads the extended-action local anaesthetic market in 2025?
Surgical analgesia leads the market with a 50% share in 2025.
Who are the major players in the extended-action local anaesthetic market?
Key players include Pacira BioSciences, Heron Therapeutics, AstraZeneca, B. Braun, and Mylan/Viatris.
Table of Content
- Executive Summary
- Regional Level Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Assessment
- Market Trade Assessment
- Products Market Assessment
- Investment Feasibility Matrix
- Value Chain Analysis
- Participants and their Roles
- Profit Margin Analysis
- Operating Margins By Region
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- By Key Regions
- By Key Countries
- Regional Parent Market Outlook
- Product Launches & Development
- Technological Innovation
- Market Dynamics
- Regional Level Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size USD Million & Units Analysis, 2020-2024
- Current and Future Market Size USD Million & Units Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Regional Level Market Analysis 2020-2024 and Forecast 2025-2035, By Product Type
- Introduction / Key Findings
- Historical Market Size USD Million & Units Analysis By Product Type, 2020-2024
- Current and Future Market Size USD Million & Units Analysis and Forecast By Product Type, 2025-2035
- Liposomal formulations
- Prolonged-release injectables
- Transdermal patches
- Device-integrated nerve-block systems
- Others
- Y-o-Y Growth Trend Analysis By Product Type, 2020-2024
- Absolute $ Opportunity Analysis By Product Type, 2025-2035
- Regional Level Market Analysis 2020-2024 and Forecast 2025-2035, By Application
- Introduction / Key Findings
- Historical Market Size USD Million & Units Analysis By Application, 2020-2024
- Current and Future Market Size USD Million & Units Analysis and Forecast By Application, 2025-2035
- Surgical analgesia
- Post-op pain management
- Chronic pain / regional blocks
- Dental & minor procedures
- Y-o-Y Growth Trend Analysis By Application, 2020-2024
- Absolute $ Opportunity Analysis By Application, 2025-2035
- Regional Level Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size USD Million & Units Analysis By End User, 2020-2024
- Current and Future Market Size USD Million & Units Analysis and Forecast By End User, 2025-2035
- Hospitals & surgical centers
- Ambulatory surgery centers
- Dental clinics
- Pain clinics
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Regional Level Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size USD Million & Units Analysis By Region, 2020-2024
- Current Market Size USD Million & Units Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- East Asia
- South Asia
- Western Europe
- Eastern Europe
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Argentina
- Rest of Latin America
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- South Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN Countries
- Australia & New Zealand
- Rest of South Asia
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- France
- Italy
- Spain
- BENELUX
- Nordic Countries
- Rest of Western Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Hungary
- Poland
- Rest of Eastern Europe
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size USD Million & Units Trend Analysis By Market Taxonomy, 2020-2024
- Market Size USD Million & Units Forecast By Market Taxonomy, 2025-2035
- By Country
- Saudi Arabia
- Other GCC Countries
- Türkiye
- South Africa
- Rest of MEA
- By Product Type
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- By End User
- Key Takeaways
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Pacira BioSciences
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Heron Therapeutics
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- AstraZeneca
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- B. Braun
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Mylan / Viatris
- Overview
- Product Portfolio
- Key Financials
- Key Developments
- Global/Regional Footprint
- Strategy Overview
- Marketing Strategy
- SWOT
- Analyst Comment
- Pacira BioSciences
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 2: Global Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 3: Global Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 4: Global Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 5: Global Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 6: Global Market Size (USD Million & Units), by Others, 2020-2035
- Table 7: Global Market Size (USD Million & Units), by Application, 2020-2035
- Table 8: Global Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 9: Global Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 10: Global Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 11: Global Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 12: Global Market Size (USD Million & Units), by End User, 2020-2035
- Table 13: Global Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 14: Global Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 15: Global Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 16: Global Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 17: North America Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 18: North America Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 19: North America Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 20: North America Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 21: North America Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 22: North America Market Size (USD Million & Units), by Others, 2020-2035
- Table 23: North America Market Size (USD Million & Units), by Application, 2020-2035
- Table 24: North America Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 25: North America Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 26: North America Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 27: North America Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 28: North America Market Size (USD Million & Units), by End User, 2020-2035
- Table 29: North America Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 30: North America Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 31: North America Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 32: North America Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 33: USA Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 34: USA Market Size (USD Million & Units), by Application, 2020-2035
- Table 35: USA Market Size (USD Million & Units), by End User, 2020-2035
- Table 36: Canada Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 37: Canada Market Size (USD Million & Units), by Application, 2020-2035
- Table 38: Canada Market Size (USD Million & Units), by End User, 2020-2035
- Table 39: Mexico Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 40: Mexico Market Size (USD Million & Units), by Application, 2020-2035
- Table 41: Mexico Market Size (USD Million & Units), by End User, 2020-2035
- Table 42: Latin America Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 43: Latin America Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 44: Latin America Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 45: Latin America Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 46: Latin America Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 47: Latin America Market Size (USD Million & Units), by Others, 2020-2035
