Mammalian Cell Fermentation Technology Market
This report covers the Mammalian Cell Fermentation Technology market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, type trends, application analysis, end trends, supply chain developments, strategic growth opportunities.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Mammalian Cell Fermentation Technology Market Size, Market Forecast and Outlook By FMI
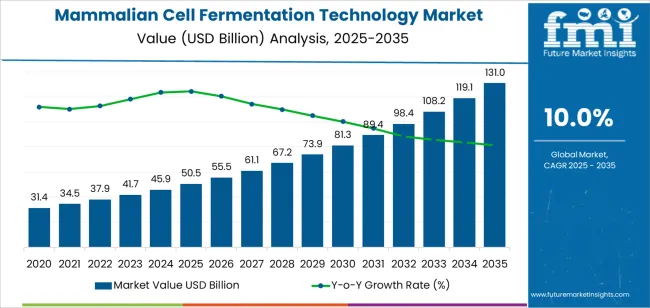
The mammalian cell fermentation technology market was valued at USD Billion 50.50 billion in 2025, projected to reach USD Billion 55.55 billion in 2026, and is forecast to expand to USD Billion 144.08 billion by 2036 at a 10.0% CAGR. CHO cell lines dominate production at 46.7% share, reflecting their established regulatory acceptance and high-titer expression capabilities for monoclonal antibody manufacturing.
Summary of Mammalian Cell Fermentation Technology Market
- Market Snapshot
- The mammalian cell fermentation technology market is valued at USD Billion 50.50 billion in 2025 and is projected to reach USD Billion 144.08 billion by 2036.
- The industry is expected to grow at a 10.0% CAGR from 2026 to 2036, creating an incremental opportunity of USD Billion 88.53 billion.
- The market is a biopharmaceutical manufacturing infrastructure category where cell line productivity, single-use bioreactor adoption, and regulatory validation requirements for biologics production define competitive positioning among bioprocess technology providers.
- Demand and Growth Drivers
- Demand is driven by the expanding global biologics pipeline, where monoclonal antibody approvals alone account for over half of mammalian cell fermentation capacity requirements.
- Single-use bioreactor adoption is accelerating as manufacturers prioritize changeover speed and cross-contamination risk reduction in multi-product facilities.
- Growth is supported by CDMO capacity expansion as biopharmaceutical companies increasingly outsource biologics manufacturing to manage pipeline diversity.
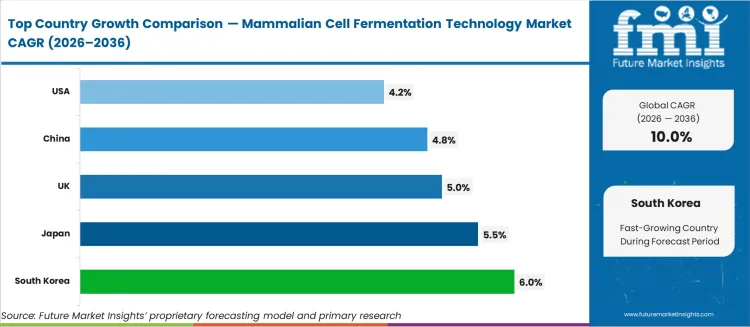
- Among key countries, South Korea at 6.0%, Japan at 5.5%, UK at 5.0%, China at 4.8%, USA at 4.2%.
- Product and Segment View
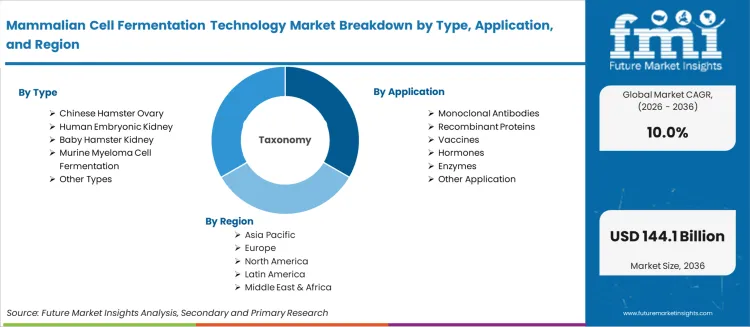
- The Mammalian Cell Fermentation Technology Market covers bioprocessing equipment, media, and consumables used for the cultivation of mammalian cell lines in biopharmaceutical manufacturing. Cell line types include Chinese Hamster Ovary (CHO), Human Embryonic Kidney (HEK), Baby Hamster Kidney (BHK), and murine myeloma systems.
