Intensive Care Consumables Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Intensive Care Consumables Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Intensive Care Consumables Market Forecast and Outlook 2026 to 2036
The intensive care consumables market is forecasted to reach USD 15.8 billion in 2026 and expand to USD 29.7 billion by 2036, at a CAGR of 6.5%. Value behavior is anchored in the non-deferrable nature of consumable usage within critical care, where single-use items are required continuously to sustain ventilation, infusion, monitoring, and infection prevention. Spending scales with ICU occupancy, length of stay, and care intensity rather than capital expansion, creating a structurally recurring demand profile. Hospitals allocate expenditure toward consumables that ensure sterility, compatibility with installed devices, and uninterrupted availability during high-acuity care delivery.
Adoption patterns are shaped by infection control mandates, reimbursement bundling, and supply chain reliability constraints. Utilization rises where invasive procedures, ventilator days, and renal support therapies increase per-patient consumption rates. Limited interchangeability across device ecosystems reinforces vendor standardization and long-term supply agreements. Cost containment pressure, waste management requirements, and vulnerability to supply disruption moderate margin expansion. Market structure remains governed by throughput-driven consumption, protocol standardization, and operational risk management, sustaining steady growth tied to critical care utilization rather than episodic demand shocks.
Quick Stats for Intensive Care Consumables Market
- Intensive Care Consumables Market Value (2026): USD 15.8 billion
- Intensive Care Consumables Market Forecast Value (2036): USD 29.7 billion
- Intensive Care Consumables Market Forecast CAGR 2026 to 2036: 6.5%
- Leading Consumable Type by Demand Share: Respiratory consumables
- Fastest-Growing Countries: India, Brazil, China, USA, Germany
- Top Players in Global Demand: Medtronic, BD, Baxter, Fresenius Medical Care, Smiths Medical
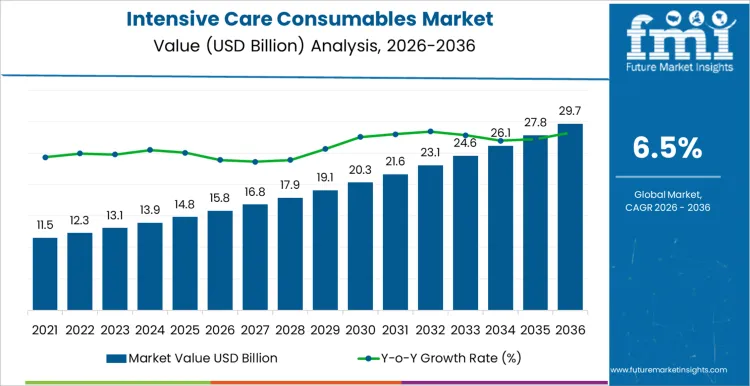
Intensive Care Consumables Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 15.8 billion |
| Market Forecast Value (2036) | USD 29.7 billion |
| Forecast CAGR 2026 to 2036 | 6.5% |
Why is the Demand for Intensive Care Consumables Growing?
Demand for intensive care consumables is increasing as hospitals and critical care units manage rising volumes of acutely ill patients requiring sustained monitoring and supportive therapies. Consumables such as ventilator circuits, infusion sets, catheter kits, suction canisters, and pressure monitoring lines are integral to life support, hemodynamic stabilization, and infection control in patients with respiratory failure, sepsis, multi-organ dysfunction, and post-operative critical needs. Clinicians specify consumables that deliver reliable performance, compatibility with advanced monitoring systems, and minimal risk of contamination to support uninterrupted care delivery. Procurement teams assess product quality, sterility assurance, and supplier reliability because consistent availability of these items directly influences ICU throughput, procedural efficiency, and overall patient safety under high-utilization conditions. These selection criteria are particularly important in environments managing complex cases where frequent changes of consumables are required to maintain therapeutic effectiveness and hygiene standards.
