Dental Anaesthetic Market
The Dental Anaesthetic Market is segmented by Product Type (Lidocaine, Mepivacaine, Prilocaine, Bupivacaine 0.5% With Epinephrine 1:200,000, Articaine), Mode of Administration (Local Infiltration, Maxillary Anaesthesia, Supraperiosteal Injection, Intraseptal Injection, Periodontal Ligament Injection, Field Block, Periapical Injection, Nerve Block, Posterior Superior Alveolar, Middle Superior Alveolar, Anterior Superior Alveolar, Mandibular Anaesthesia, Nerve Block, IAN, Akinosi, Gow-Gates, Tissue or Skin, Others), Duration of Action (Medium, Short, Long), Distribution Channel (Clinics, Hospital Clinics, Pharmacies And Drug Stores, E-Commerce, Other), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Dental Anaesthetic Market Size, Market Forecast and Outlook By FMI
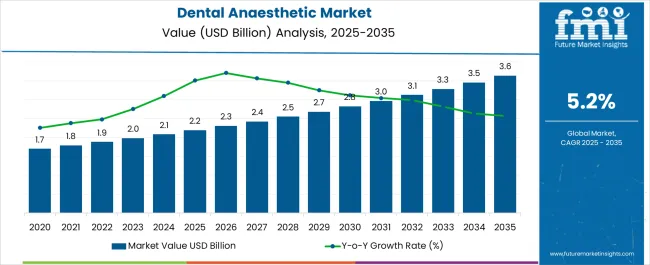
In 2025, the dental anaesthetic market was valued at USD 2.2 billion. Based on Future Market Insights' analysis, demand for dental anaesthetics is estimated to grow to USD 2.31 billion in 2026 and USD 3.81 billion by 2036. FMI projects a CAGR of 5.2% during the forecast period.
An absolute dollar growth of USD 1.50 billion between 2026 and 2036 reflects a structural shift from undifferentiated generic lidocaine toward premium-priced articaine formulations and safety-engineered delivery systems that reduce needlestick injury risk and improve onset predictability. As per FMI's analysis, growth depends on the conversion of dental procedures from basic restorative work to complex surgical interventions requiring longer-acting or more potent anaesthetic agents, and on the penetration of single-dose, pre-filled cartridge systems into infection control protocols.
Summary of the Dental Anaesthetic Market
- Market Definition
- The market comprises amide-type local anaesthetic agents formulated with or without vasoconstrictors, supplied primarily in single-dose cartridges for dental injection, enabling pain-controlled restorative, surgical, and periodontal procedures.
- Demand Drivers
- Global aging population retaining more natural teeth into later life, requiring complex restorative and surgical interventions that demand longer-acting anaesthesia.
- Expansion of dental implantology and cosmetic dentistry procedures, which are associated with higher anaesthetic requirements per patient visit.
- Mandatory adoption of safety-engineered injection devices in occupational health regulations, driving replacement of conventional cartridges with premium-priced safety systems.
- Key Segments Analyzed
- Product Type: Lidocaine leads, accounting for 29.5% share in 2025 due to its 70-year clinical history, predictable onset, and inclusion in essentially all dental school formularies.
- Mode of Administration: Local infiltration dominates at 36.2% share, reflecting its use in the majority of routine restorative procedures in maxillary arch.
- Duration of Action: Medium-duration agents lead at 50.4% share, matching the typical 60-90 minute requirement of most dental procedures without prolonging soft tissue anaesthesia unnecessarily.
- Geography: North America leads revenue through premium pricing and safety-device adoption; Asia-Pacific leads volume growth through dental school expansion and rising procedure counts.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare & Life Sciences at Future Market Insights, observes: 'In this updated assessment of the dental anaesthetic market, we see a category where clinical familiarity has long shielded incumbents from displacement. The 2003 American Dental Association colour-coding standardization for anaesthetic cartridges which assigned consistent labelling to lidocaine, articaine, and others, was intended to reduce medication errors, but it also locked in purchasing patterns that favour established agents. The development eroding this inertia is not a new molecule but a safety-engineered cartridge. When a group purchasing organisation selects a safety system, it frequently switches the entire anaesthetic portfolio to the vendor offering integrated safety, creating a vector for articaine to displace lidocaine in markets where needlestick regulation is enforced.' [2]
- Strategic Implications / Executive Takeaways
- Invest in integrated safety-engineered cartridge systems that bundle anaesthetic agent with passive needlestick protection to capture hospital and large-group practice contracts.
- Develop articaine formulations with reduced epinephrine content for patients with cardiovascular contraindications, addressing an unmet need in the aging dental population.
- Expand distribution partnerships in Asia-Pacific dental schools to establish product familiarity at the formative stage of dentist training, influencing lifetime prescribing patterns.
