Laboratory Information System Market
The Laboratory Information System Market is segmented by Product, Component, Delivery Mode, Application, End User, Functionality, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Laboratory Information System Market Size, Market Forecast and Outlook By FMI
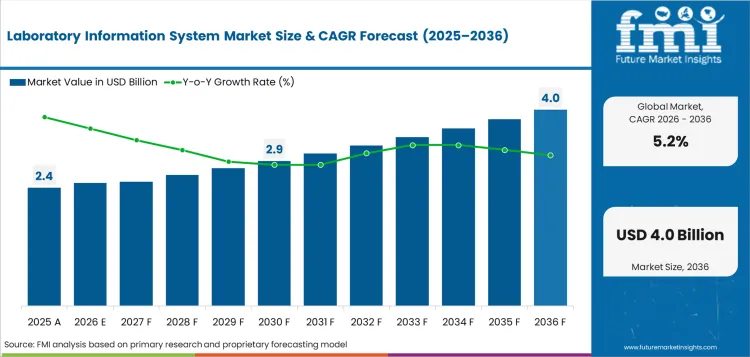
The laboratory information system market was valued at USD 2.4 billion in 2025. The market is forecast to reach USD 2.5 billion in 2026 and is forecast to expand at a CAGR of 5.2% between 2026 and 2036 to reach USD 4.0 billion by 2036. Integrated laboratory information systems are expected to dominate with 68.0% product share, and software is projected to lead the component segment with a 60.0% share.
Regulatory uncertainty and data-quality pressure now define LIS buying more than simple result storage. FDA’s 2025 reversal of the May 2024 LDT final rule shows why laboratories need systems that can adjust compliance fields without disrupting operations. CAP’s 2024 checklist changes added CLIA personnel qualification updates effective December 28, 2024.
That raises the value of credential tracking and quality sign-off inside the LIS. Interoperability adds the next pressure point because ASTP/ONC found that 46% of health information organizations map non-standard laboratory codes to LOINC. A 2025 LIS migration study involving Epic Beaker and Epic EMR shows why replacements require validation, archive access and coordinated go-live planning. Buyers are likely to favor vendors that combine compliance flexibility, terminology control and implementation support.
Summary of the Laboratory Information System Market
- Demand and Growth Drivers
- Hospital digitization is creating demand for laboratory platforms which control test orders and result release.
- Diagnostic chains need multi-site records as sample movement and reporting timelines become harder to manage.
- Compliance needs reinforce LIS spending as laboratories need audit trails and controlled data access.
- Product and Segment View
- Integrated laboratory information systems are expected to lead the product segment with 68.0% share in 2026, reflecting demand for connected laboratory records.
- Software is expected to lead the component segment with 60.0% share in 2026, reflecting high dependence on workflow rules and data control.
- Cloud-based delivery is predicted to hold 45.0% share in 2026 as diagnostic networks need remote access and lower server burden.
- Geography and Competitive Outlook
- China is expected to record a CAGR of 11.5% through 2036 as hospital networks invest in digital laboratory control.
- India is projected to expand at 6.3% CAGR from 2026 to 2036 as digital health records and diagnostic chains advance.
- Companies combining interface depth with hospital workflow knowledge are strengthening their market position during the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Associate Vice President at FMI says, “Laboratory information systems now support hospital and diagnostic chain operations through sample tracking and result release control. Strong interface engines reduce go-live risk as integration work and changeover cost shape long LIS contracts across older hospital systems.”
- Laboratory Information System Market Value Analysis
- The laboratory information system market is shifting from standalone result storage into structured workflow control across hospitals and diagnostic networks.
- Adoption is being enhanced by the need for sample traceability and audit-ready reporting across clinical laboratories.
- The market is benefiting from deeper use of electronic medical records across hospitals and outpatient care settings.
- Demand is supported by connected records that meet compliance and clinician review needs.
Laboratory Information System Market Definition
The laboratory information system market includes software platforms used to manage laboratory orders and sample workflows before results reach clinical users. Systems assess and control sample collection, test ordering, result reporting, quality records, and data exchange. Scope includes standalone LIS, integrated LIS, software, hardware, services, and hosting models. General electronic health record platforms and laboratory instruments are outside the scope.
