Cell Culture Media Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Cell Culture Media Market is segmented by Type (Serum-free Media, Classical Media, Stem Cell Media, Specialty Media, Chemically Defined Media, Other Media), Application(Cancer Research, Biopharmaceutical Production, Monoclonal Antibodies, Vaccine Production, Diagnostics, Drug Screening, Cell Therapy), End User, Form, Cell Type, Sales Channel, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Cell Culture Media Market Size, Market Forecast and Outlook By FMI
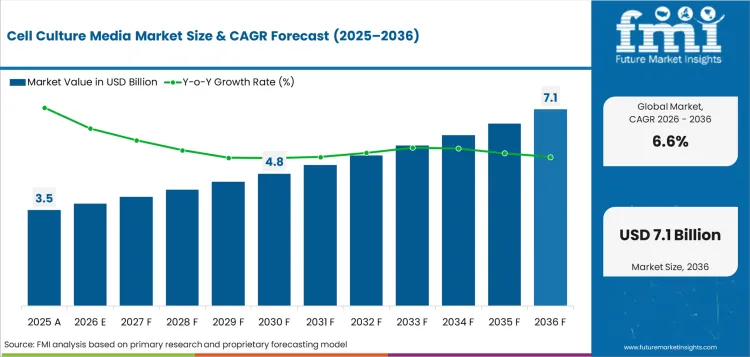
The cell culture media market was valued at USD 3.47 billion in 2025 and is projected to reach USD 3.70 billion in 2026. It is forecast to reach USD 7.10 billion by 2036 at 6.6% CAGR. Serum-free media is projected to lead the type segment with 51.0% share. Cancer research is expected to hold 43.4% share by application in 2026.
Defined formulation and production control are the main shifts behind cell culture media demand. Drug makers are reducing reliance on animal serum because regulated biologics production needs repeatable cell performance. FDA's Center for Drug Evaluation and Research approved 46 novel drugs in 2025. That pipeline activity keeps media supply connected to biologics and cell therapy work. Oncology work adds a separate research route. Europe and Asia add separate production checks to the forecast.
The European Medicines Agency recommended 114 human medicines for authorization in 2024. India reported a USD 165.7 billion bioeconomy in 2024. Those figures show why media suppliers must plan for GMP-grade output and regional supply assurance.
Summary of the Cell Culture Media Market
- Demand and Growth Drivers
- Cell culture media use is rising as biologics producers need repeatable cell growth in controlled manufacturing runs.
- Serum-free media is gaining share as it removes animal-derived variability from regulated production work.
- Cancer research leads applications due to rise in oncology studies that need stable cell line performance.
- Cell and gene therapy work is raising the need for documented GMP media and supplier quality files.
- Product and Segment View
- Serum-free media is projected to account for 51.0% share in 2026 as producers standardize controlled formulas.
- Cancer research is expected to represent 43.4% share in 2026 because oncology work uses many cell models.
- Liquid media is estimated to account for 62.0% share in 2026 as it supports ready use in routine labs.
- Mammalian cell lines are estimated to account for 68.0% share since antibody and protein production use them widely.
- Geography and Competitive Outlook
- China is expected to post 8.5% CAGR through 2036 as biologics review timelines and domestic production improve.
- India is forecast at 8.2% CAGR through 2036 as bioeconomy expansion lifts media demand in research and production.
- Merck KGaA and Thermo Fisher Scientific shape supplier expectations through GMP files and broad media portfolios.
- Sartorius and FUJIFILM Biotechnologies raise linked demand through bioprocess capacity and CDMO expansion.
- Analyst Opinion
- Sabyasachi Ghosh is Principal Consultant at FMI and says, “I do not view cell culture media as a routine lab input anymore. The purchase decision is moving toward batch record support and animal-component control. Regional supply assurance is becoming part of the same choice. Suppliers that can prove media consistency across research and pilot work will gain trust faster. GMP production proof decides broader supply roles.”
- Cell Culture Media Market Value Analysis
- The market is moving from research-led volume toward production-grade media used in biologics and cell therapy work.
- Serum-free media carries the main value share because it supports more repeatable outcomes in regulated production.
- Pricing varies by documentation level and custom formulation work. Product form also affects cost when buyers compare liquid and dry powder media.
- Bulk media demand rises when CDMOs add mammalian cell culture capacity and need repeat supply across client programs.
Cell Culture Media Market Definition
The cell culture media market covers nutrient mixtures used to grow and maintain cells outside the body. Scope includes basal media and serum-free media used in research or production. Chemically defined media and stem cell media are also included. Revenue includes liquid media and dry powder media supplied to laboratories. Custom media supplied to drug developers and biomanufacturing sites is included.
