Mammalian Transient Protein Expression Market
The Mammalian Transient Protein Expression Market is segmented by Product Category (Expression Vectors, Reagents, Competent Cells, Instruments), Application (Bio-production, Drug Discovery, Functional Assays, Vaccine Research), Expression Host (HEK293, CHO, Hybridoma, Others), End User (Biopharma Companies, CROs, Academic Institutes, Research Labs), Scale (Micro Scale, Small Scale, Pilot Scale, High Throughput). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mammalian Transient Protein Expression Market Size, Market Forecast and Outlook By FMI
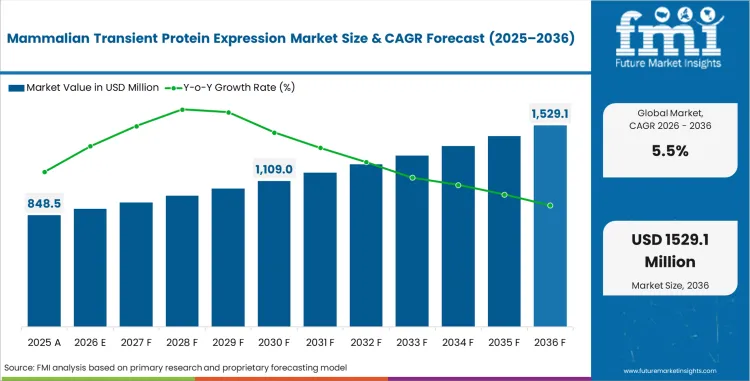
The mammalian transient protein expression market was valued at USD 804.3 million in 2025. The industry is expected to reach USD 848.5 million in 2026, and cross USD 1,449.4 million by 2036 at a 5.5% CAGR during the forecast period. Expression vectors are projected to account for 28.5% share in 2026, as plasmid design quality influences protein yield and screening speed. Bio-production is estimated to hold 40.0% share in 2026.
Summary of the Mammalian Transient Protein Expression Market
- Demand and Expansion Drivers
- Antibody discovery supports platform use as screening teams need gram-scale protein samples before stable line selection.
- Recombinant protein studies support repeat reagent purchases with vectors and transfection systems needed in every new construct cycle.
- Contract research demand supports service expansion as small biotech firms outsource protein production before asset validation.
- South Korea’s 6.4% CAGR reflects public and private biologics capacity across Seoul and Incheon clusters.
- Product and Segment View
- Expression vectors are projected to lead with 28.5% share in 2026 as construct quality affects downstream assay reliability.
- Bio-production is expected to hold 40.0% share with antibody and antigen supply supporting early drug work.
- HEK293 is projected to account for 46.0% share in 2026 as fast expression suits difficult proteins and rapid screening.
- Biopharma Companies are expected to lead end user with 48.0% share as internal teams run repeated candidate selection cycles.
- Small Scale is projected to lead Scale with 42.0% share as most projects need milligram to gram quantities.
- Geography and Competitive Outlook
- The USA is projected to record 5.8% CAGR through 2036 as research funding and biotech density support repeat purchases.
- Germany is expected to expand at 5.6% CAGR through 2036 as biologics and process science programs support steady demand.
- Thermo Fisher Scientific is estimated to dominate through its Expi systems and reagent breadth.
- Merck KGaA and Cytiva compete through cell culture depth and service access. Sartorius and Lonza support demand through bioprocess platforms.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI says, “Our team view mammalian transient protein expression less as a commodity reagent market and more as a strategic acceleration layer within biologics research. The value lies in how effectively platforms compress candidate screening timelines without introducing variability in cost or assay confidence. Rising antibody complexity and tighter protein quality expectations are fundamentally changing early discovery workflows. Teams are now required to evaluate a broader construct set before committing to stable cell‑line development.”
- Mammalian Transient Protein Expression Market Value Analysis
- Revenue is projected to increase from USD 848.5 million in 2026 to USD 1,449.4 million by 2036.
- Expression vectors lead product demand as construct design influences yield and protein quality before purification begins.
- Bio-production forms the largest application base with antibody screening and recombinant antigen work supporting repeat usage.
- Direct sales dominate as biopharma teams need technical support during construct and scale changes.
- Price pressure is highest in academic labs as reagent costs build quickly across repeated transfection runs.
Mammalian Transient Protein Expression Market Definition
The mammalian transient protein expression market covers systems used to produce recombinant proteins without stable genome integration. Scope includes plasmid vectors and transfection reagents. It includes mammalian host cells and culture media. Enhancers and small bioreactors are included with related service workflows. Early bio-production studies use these systems for fast protein supply. Scope excludes bacterial expression and yeast expression.