Laboratory Information System Market Inclusions
Market scope covers all commercially traded laboratory information system products categorized by product type, including standalone and integrated LIS platforms; components, including software, hardware and services; delivery modes, including on-premise, cloud-based and hybrid deployment; applications, including clinical diagnostics, laboratory management and drug development; end users, including hospitals, clinics, independent laboratories and research institutes; and functionalities, including workflow management, decision support and integration. Revenue scope covers 2026 to 2036. Key stakeholders include LIS software providers, hospital IT teams, diagnostic laboratory operators, healthcare system integrators, cloud hosting providers, laboratory equipment vendors, clinical data management teams, compliance officers, public health agencies and regulatory bodies.
Laboratory Information System Market Exclusions
Scope does not include laboratory instruments, general hospital billing systems, imaging software, generic EHR platforms, or patient scheduling tools without laboratory workflow control.
Laboratory Information System Market Research Methodology
- Primary Research: FMI analysts talked to hospital laboratories, diagnostic chains, healthcare IT integrators, and laboratory software specialists in priority markets.
- Desk Research: FMI combined data from digital health programs, vendor product portfolios, regulatory references, and laboratory workflow requirements.
- Market sizing and forecasting: FMI added LIS use across product categories and buyer groups from the bottom up.
- Data validation: FMI cross-checked every three months against hospital IT spending patterns and company software coverage.
Why is the Laboratory Information System Market Growing?
- Hospitals are using LIS platforms to reduce manual result handling before clinical review begins.
- Diagnostic chains are using cloud-based systems as multi-site testing needs shared records and centralized access.
- Laboratories are replacing disconnected tools with platforms offering stronger workflow and audit control.
Clinical laboratories need stronger data control as test volumes move across hospitals and outside diagnostic networks. As of July 2025, CMS reported 307,193 CLIA-certified laboratories in the United States. The CLIA compliance base supports demand for LIS platforms with controlled records and audit trails. Vendors with strong validation tools help laboratories reduce manual checks during inspection and quality review.
Electronic health record adoption raises another channel for LIS integration. The 2024 NEHRS found 95.0% EHR adoption among USA office-based physicians and 83.6% use of certified EHR systems. The clinical record base raises the need for laboratory results to move into provider workflows without manual re-entry. LIS vendors with reliable interface engines gain preference as health systems protect turnaround time and data accuracy.
Market Segmentation Analysis
- Integrated laboratory information systems are estimated to capture 68.0% product share in 2026 as hospital networks prioritize connected sample records and result release.
- Software is expected to represent 60.0% of the component mix in 2026 through its central role in workflow control and audit documentation.
- Cloud-based delivery is projected to reach 45.0% share by 2026 as diagnostic chains require shared access across multiple sites.
- Clinical diagnostics is estimated to contribute 41.0% of application revenue in 2026 since routine testing relies on accurate reporting and faster clinician review.
- Hospitals are projected to hold 42.0% end-user share in 2026 through high test volumes and stronger sample tracking needs.
Laboratory information systems are divided into six primary segment groups based on product, component, delivery mode, application, end user, and functionality. Product includes Standalone Laboratory Information Systems and Integrated Laboratory Information Systems. Component includes Software, Hardware, and Services. Delivery mode includes On-Premise, Cloud-Based, and Hybrid Deployment. Application includes Clinical Diagnostics, Laboratory Management, and Drug Development. End user includes Hospitals, Clinics, Independent Laboratories, and Others. Functionality includes Workflow Management, Decision Support, and Integration and Interoperability.
Insights into Laboratory Information System Market by Product
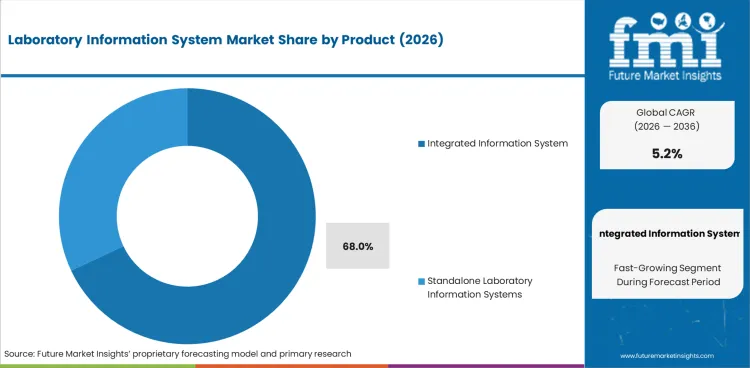
- Integrated laboratory information systems are expected to account for 68.0% of the product segment in 2026. Hospitals prefer integrated platforms as test orders and result release need one controlled workflow inside clinical laboratories.
