Multi Nutritional Supplement Market
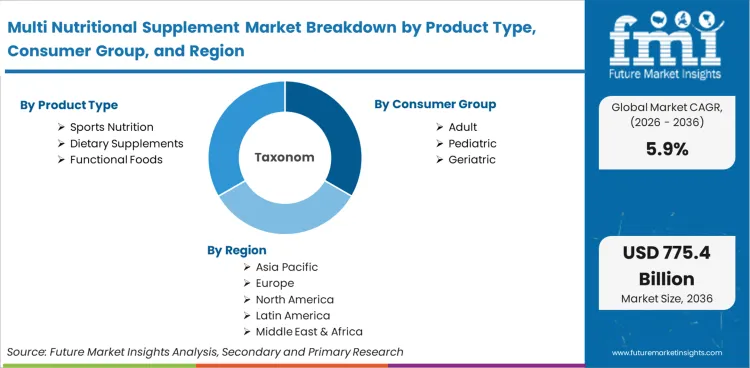
The Multi Nutritional Supplement Market is segmented by Product Type (Sports Nutrition, Dietary Supplements, Functional Foods), Consumer Group (Adults, Pediatric, Geriatric), and Region. Forecast for 2026 to 2036.
Multi Nutritional Supplement Market Size, Market Forecast and Outlook By FMI
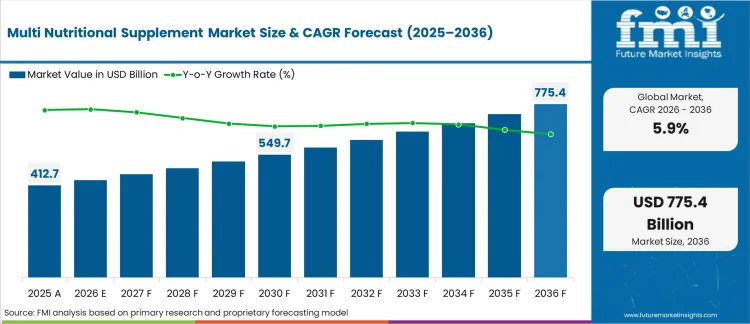
In 2025, the multi nutritional supplement market crossed a valuation of USD 412.7 billion. Based on Future Market Insights’ analysis, demand for multi nutritional supplements is estimated to grow to USD 437.1 billion in 2026 and USD 773.7 billion by 2036. FMI projects a CAGR of 5.9% during the forecast period.
Production frameworks transition from generic dietary supplements toward specialized bio-active compounds to satisfy stricter international quality mandates. Brands deploy advanced extraction technologies to maintain high-potency ingredients while navigating rigid supply chain bottlenecks. As per FMI's projection, stringent purity regulations filter out speculative entrants and consolidate volume among vertically integrated formulating entities.
Summary of Multi Nutritional Supplement Market
- Multi Nutritional Supplement Market Definition:
- Formulated nutrient compounds intended to augment daily nutritional intake via concentrated dose formats.
- Demand Drivers in the Market:
- Preventive health routines replace reactive symptom management.
- Direct-to-consumer digital subscription models improve replenishment consistency.
- Novel delivery formats reduce pill fatigue among younger demographics.
- Key Segments Analyzed in the FMI Report:
- Product Type: Sports nutrition leads (38.2%) over dietary supplements and functional foods.
- Consumer Group: Adults dominate consumption (41.5%) versus pediatric and geriatric segments.
- Geography: High-growth India and China versus compliance-intensive North America and Europe.
- Analyst Opinion at FMI:
- Nandini Roy Choudhury, Principal Consultant for Food & Beverage at Future Market Insights, opines, “In the updated Multi Nutritional Supplement Market report for 2026 to 2036, my team and I have observed that regulatory scrutiny and consumer demand for clinically backed claims are redefining competitive advantage. Brands can no longer rely on generic multivitamin positioning; precision dosing, bioavailability enhancement, and vertically integrated sourcing are becoming non-negotiable to defend margins and sustain long-term brand credibility in an increasingly compliance-driven environment.”
- Strategic Implications/Executive Takeaways:
- Vertically integrated supply chains protect margins against raw material cost volatility.
- Specialized condition-specific formulas command higher consumer loyalty than generic multivitamins.
- Manufacturing scalability determines brand survival amid shifting regional safety standards.