- Chinese Hamster Ovary leads by Type with 46.7% share in 2026.
- Monoclonal Antibodies leads by Application with 52.4% share in 2026.
- Biopharmaceutical Companies leads by End User with 58.9% share in 2026.
- Market scope covers bioreactor systems, cell culture media, single-use consumables, and process analytical technology instruments used in mammalian cell fermentation.
- The scope excludes microbial fermentation systems, plant cell culture technologies, cell therapy manufacturing platforms, and finished biologic drug products.
- Geography and Competitive Outlook
- South Korea and Japan are the fastest-growing markets, while China and USA represent mature demand bases.
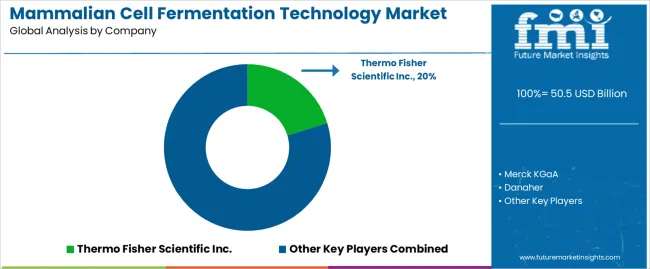
- Competition is shaped by concentrated market share among established players, with key players including Thermo Fisher Scientific Inc., Merck KGaA, Danaher, Lonza.
- Thermo Fisher Scientific Inc. holds approximately 20% market share, positioning it as the category leader.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant, opines: 'In my analysis, I have observed that the defining competitive variable in mammalian cell fermentation technology is not equipment pricing but validated supply chain continuity for single-use consumables. Biopharmaceutical manufacturers operating multi-product facilities on single-use platforms face catastrophic production risk from consumable supply disruptions. Bioprocess technology suppliers unable to guarantee multi-year consumable supply commitments backed by diversified manufacturing footprints will lose specification positions to competitors offering supply security assurances.'
- Strategic Implications / Executive Takeaways
- Bioprocess technology suppliers must establish diversified single-use consumable manufacturing footprints to provide supply continuity guarantees required by multi-product biologics facilities.
- Biopharmaceutical manufacturing directors should evaluate hybrid stainless-steel and single-use facility configurations to balance operational flexibility with supply chain resilience.
- CDMO capacity planning teams must integrate cell culture media supply agreements into facility expansion decisions to prevent consumable bottlenecks during production scale-up.
Mammalian Cell Fermentation Technology Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD Billion 55.55 billion |
| Industry Value (2036) | USD Billion 144.08 billion |
| CAGR (2026-2036) | 10.0% |
Source: Future Market Insights, 2026
FMI analysts observe a fundamental manufacturing paradigm shift underway, where fixed stainless-steel bioreactor facilities are yielding to single-use systems that reduce changeover times and cross-contamination risks. This transition is raising per-batch consumable expenditure while lowering facility capital requirements. CDMO capacity expansion is absorbing a growing share of bioprocess technology demand as biopharmaceutical companies outsource manufacturing to manage pipeline diversity without proportional capital investment.
All major markets reflect differentiated growth trajectories. South Korea at 6.0% CAGR, Japan at 5.5% CAGR, UK at 5.0% CAGR, China at 4.8% CAGR, USA at 4.2% CAGR. Each country path is shaped by local regulatory frameworks, infrastructure maturity, and industry-specific demand patterns.
Mammalian Cell Fermentation Technology Market Definition
The Mammalian Cell Fermentation Technology Market covers bioprocessing equipment, media, and consumables used for the cultivation of mammalian cell lines in biopharmaceutical manufacturing. Cell line types include Chinese Hamster Ovary (CHO), Human Embryonic Kidney (HEK), Baby Hamster Kidney (BHK), and murine myeloma systems. Applications span monoclonal antibody production, recombinant proteins, vaccines, hormones, and enzyme manufacturing across biopharmaceutical companies, CMOs/CDMOs, and academic research institutes.