- Standalone LIS is useful for smaller laboratories with limited connectivity needs and narrower reporting requirements. Its role is more selective when hospitals need direct links with clinical records and billing systems.
Insights into Laboratory Information System Market by Component
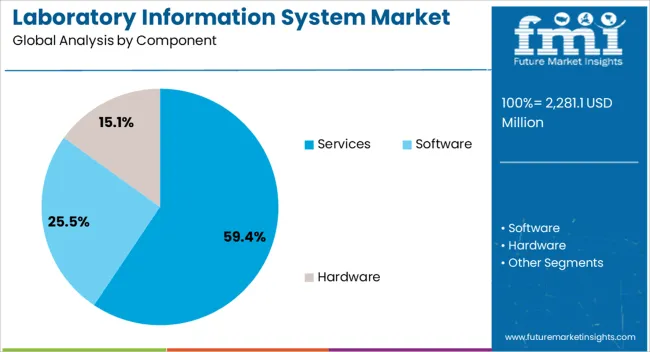
- Software is projected to hold 60.0% of the component segment in 2026. Laboratory teams rely on software to manage workflow rules, audit records and user access controls.
- Hardware and services usually follow the software decision during most LIS replacement programs. Their value improves during server upgrades, system migration and interface configuration work.
Insights into Laboratory Information System Market by Delivery Mode
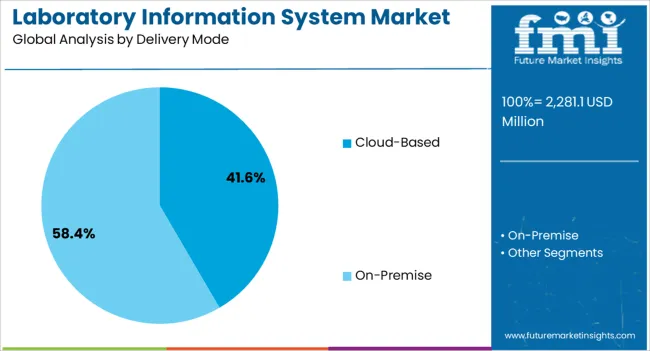
- In 2026, cloud-based delivery is expected to represent 45.0% share of the delivery mode segment. Diagnostic chains use cloud platforms to manage shared records across multiple laboratory sites.
- On premise systems continue in hospitals with strict internal data control needs and local hosting preferences. Hybrid deployment is preferred when laboratories want local control with remote access options.
Insights into Laboratory Information System Market by Application
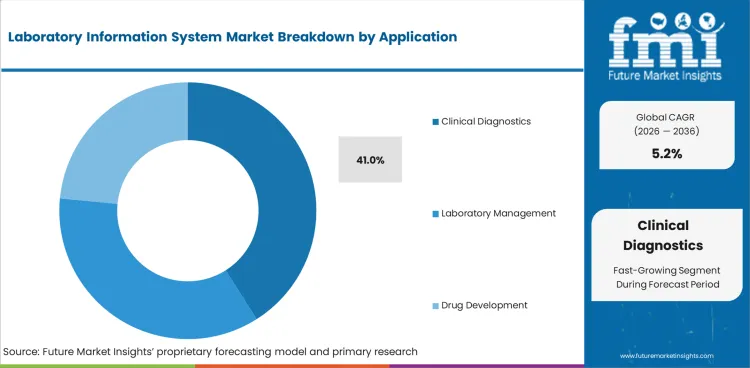
- Clinical diagnostics is forecast to account for 41.0% of the application segment in 2026. Routine testing generates high result volumes and requires accurate reporting to clinical teams.
- Laboratory management and drug development applications need deeper configuration for quality records and sample data. Their demand improves when laboratories require structured controls for review and reporting.
Insights into Laboratory Information System Market by End Use
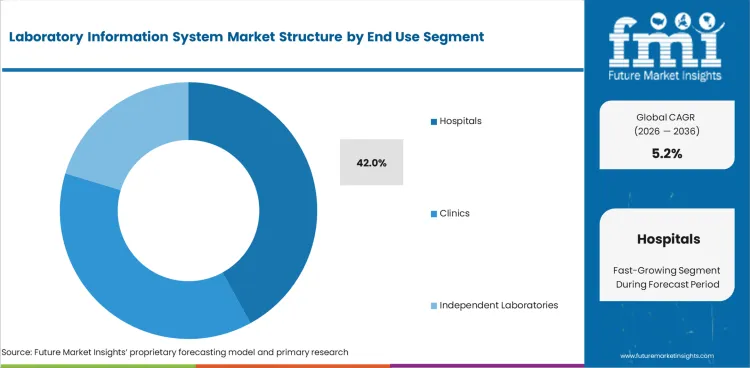
- Hospitals are projected to hold 42.0% of the end-use segment in 2026. Hospital laboratories need LIS platforms to manage test ordering, sample tracking and result release.