- Methodology:
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
“As customer interest in probiotics continues to accelerate, we are investing heavily to stay ahead of what’s next,” said Patrick Brueggman, President and CEO of Vitaquest. “This new Probiotics Suite reflects our commitment to building best-in-class capabilities that help our partners grow with confidence.” [1]
Regional growth in the multi nutritional supplement industry is expected to progress at different rates, reflecting varied consumer adoption and distribution maturity. FMI estimates indicate stronger momentum in Asia, with China projected at 5.8% CAGR and India at 6.0%. The United States and United Kingdom are forecast to expand at 5.1% and 5.2%, while Germany and Brazil are expected to grow at 5.4% and 5.3%, respectively.
Multi Nutritional Supplement Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 437.1 Billion |
| Industry Value (2036) | USD 773.7 Billion |
| CAGR (2026 to 2036) | 5.9% |
Multi Nutritional Supplement Market Definition
Defined boundaries cover concentrated nutrient sources manufactured to supplement nutritional intake beyond standard dietary consumption. Product formats include capsules, powders, gummies, and functional liquids containing vitamins, minerals, amino acids, and botanicals. Classifications depend heavily on localized regulatory frameworks dictating allowable health claims. Manufacturers deploy varying delivery mechanisms to enhance bioavailability and absorption rates among end users.
Multi Nutritional Supplement Market Inclusions
Included categories span commercially distributed multi-ingredient formulas targeting performance enhancement, immune support, and metabolic balance. Research captures sales across pharmacies, specialty health stores, and direct-to-consumer digital channels. Assessment models incorporate specialized sports nutrition powders and ready-to-drink formulations. Corporate data aggregating wholesale production volumes establishes baseline supply metrics.
Multi Nutritional Supplement Market Exclusions
Excluded product lines encompass regulated pharmaceutical medications requiring physician prescriptions for treatment of diagnosed clinical diseases. Unprocessed whole foods and standard agricultural commodities fall outside measured valuation boundaries. Research models omit single-ingredient generic vitamins manufactured solely for hospital inpatient administration. Speculative artisanal blends lacking standardized active compound concentrations remain unmeasured within formal industry datasets.
Multi Nutritional Supplement Market Research Methodology
- Primary Research: Direct interviews with formulation scientists, procurement directors, and regulatory compliance officers provided verifiable baseline pricing structures.
- Desk Research: Aggregate data from national health ministries and international trade registries confirmed macro import-export volumes.
- Market-Sizing and Forecasting: Production volume metrics correlated against regional population demographic shifts established 10-year projection models.
- Data Validation and Update Cycle: Internal peer-review processes triangulate corporate annual filings against published factory output statistics.
Segmental Analysis
Multi Nutritional Supplement Market Analysis by Product Type
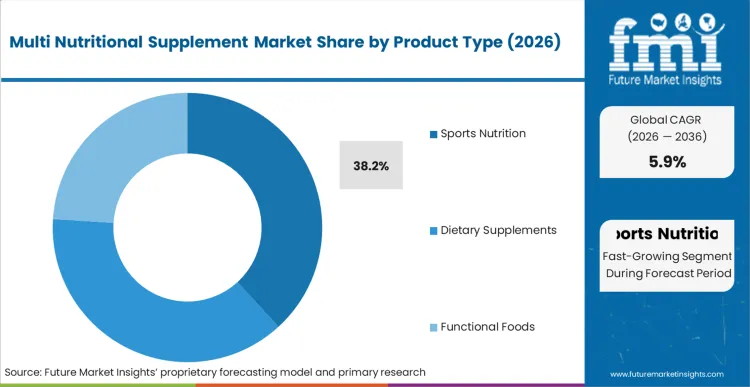
Sports nutrition holds a leading 38.2% share, supported by a growing base of fitness focused consumers and rising demand for performance targeted supplements. Procurement teams are expanding production capacity for specialized vitamin formulations with more complex amino acid profiles. FMI analysts opine that evolving formulations increase manufacturing complexity and tighten quality control requirements. Suppliers are adopting automated blending systems to reduce cross contamination risk across high potency actives. Market momentum also favors brands with verified third party testing credentials that meet scrutiny from professional athletic bodies.
- Formulation shift: Escalating athletic performance standards force brands to replace artificial additives with natural botanical extracts. Upgraded ingredient profiles secure higher retail pricing tiers. Compliance protocols alter packaging strategies immediately. Verified product efficacy guarantees recurrent buyer spending.