Cell Culture Media Market Inclusions
Market scope includes media used for cancer research and biologics production. It also covers monoclonal antibodies and vaccine production. Diagnostics and drug screening are included. Tissue engineering and cell therapy are also included. It includes media sold with related cell culture supplements where the media component is separately identified. It also includes media used with 3D cell culture systems and cell line development services when clone screening shifts into media selection. Balanced salt solutions are included when sold as a defined support input for mammalian cell work.
Cell Culture Media Market Exclusions
Scope does not include fetal bovine serum sold as a standalone product or laboratory plasticware. General reagents and instruments are excluded. It excludes bioreactors and cell culture incubators unless the media value is stated separately. Cell counting systems are excluded. Cell culture waste aspirators are also excluded when sold as standalone laboratory equipment. It also excludes food microbiology culture media and diagnostic agar plates that are not used for mammalian or therapeutic cell culture.
Cell Culture Media Market Research Methodology
- Primary Research: FMI analysts reviewed supplier portfolios and CDMO needs. Pharma user feedback and documentation needs were checked across regulated production settings.
- Desk Research: The study used public data from regulators and government bodies. Company releases and official life science statistics were checked separately.
- Market sizing and forecasting: The market size was estimated using top-down application demand and bottom-up media use across research and production sites.
- Data validation: Forecast checks compared media shares and biologics approvals. CDMO capacity additions and country bioeconomy signals were reviewed separately. Supplier quality certifications were checked in the same validation stage.
Why is the Cell Culture Media Market Growing?
- Biologics manufacturers are moving toward defined media to reduce variation between cell culture runs.
- Cancer research keeps media consumption high because oncology studies rely on stable cell line models.
- Cell therapy workflows need GMP-grade media with documentation that can move from clinical trials to production.
Cell culture media use is expanding as biologics production depends on cells that behave predictably. Automated cell culture systems increase the need for media that can move through repeat handling steps. Serum-based formulas can introduce variation that affects cell yield and quality checks. As per FMI, this makes serum-free and chemically defined media more important in regulated manufacturing.
Production capacity is another route for demand. FUJIFILM Biotechnologies announced an additional USD 1.2 billion investment in North Carolina in April 2024. The plan adds eight 20,000-liter mammalian cell culture bioreactors by 2028 and raises media consumption tied to large production runs.
Market Segmentation Analysis
- Serum-free media is projected to reach 51.0% share in 2026 as producers reduce animal-derived inputs.
- Cancer research is expected to represent 43.4% share in 2026 because oncology work uses many cellular models.
- Pharmaceutical and biotechnology companies are estimated to account for 52.0% share in 2026 through biologics and cell therapy work.
- Liquid media is estimated to account for 62.0% share in 2026 because labs need ready-to-use formats.
- Mammalian cell lines are estimated to account for 68.0% share in 2026 because antibody production uses CHO and HEK cell systems.
- Direct supplier sales are estimated to account for 56.0% share in 2026 as regulated buyers prefer direct quality support.
The market for cell culture media is divided into six primary segment groups based on type and application. End user and form explain buyer behavior. Cell type and sales channel show how media is specified. Culture media preparators affect high-volume labs because prepared media quality shapes daily cell work. Type explains formulation choice. Application shows where the media is consumed. End user and sales channel explain who buys the product and how suppliers support quality files.
Insights into the Serum-free Media Type Segment
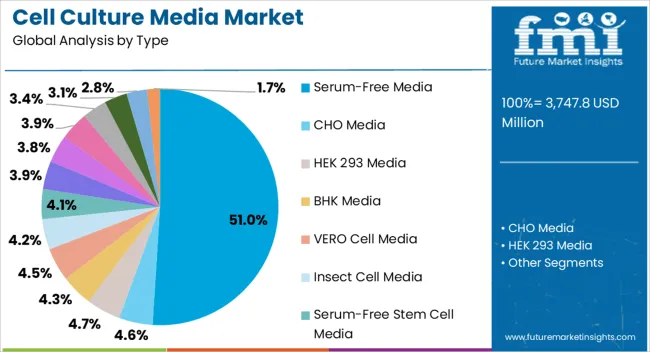
- Serum-free media is projected to account for 51.0% share of type demand in 2026. Producers use it because animal-component control lowers batch variation and supports cleaner quality records.
- Chemically defined media is gaining use in production programs that need precise ingredient lists. Classical media stays relevant in academic work and early screening.