Growth in critical care demand, driven by aging populations, higher prevalence of chronic diseases, and broader access to hospital services, reinforces sustained use of consumables that support advanced therapies. Expansion of ventilator-assisted care, continuous renal replacement therapies, and invasive hemodynamic monitoring increases per-patient consumption rates, prompting facilities to optimize inventory strategies and supplier partnerships. Infection prevention protocols also elevate demand for single-use and sterile consumables to reduce cross-contamination risks and support regulatory compliance. Technical refinements in material science and ergonomic design improve ease of use and reduce clinician fatigue during prolonged procedures. These clinical, operational, and safety-focused priorities are contributing to continued demand growth in the intensive care consumables market.
How Is the Intensive Care Consumables Industry Segmented by Consumable Type, Clinical Application, and Sales Channel?
Demand for intensive care consumables is shaped by ICU admission rates, disease acuity, and length of stay across critical care settings. Utilization patterns reflect increasing prevalence of respiratory failure, sepsis, and post-surgical complications requiring continuous monitoring and intervention. Hospitals prioritize reliable supply, standardization, and infection prevention under strict clinical protocols. Procurement behavior aligns with cost control pressures, emergency preparedness, and vendor consolidation. Segmentation highlights how product categories, care intensity, and purchasing structures determine consumption patterns within critical care environments.
Which Consumable Type Accounts for the Largest Share in ICU Utilization?
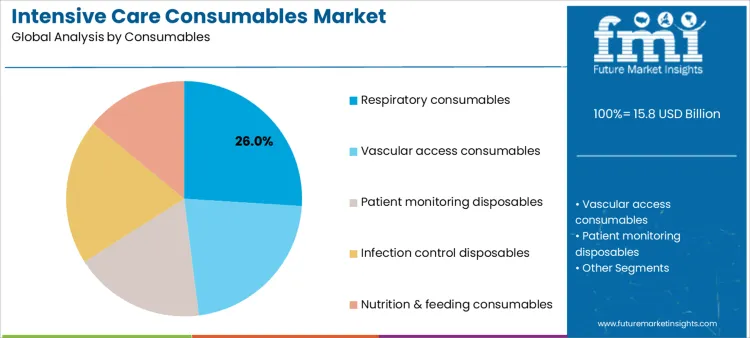
Respiratory consumables account for 26.0%, driven by widespread use in ventilation, oxygen therapy, and airway management. These products experience high turnover due to single-use protocols and infection risk mitigation. Vascular access consumables at 22.0% support fluid delivery, medication administration, and hemodynamic monitoring. Infection control disposables at 20.0% reflect heightened emphasis on preventing hospital-acquired infections in high-risk patients. Patient monitoring disposables at 18.0% enable continuous vital sign tracking. Nutrition and feeding consumables at 14.0% address metabolic support in prolonged ICU stays. Consumable segmentation reflects intensity of respiratory and invasive care.
Key Points:
- Respiratory items dominate due to ventilation reliance.
- Vascular access supports continuous therapeutic delivery.
- Infection control remains central to ICU safety protocols.
How Do Clinical Applications Influence ICU Consumable Demand?
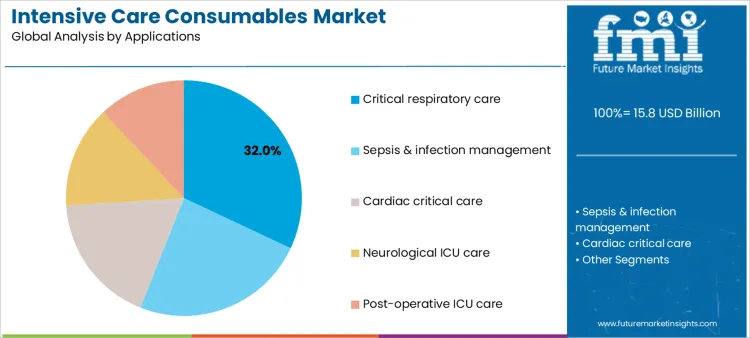
Critical respiratory care represents 32.0%, reflecting high incidence of ventilation-dependent conditions. Sepsis and infection management at 24.0% drives use of disposables linked to antimicrobial delivery and monitoring. Cardiac critical care at 18.0% relies on invasive monitoring and vascular consumables. Neurological ICU care at 14.0% focuses on monitoring and supportive devices for unstable patients. Post-operative ICU care at 12.0% supports recovery following major surgical interventions. Application-based segmentation indicates higher consumption where patient instability requires continuous intervention and monitoring.