- Capacity expansion: Rising global consumption triggers immediate facility upgrades among leading contract manufacturers. Scaled production volumes lower unit costs. Margin improvements fund future proprietary research programs. Enhanced processing environments protect sensitive biocompounds.
- Retail integration: Brands establish physical presence within specialized fitness environments to capture point-of-use transactions. Direct consumer access bypasses traditional wholesale markups. Immediate feedback loops guide future iterations. Localized distribution networks strengthen brand loyalty metrics.
Multi Nutritional Supplement Market Analysis by Consumer Group
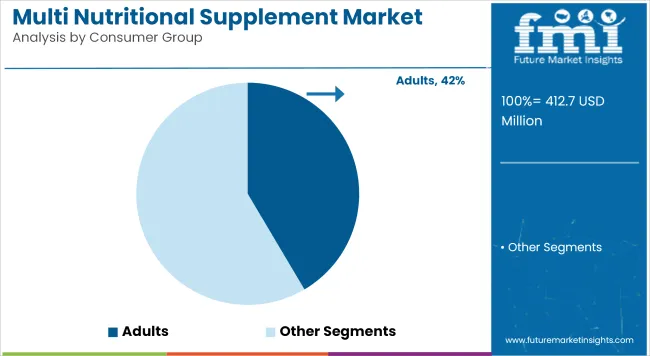
Adult formulations account for a leading 41.5% share, supported by a broad consumer base focused on long term health maintenance. Formulators increasingly tailor products for cardiovascular support, joint health, and cognitive wellness. Aging population trends are strengthening demand for preventive nutrition solutions. Procurement teams source specialized botanical ingredients to differentiate premium adult lines from standard multivitamin offerings. FMI projects that subscription based purchasing models improve revenue visibility for major nutraceutical brands. Regulatory agencies also require clear label warnings on potential interactions with medications commonly used by older adults.
- Adoption trigger: Pervasive chronic conditions compel aging populations to seek non-pharmaceutical management options. Routine consumption establishes predictable retail turnover rates. Consistent brand engagement prevents competitor switching behaviors. Active ingredient absorption remains a primary purchasing criterion.
- Procurement shift: Manufacturers prioritize scientifically validated ingredients to satisfy increasingly skeptical older buyers. Brands allocate extensive resources toward clinical efficacy trials. Substantiated health claims warrant premium price positioning. Verified outcomes strengthen practitioner recommendation rates.
- Format evolution: Swallowing difficulties among older consumers prompt transitions from traditional capsules toward functional liquids. Alternative delivery mechanisms increase manufacturing complexity. Advanced blending techniques ensure uniform active ingredient distribution. Improved user experiences generate higher monthly retention figures.
Multi Nutritional Supplement Market Drivers, Restraints, and Opportunities
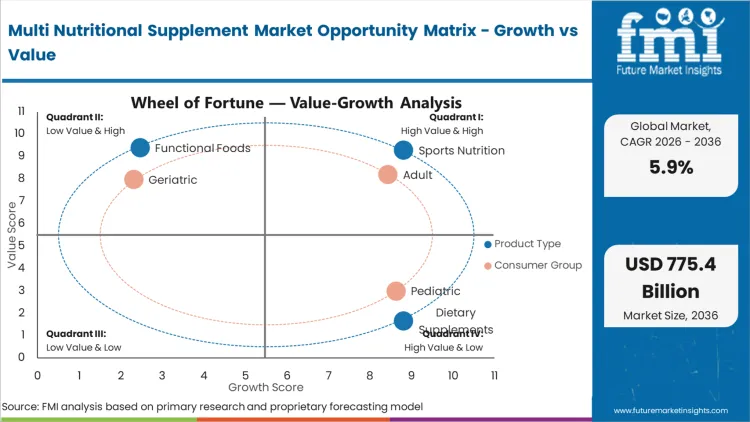
Pervasive chronic conditions establish structural reliance on formulated preventive nutrition. Approximately 1.4 billion adults exhibited hypertension globally during 2024, prompting widespread transitions toward non-pharmaceutical cardiovascular management. [2] Procurement teams source specialized botanical extracts to develop targeted vitamin and mineral supplement ranges. According to FMI's estimates, validated symptom mitigation translates into rigid daily consumption habits and predictable retail turnover. Formulators prioritize documented traceability to satisfy rapid border inspections. Transparent sourcing practices build institutional trust among regional health ministries. Brand equity relies heavily on maintaining unblemished safety profiles across multiple operating regions.