- Independent laboratories and clinics use LIS platforms for reporting speed and referral management. Their buying decisions often rely on hosting cost and integration effort.
Laboratory Information System Market Drivers, Restraints, and Opportunities
- Clinical result traceability creates structured demand for LIS platforms across high-volume diagnostic departments.
- Integration cost increases pricing pressure for smaller hospitals replacing older laboratory systems.
- Cloud-based LIS improves service demand through lower maintenance burden and easier multi-site access.
Laboratory information systems for hospitals and diagnostic chains continue to expand because clinical labs need sample tracking, result release control and connected patient records for daily testing. Cost pressure and migration work can slow broader adoption. Expansion continues through cloud-based platforms and hospital integration.
Reimbursement Timing Is Moderating LIS Price Resistance
CMS policy creates a two-speed pricing environment for laboratory information systems: less immediate discounting pressure in 2026. Buyer scrutiny is tighter for contracts that run into 2027. In May 2026, CMS stated, “there is no CLFS phase-in reduction in 2026, and payments may not be reduced by more than 15% per year from January 2027 to 2029.
The related reporting window runs from May 1 to July 31, 2026, using January 1 to June 30, 2025 data”. LIS vendors can defend pricing for compliance reporting and payer rate analytics due to regulatory and operational requirements. Buyers will likely push for staged implementation fees before reimbursement risk stabilizes and budget visibility improves.
Public Tenders Are Forcing Price Unbundling
Public-sector LIMS procurement is pushing market pricing away from opaque license quotes and toward itemized total-cost-of-ownership comparisons. In August 2025, the City of Abilene RFP assigned 30 points to price and required vendors to break out system licensing and installation costs. Vendors had to detail integration and training, along with annual maintenance and total project cost.
The RFP gave 25 points to system features and compatibility. The scoring indicates low upfront software pricing is losing value when integration depth is weak. For LIS suppliers, modular pricing gains value with clearly priced interfaces and implementation milestones across system delivery. Defined support tiers improve transparency and simplify evaluation for buyers during procurement decisions. For buyers, it strengthens negotiation leverage around maintenance escalators and paid change requests.
Analysis of Laboratory Information System Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| USA | 3.6% |
| China | 11.5% |
| Japan | 5.3% |
| Germany | 2.8% |
| Spain | 4.6% |
| India | 6.3% |
| Brazil | 5.3% |
Source: Future Market Insights, 2026.
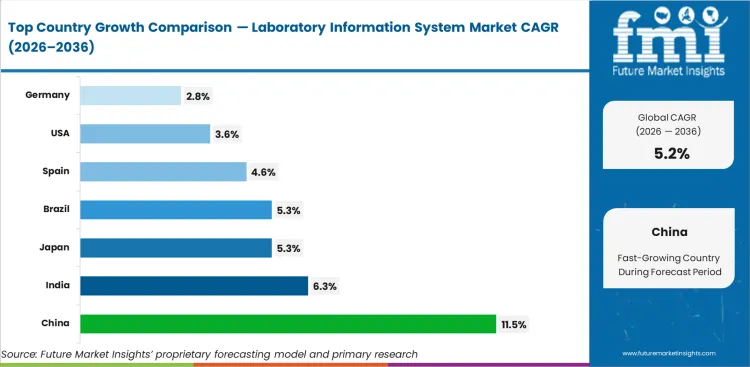
- China is projected to record 11.5% CAGR through 2036 as hospital networks expand and diagnostic capacity moves into larger digital workflows.
- India’s laboratory information system market is expected to advance at 6.3% CAGR during 2026 to 2036, backed by digital health records and private diagnostic chains.
- Japan is forecast at 5.3% CAGR for the assessment period as hospitals replace older systems and improve laboratory quality control.
- Brazil is expected to expand at 5.3% CAGR by 2036, due to public digital health investment and regional laboratory connectivity needs.