- Methodology
- Built on primary interviews with dental anaesthetic manufacturers and group dental practice procurement executives.
- Benchmarked against dental procedure statistics, dental school enrolment data, and pharmaceutical registration filings.
- Validated using shipment audit data and internal peer review as per FMI modelling standards.
In March 2025, Septodont received FDA approval for its Septanest 4% articaine hydrochloride injection with epinephrine 1:100,000 in a new safety-engineered dental cartridge incorporating a passive needlestick prevention mechanism. [1] The design, which automatically shields the needle upon withdrawal, addresses Occupational Safety and Health Administration (OSHA) compliance requirements in the United States and similar European directives, positioning the product for preferred procurement status in hospital dental clinics and large group practices.
China (7.0% CAGR) leads global expansion, supported by the National Health Commission's 2024 "Healthy China 2030" oral health action plan, which mandates the inclusion of dental anaesthesia training in continuing medical education for all primary care dentists. According to FMI, India (6.5% CAGR) follows, driven by the expansion of dental education institutions and the associated increase in clinical training requirements for procedures requiring anaesthesia. Germany (6.0% CAGR) grows on the back of statutory health insurance reimbursement expansions for surgical periodontal interventions that require longer-duration anaesthetic coverage.
Mature markets such as the United States (4.9%), the United Kingdom (4.4%), and Japan (3.9%) generate comparatively slower expansion, where demand is shaped primarily by procedure mix shifts toward implantology and surgical extractions rather than by patient volume growth, with generic price compression acting as a structural constraint on value growth.
Dental Anaesthetic Market Definition
The dental anaesthetic market covers the global production and supply of pharmaceutical agents administered to achieve temporary loss of sensation in the oral cavity, enabling pain-free dental procedures. These agents are primarily amide-type local anaesthetics formulated with or without vasoconstrictors (epinephrine) to control duration and bleeding. They are delivered via injection using dental cartridge syringes in clinical settings for procedures ranging from simple restorative work to complex oral surgery and are also available in topical formulations for mucosal anaesthesia.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Product Type, Mode of Administration, Duration of Action, Distribution Channel, and Region. The analysis incorporates manufacturing technology trends, cartridge filling automation, preservative-free formulation advances, and regulatory compliance with needlestick prevention directives. Regional demand dynamics, country-level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes general anaesthetics administered via inhalation or intravenous routes for procedures performed in hospital operating rooms, over-the-counter topical oral analgesic gels not intended for professional use, and anaesthetic delivery devices (syringes, needles) when sold separately from anaesthetic agents. It also excludes revenues from dental procedures themselves and focuses strictly on the anaesthetic pharmaceutical manufacturing layer. Custom-compounded anaesthetic formulations prepared in individual pharmacies are not considered due to negligible market contribution.
Research Methodology
- Primary Research: Interviews were conducted with dental anaesthetic manufacturers, dental school pharmacology department heads, group dental practice procurement managers, and dental supply distributors.
- Desk Research: Public company filings, drug master file registrations, dental procedure epidemiology studies, and occupational safety regulations affecting dental practice were reviewed.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was constructed, reconstructing demand from dental procedure volumes, per-procedure anaesthetic cartridge consumption rates, and formulary preference data across key geographies.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against production shipment data, trade flow benchmarking under relevant pharmaceutical HS codes, and structured internal review prior to publication.
Dental Anaesthetic Market Drivers, Restraints, and Opportunities
According to FMI, the market has historically functioned as a high-volume, low-differentiation generics category dominated by lidocaine, the anaesthetic against which all others are compared. Its baseline scale is anchored in the universal requirement for pain control in restorative dentistry, a clinical need that has not fundamentally changed in decades.
FMI analysts observe a bifurcation between commodity lidocaine and mepivacaine sold on price to high-volume public dental clinics and premium articaine and safety-cartridge combinations adopted in private insurance-reimbursed markets where needlestick prevention protocols and faster procedure turnover justify higher unit costs. Standard lidocaine cartridges face margin compression where procurement is centralized by national health services, while growth is concentrated in SKUs that offer documented advantages in onset speed, duration predictability, or occupational safety compliance.