Insights into the Cancer Research Application Segment
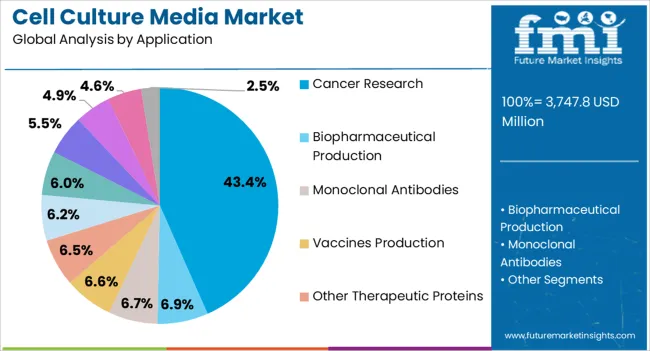
- Cancer research is expected to represent 43.4% share of application demand in 2026. Oncology studies need tumor cell lines and immune cell models. Culture systems must support repeat testing across many lab runs.
- Drug screening and development stays close behind because cell models help teams test drug response before animal work. Cell isolation workflows support this step when primary cells are used.
Insights into the Pharmaceutical and Biotechnology Companies End User Segment
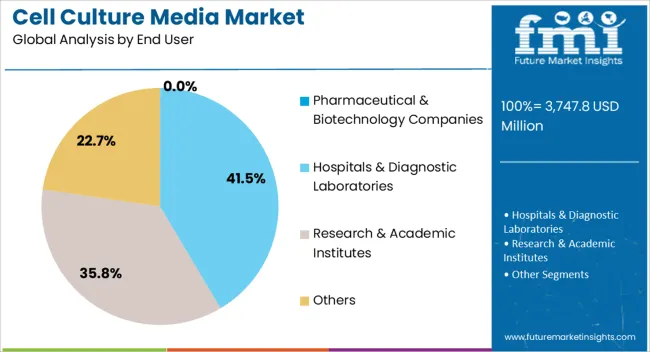
- Pharmaceutical and biotechnology companies are estimated to account for 52.0% share of end-user demand in 2026. Their buying is tied to biologics development and clinical batches. Production scale-up adds larger repeat orders.
- Research institutes buy many smaller lots for discovery work. Hospitals and diagnostic laboratories use media in more controlled volumes linked with testing and clinical research.
Insights into the Liquid Media Form Segment
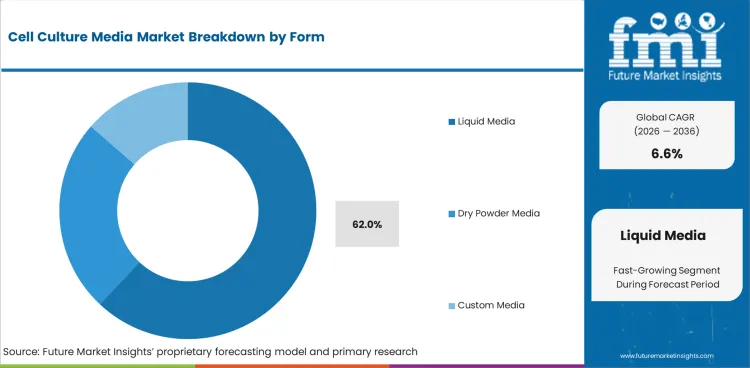
- Liquid media is estimated to account for 62.0% share of form demand in 2026. Routine users prefer ready-to-use formats because preparation steps and mixing errors are reduced.
- Dry powder formats matter in high-volume supply because freight and storage needs are lower. Dehydrated culture media becomes more relevant when buyers operate many production batches.
Insights into the Mammalian Cell Lines Cell Type Segment
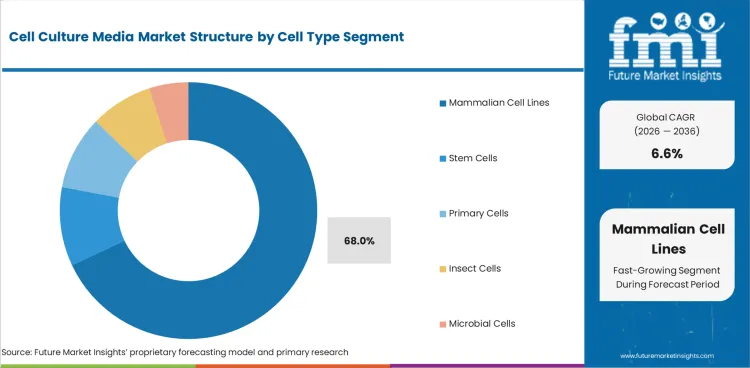
- Mammalian cell lines are estimated to account for 68.0% share of cell type demand in 2026. Antibody and protein production relies on CHO and HEK cell systems. Related mammalian systems add further media use.