Mammalian Transient Protein Expression Market Inclusions
Scope includes HEK293 systems and CHO systems. It also includes expression vectors and transfection reagents. Suspension cells and culture media are included with process enhancers. Small bioreactors and contract expression services are included across global demand.
Mammalian Transient Protein Expression Market Exclusions
Scope excludes bacterial expression and yeast expression. It excludes insect expression and stable clone selection. Commercial fill-finish work and large-scale GMP manufacturing are outside scope. Protein purification-only services and diagnostic kit production are excluded.
Mammalian Transient Protein Expression Market Research Methodology
- Primary Research: FMI analysts reviewed input from biopharma research teams and CRO service managers. Reagent suppliers and cell culture specialists informed the second review layer. Platform users added feedback on practical workflow issues. The review focused on yield reliability and turnaround time. Cell viability and protocol transfer support shaped the final buying criteria.
- Desk Research: Desk work used official company product information and scientific literature. Regulatory references and public funding sources supported the scope review. Product scope was defined around mammalian systems used for transient expression of proteins and antibodies.
- Market Sizing and Forecasting: The model combined reagent use frequency and project count. It reviewed service pricing and platform penetration. Country-level research intensity supported the final forecast.
- Data Validation: Forecasts were validated through comparison with antibody research activity and recombinant DNA use. Protein purification demand supported the final cross-check. Company portfolios were reviewed to confirm active participation in vectors and expression services.
Why is the Mammalian Transient Protein Expression Market Expanding?
- Antibody screening programs need faster protein batches before teams commit resources to stable cell-line work.
- Recombinant protein studies use mammalian hosts as folding and glycosylation can determine assay value.
- CRO service demand is increasing as small biotech firms outsource expression work before funding milestones.
- HEK293 workflows support fast turnaround as many discovery teams need rapid samples for functional assays.
- CHO workflows support comparability with later manufacturing hosts and reduce technical change during scale work.
Antibody discovery creates repeat demand for transient expression systems as each candidate panel needs fresh protein material. FDA’s Center for Drug Evaluation and Research approved 50 new drugs in 2024, with biologics and targeted therapies keeping early protein work active. This approval process puts pressure on discovery teams to generate decision-ready protein material before clinical candidates reach filing stage. Expression vendors benefit as research teams screen more antibody forms and need quick protein lots for binding tests.
Recombinant protein work favors mammalian systems when protein folding affects assay reliability and therapeutic relevance. Scientific literature from 2024 and 2025 continues to identify CHO and HEK293 systems as core hosts for transient gene expression in antibodies and complex proteins. This reinforces use in early testing as bacterial systems often miss mammalian post-translational patterns. Suppliers with vector design and transfection chemistry reduce troubleshooting time. Media optimization improves output stability during difficult expression programs. The result is higher repeat use among labs developing complex antibodies and glycoproteins.
Market Segmentation Analysis
- Expression vectors are projected to account for 28.5% share in 2026 as construct quality shapes yield and assay output.
- Bio-production is expected to account for 40.0% share in 2026 with antibody and antigen batches driving demand.
- HEK293 is projected to represent 46.0% share in 2026 as fast expression supports discovery timelines.
- Biopharma Companies are expected to hold 48.0% share in 2026 with internal discovery teams using repeated runs.
- Small Scale is projected to represent 42.0% share in 2026 as most projects need limited protein quantities.
The market is segmented by product category into expression vectors, reagents, competent cells, and instruments, each supporting different stages of transient protein production. It is further segmented by application across bio‑production, drug discovery, functional assays, and vaccine research, reflecting broad use in early biologics workflows. Segmentation by expression host includes HEK293, CHO, hybridoma, and other mammalian systems, with HEK293 leading due to rapid transfection performance. By end user and scale, the market spans biopharma companies, CROs, academic institutes, and research labs, operating from micro‑scale to high‑throughput production.
Mammalian Transient Protein Expression Market Analysis By Product Category
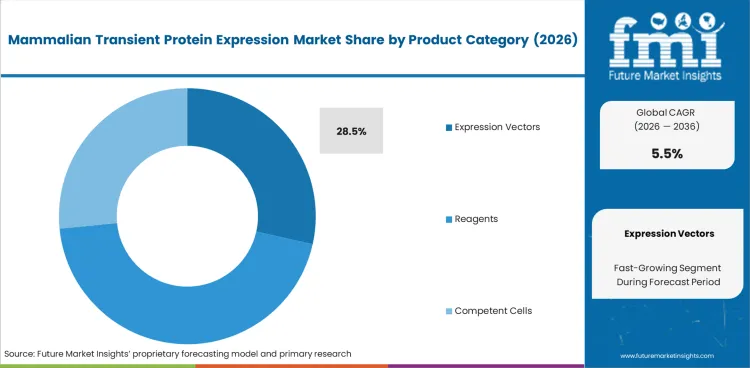
- Expression vectors are projected to account for 28.5% of product category in 2026 as plasmid architecture affects expression yield and protein recovery during early assay work.