- Spain’s market is projected to register 4.6% CAGR across the forecast years as public hospitals manage heavier workloads and improve result routing.
- USA demand is expected to rise at 3.6% CAGR over the decade as mature EHR use shifts LIS spending toward replacement programs.
- Germany is forecast to post 2.8% CAGR between 2026 and 2036 as established hospital IT estates follow careful upgrade cycles.
Demand for laboratory information systems is forecast to rise at 5.2% CAGR from 2026 to 2036. Country-level analysis covers the markets contributing to this forecast, with the key markets listed below.
Demand Outlook for Laboratory Information System Market in China
China’s laboratory information system industry is projected to record a CAGR of 11.5% through 2036. China’s healthcare network is raising need for laboratory information system platforms across hospitals and diagnostic settings. Local hospitals require better sample control and result release as testing activity becomes more connected with clinical records. LIS vendors with practical implementation support can help providers improve reporting discipline and reduce manual workflow gaps.
- Large hospital networks raise demand for integrated laboratory records and workflow management.
- Private hospitals need LIS platforms which improve turnaround time and reporting quality.
- Diagnostic data growth enhances the use of digital pathology tools alongside LIS platforms.
Sales Analysis of Laboratory Information System Market in India
The laboratory information system sector in India is expected to expand at a CAGR of 6.3% during the assessment period. As of February 6, 2025, India recorded over 73.98 crore ABHA accounts and over 49.06 crore linked health records. The ABDM digital base supports demand for India laboratory information system platforms. Hospitals and diagnostic chains need LIS tools to connect test results with patient records.
- Private diagnostic chains need cloud-based LIS platforms for multi-city testing operations.
- Hospitals using ABHA workflows need structured laboratory results for digital patient records.
- Public digital health expansion boosts demand for digital transformation in healthcare across diagnostic workflows.
Demand Outlook for Laboratory Information System Market in Japan
Japan is forecast to grow at a CAGR of 5.3% by 2036. Japan’s hospital base was reported at 8,110 facilities in 2025 MHLW-based research. Mature hospital networks value LIS platforms with low disruption and strong data quality. Replacement demand is strongest where older systems limit workflow control and clinical connectivity.
- Urban hospitals need LIS platforms to connect laboratory results with clinical records.
- Regional hospitals value staged migration from older laboratory software.
- Diagnostic departments use laboratory information management systems where research and clinical workflows overlap.
Opportunity Analysis of Laboratory Information System Market in Brazil
The laboratory information system segment in Brazil is forecast to advance at 5.3% CAGR over the forecast years. Brazil announced USD 84 million in informatics and digital solutions in 2024 for North and Northeast regions. The public digital health investment supports demand for Brazil laboratory information system platforms. Regional networks need affordable implementation and remote service support.
- Public hospitals need LIS platforms which work across uneven infrastructure and limited IT staffing.
- Diagnostic providers in major cities value stronger links with private hospital networks.
- Cloud hosting can reduce maintenance pressure for laboratories outside major urban centers.
Future Outlook for Laboratory Information System Market in Spain
The Spain laboratory information system industry is expected to post 4.6% CAGR through 2036. INE reported 4,979,678 hospital discharges in 2024, up 2.3% from 2023. The inpatient workload supports demand for Spain laboratory information system platforms as hospitals manage result flow and discharge timing. Vendors with fast configuration and integration tools can serve public hospital networks.
- Public hospitals need LIS tools to reduce manual result entry and reporting delays.
- Regional health services value platforms connecting laboratories across hospital networks.
- Inpatient workload raises demand for better sample tracking and clinical result routing.
Demand Outlook for Laboratory Information System Market in the United States
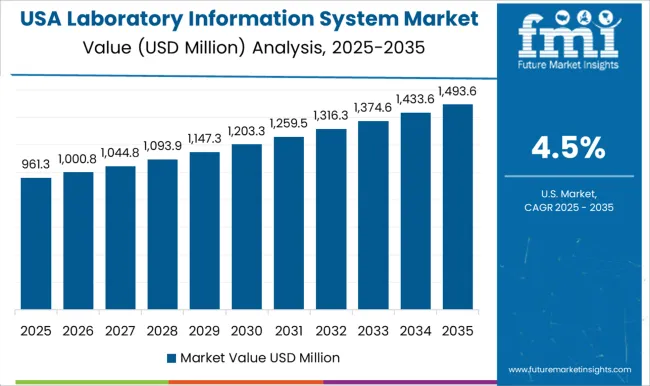
The USA laboratory information system industry is projected to record a CAGR of 3.6% through 2036. USA demand is shaped by mature hospital IT estates and strict laboratory quality requirements. Certified laboratories need accurate test records and dependable audit trails across routine diagnostic workflows. LIS providers need proven migration tools and strong interface depth to win replacement programs across established healthcare networks.