- Table 48: Latin America Market Size (USD Million & Units), by Application, 2020-2035
- Table 49: Latin America Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 50: Latin America Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 51: Latin America Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 52: Latin America Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 53: Latin America Market Size (USD Million & Units), by End User, 2020-2035
- Table 54: Latin America Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 55: Latin America Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 56: Latin America Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 57: Latin America Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 58: Brazil Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 59: Brazil Market Size (USD Million & Units), by Application, 2020-2035
- Table 60: Brazil Market Size (USD Million & Units), by End User, 2020-2035
- Table 61: Argentina Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 62: Argentina Market Size (USD Million & Units), by Application, 2020-2035
- Table 63: Argentina Market Size (USD Million & Units), by End User, 2020-2035
- Table 64: Rest of Latin America Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 65: Rest of Latin America Market Size (USD Million & Units), by Application, 2020-2035
- Table 66: Rest of Latin America Market Size (USD Million & Units), by End User, 2020-2035
- Table 67: East Asia Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 68: East Asia Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 69: East Asia Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 70: East Asia Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 71: East Asia Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 72: East Asia Market Size (USD Million & Units), by Others, 2020-2035
- Table 73: East Asia Market Size (USD Million & Units), by Application, 2020-2035
- Table 74: East Asia Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 75: East Asia Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 76: East Asia Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 77: East Asia Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 78: East Asia Market Size (USD Million & Units), by End User, 2020-2035
- Table 79: East Asia Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 80: East Asia Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 81: East Asia Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 82: East Asia Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 83: China Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 84: China Market Size (USD Million & Units), by Application, 2020-2035
- Table 85: China Market Size (USD Million & Units), by End User, 2020-2035
- Table 86: Japan Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 87: Japan Market Size (USD Million & Units), by Application, 2020-2035
- Table 88: Japan Market Size (USD Million & Units), by End User, 2020-2035
- Table 89: South Korea Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 90: South Korea Market Size (USD Million & Units), by Application, 2020-2035
- Table 91: South Korea Market Size (USD Million & Units), by End User, 2020-2035
- Table 92: South Asia Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 93: South Asia Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 94: South Asia Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 95: South Asia Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 96: South Asia Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 97: South Asia Market Size (USD Million & Units), by Others, 2020-2035
- Table 98: South Asia Market Size (USD Million & Units), by Application, 2020-2035
- Table 99: South Asia Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 100: South Asia Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 101: South Asia Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 102: South Asia Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 103: South Asia Market Size (USD Million & Units), by End User, 2020-2035
- Table 104: South Asia Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 105: South Asia Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 106: South Asia Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 107: South Asia Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 108: India Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 109: India Market Size (USD Million & Units), by Application, 2020-2035
- Table 110: India Market Size (USD Million & Units), by End User, 2020-2035
- Table 111: ASEAN Countries Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 112: ASEAN Countries Market Size (USD Million & Units), by Application, 2020-2035
- Table 113: ASEAN Countries Market Size (USD Million & Units), by End User, 2020-2035
- Table 114: Australia & New Zealand Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 115: Australia & New Zealand Market Size (USD Million & Units), by Application, 2020-2035
- Table 116: Australia & New Zealand Market Size (USD Million & Units), by End User, 2020-2035
- Table 117: Rest of South Asia Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 118: Rest of South Asia Market Size (USD Million & Units), by Application, 2020-2035
- Table 119: Rest of South Asia Market Size (USD Million & Units), by End User, 2020-2035
- Table 120: Western Europe Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 121: Western Europe Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 122: Western Europe Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 123: Western Europe Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 124: Western Europe Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 125: Western Europe Market Size (USD Million & Units), by Others, 2020-2035
- Table 126: Western Europe Market Size (USD Million & Units), by Application, 2020-2035
- Table 127: Western Europe Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 128: Western Europe Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 129: Western Europe Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 130: Western Europe Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 131: Western Europe Market Size (USD Million & Units), by End User, 2020-2035
- Table 132: Western Europe Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 133: Western Europe Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 134: Western Europe Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 135: Western Europe Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 136: Germany Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 137: Germany Market Size (USD Million & Units), by Application, 2020-2035
- Table 138: Germany Market Size (USD Million & Units), by End User, 2020-2035
- Table 139: UK Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 140: UK Market Size (USD Million & Units), by Application, 2020-2035
- Table 141: UK Market Size (USD Million & Units), by End User, 2020-2035