Elevated integration costs present substantial barriers for emerging formulators attempting scale operations. Complex multi-ingredient formulas exhibit unpredictable stability behaviors during large-scale blending procedures. Bioactive compounds frequently degrade when exposed to standard thermal processing environments. Manufacturers mitigate vulnerabilities through costly microencapsulation technologies and climate-controlled infrastructure. Specialized supplements and nutrition packaging must prevent moisture ingress and oxygen exposure to guarantee stated shelf-life durations. Capital-intensive prerequisites restrict market entry exclusively to well-funded corporate entities.
- E-commerce Expansion: Digital distribution channels remove geographic barriers preventing niche brand discovery among highly specific patient demographics. Online platforms facilitate recurring subscription revenues. Predictable cash flows fund continuous product innovation cycles.
- Personalized Formulation: Diagnostic testing advances allow brands to formulate highly specific nutrient blends matching individual genetic profiles. Targeted therapies command substantial price premiums. Custom manufacturing lines replace mass-production equipment. Patient outcome improvements guarantee absolute brand loyalty.
- Merger Consolidation: Pharmaceutical conglomerates acquire agile consumer brands to capture established digital audiences and modern branding techniques. Financial backing propels global distribution expansion plans. Combined operational efficiency lowers aggregate production costs.
Regional Analysis
Based on the regional analysis, the Multi Nutritional Supplement market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 6.0% |
| China | 5.8% |
| Germany | 5.4% |
| Brazil | 5.3% |
| United Kingdom | 5.2% |
| United States | 5.1% |
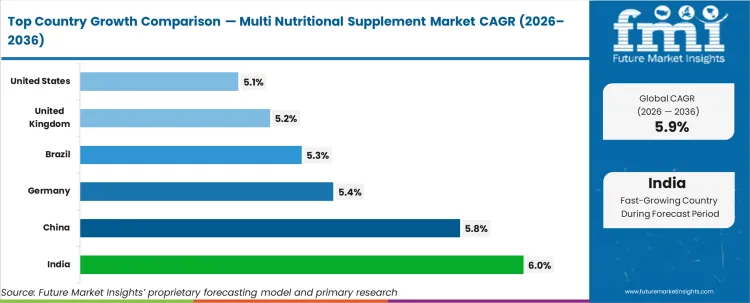
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Multi Nutritional Supplement Market Analysis
North American procurement cycles favor scientifically validated formulations promising specific physiological outcomes rather than general wellness maintenance. Mature retail networks provide extensive physical shelf space for diverse product categories ranging from specialized bio-active blends to advanced sports performance powders. FMI analysts opine that rigid regulatory enforcement by federal agencies eliminates non-compliant formulations from regional supply chains. Consumers dedicate substantial discretionary income toward proactive health management. Leading formulators continuously upgrade facility infrastructure to maintain domestic production advantages. Tightened raw material sourcing protocols protect premium brand reputations.
- United States: Demand for targeted interventions in the USA is set to grow at 5.1% CAGR through 2036, supported by an adult obesity prevalence of 40.3% before procurement and budgeting cycles translate into higher deployment volumes. [3] Domestic manufacturers upgrade extraction technologies to secure specialized botanical compounds. Stricter formulation mandates eliminate speculative imports, allowing established nutraceutical contract manufacturing services to capture immense contract volumes.
FMI's report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA and Canada. Emerging opportunities exist across Canada and Mexico as cross-border trade agreements normalize ingredient standards. Brands actively monitor localized retail shifts involving premium dietary components to capture secondary household expenditure.
Europe Multi Nutritional Supplement Market Analysis
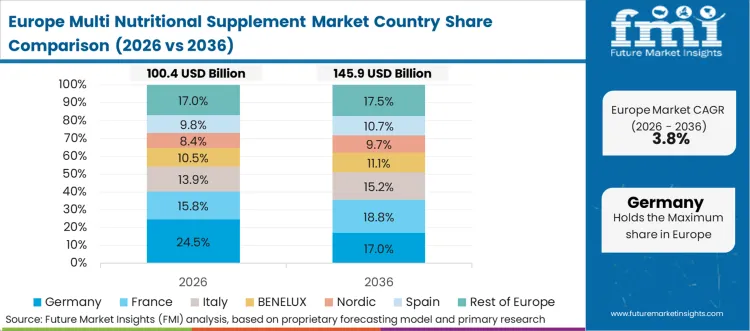
European regulatory frameworks impose stringent documentation requirements verifying ingredient safety and efficacy before granting commercial retail access. Consumer preferences favor natural botanical extracts and clean-label manufacturing processes devoid of synthetic preservatives. Pharmacy channels maintain outsized influence regarding consumer purchasing decisions and brand recommendations. Regional health ministries actively monitor population nutrient deficiencies to guide public health initiatives. Established chemical conglomerates divest non-core nutritional ingredient portfolios to specialized entities. Strict environmental mandates govern packaging material selection among regional producers.