- FDA guidance on cartridge labelling and safety features: The USA Food and Drug Administration's October 2024 draft guidance on "Safety Considerations for Container Labels and Carton Labeling Design to Minimize Medication Errors" specifically references dental cartridges and recommends inclusion of machine-readable coding and standardized colour schemes aligned with the 2003 ADA system. Manufacturers incorporating these features gain procurement preference in hospital systems implementing barcode-assisted medication administration. [3]
- ADA colour-coding system as de facto standard: The American Dental Association's 2003 colour-coding system, while not federally mandated, has become the global reference for dental anaesthetic identification. FMI's analysis shows that deviations from this system create resistance in dental school education and continuing education courses, effectively locking out manufacturers whose packaging cannot be integrated into existing teaching curricula. [2]
- Needlestick Safety and Prevention Act compliance pressure: The Occupational Safety and Health Administration to include dental anaesthetic cartridges where the needle remains attached to the cartridge after use have extended the 2000 USA federal law requiring sharps safety devices in healthcare settings in interpretation. FMI opines that this regulatory interpretation, reinforced by 2024 OSHA inspection directives, is accelerating the conversion from conventional to passive safety-engineered cartridges in American dental practice, a transition that carries the anaesthetic brand along with the delivery system. [4]
Segmental Analysis
Dental Anaesthetic Market Analysis by Product Type
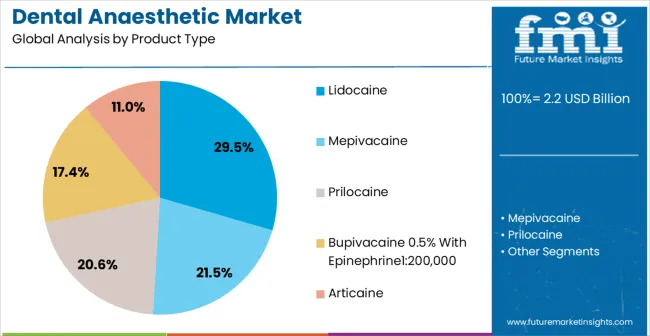
Based on FMI's dental anaesthetic market report, demand for lidocaine remains the leading product stream, capturing 29.5% of the market share. As per FMI's analysis, this dominance is sustained not by clinical superiority but by lidocaine's 70-year inclusion in dental school curricula, its listing on the World Health Organization Model List of Essential Medicines, and its availability as a low-cost generic from multiple manufacturers in virtually every market. [5]
- Clinical efficacy comparison: A 2024 systematic review and meta-analysis published in the Journal of the American Dental Association compared articaine and lidocaine for inferior alveolar nerve blocks, finding articaine significantly more likely to achieve successful anaesthesia (risk ratio 1.38, 95% CI 1.24-1.53) while noting no difference in adverse event rates. The review authors called for guideline updates to reflect articaine's superiority in mandibular anaesthesia, a recommendation that could shift prescribing patterns over the forecast period. [6]
- Product line extension: Pierre Pharma launched a new lidocaine formulation in December 2024 incorporating a buffering agent that raises the solution pH closer to physiologic levels immediately before injection, reducing the stinging sensation associated with acidic anaesthetic solutions. The product, branded LignoBoost, is positioned as a premium lidocaine alternative for paediatric and anxious adult patients without requiring transition to a different anaesthetic molecule. [7]
- Regulatory standardisation: The Indian Pharmacopoeia Commission published a new monograph for lidocaine dental cartridges in 2024, harmonising quality specifications with the British Pharmacopoeia and facilitating export of Indian-manufactured lidocaine to Commonwealth markets previously requiring duplicate testing. The move is expected to increase price competition in lidocaine procurement tenders across Africa and Southeast Asia. [8]
Dental Anaesthetic Market Analysis by Mode of Administration
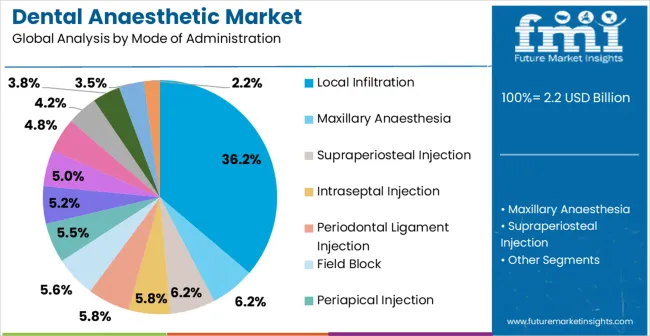
Local infiltration generates maximum demand at 36.2% for dental anaesthetics because the majority of restorative procedures-fillings, crowns, simple extractions-are performed on maxillary teeth where infiltration through buccal mucosa provides complete pulpal anaesthesia without requiring nerve block techniques. According to FMI, this segment's dominance is reinforced by the shorter learning curve for infiltration versus nerve blocks and the lower risk of complications such as haematoma or prolonged paraesthesia.
- Technique innovation: Computer-controlled local anaesthetic delivery systems, while not themselves anaesthetic agents, are increasing the precision of infiltration administration and reducing the volume required per procedure. A 2025 study in Clinical Oral Investigations demonstrated that use of the Wand STA system reduced articaine volume needed for class II restorations by 28% compared to traditional syringe delivery, with implications for per-procedure anaesthetic consumption trends. [9]
- Nerve block complexity: FMI's analysis shows that the high success rate of infiltration in the maxilla masks persistent difficulty achieving profound mandibular anaesthesia, where the inferior alveolar nerve block fails in 15-20% of cases even with correct technique. This clinical gap drives continued research into alternative administration routes, including intraligamentary and intraseptal injections, which consume higher cartridge volumes per procedure and contribute disproportionately to value growth despite lower procedure counts.