- Buyers often choose premium vectors for difficult proteins. Weak signal or poor secretion can waste weeks of screening time across antibody programs.
Mammalian Transient Protein Expression Market Analysis By Application
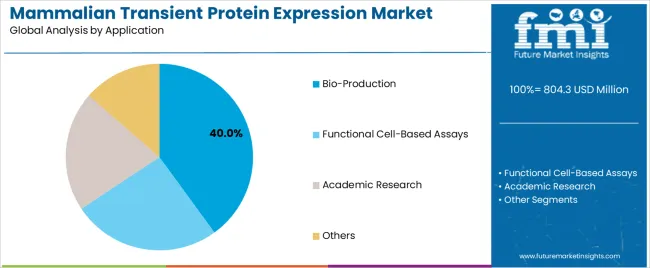
- In 2026, bio-production is expected to represent 40.0% of application segment with antibody and antigen sample needs supporting repeated transient expression cycles.
- Drug discovery teams often use bio-production batches before stable clone investment. Early purity and activity data decide which molecules advance.
Mammalian Transient Protein Expression Market Analysis By Expression Host
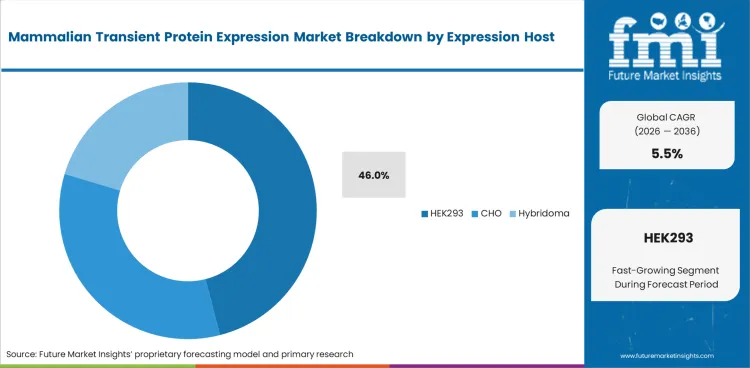
- HEK293 is anticipated to represent 46.0% of expression host in 2026 as fast transfection and high protein output suit screening projects.
- CHO is preferred in later comparability work since many commercial biologics use CHO manufacturing paths and need closer process alignment.
Mammalian Transient Protein Expression Market Analysis By End User
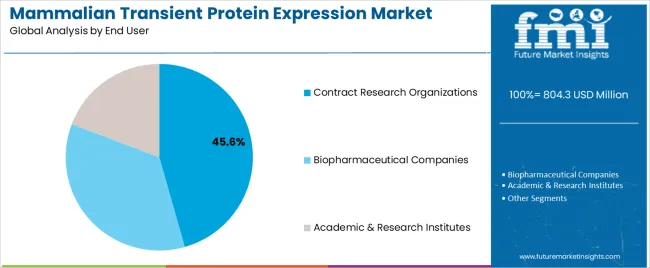
- Biopharma Companies are projected to account for 48.0% market share in 2026 with internal antibody and protein engineering teams using repeat workflows.
- CROs win overflow work from smaller biotech firms as service contracts reduce the need for in-house cell culture teams.
Mammalian Transient Protein Expression Market Analysis By Scale
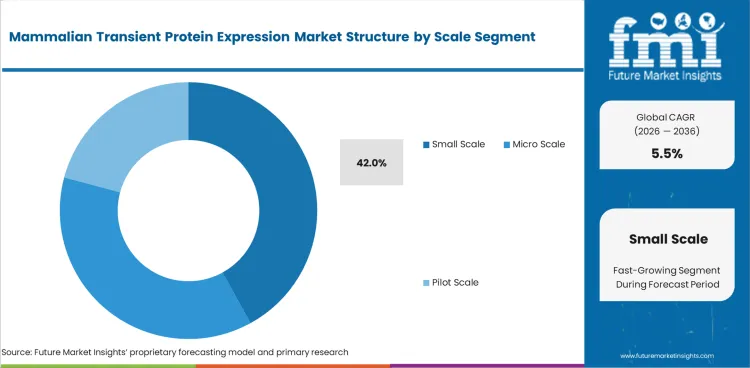
- Small Scale is expected to represent 42.0% share in 2026 as most discovery programs need milligram to gram output.