Mammalian Cell Fermentation Technology Market Inclusions
Market scope covers bioreactor systems, cell culture media, single-use consumables, and process analytical technology instruments used in mammalian cell fermentation. Global and regional market sizes for the forecast period 2026 to 2036 are covered, segmented by cell line type, application, and end user.
Mammalian Cell Fermentation Technology Market Exclusions
The scope excludes microbial fermentation systems, plant cell culture technologies, cell therapy manufacturing platforms, and finished biologic drug products. Laboratory-scale cell culture consumables for non-manufacturing research are also excluded.
Mammalian Cell Fermentation Technology Market Research Methodology
- Primary Research: Analysts engaged with biopharmaceutical manufacturing directors, bioprocess engineering leads, and CDMO capacity planning managers across major biologics production regions.
- Desk Research: Data collection included biologics manufacturing capacity databases, FDA and EMA biologic drug approval pipelines, and bioprocessing industry association production statistics.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of global biologics manufacturing capacity data, applying single-use technology penetration rates and cell culture media consumption volumes to project bioprocessing expenditure through 2036.
- Data Validation and Update Cycle: Projections are cross-validated against publicly reported bioprocessing revenue data from leading life science tools companies and biopharmaceutical manufacturer capital expenditure disclosures.
Mammalian Cell Fermentation Technology Market Growth Drivers
- The rising prevalence of chronic diseases, such as cancer, autoimmune disorders, and metabolic diseases, has led to a growing demand for biopharmaceuticals.
- Mammalian cell fermentation technology is crucial for producing complex biologics like monoclonal antibodies and recombinant proteins, driving market growth.
- Investments in R&D to develop innovative therapies like developing new cell lines, optimizing culture media formulations, and enhancing bioreactor designs, all of which contribute to the advancement of mammalian cell fermentation technology.
- Mammalian cell fermentation technology enables the production of personalized therapeutics, driving its adoption in this emerging field and contributing to market growth.
Mammalian Cell Fermentation Technology Market Key Challenges
- Scaling up mammalian cell fermentation from laboratory to commercial production levels presents numerous technical challenges.
- Maintaining consistent cell growth, productivity, and product quality across larger bioreactors while minimizing the risk of contamination and process variability requires careful optimization and validation.
- Mammalian cell fermentation is typically more expensive than microbial fermentation due to higher culture media costs, longer production times, and the need for specialized equipment and facilities.
- The global supply chain for cell culture media, reagents, and raw materials used in mammalian cell fermentation is complex and can be vulnerable to disruptions, such as shortages or price fluctuations. Ensuring a reliable supply of high-quality materials is essential for uninterrupted biopharmaceutical production.
Country-wise Analysis
The provided table highlights the top five countries in terms of revenue, with Japan and South Korea leading the list. Japan and South Korea have been focusing on the development of innovative therapies to address unmet medical needs, particularly in areas such as oncology, immunology, and rare diseases.
Mammalian cell fermentation technology plays a crucial role in the production of these advanced biologics, driving market growth in both countries.
.webp)
| Countries | Forecast CAGRs from 2026 to 2036 |
|---|---|
| The United States | 4.2% |
| The United Kingdom | 5.0% |
| China | 4.8% |
| Japan | 5.5% |
| South Korea | 6.0% |
The Mammalian Cell Fermentation Technology Market in United States Propelled by the Increasing Demand for Biopharmaceuticals
The mammalian cell fermentation technology market in the United States is expected to expand with a CAGR of 4.2% from 2026 to 2036. The United States has a high demand for biologic drugs to treat various diseases, including cancer, autoimmune disorders, and infectious diseases. Mammalian cell fermentation technology is essential for producing these biopharmaceuticals, driving market growth.
The United States biopharmaceutical industry invests heavily in research and development to develop innovative therapies and improve manufacturing processes. Investments in cell line engineering, process optimization, and bioprocessing technologies contribute to the growth of the mammalian cell fermentation technology market.
Government Support Contribute to the Growth of the Mammalian Cell Fermentation Technology Market in China
The mammalian cell fermentation technology market in China is expected to expand with a CAGR of 4.8% from 2026 to 2036. China's biopharmaceutical industry has experienced rapid growth in recent years, fueled by government initiatives, investments in research and development, and a growing demand for innovative therapies.