Key Points:
- Respiratory care drives the highest consumable intensity.
- Sepsis management increases disposable utilization.
- Cardiac and neurological ICUs require specialized monitoring.
Which Sales Channels Dominate Distribution of Intensive Care Consumables?
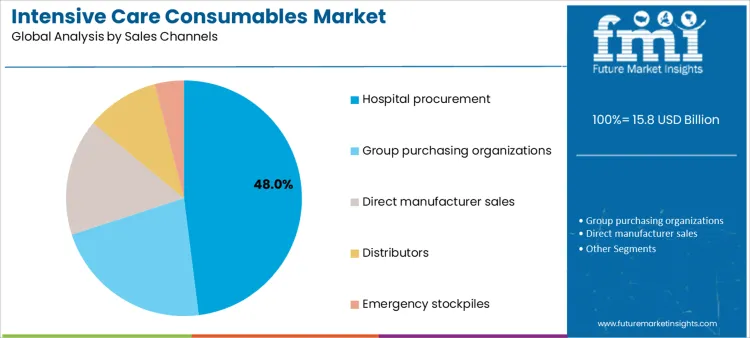
Hospital procurement accounts for 48.0%, reflecting centralized purchasing aligned with internal formularies and usage forecasting. Group purchasing organizations at 22.0% aggregate demand across facilities to manage costs and standardize products. Direct manufacturer sales at 16.0% support high-volume or specialized supply agreements. Distributors at 10.0% provide logistical support and inventory flexibility. Emergency stockpiles at 4.0% address surge preparedness and crisis response. Sales channel segmentation shows concentration where purchasing scale, continuity of supply, and cost control drive distribution strategies.
Key Points:
- Hospitals rely on centralized procurement models.
- GPOs consolidate volume to manage pricing.
- Emergency stockpiles support surge readiness.
What are the Key Dynamics in the Intensive Care Consumables Market?
Demand for intensive care consumables reflects continuous use requirements for single-use medical products supporting critical patient management. Adoption concentrates in hospital intensive care units, trauma centers, and high-acuity wards. Global scope aligns with expansion of critical care capacity and infection control standards. Usage spans respiratory disposables, vascular access items, wound care materials, monitoring accessories, and fluid management consumables required for uninterrupted bedside care.
How do patient acuity intensity and infection control priorities shape adoption?
Critical care patients require constant intervention involving ventilation, infusion, monitoring, and invasive procedures. Demand increases where high device utilization rates drive frequent replacement of tubing, catheters, filters, and sensors. Single-use consumables reduce cross-contamination risk and support strict infection prevention protocols. Ventilated patients generate sustained demand for circuits, humidification components, and suction products. Hemodynamic instability necessitates reliable access consumables to maintain continuous drug delivery and monitoring. Standardized consumable sets improve workflow efficiency during emergencies and shift changes. Procurement decisions emphasize availability, compatibility with installed equipment, and consistency under high turnover conditions. Adoption reflects operational dependence on consumables as essential enablers of safe, uninterrupted intensive care delivery.
How do supply continuity risk, cost exposure, and standardization limits influence market scalability?
Intensive care consumables generate recurring expenditure due to high daily usage per patient. Demand sensitivity rises where budget pressure affects stocking depth and brand selection. Supply disruptions create immediate clinical risk due to lack of reusable alternatives. Product standardization varies across equipment vendors, complicating inventory management. Waste volume and disposal compliance add operational burden. Training requirements increase where multiple product variants coexist. Reimbursement structures often bundle consumables into fixed care payments, tightening cost control. Scalability remains constrained by supply chain resilience requirements, cost containment pressure, and limited interchangeability across critical care device ecosystems.