- High throughput formats support screening campaigns with many constructs and limited protein volume per candidate.
Mammalian Transient Protein Expression Market Drivers, Restraints, and Opportunities
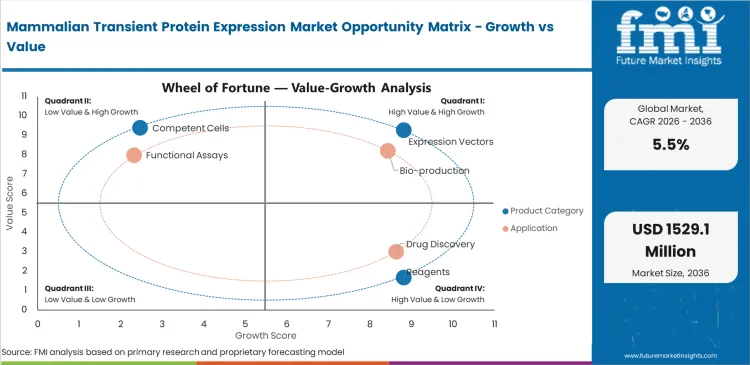
- Antibody complexity drives demand for mammalian hosts since protein folding can change binding and assay results.
- Screening volume supports repeat reagent use as each new construct needs vectors and transfection chemistry.
- High reagent cost limits broad use in academic labs with tight project budgets and frequent failed runs.
- CRO outsourcing creates opportunity as small biotech firms need fast protein samples without expanding internal capacity.
- CHO transient systems create opportunity as projects seek closer alignment with later production hosts.
Antibody Screening Workloads
Antibody screening is the most direct demand path for mammalian transient expression systems. Buyers need fast protein lots for binding assays and functional testing. Developability review happens before stable cell-line spending begins. Research antibodies demand supports this workflow as validated binding tools shape laboratory decisions. Expression suppliers gain advantage when systems reduce failed runs and support difficult molecules. This creates a practical buying test around turnaround time and yield range. Protein activity after purification shapes final supplier choice.
Biological Output Prices Are Supporting Selective Pass-Through
Price movement in the Mammalian Transient Protein Expression Market is directly linked to the selling-price environment for biologics and pharmaceutical preparation outputs, because transient expression services are commonly priced around cell-line platform use and delivery timelines. In BLS’ Producer Price Indexes, updated in March 2026, biological products including diagnostics increased 3.2% from March 2025 to March 2026. Pharmaceutical preparations increased 0.8% over the same period. This does not create blanket pricing power. CDMOs and expression-service providers can defend pass-through for higher-grade plasmids and analytical QA where clients require regulated-development readiness.
Consumables Costs Are Creating Quote Volatility
Transient mammalian expression pricing is exposed to upstream consumables rather than only final biologics pricing. BLS’ March 2026 PPI table shows industrial gases up 12.6% from March 2025 to March 2026 - unsupported plastic film and other shapes up 4.8%. Industrial chemicals down 0.5% and plastic resins and materials down 3.5%. For expression-service buyers, the mixed signal is important: some inputs used around bioreactor operations and single-use systems remain inflationary. Selected chemical and resin categories are softer. Suppliers should avoid long fixed-price quotes unless volumes and expedited timelines are tightly defined.
Analysis of Mammalian Transient Protein Expression By Key Countries
.webp)
| Country | CAGR |
|---|---|
| South Korea | 6.4% |
| China | 6.2% |
| India | 6.0% |
| USA | 5.8% |
| Germany | 5.6% |
| Japan | 5.2% |
| UK | 5.0% |
Source: FMI analysis based on primary research and proprietary forecasting model.
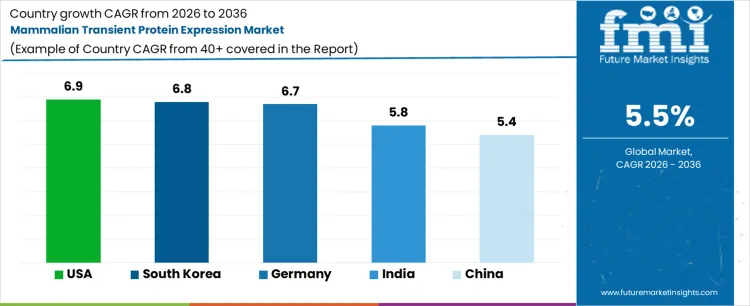
Mammalian Transient Protein Expression CAGR Analysis By Country
- South Korea is projected to record a 6.4% CAGR through 2036 as biologics investment supports advanced expression workflows.
- China is expected to expand at 6.2% CAGR through 2036 with domestic antibody discovery and CRO activity supporting demand.