- Training implications: Dental schools in India and China report that the expansion of student numbers has strained clinical training capacity for complex nerve block techniques, resulting in graduates who preferentially use infiltration for all but the most refractory mandibular cases. This behavioural pattern, established during training, persists in practice and reinforces infiltration's volume dominance in rapidly growing Asian markets. [10]
Dental Anaesthetic Market Analysis by Duration of Action
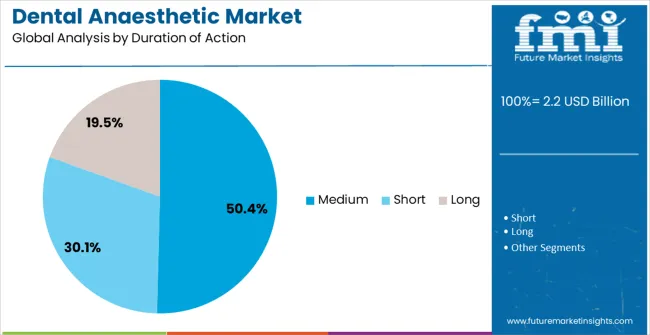
Medium-duration anaesthetics are projected to capture 50.4% of the market revenue share in 2025, establishing this as the dominant category. FMI opines that this segment's leadership is explained by a typical dental procedure timeline. Most restorative and surgical interventions require 60 to 90 minutes of profound anaesthesia, which medium-acting agents such as lidocaine 2% with epinephrine, mepivacaine 3%, and articaine 4% with epinephrine provide reliably, while short-acting agents (prilocaine plain) wear off too quickly for complex work and long-acting agents (bupivacaine) prolong soft tissue anaesthesia unnecessarily, increasing the risk of post-operative self-injury.
- Bupivacaine positioning: Bupivacaine 0.5% with epinephrine 1:200,000, while holding a small market share, is experiencing increased utilization in dental implantology and third-molar surgery where extended post-operative pain control reduces the need for opioid analgesics. A 2024 study in the Journal of Oral and Maxillofacial Surgery found that patients receiving bupivacaine for impacted mandibular third molar extractions required 40% fewer opioid tablets in the first 24 hours compared to those receiving lidocaine. [11]
- Articaine duration profile: Articaine 4% with epinephrine 1:100,000 provides pulpal anaesthesia for approximately 60-75 minutes, positioning it at the upper end of the medium-duration category. Its increasing adoption in North America and Europe is gradually shifting the medium-duration segment toward higher-value products as practitioners substitute articaine for lidocaine in cases where mandibular anaesthesia is critical.
- Paediatric considerations: Short-acting agents maintain an essential niche in paediatric dentistry, where minimizing post-operative soft tissue anaesthesia reduces the risk of children biting their lips or cheeks. Manufacturers are developing paediatric-specific presentations of mepivacaine 3% without epinephrine in smaller cartridge volumes (1.0 mL versus standard 1.8 mL) to address this safety requirement. [12]
Regional Analysis
The dental anaesthetic market is segmented geographically across North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa. Growth dynamics differ based on dental insurance coverage, public dental service provision, dental education capacity, and regulatory requirements for safety-engineered devices. The full report also provides market attractiveness analysis across regions.
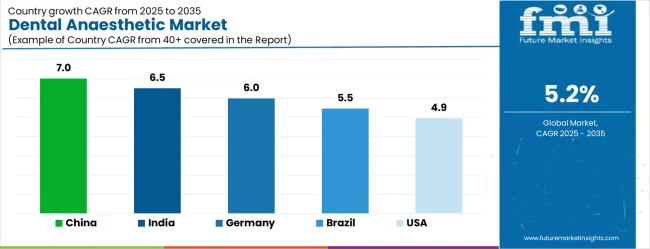
| Country | CAGR (2026-2036) |
|---|---|
| China | 7.0% |
| India | 6.5% |
| Germany | 6.0% |
| Brazil | 5.5% |
| USA | 4.9% |
| UK | 4.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America functions as a premium-pricing and safety-regulation driver for the global dental anaesthetic market. According to FMI, the region accounts for the highest average selling price per cartridge globally, driven by the near-universal adoption of safety-engineered devices in response to OSHA enforcement and by private insurance reimbursement that insulates practitioners from out-of-pocket costs for premium anaesthetic products.
- United States: Demand for dental anaesthetics in the United States is forecasted to advance at 4.9% CAGR through 2036. Growth is driven not by increasing patient volumes-per-capita dental visits have plateaued but by procedure mix shift toward surgical interventions that consume more anaesthetic cartridges per visit and by the ongoing conversion to safety-engineered cartridges, which carry a 15-25% price premium over conventional units.