- Stem cells and primary cells need more specialized formulas. Pure suspension cell culture medium supports large batch work where cells grow without attachment surfaces. Cell freezing media links with storage programs after cells are grown and banked.
Insights into the Direct Supplier Sales Channel Segment
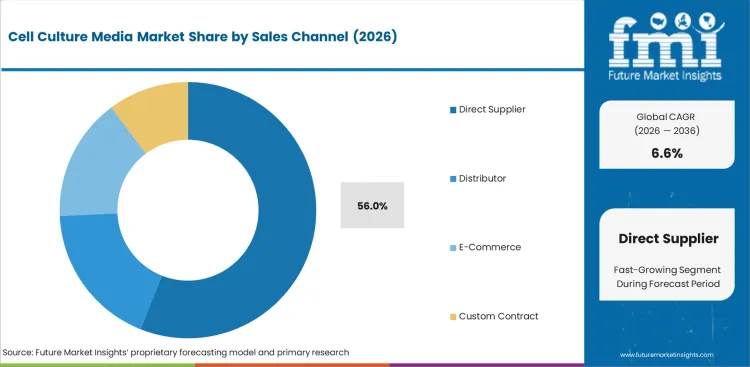
- Direct supplier sales are estimated to account for 56.0% share of channel demand in 2026. Regulated buyers prefer direct access to certificates and change control notes. Technical support helps when media enters production runs.
- Distributors remain useful for smaller research orders and urgent replacement stock. 3D cell culture plasticware adds related pull when labs shift from flat cultures to tissue-like cell models.
Cell Culture Media Market Drivers, Restraints, and Opportunities
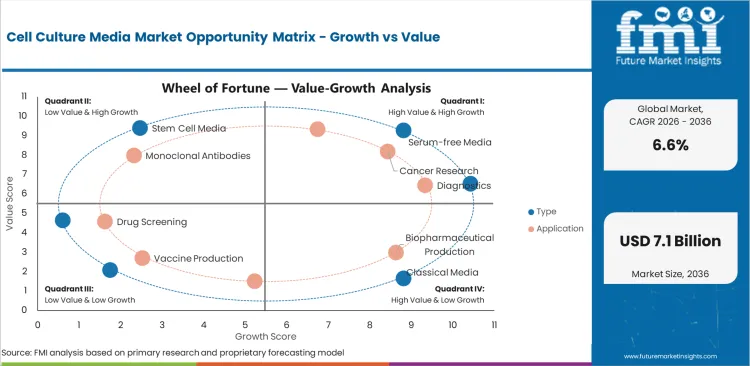
- Defined-media demand is raising share for serum-free and chemically defined formats in regulated cell production.
- Switching cost slows replacement of established formulas because users must revalidate cell performance after a change.
- CDMO scale creates opportunity for bulk media suppliers with GMP records and regional production support.
Media choice is moving closer to the manufacturing decision because each formula affects cell yield and purity. Process repeatability is part of the same decision. The European Medicines Agency recommended 114 human medicines for authorization in 2024. This reinforces the need for inputs that can support drug quality files as production moves forward.
Validation remains is one of the main restraint in the market. A media change can alter growth rate and cell viability. Protein yield can change after the same switch. Buyers will not switch suppliers unless the new source can support testing and documentation. Stable supply over repeat batches is also needed.
Analysis of Cell Culture Media Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| United States | 7.1% CAGR |
| China | 8.5% CAGR |
| India | 8.2% CAGR |
| Germany | 7.4% CAGR |
| Japan | 7.7% CAGR |
| South Korea | 7.9% CAGR |
| United Kingdom | 7.0% CAGR |
Source: Future Market Insights, 2026.
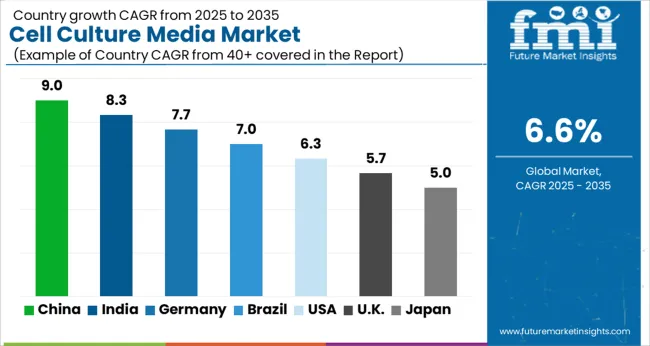
Cell Culture Media Market CAGR Analysis by Country
- China is expected to post 8.5% CAGR by 2036 as biologics review reform and domestic supply raise production needs.