The Chinese government has implemented policies to support the development of the biopharmaceutical sector, including incentives for research, investment, and innovation. Government funding and support for biotechnology infrastructure and talent development contribute to the growth of the mammalian cell fermentation technology market.
Leading global companies in the mammalian cell fermentation technology market, such as Thermo Fisher Scientific, Merck KGaA, and Lonza, are expanding their presence in China. Partnerships, collaborations, and investments by these companies contribute to market growth and technology transfer in the country.
Thriving Biopharmaceutical Industry Drives Market in United Kingdom
The mammalian cell fermentation technology market in United Kingdom is expected to expand with a CAGR of 5.0% from 2026 to 2036. The United Kingdom has a strong biopharmaceutical sector, with a well-established ecosystem of pharmaceutical companies, research institutions, and academic centers.
The demand for biologic drugs in the UK, driven by an aging population and increasing prevalence of chronic diseases, fuels growth in mammalian cell fermentation technology.
The United Kingdom government has been actively promoting investment in life sciences and biotechnology through funding initiatives, tax incentives, and research grants. This support encourages innovation and stimulates growth in the mammalian cell fermentation technology market.
Advancements in Biotechnology to Spur the Market Demand in Japan
The mammalian cell fermentation technology market in Japan is expected to expand with a CAGR of 5.5% from 2026 to 2036.
Japan is known for its expertise in biotechnology and has made significant advancements in cell line engineering, process optimization, and bioprocessing technologies. These advancements have led to improvements in productivity, scalability, and cost-effectiveness in mammalian cell fermentation, driving market growth.
The Japanese government has implemented initiatives to promote biotechnology and innovation in healthcare, including funding for research and development in the biopharmaceutical sector. Investments in infrastructure, technology transfer, and talent development have contributed to the growth of the mammalian cell fermentation technology market.
Rising Demand for Biologic Drugs to Drive Market Growth in South Korea
South Korea is home to world-class research institutions and universities with expertise in biotechnology and cell culture technologies. Collaborations between academia and industry facilitate technology transfer, innovation, and skill development in mammalian cell fermentation. The mammalian cell fermentation technology market in South Korea is expected to expand with a CAGR of 6.0% from 2026 to 2036.
The increasing prevalence of chronic diseases, aging population, and changing healthcare landscape in South Korea have driven the demand for biologic drugs. Mammalian cell fermentation technology enables the production of high-quality biologics such as monoclonal antibodies and recombinant proteins to address these healthcare needs.
Category-wise Insights
The below section shows the leading segment. The Chinese Hamster Ovary (CHO) Cell Fermentation segment is to rise at a CAGR of 3.8% from 2026 to 2036. Based on application, the monoclonal antibodies segment is anticipated to hold a dominant share through 2036. It is set to exhibit a CAGR of 3.6% from 2026 to 2036.
| Category | CAGR from 2026 to 2036 |
|---|---|
| Chinese Hamster Ovary (CHO) Cell Fermentation | 3.8% |
| Monoclonal Antibodies | 3.6% |
Chinese Hamster Ovary (CHO) Cell Fermentation Emerges as a Significant Segment in the Market
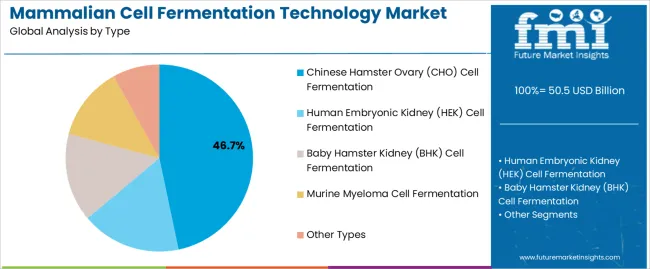
Based on the type, the Chinese Hamster Ovary (CHO) Cell Fermentation segment is anticipated to thrive at a CAGR of 3.8% from 2026 to 2036. CHO cells are widely used for biopharmaceutical production because they have the machinery to properly fold and post-translationally modify complex proteins, making them suitable for producing human therapeutic proteins like monoclonal antibodies and recombinant proteins.
CHO cells have a long history of use in biopharmaceutical manufacturing and are well-characterized in terms of safety, stability, and regulatory acceptance. Their established safety profile reduces regulatory hurdles and accelerates the development and approval of biologic drugs produced using CHO cell fermentation.