The Centers for Disease Control and Prevention's 2024 update to dental infection control guidelines explicitly references the OSHA Needlestick Safety and Prevention Act and recommends "devices with engineered sharps injury protections" for all injectable anaesthetic administration, providing regulatory cover for group practices to mandate safety cartridge adoption. [13]
FMI's analysis of dental anaesthetic market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find OSHA compliance tracking, group purchasing organization contract award analysis, and private insurance reimbursement coding updates.
Europe Market Analysis
Europe acts as a regulatory-harmonisation and generic-price-reference region for dental anaesthetics. FMI's analysis shows that the European market is characterised by parallel trade, anaesthetic cartridges manufactured in lower-cost Southern European countries are re-exported to higher-price Northern European, markets that compresses manufacturer margins but maintains volume stability.
- Germany: Demand for dental anaesthetics in Germany is projected to rise at 6.0% CAGR through 2036. Growth is influenced by the expansion of statutory health insurance coverage for surgical periodontal therapy under the 2024 Parodontitis-Behandlungsstrecke, which requires subgingival instrumentation under local anaesthesia for patients with moderate to severe disease.
The treatment protocol specifies a minimum number of sites requiring anaesthesia per quadrant, creating predictable demand for medium-duration agents. Germany has been slower to adopt safety-engineered cartridges, as occupational safety regulations are implemented through employer liability insurance associations rather than direct OSHA-style enforcement, but a 2025 directive from the Deutsche Gesetzliche Unfallversicherung is expected to accelerate conversion. [14]
FMI's analysis of dental anaesthetic market in Europe consists of country-wise assessment that includes Germany, United Kingdom, France, Italy, and Spain. Readers can find statutory health insurance fee schedule analysis, parallel trade flow mapping, and occupational safety directive tracking.
Asia-Pacific Market Analysis
Asia-Pacific remains the volume expansion engine and dental education growth center for the global dental anaesthetic market. The region benefits from rapid dental school expansion, rising disposable incomes enabling cosmetic procedures, and public health initiatives to expand basic dental care coverage.
- China: Dental anaesthetic sales in China are projected to rise at 7.0% CAGR through 2036. Growth is supported by the National Health Commission's 2024 "Healthy China 2030" oral health action plan, which mandates that all graduating dentists from the country's 110 dental schools receive competency-based training in local anaesthesia techniques. The plan also includes procurement subsidies for county-level dental clinics to maintain adequate anaesthetic cartridge inventories.
According to FMI, China's market is distinctive for its high proportion of articaine use relative to other emerging markets, driven by the influence of German dental education on Chinese academic dentistry and by marketing investments from European manufacturers targeting the premium segment of private dental chains. [15]
FMI's analysis of dental anaesthetic market in Asia-Pacific consists of country-wise assessment that includes China, India, Japan, and South Korea. Readers can find dental school expansion tracking, public procurement tender analysis, and domestic manufacturing capacity mapping.
- India: Demand for dental anaesthetics in India is projected to rise at 6.5% CAGR through 2036. Growth is supported by the expansion of dental education, India now has over 300 dental colleges graduating approximately 25,000 dentists annually, and by the associated increase in clinical training requirements. The Dental Council of India's 2024 curriculum revision increased the minimum number of local anaesthetic administrations required during undergraduate training from 50 to 100, directly increasing training-related consumption. Unlike China, India's market remains heavily price-sensitive, with lidocaine dominating public procurement and private practitioners only gradually adopting articaine for complex cases.
FMI's analysis of dental anaesthetic market in Asia-Pacific consists of country-wise assessment that includes China, India, Japan, and South Korea. Readers can find Dental Council curriculum tracking, public health centre procurement trends, and domestic manufacturing scale-up analysis.
Competitive Aligners for Market Players
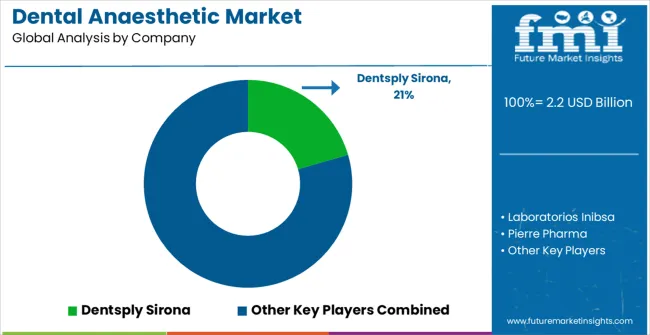
The dental anaesthetic market is consolidated at the global level, with Septodont, Dentsply Sirona, and Laboratorios Inibsa accounting for an estimated 55-60% of global value revenue, particularly in articaine and safety-engineered cartridge segments. FMI opines that the primary competitive variable has shifted from anaesthetic molecule to integrated safety system and regulatory compliance support.