- Table 142: France Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 143: France Market Size (USD Million & Units), by Application, 2020-2035
- Table 144: France Market Size (USD Million & Units), by End User, 2020-2035
- Table 145: Italy Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 146: Italy Market Size (USD Million & Units), by Application, 2020-2035
- Table 147: Italy Market Size (USD Million & Units), by End User, 2020-2035
- Table 148: Spain Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 149: Spain Market Size (USD Million & Units), by Application, 2020-2035
- Table 150: Spain Market Size (USD Million & Units), by End User, 2020-2035
- Table 151: BENELUX Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 152: BENELUX Market Size (USD Million & Units), by Application, 2020-2035
- Table 153: BENELUX Market Size (USD Million & Units), by End User, 2020-2035
- Table 154: Nordic Countries Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 155: Nordic Countries Market Size (USD Million & Units), by Application, 2020-2035
- Table 156: Nordic Countries Market Size (USD Million & Units), by End User, 2020-2035
- Table 157: Rest of Western Europe Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 158: Rest of Western Europe Market Size (USD Million & Units), by Application, 2020-2035
- Table 159: Rest of Western Europe Market Size (USD Million & Units), by End User, 2020-2035
- Table 160: Eastern Europe Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 161: Eastern Europe Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 162: Eastern Europe Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 163: Eastern Europe Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 164: Eastern Europe Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 165: Eastern Europe Market Size (USD Million & Units), by Others, 2020-2035
- Table 166: Eastern Europe Market Size (USD Million & Units), by Application, 2020-2035
- Table 167: Eastern Europe Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 168: Eastern Europe Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 169: Eastern Europe Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 170: Eastern Europe Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 171: Eastern Europe Market Size (USD Million & Units), by End User, 2020-2035
- Table 172: Eastern Europe Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 173: Eastern Europe Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 174: Eastern Europe Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 175: Eastern Europe Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 176: Russia Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 177: Russia Market Size (USD Million & Units), by Application, 2020-2035
- Table 178: Russia Market Size (USD Million & Units), by End User, 2020-2035
- Table 179: Hungary Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 180: Hungary Market Size (USD Million & Units), by Application, 2020-2035
- Table 181: Hungary Market Size (USD Million & Units), by End User, 2020-2035
- Table 182: Poland Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 183: Poland Market Size (USD Million & Units), by Application, 2020-2035
- Table 184: Poland Market Size (USD Million & Units), by End User, 2020-2035
- Table 185: Rest of Eastern Europe Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 186: Rest of Eastern Europe Market Size (USD Million & Units), by Application, 2020-2035
- Table 187: Rest of Eastern Europe Market Size (USD Million & Units), by End User, 2020-2035
- Table 188: Middle East & Africa Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 189: Middle East & Africa Market Size (USD Million & Units), by Liposomal formulations, 2020-2035
- Table 190: Middle East & Africa Market Size (USD Million & Units), by Prolonged-release injectables, 2020-2035
- Table 191: Middle East & Africa Market Size (USD Million & Units), by Transdermal patches, 2020-2035
- Table 192: Middle East & Africa Market Size (USD Million & Units), by Device-integrated nerve-block systems, 2020-2035
- Table 193: Middle East & Africa Market Size (USD Million & Units), by Others, 2020-2035
- Table 194: Middle East & Africa Market Size (USD Million & Units), by Application, 2020-2035
- Table 195: Middle East & Africa Market Size (USD Million & Units), by Surgical analgesia, 2020-2035
- Table 196: Middle East & Africa Market Size (USD Million & Units), by Post-op pain management, 2020-2035
- Table 197: Middle East & Africa Market Size (USD Million & Units), by Chronic pain / regional blocks, 2020-2035
- Table 198: Middle East & Africa Market Size (USD Million & Units), by Dental & minor procedures, 2020-2035
- Table 199: Middle East & Africa Market Size (USD Million & Units), by End User, 2020-2035
- Table 200: Middle East & Africa Market Size (USD Million & Units), by Hospitals & surgical centers, 2020-2035
- Table 201: Middle East & Africa Market Size (USD Million & Units), by Ambulatory surgery centers, 2020-2035
- Table 202: Middle East & Africa Market Size (USD Million & Units), by Dental clinics, 2020-2035
- Table 203: Middle East & Africa Market Size (USD Million & Units), by Pain clinics, 2020-2035
- Table 204: Saudi Arabia Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 205: Saudi Arabia Market Size (USD Million & Units), by Application, 2020-2035
- Table 206: Saudi Arabia Market Size (USD Million & Units), by End User, 2020-2035
- Table 207: Other GCC Countries Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 208: Other GCC Countries Market Size (USD Million & Units), by Application, 2020-2035
- Table 209: Other GCC Countries Market Size (USD Million & Units), by End User, 2020-2035
- Table 210: Türkiye Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 211: Türkiye Market Size (USD Million & Units), by Application, 2020-2035
- Table 212: Türkiye Market Size (USD Million & Units), by End User, 2020-2035
- Table 213: South Africa Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 214: South Africa Market Size (USD Million & Units), by Application, 2020-2035
- Table 215: South Africa Market Size (USD Million & Units), by End User, 2020-2035
- Table 216: Rest of MEA Market Size (USD Million & Units), by Product Type, 2020-2035
- Table 217: Rest of MEA Market Size (USD Million & Units), by Application, 2020-2035
- Table 218: Rest of MEA Market Size (USD Million & Units), by End User, 2020-2035
List of Figures