CHO cells can be adapted to grow in serum-free or chemically defined media, which eliminates the risk of introducing contaminants from animal-derived components and simplifies downstream purification processes. Serum-free media also enhance product consistency and facilitate regulatory compliance.
Monoclonal Antibodies Segment is expected to Expand at the Fastest CAGR over the Forecast Period
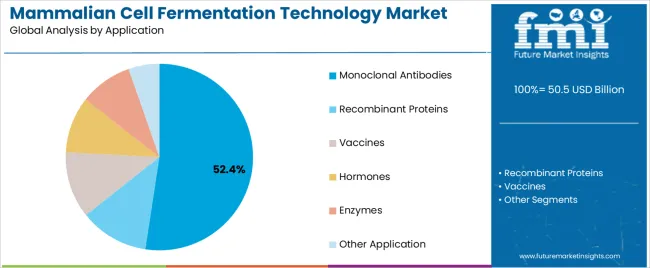
Based on application, the monoclonal antibodies segment is anticipated to thrive at a CAGR of % from 2026 to 2036. Monoclonal antibodies are highly effective in treating a wide range of diseases, including cancer, autoimmune disorders, and infectious diseases. Their specificity for target antigens allows for precise targeting of disease pathways, resulting in potent therapeutic effects with minimal off-target effects.
Monoclonal antibodies have a broad spectrum of therapeutic applications, including cancer immunotherapy, autoimmune disease management, and infectious disease treatment. Their versatility makes them valuable tools in addressing various unmet medical needs, driving demand for their production through mammalian cell fermentation.
Ongoing advancements in mammalian cell culture techniques, cell line engineering, and bioprocessing technologies have further enhanced the productivity, yield, and cost-effectiveness of monoclonal antibody production through mammalian cell fermentation. These technological innovations drive continued growth and innovation in the field.
Competitive Landscape
Leading companies allocate significant resources to research and development efforts aimed at enhancing fermentation processes, improving cell line productivity, and developing novel technologies. Investing in R&D enables companies to stay ahead of technological advancements and address evolving market demands.
- In July 2025, Lonza introduced the TheraPRO® CHO Media System, a cutting-edge cell culture platform designed to enhance productivity and protein quality in conjunction with GS-CHO cell lines. This innovative launch caters to the needs of pharmaceutical and biotechnology firms producing therapeutic proteins, aiming to elevate product quality and accelerate time-to-market through streamlined processes.
- In the mammalian cell fermentation technology market, Thermo Fisher offers a comprehensive portfolio of bioprocessing solutions, including bioreactors, cell culture media, purification systems, and analytical instruments. The company focuses on innovation, continuously developing new technologies and improving existing products to meet the evolving needs of biopharmaceutical manufacturers.
- Merck KGaA, known as MilliporeSigma in the United States and Canada, is a leading supplier of life science tools, technologies, and services. The company offers a wide range of products for mammalian cell fermentation, including cell culture media, cell culture supplements, single-use bioreactors, and purification technologies.
Key Coverage in Precision Plating Market Report
- Demand for Mammalian Cell Fermentation Technology in the United Kingdom
- Strategies of Key Players of the Mammalian Cell Fermentation Technology Market
- Demand for Mammalian Cell Fermentation Technology in East Asia
- Recent Advances in Mammalian Cell Fermentation Technology Market
- Promising Opportunities in Mammalian Cell Fermentation Technology in the Middle East and Africa Market
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD Billion 55.55 to USD Billion 144.08 billion, at a CAGR of 10.0% |
| Market Definition | The Mammalian Cell Fermentation Technology Market covers bioprocessing equipment, media, and consumables used for the cultivation of mammalian cell lines in biopharmaceutical manufacturing. Cell line types include Chinese Hamster Ovary (CHO), Human Embryonic Kidney (HEK), Baby Hamster Kidney (BHK), and murine myeloma systems. |
| Segmentation | Type: Chinese Hamster Ovary, Human Embryonic Kidney, Baby Hamster Kidney, Murine Myeloma Cell Fermentation, Other Types; Application: Monoclonal Antibodies, Recombinant Proteins, Vaccines, Hormones, Enzymes, Other Application; End User: Biopharmaceutical Companies, CMOs & CDMOs, Academic & Research Institutes |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | South Korea, Japan, UK, China, USA, and 40 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific Inc., Merck KGaA, Danaher, Lonza |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline values derive from a bottom-up aggregation of global biologics manufacturing capacity data, applying single-use technology penetration rates and cell culture media consumption volumes to project bioprocessing expenditure through 2036. |
Mammalian Cell Fermentation Technology Market - Key Segments
By Type:
- Chinese Hamster Ovary (CHO) Cell Fermentation
- Human Embryonic Kidney (HEK) Cell Fermentation
- Baby Hamster Kidney (BHK) Cell Fermentation
- Murine Myeloma Cell Fermentation
- Other Types
By Application:
- Monoclonal Antibodies
- Recombinant Proteins
- Vaccines
- Hormones
- Enzymes
- Other Application
By End Use:
- Biopharmaceutical Companies
- CMOs & CDMOs
- Academic & Research Institutes
By Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- South Asia and Pacific
- East Asia
- The Middle East and Africa
Bibliography
- 1. USA Food and Drug Administration. (2024). Biologics license application approval statistics. FDA.