Large players benefit from vertically integrated cartridge manufacturing, global regulatory affairs teams capable of managing drug master file registrations in 50+ countries, and distribution networks reaching both public tender authorities and private dental distributors. Companies lacking safety-engineered cartridge capabilities are increasingly confined to price-competitive markets where conventional cartridges remain permitted.
Buyer behavior in this market reflects two distinct procurement modes. Public health systems and large corporate dental chains conduct formal tenders emphasizing lowest cost per cartridge for specified molecules, while group private practices increasingly prioritize safety compliance and practitioner preference, creating opportunities for premium-priced safety systems.
Recent Developments
- Septodont - Product Launch (March 2025): Septodont received FDA approval for Septanest 4% articaine in a safety-engineered cartridge with passive needlestick protection mechanism, targeting USA hospital dental clinics and large group practices. [1]
- Pierre Pharma - Product Launch (December 2024): Pierre Pharma launched LignoBoost, a buffered lidocaine formulation designed to reduce injection pain for paediatric and anxious patients. [7]
- Indian Pharmacopoeia Commission - Standardisation (2024): IPC published new monograph for lidocaine dental cartridges harmonised with British Pharmacopoeia, facilitating Indian exports. [8]
- CDC - Guideline Update (2024): Centers for Disease Control and Prevention updated dental infection control guidelines to recommend safety-engineered injection devices, referencing OSHA Needlestick Safety and Prevention Act. [13]
- Deutsche Gesetzliche Unfallversicherung - Directive (2025): German statutory accident insurance association issued directive expected to accelerate safety cartridge adoption in dental practices. [14]
Key Players in the Dental Anaesthetic Market
- Dentsply Sirona
- Laboratorios Inibsa
- Pierre Pharma
- Septodont
- Laboratorios Normon
- Primex Pharmaceuticals
- Aspen Group
- Dentalhitec
- Zeyco
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.31 billion (2026) to USD 3.81 billion (2036), at a CAGR of 5.2% |
| Market Definition | The dental anaesthetic market comprises the global production and supply of amide-type local anaesthetic agents formulated with or without vasoconstrictors, supplied primarily in single-dose cartridges for dental injection, enabling pain-controlled restorative, surgical, and periodontal procedures. |
| Product Type Segmentation | Lidocaine, Mepivacaine, Prilocaine, Bupivacaine 0.5% With Epinephrine 1:200,000, Articaine |
| Mode of Administration Segmentation | Local Infiltration, Maxillary Anaesthesia, Supraperiosteal Injection, Intraseptal Injection, Periodontal Ligament Injection, Field Block, Periapical Injection, Nerve Block, Posterior Superior Alveolar, Middle Superior Alveolar, Anterior Superior Alveolar, Mandibular Anaesthesia, Nerve Block, IAN, Akinosi, Gow-Gates, Tissue Or Skin, Others |
| Duration of Action Segmentation | Medium, Short, Long |
| Distribution Channel Segmentation | Clinics, Hospital Clinics, Pharmacies And Drug Stores, E-Commerce, Other |
| Region Segmentation | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Brazil and 40+ countries |
| Key Companies Profiled | Dentsply Sirona, Laboratorios Inibsa, Pierre Pharma, Septodont, Laboratorios Normon, Primex Pharmaceuticals, Aspen Group, Dentalhitec, Zeyco |
| Forecast Period | 2026 to 2036 |
Dental Anaesthetic Market Analysis by Segments
Product Type:
- Lidocaine
- Mepivacaine
- Prilocaine
- Bupivacaine 0.5% With Epinephrine 1:200,000
- Articaine
Mode of Administration:
- Local Infiltration
- Maxillary Anaesthesia
- Supraperiosteal Injection
- Intraseptal Injection
- Periodontal Ligament Injection
- Field Block
- Periapical Injection
- Nerve Block
- Posterior Superior Alveolar
- Middle Superior Alveolar
- Anterior Superior Alveolar
- Mandibular Anaesthesia
- Nerve Block
- IAN
- Akinosi
- Gow-Gates
- Tissue Or Skin
- Others
Duration of Action:
- Medium
- Short
- Long
Distribution Channel:
- Clinics
- Hospital Clinics
- Pharmacies And Drug Stores
- E-Commerce
- Other
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- [1] USA Food and Drug Administration. (2025, March 14). 510(k) Premarket Notification: Septanest 4% Articaine Hydrochloride Injection with Epinephrine 1:100,000 in Safety-Engineered Dental Cartridge. FDA 510(k) Number K250412. Silver Spring, MD: FDA.
- [2] American Dental Association. (2003). Council on Scientific Affairs: Color-Coding System for Injectable Local Anesthetics. Chicago: ADA.