- Hospital outreach laboratories need LIS platforms to manage payer rules and performing lab identifiers.
- Reference laboratories prioritize fast interface work with hospital clients and physician records.
- Large health systems favor vendors with stable migration tools and cybersecurity documentation.
In-depth Analysis of Laboratory Information System Market in Germany
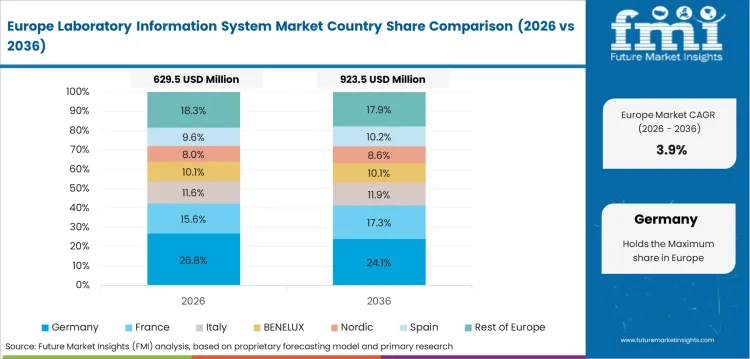
Germany’s laboratory information system industry is expected to post 2.8% CAGR through 2036. Established hospitals use mature IT estates, so replacement cycles reflect integration cost and downtime risk. Health expenditure reached EUR 538.2 billion in 2024, signaling a large system with careful budget control. LIS vendors must prove implementation discipline and reliable service capacity.
- University hospitals need LIS platforms to handle complex testing and research workflows.
- Regional labs emphasize validated interfaces with hospital records and billing systems.
- Buyers prefer phased implementation plans to avoid laboratory downtime during platform change.
Competitive Landscape and Strategic Positioning
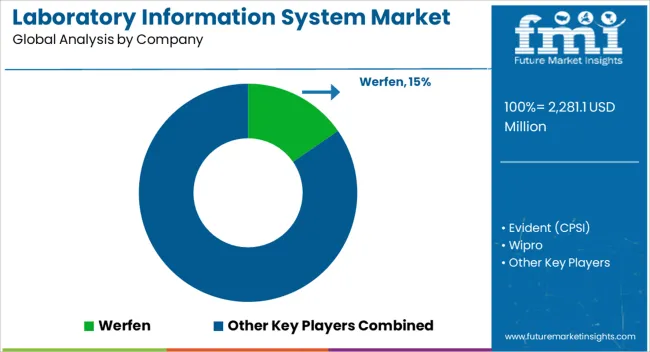
Laboratory Information System Market Analysis By Company
- Werfen holds a leading position through diagnostic workflow depth and close alignment with laboratory software needs.
- LabWare and LabVantage use broad informatics coverage and flexible workflow systems for regulated laboratories.
- Specialist providers concentrate on diagnostic chains and hospital laboratory operations with service teams built around workflow-specific rollout.
Competition in the laboratory information system market is influenced by workflow control and integration reliability across hospital and diagnostic networks. Werfen combines diagnostic system alignment with laboratory workflow knowledge for health systems needing practical LIS configuration. LabWare brings strong informatics coverage across laboratory data management and regulated workflows. LabVantage and Sunquest focus on the enterprise LIS capability and implementation depth. Epic Systems and Oracle Health add hospital platform reach through clinical record integration.
High migration cost and strict documentation needs create barriers across regulated laboratory environments. Interface quality and faster implementation are central priorities across long hospital IT cycles. Health systems favor vendors offering workflow control and reliable data exchange with less go-live uncertainty. Vendors with broader service coverage are better placed to reduce coordination burden for hospitals.
Key Companies in the Laboratory Information System Market
Global companies active in the laboratory information system market include:
- Werfen, LabWare, and LabVantage have strong laboratory workflow coverage and system configuration depth.
- Oracle Health, Epic Systems Corporation, and MEDITECH serve hospitals through broader clinical record integration.
- Sunquest, Soft Computer Consultants, Xifin, CompuGroup Medical, and Sysmex Corporation hold specialist positions across diagnostic workflow and laboratory data exchange.