- 2. European Medicines Agency. (2024). Advanced therapy medicinal products manufacturing guidance. EMA.
- 3. International Society for Pharmaceutical Engineering. (2024). ISPE baseline guide for biopharmaceutical manufacturing facilities. ISPE.
- 4. Thermo Fisher Scientific Inc. (2025). 2024 annual report. Thermo Fisher Scientific.
- 5. Organisation for Economic Co-operation and Development. (2024). Biotechnology industry statistics report. OECD.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Mammalian Cell Fermentation Technology Market in the global market in 2026?
Demand for the mammalian cell fermentation technology market in the global market is estimated to be valued at USD Billion 55.55 billion in 2026.
What will be the market size of Mammalian Cell Fermentation Technology Market by 2036?
Market size for the mammalian cell fermentation technology market is projected to reach USD Billion 144.08 billion by 2036.
What is the expected demand growth for Mammalian Cell Fermentation Technology Market between 2026 and 2036?
Demand for the mammalian cell fermentation technology market is expected to grow at a CAGR of 10.0% between 2026 and 2036.
Which Type segment is poised to lead by 2026?
Chinese Hamster Ovary accounts for 46.7% of type share in 2026.
Which Application segment is poised to lead by 2026?
Monoclonal Antibodies accounts for 52.4% of application share in 2026.
Which End User segment is poised to lead by 2026?
Biopharmaceutical Companies accounts for 58.9% of end user share in 2026.
What is driving demand in South Korea?
South Korea is projected to grow at a CAGR of 6.0% during 2026 to 2036, supported by local industry expansion and regulatory developments.
What is Mammalian Cell Fermentation Technology Market and what is it mainly used for?
The Mammalian Cell Fermentation Technology Market covers bioprocessing equipment, media, and consumables used for the cultivation of mammalian cell lines in biopharmaceutical manufacturing. Cell line types include Chinese Hamster Ovary (CHO), Human Embryonic Kidney (HEK), Baby Hamster Kidney (BHK), and murine myeloma systems.
How does FMI build and validate the Mammalian Cell Fermentation Technology Market forecast?
Baseline values derive from a bottom-up aggregation of global biologics manufacturing capacity data, applying single-use technology penetration rates and cell culture media consumption volumes to project bioprocessing expenditure through 2036. Projections are cross-validated against publicly reported bioprocessing revenue data from leading life science tools companies and biopharmaceutical manufacturer capital expenditure disclosures.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Chinese Hamster Ovary
- Human Embryonic Kidney
- Baby Hamster Kidney
- Murine Myeloma Cell Fermentation
- Other Types
- Chinese Hamster Ovary
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Monoclonal Antibodies
- Recombinant Proteins
- Vaccines
- Hormones
- Enzymes
- Other Application
- Monoclonal Antibodies
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End, 2026 to 2036
- Biopharmaceutical Companies
- CMOs & CDMOs
- Academic & Research Institutes
- Biopharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End, 2021 to 2025
- Absolute $ Opportunity Analysis By End, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By End
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- By End
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Merck KGaA
- Danaher
- Lonza
- Thermo Fisher Scientific Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Type
- Figure 26: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Application
- Figure 29: North America Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Application
- Figure 39: Latin America Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Application
- Figure 49: Western Europe Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Application
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Application
- Figure 69: East Asia Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