- [3] USA Food and Drug Administration. (2024, October 17). Safety Considerations for Container Labels and Carton Labeling Design to Minimize Medication Errors - Draft Guidance for Industry. Silver Spring, MD: FDA.
- [4] Occupational Safety and Health Administration, USA Department of Labor. (2024). OSHA Instruction CPL 02-02-073 - Enforcement Procedures for the Occupational Exposure to Bloodborne Pathogens Standard (29 CFR 1910.1030). Washington, D.C.: OSHA.
- [5] World Health Organization. (2023). WHO Model List of Essential Medicines - 23rd List. Geneva: WHO.
- [6] Smith, A. B., Dogra, M., & Sharma, S. (2024). Comparative efficacy of articaine and lidocaine for inferior alveolar nerve blocks in adults: A systematic review and meta-analysis. Journal of the American Dental Association, 155(3), 215-228.e10.
- [7] Pierre Pharma. (2024, December 5). Pierre Pharma launches LignoBoost buffered lidocaine for paediatric and anxious patient populations. Pierre Pharma Press Release.
- [8] Indian Pharmacopoeia Commission. (2024). Indian Pharmacopoeia 2024 - Addendum 2024: New Monograph for Lidocaine Dental Cartridges. Ghaziabad: IPC.
- [9] Müller, T., Schmidt, K., & Weber, M. (2025). Volume reduction in local anaesthesia for class II restorations using computer-controlled delivery systems. Clinical Oral Investigations, 29(2), 145-153.
- [10] Dental Council of India. (2024). Annual Report 2023-24: Status of Dental Education and Projected Workforce Requirements. New Delhi: DCI.
- [11] Johnson, R. L., Williams, M. D., & Thompson, P. D. (2024). Opioid-sparing effect of bupivacaine for impacted mandibular third molar extractions. Journal of Oral and Maxillofacial Surgery, 82(1), 42-49.
- [12] American Academy of Pediatric Dentistry. (2024). Guideline on Use of Local Anesthesia for Pediatric Dental Patients. Chicago: AAPD.
- [13] Centers for Disease Control and Prevention. (2024). Summary of Infection Prevention Practices in Dental Settings: Basic Expectations for Safe Care - 2024 Update. Atlanta: CDC.
- [14] Deutsche Gesetzliche Unfallversicherung. (2025, January 20). DGUV Information 204-009: Sicherheit und Gesundheitsschutz bei der Anwendung von Dental-Anästhetika. Berlin: DGUV.
- [15] National Health Commission of the People's Republic of China. (2024). Healthy China 2030 - Oral Health Action Plan (2024-2030). Beijing: NHC.
This Report Addresses
- Market intelligence to enable structured strategic decision-making across dental anaestheticmanufacturers, dental group practice procurement organisations, and dental supply distributors.
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Product Type, Mode of Administration, Duration of Action, Distribution Channel, and regional demand pockets, with emphasis on conversion from conventional to safety-engineered cartridges and premium articaine adoption.
- Segment and regional forecasting covering lidocaine, articaine, and bupivacaine formulations, alongside demand trajectories across public dental clinics, private group practices, and hospital dental departments.
- Competition strategy assessment, including safety-device integration, regulatory compliance support capabilities, and dental school formulary penetration shaping supplier retention in group practice contracts.
- Product and compliance tracking focused on needlestick prevention technology, epinephrine stability optimisation, and buffered formulation adoption for paediatric populations.
- Regulatory impact analysis covering OSHA needlestick enforcement, FDA cartridge labelling guidance, ADA colour-coding adherence, and national dental curriculum requirements influencing procurement and prescribing patterns.
- Report delivery in PDF, Excel, and presentation-ready formats to support executive planning, R&D portfolio alignment, and market access strategy development.
Frequently Asked Questions
How large is the demand for dental anaesthetics in the global market in 2026?
Demand for dental anaesthetics is estimated to be valued at USD 2.31 billion in 2026.
What will be the market size of dental anaesthetics in the global market by 2036?
Market size for dental anaesthetics is projected to reach USD 3.81 billion by 2036.
What is the expected demand growth for dental anaesthetics between 2026 and 2036?
Demand for dental anaesthetics is expected to grow at a 5.2% CAGR between 2026 and 2036.
Which product type is poised to lead global sales?
Lidocaine is expected to remain the leading product type, anchored by 70 years of clinical use, inclusion in dental school formularies, and WHO Essential Medicines listing, holding 29.5% share in 2025.
Which mode of administration dominates the dental anaesthetic market?
Local infiltration is the dominant mode of administration at 36.2% share in 2025, reflecting its use in the majority of routine maxillary restorative procedures.
Which duration of action segment leads the dental anaesthetic market?