- Figure 1: Global Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 2: Global Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 3: Global Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 4: Global Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 5: Global Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 6: Global Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 7: Global Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 8: Global Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 9: Global Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 10: Global Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 11: Global Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 12: Global Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 13: Global Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 14: Global Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 15: Global Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 16: Global Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 17: North America Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 18: North America Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 19: North America Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 20: North America Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 21: North America Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 22: North America Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 23: North America Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 24: North America Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 25: North America Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 26: North America Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 27: North America Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 28: North America Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 29: North America Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 30: North America Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 31: North America Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 32: North America Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 33: USA Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 34: USA Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 35: USA Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 36: USA Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 37: USA Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 38: USA Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 39: Canada Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 40: Canada Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 41: Canada Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 42: Canada Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 43: Canada Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 44: Canada Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 45: Mexico Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 46: Mexico Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 47: Mexico Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 48: Mexico Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 49: Mexico Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 50: Mexico Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 51: Latin America Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 52: Latin America Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 53: Latin America Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 54: Latin America Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 55: Latin America Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 56: Latin America Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 57: Latin America Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 58: Latin America Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 59: Latin America Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 60: Latin America Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 61: Latin America Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 62: Latin America Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 63: Latin America Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 64: Latin America Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 65: Latin America Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 66: Latin America Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 67: Brazil Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 68: Brazil Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 69: Brazil Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 70: Brazil Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 71: Brazil Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 72: Brazil Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 73: Argentina Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 74: Argentina Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 75: Argentina Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 76: Argentina Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 77: Argentina Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 78: Argentina Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 79: Rest of Latin America Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 80: Rest of Latin America Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 81: Rest of Latin America Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 82: Rest of Latin America Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 83: Rest of Latin America Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 84: Rest of Latin America Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 85: East Asia Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 86: East Asia Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 87: East Asia Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 88: East Asia Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 89: East Asia Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 90: East Asia Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 91: East Asia Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 92: East Asia Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 93: East Asia Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 94: East Asia Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 95: East Asia Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 96: East Asia Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 97: East Asia Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 98: East Asia Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 99: East Asia Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 100: East Asia Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 101: China Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 102: China Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 103: China Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 104: China Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 105: China Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 106: China Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 107: Japan Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 108: Japan Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 109: Japan Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 110: Japan Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 111: Japan Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 112: Japan Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 113: South Korea Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 114: South Korea Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 115: South Korea Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 116: South Korea Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 117: South Korea Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 118: South Korea Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 119: South Asia Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 120: South Asia Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 121: South Asia Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 122: South Asia Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 123: South Asia Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 124: South Asia Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 125: South Asia Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 126: South Asia Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 127: South Asia Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 128: South Asia Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 129: South Asia Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 130: South Asia Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 131: South Asia Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 132: South Asia Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 133: South Asia Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 134: South Asia Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 135: India Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 136: India Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 137: India Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 138: India Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 139: India Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 140: India Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 141: ASEAN Countries Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 142: ASEAN Countries Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 143: ASEAN Countries Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 144: ASEAN Countries Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 145: ASEAN Countries Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 146: ASEAN Countries Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 147: Australia & New Zealand Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 148: Australia & New Zealand Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 149: Australia & New Zealand Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 150: Australia & New Zealand Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 151: Australia & New Zealand Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 152: Australia & New Zealand Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 153: Rest of South Asia Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 154: Rest of South Asia Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 155: Rest of South Asia Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 156: Rest of South Asia Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 157: Rest of South Asia Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 158: Rest of South Asia Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 159: Western Europe Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 160: Western Europe Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 161: Western Europe Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 162: Western Europe Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 163: Western Europe Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 164: Western Europe Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 165: Western Europe Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 166: Western Europe Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 167: Western Europe Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 168: Western Europe Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 169: Western Europe Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 170: Western Europe Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 171: Western Europe Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 172: Western Europe Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 173: Western Europe Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 174: Western Europe Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 175: Germany Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 176: Germany Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 177: Germany Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 178: Germany Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 179: Germany Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 180: Germany Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 181: UK Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 182: UK Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 183: UK Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 184: UK Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 185: UK Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 186: UK Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 187: France Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 188: France Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 189: France Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 190: France Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 191: France Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 192: France Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 193: Italy Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 194: Italy Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 195: Italy Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 196: Italy Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 197: Italy Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 198: Italy Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 199: Spain Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 200: Spain Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 201: Spain Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 202: Spain Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 203: Spain Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 204: Spain Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 205: BENELUX Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 206: BENELUX Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 207: BENELUX Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 208: BENELUX Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 209: BENELUX Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 210: BENELUX Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 211: Nordic Countries Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 212: Nordic Countries Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 213: Nordic Countries Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 214: Nordic Countries Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 215: Nordic Countries Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 216: Nordic Countries Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 217: Rest of Western Europe Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 218: Rest of Western Europe Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 219: Rest of Western Europe Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 220: Rest of Western Europe Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 221: Rest of Western Europe Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 222: Rest of Western Europe Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 223: Eastern Europe Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 224: Eastern Europe Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 225: Eastern Europe Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 226: Eastern Europe Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 227: Eastern Europe Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 228: Eastern Europe Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 229: Eastern Europe Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 230: Eastern Europe Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 231: Eastern Europe Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 232: Eastern Europe Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 233: Eastern Europe Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 234: Eastern Europe Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 235: Eastern Europe Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 236: Eastern Europe Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 237: Eastern Europe Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 238: Eastern Europe Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 239: Russia Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 240: Russia Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 241: Russia Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 242: Russia Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 243: Russia Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 244: Russia Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 245: Hungary Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 246: Hungary Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 247: Hungary Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 248: Hungary Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 249: Hungary Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 250: Hungary Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 251: Poland Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 252: Poland Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 253: Poland Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 254: Poland Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 255: Poland Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 256: Poland Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 257: Rest of Eastern Europe Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 258: Rest of Eastern Europe Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 259: Rest of Eastern Europe Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 260: Rest of Eastern Europe Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 261: Rest of Eastern Europe Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 262: Rest of Eastern Europe Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 263: Middle East & Africa Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 264: Middle East & Africa Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 265: Middle East & Africa Market (USD Million & Units) Forecast, by Prolonged-release injectables, 2020-2035
- Figure 266: Middle East & Africa Market (USD Million & Units) Forecast, by Transdermal patches, 2020-2035
- Figure 267: Middle East & Africa Market (USD Million & Units) Forecast, by Device-integrated nerve-block systems, 2020-2035
- Figure 268: Middle East & Africa Market (USD Million & Units) Forecast, by Others, 2020-2035
- Figure 269: Middle East & Africa Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 270: Middle East & Africa Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 271: Middle East & Africa Market (USD Million & Units) Forecast, by Post-op pain management, 2020-2035
- Figure 272: Middle East & Africa Market (USD Million & Units) Forecast, by Chronic pain / regional blocks, 2020-2035
- Figure 273: Middle East & Africa Market (USD Million & Units) Forecast, by Dental & minor procedures, 2020-2035
- Figure 274: Middle East & Africa Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 275: Middle East & Africa Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 276: Middle East & Africa Market (USD Million & Units) Forecast, by Ambulatory surgery centers, 2020-2035
- Figure 277: Middle East & Africa Market (USD Million & Units) Forecast, by Dental clinics, 2020-2035
- Figure 278: Middle East & Africa Market (USD Million & Units) Forecast, by Pain clinics, 2020-2035
- Figure 279: Saudi Arabia Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 280: Saudi Arabia Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 281: Saudi Arabia Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 282: Saudi Arabia Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 283: Saudi Arabia Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 284: Saudi Arabia Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 285: Other GCC Countries Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 286: Other GCC Countries Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 287: Other GCC Countries Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 288: Other GCC Countries Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 289: Other GCC Countries Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 290: Other GCC Countries Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 291: Türkiye Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 292: Türkiye Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 293: Türkiye Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 294: Türkiye Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 295: Türkiye Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 296: Türkiye Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 297: South Africa Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 298: South Africa Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 299: South Africa Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 300: South Africa Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 301: South Africa Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 302: South Africa Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 303: Rest of MEA Market (USD Million & Units) Forecast, by Product Type, 2020-2035
- Figure 304: Rest of MEA Market (USD Million & Units) Forecast, by Liposomal formulations, 2020-2035
- Figure 305: Rest of MEA Market (USD Million & Units) Forecast, by Application, 2020-2035
- Figure 306: Rest of MEA Market (USD Million & Units) Forecast, by Surgical analgesia, 2020-2035
- Figure 307: Rest of MEA Market (USD Million & Units) Forecast, by End User, 2020-2035
- Figure 308: Rest of MEA Market (USD Million & Units) Forecast, by Hospitals & surgical centers, 2020-2035
- Figure 309: Pacira BioSciences - Company Snapshot
- Figure 310: Pacira BioSciences - SWOT Analysis
- Figure 311: Heron Therapeutics - Company Snapshot
- Figure 312: Heron Therapeutics - SWOT Analysis
- Figure 313: AstraZeneca - Company Snapshot
- Figure 314: AstraZeneca - SWOT Analysis
- Figure 315: B. Braun - Company Snapshot
- Figure 316: B. Braun - SWOT Analysis
- Figure 317: Mylan / Viatris - Company Snapshot
- Figure 318: Mylan / Viatris - SWOT Analysis
- Figure 319: Others - Company Snapshot
- Figure 320: Others - SWOT Analysis