- India is forecast at 8.2% CAGR by 2036 as a larger bioeconomy lifts media use in research and manufacturing.
- South Korea is projected to rise at 7.9% CAGR by 2036 as cell culture media production capacity expands locally.
- Japan is forecast to grow at 7.7% CAGR by 2036 as antibody and regenerative medicine work sustains GMP media demand.
- Germany and the United States remain core quality markets through supplier certification and drug development depth.
Demand for cell culture media is forecast to rise at 6.6% CAGR from 2026 to 2036. Country-level analysis covers the markets where biologics manufacturing and research depth shape media demand. Supplier presence and regulatory change explain country differences.
Demand Outlook for Cell Culture Media in the United States
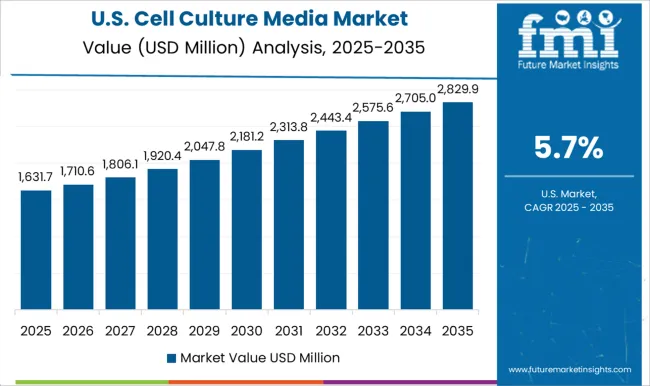
Drug development volume is estimated to keep the United States central to media demand. The United States is projected to record a 7.1% CAGR through 2036, supported by biologics pipelines and cell therapy trials. FDA's Center for Drug Evaluation and Research approved 46 novel drugs in 2025. Suppliers with USA technical support can respond faster when buyers need formulation help during scale-up.
- North Carolina biomanufacturing sites need bulk media for mammalian cell culture capacity additions.
- Massachusetts research clusters create repeat demand for oncology and cell therapy media.
- California cell therapy companies need GMP media files before clinical manufacturing starts.
Sales Analysis of Cell Culture Media in China
China is changing the review path for drug and medical device programs. The National Medical Products Administration said pilot programs can shorten clinical trial review from 60 working days to 30 working days. China is expected to expand at 8.5% CAGR through 2036 as faster reviews raise demand for media used in biologics and cell therapy work.
- Shanghai drug developers need defined media for oncology and antibody work.
- Suzhou and Hangzhou manufacturing clusters create demand for bulk liquid and dry powder media.
- Guangdong cell therapy work favors suppliers that can support clinical trial documentation.
Demand Outlook for Cell Culture Media in India
India is building a wider life science base beyond low-cost services. India is forecast to grow at 8.2% CAGR through 2036 as its bioeconomy expands into biologics and cell therapy inputs. The Press Information Bureau reported India’s bioeconomy at USD 165.7 billion in 2024. Media suppliers that offer training and local stock can reduce delays for smaller laboratories.
- Hyderabad biologics developers need serum-free media for antibody and vaccine process work.
- Pune and Bengaluru research centers use liquid media for screening and cell line work.
- Ahmedabad and Gujarat biomanufacturing projects create demand for qualified media distributors.
Opportunity Analysis of Cell Culture Media in Germany
Supplier quality systems have a direct effect on German buying decisions. Germany is expected to advance at 7.4% CAGR through 2036 as certified media production supports pharma and biotech users. Merck received EXCiPACT cGMP certification for cell culture media manufacturing in September 2024. The certification covered sites in Germany and the United Kingdom. China and the United States were covered under the same certification.
- Darmstadt remains important because Merck links media production with quality files.
- Munich and Heidelberg research clusters need media for translational research and biologics work.
- German buyers compare change-control support before they approve new media suppliers.
Future Outlook for Cell Culture Media in Japan
Japan’s media demand is linked with careful production programs and regenerative medicine work. FUJIFILM completed its first antibody drug manufacturing plant at Toyama Second Factory in December 2025. Japan is forecast at 7.7% CAGR through 2036 as new antibody capacity uses mammalian cell culture media. The plant includes two 5,000-liter mammalian cell culture bioreactors. It also includes two 2,000-liter mammalian cell culture bioreactors.