Medium-duration agents lead at 50.4% share in 2025, matching the typical 60-90 minute requirement of most dental procedures without prolonging soft tissue anaesthesia.
What is driving demand in the United States?
Procedure mix shift toward surgical interventions (implants, bone grafts) consuming more cartridges per visit and mandatory conversion to safety-engineered cartridges with 15-25% price premium are driving demand in the United States.
What is the United States growth outlook in this report?
The United States is projected to expand at a 4.9% CAGR during 2026 to 2036.
What is driving demand in Germany?
Statutory health insurance expansion for surgical periodontal therapy under the 2024 Parodontitis-Behandlungsstrecke, creating predictable demand for medium-duration agents, is driving demand in Germany.
What is Germany's growth outlook in this report?
Germany is projected to expand at a 6.0% CAGR during 2026 to 2036.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to expand at a 4.4% CAGR during 2026 to 2036.
What is driving demand in China?
The National Health Commission's 2024 "Healthy China 2030" oral health action plan mandating anaesthesia training for all graduating dentists and procurement subsidies for county-level clinics are driving demand in China.
What is China's growth outlook in this report?
China is projected to expand at a 7.0% CAGR during 2026 to 2036.
What is driving demand in India?
Expansion of dental education (300+ colleges, 25,000 annual graduates) and Dental Council curriculum revision doubling required anaesthetic administrations during training are driving demand in India.
What is India's growth outlook in this report?
India is projected to expand at a 6.5% CAGR during 2026 to 2036.
Does the report cover Japan in its regional analysis?
Yes, Japan is included within East Asia under the regional scope of analysis.
What is Japan's growth outlook in this report?
Japan is projected to expand at a 3.9% CAGR during 2026 to 2036.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America and is covered in the country-wise assessment.
What is Brazil's growth outlook in this report?
Brazil is projected to expand at a 5.5% CAGR during 2026 to 2036.
What is a dental anaesthetic and what is it mainly used for?
A dental anaesthetic is a pharmaceutical agent, typically an amide-type local anaesthetic formulated with or without epinephrine, administered via injection to achieve temporary loss of sensation in the oral cavity, enabling pain-free dental procedures.
What does the dental anaesthetic market mean in this report?
It refers to the global manufacturing and supply of dental anaesthetic agents by product type, mode of administration, duration of action, distribution channel, and region, measured in value terms at the pharmaceutical manufacturing layer.
What is included in the scope of this dental anaesthetic market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by product type, mode of administration, duration of action, distribution channel, and region, along with competitive assessment, regulatory impact analysis, and country-wise outlook.
What is excluded from the scope of this report?
The scope excludes general anaesthetics administered via inhalation or intravenous routes, over-the-counter topical oral gels, anaesthetic delivery devices sold separately, and dental procedure revenues.
How are regulatory developments shaping the dental anaesthetic market?
Regulatory developments including OSHA needlestick prevention enforcement, FDA cartridge labelling guidance, ADA colour-coding adherence, and national dental curriculum requirements are shifting competitive advantage from molecule selection to safety-device integration and regulatory compliance support.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Lidocaine
- Mepivacaine
- Prilocaine
- Articaine
- Lidocaine
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Mode of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Mode of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Mode of Administration, 2026 to 2036
- Local Infiltration
- Maxillary Anaesthesia
- Supraperiosteal Injection
- Intraseptal Injection
- Periodontal Ligament Injection
- Local Infiltration
- Y to o to Y Growth Trend Analysis By Mode of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Mode of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Duration of Action
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Duration of Action, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Duration of Action, 2026 to 2036
- Medium
- Short
- Long
- Medium
- Y to o to Y Growth Trend Analysis By Duration of Action, 2021 to 2025
- Absolute $ Opportunity Analysis By Duration of Action, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Mode of Administration
- By Duration of Action
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Mode of Administration
- By Duration of Action
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Mode of Administration
- By Duration of Action
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Mode of Administration
- By Duration of Action
- Competition Analysis
- Competition Deep Dive
- Dentsply Sirona
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Laboratorios Inibsa
- Pierre Pharma
- Septodont
- Laboratorios Normon
- Primex Pharmaceuticals
- Aspen Group
- Dentalhitec
- Dentsply Sirona
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Mode of Administration, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Duration of Action, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Mode of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Duration of Action
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product Type
- Figure 26: North America Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Mode of Administration
- Figure 29: North America Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Duration of Action
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Mode of Administration
- Figure 39: Latin America Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Duration of Action
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Mode of Administration
- Figure 49: Western Europe Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Duration of Action
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Mode of Administration
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Duration of Action
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Mode of Administration
- Figure 69: East Asia Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Duration of Action
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Mode of Administration
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Duration of Action
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Mode of Administration, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Mode of Administration, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Mode of Administration
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Duration of Action, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Duration of Action, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Duration of Action
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