How Is Demand for Intensive Care Consumables Evolving Globally?
Demand for intensive care consumables is expanding globally as hospitals emphasize readiness, infection control, and continuity of critical care delivery. Ventilation circuits, catheters, tubing, disposables, and monitoring accessories see steady usage aligned with ICU occupancy and protocol intensity. Standardized care bundles and accreditation requirements increase per-patient consumption. Supply reliability and compatibility with installed equipment influence procurement decisions. Growth rates in India at 9.0%, Brazil at 8.5%, China at 8.1%, the USA at 5.0%, and Germany at 4.9% indicate sustained expansion driven by critical care utilization, protocol standardization, and quality assurance rather than episodic surge preparedness.
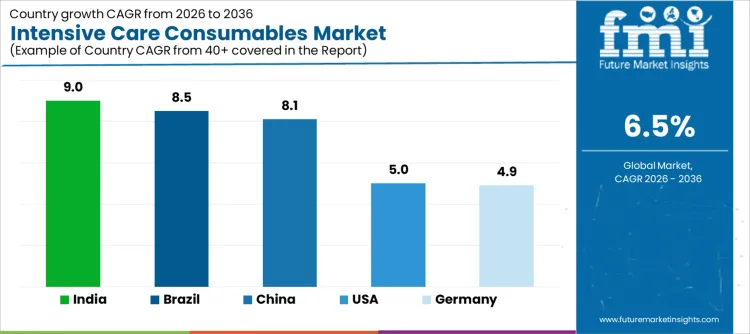
| Country | CAGR (%) |
|---|---|
| India | 9.0% |
| Brazil | 8.5% |
| China | 8.1% |
| USA | 5.0% |
| Germany | 4.9% |
What Is Driving Growth of Intensive Care Consumable Demand in India?
Demand for intensive care consumables in India is growing at a CAGR of 9.0%, supported by expansion of ICU beds across public and private hospitals. Rising case complexity increases use of single-use disposables for ventilation, vascular access, and renal support. Private hospital chains standardize ICU protocols, elevating per-patient consumable utilization. Infection prevention policies favor disposable over reusable components. Domestic manufacturers improve availability across price tiers. Growth reflects structural ICU capacity expansion, protocol formalization, and infection control priorities rather than temporary utilization spikes.
- Expansion of ICU bed capacity nationwide
- Higher acuity driving disposable usage intensity
- Standardized ICU protocols in private hospitals
- Improved domestic consumable manufacturing
Why Is Brazil Showing Strong Momentum in Intensive Care Consumables?
Intensive care consumable demand in Brazil is expanding at a CAGR of 8.5%, driven by modernization of hospital critical care services. Public hospitals increase disposable usage to meet infection control and audit requirements. Regional ICUs expand access beyond major urban centers, increasing baseline consumption. Procurement frameworks prioritize compatibility and cost control. Training programs reinforce adherence to standardized care bundles. Growth reflects system-wide quality reinforcement and ICU service expansion rather than emergency-driven procurement.
- Modernization of public hospital ICUs
- Regional expansion of critical care services
- Infection control compliance requirements
- Standardized procurement and training programs
How Is China Sustaining Growth in Intensive Care Consumable Utilization?
Demand for intensive care consumables in China is growing at a CAGR of 8.1%, aligned with scale of hospital networks and rising critical care intensity. High patient throughput elevates routine consumption of disposables. Central hospitals implement standardized ICU pathways that increase consumable consistency. Domestic suppliers provide cost-effective products at scale. Regulatory oversight supports adherence to infection prevention standards. Growth reflects volume-driven utilization and protocol standardization rather than expansion of ICU numbers alone.
- High ICU throughput increasing routine consumption
- Standardized critical care pathways adoption
- Scaled domestic supply availability
- Regulatory focus on infection prevention
What Factors Are Supporting Intensive Care Consumable Demand in United States?