- Toyama biomanufacturing capacity raises demand for production-grade media.
- Tokyo research hospitals use specialty media for cell therapy and translational work.
- Osaka drug developers need supplier support when formulas move from lab testing to GMP batches.
Sales Analysis of Cell Culture Media in South Korea
Local production is becoming more important to South Korean buyers. South Korea is projected to rise at 7.9% CAGR through 2036 as media supply and bioprocess capacity move closer to drug manufacturers. Merck announced more than EUR 300 million for a Daejeon production center in March 2024. The site includes dry powder cell culture media and process liquid capability.
- Daejeon gives suppliers a local route for dry powder media and process liquids.
- Songdo biomanufacturing capacity supports demand for bulk media and single-use handling.
- Seoul research centers need documented media for oncology and cell therapy programs.
Future Outlook for Cell Culture Media in the United Kingdom
The United Kingdom has a dense life science base that supports repeat media purchasing. Biopharmaceutical companies generated GBP 98.9 billion in turnover in 2023 and 2024. The United Kingdom is expected to post 7.0% CAGR through 2036 as research and production sites use GMP-grade media.
- Cambridge and Oxford laboratories create demand for stem cell and oncology media.
- Teesside biomanufacturing capacity adds demand for large-volume cell culture inputs.
- London research buyers need suppliers that can handle documentation and cold-chain delivery.
Competitive Landscape and Strategic Positioning
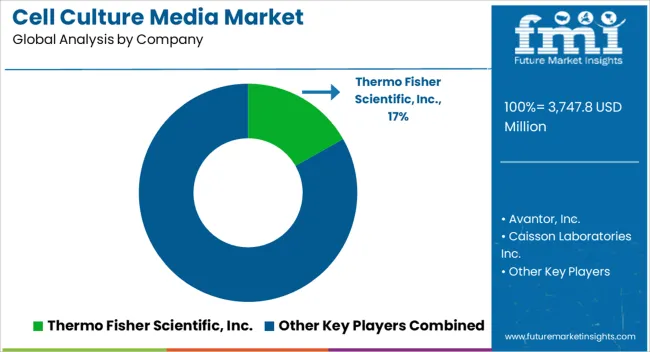
- The market is organized around formula breadth and GMP documentation. Regional supply reliability is a separate purchase filter.
- Large suppliers compete by linking media with supplements and process liquids. Quality support decides whether buyers approve repeat orders.
- Specialist suppliers compete where custom media or cell-specific formulas matter more than scale.
- Entry barriers include sterile production and change control. Raw material traceability and buyer validation time raise the barrier further.
Key players such as Merck KGaA is shaping supplier expectations through cell culture media certification and regional production investment. Its September 2024 EXCiPACT certification covers media manufacturing sites in four countries. Buyers using regulated production can treat this as a signal of repeatable quality documentation.
Thermo Fisher Scientific remains important through Gibco media and supplements. Cell therapy workflow tools add another route for supplier contact. In January 2025, the company highlighted additions to its Gibco CTS Detachable Dynabeads platform. That activity supports demand around cell therapy workflows that also require qualified media.
Bioprocess capacity additions create a second competitive layer. FUJIFILM Biotechnologies and Sartorius are expanding production networks that consume or support cell culture inputs. Suppliers that coordinate media with upstream bioprocessing equipment and fluid transfer solutions can serve production programs with fewer handoffs.
Key Companies in the Cell Culture Media Market
Competition is best read by role because media formulation and GMP production differ sharply. Bioprocess integration adds a second layer.
- Scale and Portfolio Suppliers: Thermo Fisher Scientific and Merck KGaA hold broad portfolios across basal media and serum-free media. Cytiva and Avantor add supplements and production support.
- Bioprocess and Manufacturing Specialists: Sartorius and Lonza connect media demand with mammalian cell culture capacity. FUJIFILM Biotechnologies adds process development work.
- Specialist and Regional Suppliers: Caisson Labs and PromoCell support focused applications. Bio-Rad Laboratories and HiMedia Laboratories serve regional laboratory needs. Becton Dickinson supports related cell culture workflows.