- India is forecast to grow at 6.0% CAGR through 2036 as biosimilar research and contract services increase expression use.
- The USA is expected to advance at 5.8% CAGR through 2036 as biotech clusters support repeat research purchases.
- Germany is projected to rise at 5.6% CAGR through 2036 with process science and biologics research supporting steady demand.
- Japan is forecast at 5.2% CAGR through 2036 as domestic biologics programs sustain platform use.
- The UK is expected to post 5.0% CAGR through 2036 as life science hubs support early protein research.
Demand Outlook for Mammalian Transient Protein Expression in South Korea
South Korea has a established biologics production base and a fast-expanding research services base across Incheon and Seoul. The country is expected to post 6.4% CAGR through 2036 as domestic biologics firms focus on early research and scale-up capability. Public support for bioeconomy programs strengthens supplier confidence in advanced cell culture tools.
- Incheon’s Songdo cluster supports biologics activity through large-scale manufacturing and supplier access for cell culture systems.
- South Korea’s Ministry of Health and Welfare supports biohealth policy programs with emphasis on drugs and medical technology.
- Seoul research hospitals and biotech firms create repeat demand for bioprocess technology used in early protein work.
Sales Analysis of Mammalian Transient Protein Expression in China
China’s demand is shaped by domestic antibody pipelines and CRO capacity serving local and overseas clients. The country is projected to expand at 6.2% CAGR through 2036 with Shanghai and Suzhou supporting high research density. Local service providers compete through speed and bundled gene-to-protein workflows. Imported premium systems serve complex projects needing reproducibility across sites. Any increase in domestic reagent quality would shift more spending toward local suppliers and improve price access.
- Shanghai Zhangjiang remains a major life science base with antibody discovery firms and service providers.
- Suzhou BioBAY supports biotech firms needing expression services and early protein testing capacity.
- China’s biologics firms use recombinant DNA tools across discovery and process development work.
Demand Analysis of Mammalian Transient Protein Expression in India
India’s market is supported by biosimilar research and contract service expansion across Hyderabad and Bengaluru. The country is forecast to grow at 6.0% rate through 2036 as research teams expand antibody and recombinant protein workflows. Buyers often compare imported kits with lower-cost service options from local providers. Budget pressure remains a constraint in academic labs. Stronger CRO documentation and faster delivery could increase outsourced expression volumes across domestic biotech programs.
- Hyderabad’s Genome Valley supports biologics research and contract service activity across Telangana.
- Bengaluru life science firms use expression workflows for antibody discovery and recombinant protein studies.
- India’s BioE3 policy focus supports bio-manufacturing and related research capacity across national programs.
Opportunity Analysis of Mammalian Transient Protein Expression in the USA
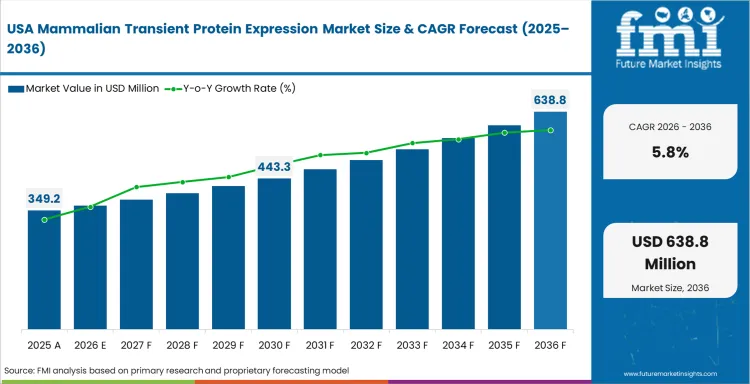
The USA has the deepest buyer base due to biotech clusters and academic labs. Large pharmaceutical research centers further increases demand. The country is expected to advance at 5.8% CAGR through 2036 as discovery programs keep using transient expression before stable-line work. Demand is concentrated around Boston and San Diego. The San Francisco Bay Area is another major research base. Suppliers win through local support and validated protocols across HEK293 and CHO systems. Faster sample-to-assay cycles could lift premium platform use in early biologics work.
- Boston-Cambridge supports repeated antibody and protein engineering work across universities and biotech firms.
- San Diego firms rely on CRO access and local supplier networks for fast research material.
- NIH-funded biomedical research supports demand for protein expression tools across academic labs.
Future Outlook for Mammalian Transient Protein Expression in Germany
Germany’s demand reflects process science strength and established biologics research across Munich. Heidelberg and North Rhine-Westphalia enhance research depth. Germany is projected to rise at 5.6% CAGR through 2036 with pharma and research institutes using expression systems for protein characterization. Higher CHO workflow use would support premium cell line and media demand across process-linked research.