Intensive care consumable demand in the United States is expanding at a CAGR of 5.0%, supported by mature ICU infrastructure and protocol-driven care. Per-patient consumption remains high due to complex case management. Replacement demand sustains steady purchasing across established hospitals. Emphasis on safety and traceability reinforces disposable use. Group purchasing organizations influence product standardization. Growth reflects utilization stability, protocol adherence, and replacement cycles rather than expansion of ICU capacity.
- High per-patient consumable intensity
- Replacement-driven purchasing patterns
- Safety and traceability requirements
- Group purchasing organization standardization
Why Is Germany Experiencing Measured Expansion in Intensive Care Consumables?
Intensive care consumable demand in Germany is growing at a CAGR of 4.9%, shaped by disciplined healthcare governance and efficiency focus. Certified ICUs apply standardized care bundles with predictable consumable usage. Emphasis on cost containment moderates volume growth. High-quality reusable components coexist with disposables where clinically justified. Central procurement frameworks guide product selection. Growth remains measured, reflecting utilization optimization and compliance stability rather than aggressive ICU expansion.
- Certified ICU care bundle implementation
- Emphasis on cost and utilization efficiency
- Balanced use of reusable and disposable items
- Centralized procurement frameworks
What is the competitive landscape of demand for the intensive care consumables market globally?
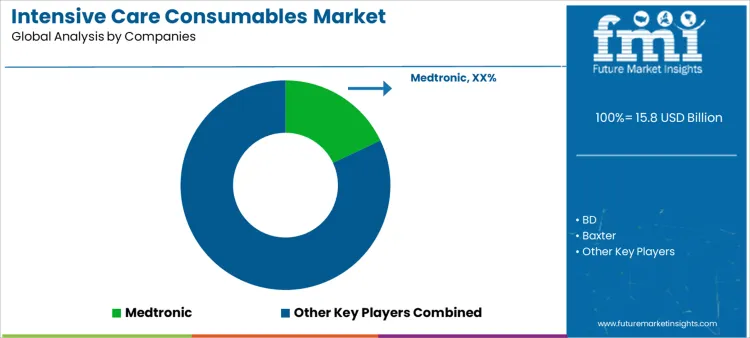
Demand for intensive care consumables is driven by critical care admissions, procedure intensity, and infection control requirements across hospitals. ICU clinicians and procurement teams evaluate product reliability, clinical safety, compatibility with monitoring and infusion systems, and consistency under high utilization rates. Buyer assessment includes sterility assurance, ease of use in emergency settings, standardization across care units, and supply continuity during demand surges. Procurement behavior reflects long-term supplier contracts, inventory optimization strategies, and preference for vendors supporting bundled consumable portfolios. Trend in the intensive care consumables market shows steady volume growth tied to aging populations and expanding critical care capacity.
Medtronic leads competitive positioning through a broad consumables portfolio integrated with critical care devices and monitoring systems. BD competes with vascular access, infusion, and infection prevention consumables widely standardized in ICU protocols. Baxter supports demand with fluid delivery, renal care, and infusion-related consumables aligned with critical care workflows. Fresenius Medical Care maintains relevance through consumables supporting continuous renal replacement therapy in intensive care settings. Smiths Medical participates by supplying airway management and infusion consumables designed for high-acuity environments. Competitive differentiation centers on clinical reliability, portfolio breadth, supply resilience, and alignment with standardized ICU care pathways.