Competitive Benchmarking: Cell Culture Media Market
| Company | GMP Media Documentation | Bulk Media Supply | Cell Therapy Support | Geographic Footprint |
|---|---|---|---|---|
| Thermo Fisher Scientific | Strong | Strong | Strong | Global with deep Gibco portfolio and cell therapy workflow reach |
| Merck KGaA | Strong | Strong | Medium | Global with certified media sites and South Korea expansion |
| Cytiva | Strong | Medium | Strong | Global with bioprocess systems and media portfolio depth |
| Avantor | Medium | Strong | Medium | Global laboratory supply and production input reach |
| Sartorius | Medium | Strong | Medium | Europe and Asia depth with bioprocess capacity additions |
| Lonza Group | Medium | Medium | Strong | Global CDMO and cell therapy process knowledge |
| FUJIFILM Biotechnologies | Medium | Strong | Medium | United States, United Kingdom, Denmark, and Japan CDMO footprint |
| Caisson Labs | Medium | Medium | Medium | United States centered specialist media supply |
| HiMedia Laboratories | Medium | Medium | Low | India centered with broad laboratory media supply |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on GMP media documentation and bulk media supply. Cell therapy formulation support is assessed separately.
Key Developments in Cell Culture Media Market
-
In September 2024, Merck KGaA received EXCiPACT cGMP certification for cell culture media manufacturing. The certification covered sites in Germany and the United Kingdom. China and the United States were covered under the same certification. The certification raises buyer confidence for production-grade media.
- In February 2026, FUJIFILM Biotechnologies unveiled a United Kingdom expansion. The site has up to 19,000 liters of total single-use bioreactor capacity. The site supports process development and biomanufacturing.
- In April 2026, FUJIFILM Biotechnologies opened a 2,000-square-meter quality control laboratory in Denmark. The site supports mammalian cell culture production and release testing.
Key Players in the Cell Culture Media Market
Major Global Players:
- Thermo Fisher Scientific
- Merck KGaA
- Cytiva
- Avantor
- Lonza Group
- Sartorius
- FUJIFILM Biotechnologies
Specialist and Regional Players:
- Caisson Labs
- PromoCell
- Bio-Rad Laboratories
- HiMedia Laboratories
- Becton Dickinson
Report Scope and Coverage

| Parameter | Details |
|---|---|
| Quantitative Units | USD 3.70 billion to USD 7.10 billion, at 6.6% CAGR |
| Market Definition | Nutrient formulations used to grow and maintain cells for research, biologics production, and therapy workflows |
| Type | Serum-free Media, Classical Media, Stem Cell Media, Specialty Media, Chemically Defined Media, Other Media |
| Application | Cancer Research, Biopharmaceutical Production, Monoclonal Antibodies, Vaccine Production, Diagnostics, Drug Screening, Cell Therapy |
| End User | Pharmaceutical and Biotechnology Companies, Hospitals and Diagnostic Laboratories, Research and Academic Institutes, Others |
| Form | Liquid Media, Dry Powder Media, Custom Media |
| Cell Type | Mammalian Cell Lines, Stem Cells, Primary Cells, Insect Cells, Microbial Cells |
| Sales Channel | Direct Supplier, Distributor, E-Commerce, Custom Contract |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, Japan, South Korea, United Kingdom, and 30 plus countries |
| Key Companies Profiled | Thermo Fisher Scientific, Merck KGaA, Cytiva, Avantor, Lonza Group, Sartorius, FUJIFILM Biotechnologies |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up model using application demand, supplier evidence, and official statistics |
Market Analysis by Segments
By Type
- Serum-free Media
- Classical Media
- Stem Cell Media
- Specialty Media
- Chemically Defined Media
- Other Media
By Application
- Cancer Research
- Biopharmaceutical Production
- Monoclonal Antibodies
- Vaccine Production
- Diagnostics
- Drug Screening
- Tissue Engineering
- Cell Therapy
By End User
- Pharmaceutical and Biotechnology Companies
- Hospitals and Diagnostic Laboratories
- Research and Academic Institutes
- Others
By Form
- Liquid Media
- Dry Powder Media
- Custom Media
By Cell Type
- Mammalian Cell Lines
- Stem Cells
- Primary Cells
- Insect Cells
- Microbial Cells
By Sales Channel
- Direct Supplier
- Distributor
- E-Commerce
- Custom Contract
By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Research Sources and Bibliography
- United States Food and Drug Administration. (2026). Novel drug approvals for 2025.
- European Medicines Agency. (2025, January 16). Human medicines in 2024.
- Press Information Bureau, Government of India. (2025, March 27). The rise of India bioeconomy.
- National Medical Products Administration. (2025, March 25). Opinions on comprehensively deepening regulatory reform for drugs and medical devices.
- Department for Science, Innovation and Technology. (2025, October 2). Bioscience and health technology sector statistics 2023 to 2024.
- Merck KGaA. (2024, March 20). Merck invests more than EUR 300 million in new life science production site in South Korea.