- Munich’s biotech cluster supports antibody research and translational protein work across academic and private labs.
- Heidelberg research institutions support structural biology and recombinant protein demand.
- North Rhine-Westphalia gives suppliers access to pharma users and protein purification workflows.
Demand Outlook for Mammalian Transient Protein Expression in Japan
Japan’s demand is shaped by steady biologics research and careful platform qualification across Tokyo and Osaka. Japan is forecast at 5.2% CAGR through 2036 as domestic pharma companies keep using transient expression for early screening. Buyers often prefer systems with documented reproducibility and documented technical support. Adoption can move slowly due to validation routines inside established research teams. Better integration between expression and purification would support higher spend per project.
- Tokyo research hubs support antibody and recombinant protein work across pharma and university labs.
- Osaka’s pharmaceutical base creates demand for validated mammalian expression workflows.
- AMED funding programs support medical research connected with biologics and advanced therapy development.
In-depth Analysis of Mammalian Transient Protein Expression in the UK
The UK demand base is shaped by academic science and biotech clusters. Contract research activity around Cambridge and Oxford increases demand. The UK is expected to post 5.0% CAGR through 2036 as translational programs need assay-grade proteins. Buyers face funding cycles that can delay large kit purchases. CROs gain share by offering packaged gene-to-protein services. Stable public funding and private biotech investment would support broader use of antibody therapy research workflows.
- Cambridge supports antibody discovery and protein engineering through university spinouts and biotech firms.
- Oxford research groups create demand for recombinant proteins used in structural biology and immunology.
- UKRI funding supports bioscience research and reinforces demand for advanced lab workflows.
Competitive Landscape and Strategic Positioning
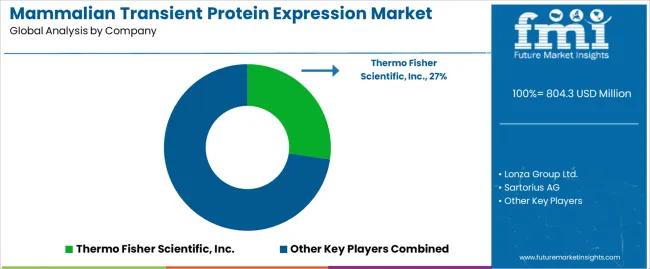
- Market concentration is moderate as large suppliers control platform depth and smaller firms compete through service speed.
- Established companies lead through expression kits and technical support for failed runs.
- Specialist CROs compete through bundled gene-to-protein services and faster antibody delivery timelines.
- Entry barriers include cell culture know-how and validated protocols. Reagent reliability and customer trust shape repeat projects.
Competition is built around platform reliability during early biologics work. Thermo Fisher Scientific holds a clear position with Expi293 and ExpiCHO systems across transient protein production. The company introduced Gibco Expi293 PRO in August 2025 as a next-generation HEK293 transient expression system for difficult proteins. This development holds importance as buyers want higher yield without rebuilding protocols. Suppliers with broad cell separation and protein workflows gain account access across research teams.
Capability depth separates established firms from smaller suppliers. Sartorius expanded production of GMP transfection reagents in Illkirch, France in September 2025. The move strengthens supply for cell and gene therapy workflows and supports reagent confidence in adjacent expression work. Cytiva introduced transient cell lines for AAV work in April 2024, which reinforces its position across viral vector and mammalian cell workflows. Buyers favor vendors able to support transfection and culture. Purification support reduces extra vendor handoffs.
Specialist service providers win through turnaround time and custom support. GenScript and Bioelectronica announced a strategic collaboration in August 2025 to link HyperCell screening with GenScript’s TurboCHO transient expression workflow. The TurboCHO platform supports purified antibodies in as little as five business days. Sino Biological competes through recombinant protein and antibody supply across research clients. Lonza supports customers through mammalian expression and process development strength. Entry remains hard as customers avoid switching after a platform produces reliable assays.
Companies in the Mammalian Transient Protein Expression Market
Competitive structure favors suppliers with expression systems, cell culture depth, and documented support across antibody programs.
- Global Leaders: Thermo Fisher Scientific and Merck KGaA compete through broad portfolios covering vectors and media. Cytiva and Sartorius compete through reagents and host-cell support. Lonza adds bioprocess support across research and development work.
- Specialist Service Providers: GenScript and Sino Biological compete through protein expression services and recombinant protein supply. Their position is clearest among biotech firms and academic labs needing purified proteins without building internal cell culture capacity.
- Regional and Emerging Players: Smaller CROs and reagent suppliers compete through local access and lower project cost. Custom antibody workflows help them win early-stage projects. These firms often win early-stage orders from academic groups and small biotech teams.