Competitive Benchmarking: Laboratory Information System Market
| Company | Workflow Configuration Depth | Interface Engine Strength | Hospital Network Reach | Geographic Footprint |
|---|---|---|---|---|
| Werfen | High | High | Strong | Direct operations across over 30 countries and distributor presence in over 100 territories. |
| LabWare | High | Medium | Moderate | Offices across 25 countries with customers in 125 countries and over 40 offices on six continents. |
| LabVantage | High | Medium | Moderate | Nearly 40 global locations across the Americas, Europe and Asia Pacific. |
| Oracle Health | Medium | High | Strong | Broad global Oracle presence with healthcare operations across North America, Europe and Asia Pacific. |
| Epic Systems Corporation | Medium | High | Strong | Strong USA base with international activity across Canada, Europe, Asia Pacific and the Middle East. |
| MEDITECH | Medium | High | Strong | Strong North American base with international business across Africa, Asia, Australia and Europe. |
| Sunquest Information Systems | High | High | Strong | Operates under Clinisys with offices across the USA, UK, India and several European markets. |
| Soft Computer Consultants | High | Medium | Moderate | USA-centered presence with global clinical laboratory information system reach. |
| Xifin | Medium | High | Moderate | Primarily USA-focused with strongest presence in diagnostic and laboratory billing workflows. |
| CompuGroup Medical | Medium | Medium | Strong | Active in 60 countries with company locations across 19 countries. |
Source: Future Market Insights, 2026.
Key Developments in Laboratory Information System Market
- In April 2025, LabWare launched LabWare ASSURE as a SaaS platform for laboratory quality and compliance workflows.
- In August 2025, Oracle made its modern Oracle Health EHR available for ambulatory providers in the United States.
- In October 2025, Oracle introduced Oracle Health Connection Hub for data sharing control and interoperability governance.
- In July 2025, Sysmex America presented informatics solutions as part of its laboratory portfolio at ADLM 2025.
Key Players in the Laboratory Information System Market
Major Global Players:
- Werfen
- Evident
- Wipro
- CompuGroup Medical
- LabWare
- Roper Technologies Inc.
- Medical Information Technology, Inc.
- Xifin Inc.
- Epic Systems Corporation
- Oracle Health
Specialist Players:
- Soft Computer Consultants, Inc.
- Allscripts Lab
- LigoLab
- Sysmex Corporation
- Ilex Medical
- LabVantage Medical Suite
- McKesson Corporation
Report Scope and Coverage

| Parameter | Details |
|---|---|
| Quantitative Units | USD 2.5 billion to USD 4.0 billion, at a CAGR of 5.2% |
| Market Definition | The laboratory information system market encompasses software platforms used to manage laboratory samples, test orders, results, and workflow records. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, China, Japan, Germany, Spain, India, Brazil, and 30+ countries |
| Key Companies Profiled | Werfen, LabWare, LabVantage, Oracle Health, Epic Systems Corporation, MEDITECH, Sunquest, Xifin, CompuGroup Medical, Sysmex Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with product demand and verified laboratory workflow use patterns. |
Source: Future Market Insights, 2026.
Laboratory Information System Market by Segments
By Product
- Standalone Laboratory Information Systems
- Integrated Laboratory Information Systems
By Component
- Software
- Hardware
- Services
By Delivery Mode
- On-Premise
- Cloud-Based
- Hybrid Deployment
By Application
- Clinical Diagnostics
- Laboratory Management
- Drug Development
By End Use
- Hospitals
- Clinics
- Independent Laboratories
- Others
By Functionality
- Workflow Management
- Decision Support
- Integration and Interoperability
Laboratory Information System Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- Centers for Medicare & Medicaid Services. (2026, May 1). Clinical Laboratory Fee Schedule. USA Department of Health & Human Services.
- City of Abilene. (2025, August). Request for proposal for laboratory information management system: RFP #CB-2558. City of Abilene.
- Centers for Medicare & Medicaid Services. (2025, September). CLIA proficiency testing (PT) (CMS-10690). Regulations.gov.
- Centers for Disease Control and Prevention. (2025, December). NEHRS results and publications.
- Press Information Bureau. (2025, February). Update on the implementation of Ayushman Bharat Digital Mission. Government of India.
- Instituto Nacional de Estadística. (2026, March). Hospital Morbidity Survey. Year 2024.