- Merck KGaA. (2024, September 5). Merck receives industry first EXCiPACT certification for cell culture media manufacturing.
- FUJIFILM Biotechnologies. (2024, April 11). Fujifilm to invest additional USD 1.2 billion to expand its large-scale cell culture CDMO business in North Carolina.
- FUJIFILM Biotechnologies. (2026, February 11). FUJIFILM Biotechnologies unveils biomanufacturing and process development expansion in the United Kingdom.
- FUJIFILM Biotechnologies. (2026, April 15). FUJIFILM Biotechnologies celebrates the opening of its new quality control laboratory in Denmark.
- Thermo Fisher Scientific Inc. (2025, January 30). Thermo Fisher Scientific reports fourth quarter and full year 2024 results.
- FUJIFILM Corporation. (2025, December 23). Fujifilm celebrates the completion of one of Japan’s largest bio CDMO facilities.
This Report Answers
- What is the expected value of the cell culture media market by 2036?
- Which type segment accounts for the largest share of cell culture media demand?
- How does cancer research affect cell culture media consumption?
- Which countries have higher CAGR outlook than the global average?
- Which suppliers are best placed for GMP-grade media demand?
- What restraints slow switching between media suppliers?
- How do CDMO capacity additions affect bulk media supply planning?
Frequently Asked Questions
What is the cell culture media market size in 2026?
The cell culture media market is estimated at USD 3.70 billion in 2026 and is projected to reach USD 7.10 billion by 2036.
What CAGR is expected for the cell culture media market?
The cell culture media market is projected to expand at 6.6% CAGR from 2026 to 2036 based on FMI analysis.
Which type leads the cell culture media market?
Serum-free media leads the cell culture media market with 51.0% share in 2026 because regulated production needs repeatable formulas.
Which application leads the cell culture media market?
Cancer research leads the cell culture media market with 43.4% share in 2026 because oncology studies use many cell models.
Which country grows fastest in the cell culture media market?
China grows fastest in the cell culture media market at 8.5% CAGR through 2036 because biologics review timelines are shortening.
Who are the key suppliers in the cell culture media market?
Thermo Fisher Scientific and Merck KGaA are key suppliers in this market. Cytiva and Avantor also compete in this market. Lonza and Sartorius compete through linked production roles. FUJIFILM Biotechnologies competes through CDMO capacity.
What does the cell culture media market include?
The cell culture media market includes basal media and serum-free media. It also covers stem cell media and custom formulations.
How does FMI estimate the cell culture media market forecast?
FMI estimates the forecast through application demand checks, supplier evidence, biologics approvals, CDMO capacity, and country-level bioeconomy indicators.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Serum-free Media
- Classical Media
- Stem Cell Media
- Specialty Media
- Chemically Defined Media
- Other Media
- Serum-free Media
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Cancer Research
- Biopharmaceutical Production
- Monoclonal Antibodies
- Vaccine Production
- Diagnostics
- Drug Screening
- Tissue Engineering
- Cell Therapy
- Cancer Research
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharmaceutical and Biotechnology Companies
- Hospitals and Diagnostic Laboratories
- Research and Academic Institutes
- Others
- Pharmaceutical and Biotechnology Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Liquid Media
- Dry Powder Media
- Custom Media
- Liquid Media
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Cell Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Cell Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Cell Type, 2026 to 2036
- Mammalian Cell Lines
- Stem Cells
- Primary Cells
- Insect Cells
- Microbial Cells
- Mammalian Cell Lines
- Y to o to Y Growth Trend Analysis By Cell Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Cell Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Direct Supplier
- Distributor
- E-Commerce
- Custom Contract
- Direct Supplier
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Application
- By End User
- By Form
- By Cell Type
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Merck KGaA
- Cytiva
- Avantor
- Sartorius
- Lonza Group
- FUJIFILM Biotechnologies
- Caisson Labs
- HiMedia Laboratories
- Thermo Fisher Scientific
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Cell Type, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Form
- Figure 15: Global Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Cell Type
- Figure 18: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Sales Channel
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Type
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Form
- Figure 44: North America Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Cell Type
- Figure 47: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Sales Channel
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Application
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Form
- Figure 63: Latin America Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Cell Type
- Figure 66: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by End User
- Figure 79: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Form
- Figure 82: Western Europe Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Cell Type
- Figure 85: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Type
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Application
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by End User
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Form
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Cell Type
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Type
- Figure 111: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Application
- Figure 114: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by End User
- Figure 117: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Form
- Figure 120: East Asia Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Cell Type
- Figure 123: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Cell Type
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Cell Type, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Cell Type, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Cell Type
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