Key Players in the Intensive Care Consumables Market
- Medtronic
- BD
- Baxter
- Fresenius Medical Care
- Smiths Medical
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Consumables | Respiratory consumables; Vascular access consumables; Patient monitoring disposables; Infection control disposables; Nutrition & feeding consumables |
| Applications | Critical respiratory care; Sepsis & infection management; Cardiac critical care; Neurological ICU care; Post-operative ICU care |
| Sales Channels | Hospital procurement; Group purchasing organizations; Direct manufacturer sales; Distributors; Emergency stockpiles |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Medtronic; BD; Baxter; Fresenius Medical Care; Smiths Medical; Others |
| Additional Attributes | Dollar sales by consumable category, application, and sales channel; usage intensity linked to ICU admission rates, ventilator days, and infection control protocols; single-use adoption driven by cross-contamination risk reduction; demand volatility during public health emergencies; supply resilience influenced by regional manufacturing capacity and stockpiling policies; procurement behavior shaped by GPO contracts, hospital formularies, and emergency preparedness programs. |
Intensive Care Consumables Market by Segment
Consumables:
- Respiratory consumables
- Vascular access consumables
- Patient monitoring disposables
- Infection control disposables
- Nutrition & feeding consumables
Applications:
- Critical respiratory care
- Sepsis & infection management
- Cardiac critical care
- Neurological ICU care
- Post-operative ICU care
Sales Channels:
- Hospital procurement
- Group purchasing organizations
- Direct manufacturer sales
- Distributors
- Emergency stockpiles
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2023). Clinical management of severe acute respiratory infections in hospital settings. World Health Organization.
- World Health Organization. (2024). WHO global report on infection prevention and control. World Health Organization.
- Centers for Disease Control and Prevention. (2023). Guideline for isolation precautions: Preventing transmission of infectious agents in healthcare settings. U.S. Department of Health and Human Services.
- United States Food and Drug Administration. (2023). Medical device sterility, biocompatibility, and single-use device considerations: Regulatory guidance. U.S. Department of Health and Human Services.
- Organisation for Economic Co-operation and Development. (2023). Health at a Glance 2023: OECD indicators – Intensive care capacity, utilization, and hospital resources. Organisation for Economic Co-operation and Development.
- International Organization for Standardization. (2023). ISO 17665-1: Sterilization of health care products – Moist heat. ISO.
Frequently Asked Questions
How big is the intensive care consumables market in 2026?
The global intensive care consumables market is estimated to be valued at USD 15.8 billion in 2026.
What will be the size of intensive care consumables market in 2036?
The market size for the intensive care consumables market is projected to reach USD 29.7 billion by 2036.
How much will be the intensive care consumables market growth between 2026 and 2036?
The intensive care consumables market is expected to grow at a 6.5% CAGR between 2026 and 2036.
What are the key product types in the intensive care consumables market?
The key product types in intensive care consumables market are respiratory consumables, vascular access consumables, patient monitoring disposables, infection control disposables and nutrition & feeding consumables.
Which applications segment to contribute significant share in the intensive care consumables market in 2026?
In terms of applications, critical respiratory care segment to command 32.0% share in the intensive care consumables market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Consumables
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Consumables , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Consumables , 2026 to 2036
- Respiratory consumables
- Vascular access consumables
- Patient monitoring disposables
- Infection control disposables
- Nutrition & feeding consumables
- Respiratory consumables
- Y to o to Y Growth Trend Analysis By Consumables , 2021 to 2025
- Absolute $ Opportunity Analysis By Consumables , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Critical respiratory care
- Sepsis & infection management
- Cardiac critical care
- Neurological ICU care
- Post-operative ICU care
- Critical respiratory care
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channels
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channels, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channels, 2026 to 2036
- Hospital procurement
- Group purchasing organizations
- Direct manufacturer sales
- Distributors
- Emergency stockpiles
- Hospital procurement
- Y to o to Y Growth Trend Analysis By Sales Channels, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channels, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Consumables
- By Applications
- By Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Consumables
- By Applications
- By Sales Channels
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Consumables
- By Applications
- By Sales Channels
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Consumables
- By Applications
- By Sales Channels
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- BD
- Baxter
- Fresenius Medical Care
- Smiths Medical
- Others
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Consumables , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Sales Channels, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Consumables
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Sales Channels
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Consumables
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by Sales Channels
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Consumables
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by Sales Channels
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Consumables
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by Sales Channels
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Consumables
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Sales Channels
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Consumables
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by Sales Channels
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Consumables
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Sales Channels
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Consumables , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Consumables , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Consumables
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Sales Channels, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channels, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Sales Channels
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