- International Trade Administration. (2025, August). Brazil - Healthcare. USA Department of Commerce.
- Federal Statistical Office of Germany. (2026). Health expenditure by functions of health care. Destatis.
- Yasui, T., et al. (2025). Impact of drug shortages on the work of hospital pharmacists in Japan. Journal of Pharmaceutical Policy and Practice.
This Report Answers
- What is the current and future size of the laboratory information system market?
- How fast is the laboratory information system market expected to expand between 2026 and 2036?
- Which product segment is expected to lead the market by 2026?
- Which component segment is expected to account for the highest demand by 2026?
- What factors are driving demand for laboratory information systems globally?
- How are hospital digitization and data traceability influencing LIS adoption?
- Why are integrated laboratory information systems the main product base for demand?
- How are cloud-based systems creating demand for multi-site diagnostic networks?
- Which countries are projected to record faster expansion through 2036?
- What is driving market expansion in China and India?
- Who are the key companies active in the laboratory information system market?
- How does FMI estimate and validate the market forecast?
Frequently Asked Questions
What is the global market demand for Laboratory Information System in 2026?
In 2026, the global market for laboratory information systems is expected to reach USD 2.5 billion.
How big will the market for Laboratory Information System be in 2036?
By 2036, the market for laboratory information systems is expected to reach USD 4.0 billion.
How much is demand for Laboratory Information System expected to expand between 2026 and 2036?
Between 2026 and 2036, demand for laboratory information systems is expected to expand at a CAGR of 5.2%.
Which product segment is expected to lead globally by 2026?
Integrated laboratory information systems will make up 68.0% of the product segment in 2026.
What is causing demand to rise in China?
The China laboratory information system market is projected to record 11.5% CAGR through 2036, due to hospital digitization and diagnostic capacity.
What is causing demand to rise in India?
The India laboratory information system market is projected to expand at 6.3% CAGR through 2036, due to digital health records and diagnostic chains.
What does this report mean by Laboratory Information System Market definition?
The laboratory information system market includes software used to manage laboratory samples, orders, results, workflows, and data exchange.
How does FMI make the Laboratory Information System forecast and check it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with product demand and checking it against workflow use patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Chemistry Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Chemistry Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Chemistry Type , 2026 to 2036
- Waterborne systems
- Powder systems
- High-solids systems
- Waterborne systems
- Y to o to Y Growth Trend Analysis By Chemistry Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Chemistry Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Model, 2026 to 2036
- On-site
- Shop-floor conversion
- Audit support
- On-site
- Y to o to Y Growth Trend Analysis By Service Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Service Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Substrate Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Substrate Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Substrate Type, 2026 to 2036
- Aluminum Surfaces
- Steel surfaces
- Composite surfaces
- Aluminum Surfaces
- Y to o to Y Growth Trend Analysis By Substrate Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Substrate Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Industrial metal
- Automotive refinish
- Aerospace parts
- Industrial metal
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Compliance Need
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Compliance Need, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Compliance Need, 2026 to 2036
- VOC reduction
- Cr(VI) substitution
- PFAS avoidance
- VOC reduction
- Y to o to Y Growth Trend Analysis By Compliance Need, 2021 to 2025
- Absolute $ Opportunity Analysis By Compliance Need, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- By Country
- Market Attractiveness Analysis
- By Country
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Chemistry Type
- By Service Model
- By Substrate Type
- By End Use
- By Compliance Need
- Competition Analysis
- Competition Deep Dive
- PPG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- AkzoNobel
- Axalta Coating Systems
- Sherwin-Williams
- Nippon Paint
- PPG
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Chemistry Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Service Model, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Substrate Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Compliance Need, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Chemistry Type
- Figure 6: Global Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Service Model
- Figure 9: Global Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Substrate Type
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Compliance Need
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Chemistry Type
- Figure 32: North America Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Service Model
- Figure 35: North America Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Substrate Type
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Compliance Need
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Chemistry Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Service Model
- Figure 51: Latin America Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Substrate Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Compliance Need
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Chemistry Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Service Model
- Figure 67: Western Europe Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Substrate Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Compliance Need
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Chemistry Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Service Model
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Substrate Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Compliance Need
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Chemistry Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Service Model
- Figure 99: East Asia Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Substrate Type
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Compliance Need
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Chemistry Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Service Model
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Substrate Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Compliance Need
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Chemistry Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Chemistry Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Chemistry Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Service Model, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Service Model, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Service Model
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Substrate Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Substrate Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Substrate Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Compliance Need, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Compliance Need, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Compliance Need
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