Competitive Benchmarking: Mammalian Transient Protein Expression Market
| Company | Vector-System Depth | Mammalian Host Coverage | Protein Delivery Support | Geographic Footprint |
|---|---|---|---|---|
| Thermo Fisher Scientific | High | High | Strong | Global |
| Merck KGaA | High | Strong | Strong | Global |
| Cytiva | Strong | High | Strong | Global |
| Sartorius | Strong | High | Strong | Global |
| Lonza | Strong | High | Strong | Global |
| GenScript | Medium | Strong | High | Global |
| Sino Biological | Medium | Medium | High | Global |
| Takara Bio | Medium | Medium | Moderate | Asia and North America |
Source: Future Market Insights competitive analysis 2026. Ratings reflect relative positioning based on vector-system depth and mammalian host coverage.
Developments in Mammalian Transient Protein Expression Market
- In August 2025, Thermo Fisher Scientific introduced Gibco Expi293 PRO Expression System for next-generation HEK293 transient expression. The system supports difficult protein research and higher throughput workflows.
- In September 2025, Sartorius expanded production of GMP transfection reagents in Illkirch, France. The expansion strengthens supply for cell and gene therapy workflows connected with transient transfection demand.
- In April 2024, Cytiva introduced transient cell lines supporting AAV workflow flexibility. The launch strengthens Cytiva’s position across cell culture and expression-adjacent research systems.
- In August 2025, GenScript and Bioelectronica announced a strategic collaboration linking antibody screening with TurboCHO transient expression. The collaboration supports faster validation after single-cell screening.
- In March 2026, Sartorius launched a rational-design CHO host cell line with higher productivity. The launch reinforces supplier focus on CHO performance and scale confidence.
Players in the Mammalian Transient Protein Expression Market
Global Leaders
- Thermo Fisher Scientific
- Merck KGaA
- Cytiva
- Sartorius
- Lonza
Specialist Service Providers
- GenScript
- Sino Biological
Regional and Emerging Players
- Takara Bio
- Creative Biolabs
- Leinco Technologies
Report Scope and Coverage
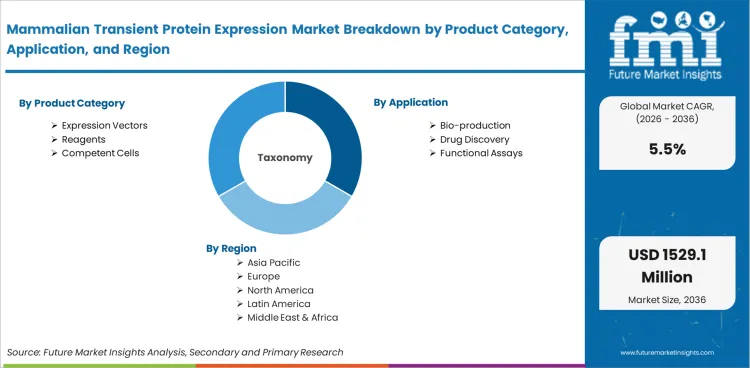
| Parameter | Details |
|---|---|
| Quantitative Units | USD 848.5 million in 2026 to USD 1,449.4 million by 2036 at 5.5% CAGR |
| Market Definition | Systems used to produce recombinant proteins in mammalian cells without stable genome integration |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, China, India, Germany, Japan, UK, South Korea, and 30+ countries |
| Key Companies Profiled | Thermo Fisher Scientific, Merck KGaA, Cytiva, Sartorius, Lonza, GenScript, Sino Biological |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology using verified demand signals, project frequency, pricing, and platform use rates |
Mammalian Transient Protein Expression Breakdown By Product Category, Application, Host, End User, Scale, and Region
Mammalian Transient Protein Expression Market Segmented by Product Category:
- Expression Vectors
- Reagents
- Competent Cells
- Instruments
Mammalian Transient Protein Expression Market Segmented by Application:
- Bio-production
- Drug Discovery
- Functional Assays
- Vaccine Research
Mammalian Transient Protein Expression Market Segmented by Expression Host:
- HEK293
- CHO
- Hybridoma
- Others
Mammalian Transient Protein Expression Market Segmented by End User:
- Biopharma Companies
- CROs
- Academic Institutes
- Research Labs
Mammalian Transient Protein Expression Market Segmented by Scale
- Micro Scale
- Small Scale
- Pilot Scale
- High Throughput
Mammalian Transient Protein Expression Market by Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- United States Bureau of Labor Statistics. (2026, April 14). Producer Price Indexes - March 2026.
- Thermo Fisher Scientific. (2025). Expi293 PRO protein expression services. Thermo Fisher Scientific.
- Sartorius. (2025, September 18). Sartorius expands production of key components for cell and gene therapies in France. Sartorius.
- Sartorius. (2026, March 3). Sartorius launches rational-design CHO host cell line with up to three-fold increase in productivity. Sartorius.
- Cytiva. (2024, April 30). Cytiva introduces new cell lines to meet the challenges of AAV production. Cytiva.
- GenScript. (2025, August 30). GenScript and Bioelectronica announce strategic collaboration to accelerate antibody discovery. GenScript.
- Fu, Y., et al. (2024). Improvement strategies for transient gene expression in mammalian cells. National Library of Medicine.
- Chavalparit, T., et al. (2025). Rapid expression of therapeutic antibodies in mammalian cells. National Library of Medicine.
- Hederoth, G., et al. (2025). Updated transient gene expression protocol in Expi293F cells. National Library of Medicine.
This bibliography is provided for reader reference and includes market pages, official company sources, and scientific publications used for article development.
This Report Answers
- What is the expected market size for mammalian transient protein expression systems in 2026 and 2036?
- What CAGR is projected for mammalian transient protein expression demand during the 2026 to 2036 period?
- Which product category is projected to lead demand in mammalian transient protein expression workflows?
- Which application area is expected to account for the largest share of transient expression spending?
- What demand factors support wider use of HEK293 and CHO transient expression systems?
- Which countries are expected to record faster expansion in mammalian transient protein expression demand?
- Which companies compete through vectors, reagents, host cells, expression systems, and protein services?
- What products and workflows are included in mammalian transient protein expression scope?
- What methodology supports the forecast for mammalian transient protein expression demand?
Frequently Asked Questions
What is the global market demand for mammalian transient protein expression in 2026?
In 2026, the global mammalian transient protein expression market is expected to be worth USD 848.5 million with steady research use.
What is the expected value of the mammalian transient protein expression market by 2036?
By 2036, the market is projected to reach USD 1,449.4 million as antibody and recombinant protein workflows expand.
What CAGR is projected for mammalian transient protein expression from 2026 to 2036?
The market is projected to expand at 5.5% CAGR between 2026 and 2036 across biopharma and contract research demand.
Which product category is projected to lead the market in 2026?
Expression vectors are projected to lead with 28.5% share in 2026 as construct quality affects yield and assay output.
Which application is expected to lead demand in 2026?
Bio-production is expected to lead with 40.0% share in 2026 due to antibody and recombinant antigen sample needs.
Which country is expected to expand fastest through 2036?
South Korea is expected to post 6.4% CAGR through 2036 as biologics investment supports advanced expression workflows.
What does mammalian transient protein expression include?
The market includes vectors and reagents used for temporary protein production. Mammalian host cells and media support the same workflow. Enhancers and small systems are included with services.
How was the mammalian transient protein expression forecast prepared?
The forecast used project frequency and reagent use. Service pricing and host selection shaped the next layer. Application mix and country-level research demand supported validation.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Primary Modes
- Desk Research Programme (Secondary Evidence)
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Category
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Category , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Category , 2026 to 2036
- Expression Vectors
- Reagents
- Competent Cells
- Y to o to Y Growth Trend Analysis By Product Category , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Category , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Bio-production
- Drug Discovery
- Functional Assays
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Expression Host
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Expression Host, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Expression Host, 2026 to 2036
- HEK293
- CHO
- Hybridoma
- Y to o to Y Growth Trend Analysis By Expression Host, 2021 to 2025
- Absolute $ Opportunity Analysis By Expression Host, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma Companies
- CROs
- Research Labs
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Scale
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Scale, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Scale, 2026 to 2036
- Small Scale
- Micro Scale
- Pilot Scale
- Y to o to Y Growth Trend Analysis By Scale, 2021 to 2025
- Absolute $ Opportunity Analysis By Scale, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Category
- By Application
- By Expression Host
- By End User
- By Scale
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Merck KGaA
- Cytiva
- Sartorius
- Lonza
- Thermo Fisher Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Scale, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Category , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Expression Host, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Scale, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Category
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Expression Host
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Scale
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Category
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Expression Host
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Scale
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Category
- Figure 48: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Application
- Figure 51: Latin America Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Expression Host
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Scale
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Category
- Figure 64: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Application
- Figure 67: Western Europe Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Expression Host
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Scale
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Category
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Application
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Expression Host
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Scale
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Category
- Figure 96: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Application
- Figure 99: East Asia Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Expression Host
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Scale
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Category
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Expression Host
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Scale
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Category , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Category , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Category
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Expression Host, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Expression Host, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Expression Host
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Scale, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Scale, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Scale
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