- United Kingdom: UK sales are projected to rise at 5.2% CAGR from 2026 to 2036 as national surveys indicate serum vitamin D deficiency in 18% of adults aged 19 to 64, creating a clearer pathway from policy or spending intent to purchase orders. [4] Pharmacy-led distribution networks establish high consumer trust levels. Subscription delivery models stabilize recurring revenue streams for premium wellness brands.
- Germany: The Germany sector is poised to expand at 5.4% CAGR during 2026 to 2036 because RSG Group formalized an exclusive partnership with ESN to offer targeted sports nutrition on-site, which lifts utilisation rates and justifies technology refresh decisions. [5] Localized distribution shortens supply chain timelines. Immediate consumer access bypasses traditional retail bottlenecks.
FMI's report includes deep structural examination of continental purchasing dynamics. Expanding commercial prospects surface within France and Italy as aging demographics seek prophylactic health maintenance. Regional compliance officers track shifting ingredient approvals affecting demand and trends analysis of dietary supplements in western Europe to maintain uninterrupted supply operations.
East Asia Multi Nutritional Supplement Market Analysis
East Asian manufacturing hubs dominate global active pharmaceutical ingredient production through unparalleled scale and technical proficiency. Government policies actively support domestic biotechnology sectors to reduce reliance on imported chemical inputs. Rapid urbanization alters traditional dietary habits, prompting widespread reliance on formulated nutritional support. As per FMI's projection, streamlined regulatory pathways speed novel product introductions across vast consumer networks. Rising middle-class populations demonstrate fierce loyalty toward premium imported brands possessing unblemished safety records. Cross-border e-commerce platforms bypass restrictive physical retail regulations. Domestic formulators upgrade internal quality control mechanisms to compete against international conglomerates.
- China: China is expected to record 5.8% CAGR in sector demand over 2026 to 2036 as Solabia acquired Mibelle Biochemistry to enhance localized active ingredient availability, tightening the link between operational constraints and adoption-led investment. [6] Increased market access intensifies domestic brand competition. Winners industrialize speed-to-approval mechanisms.
FMI's report includes comprehensive evaluation of regional supply chain mechanics. Japan and South Korea demonstrate intense consumer demand for anti-aging functional formulations. Corporate strategy teams monitor evolving multivitamin melt technologies to capture affluent older demographics requiring simplified dosing formats.
South Asia Multi Nutritional Supplement Market Analysis
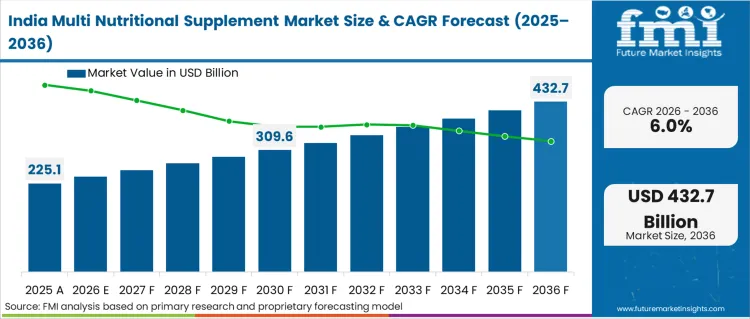
South Asian populations exhibit rising disposable incomes and expanding awareness regarding preventive healthcare practices. Digital penetration allows emerging direct-to-consumer brands to bypass fragmented traditional retail distribution networks. Widespread nutritional deficits compel localized government bodies to encourage fortified food consumption. Pharmaceutical leaders acquire agile digital-first brands to rapidly capture youthful consumer segments. Modern manufacturing facilities deploy advanced extraction techniques to process regional Ayurvedic botanicals for global export. Influencer marketing campaigns effectively educate younger demographics regarding specialized sports nutrition protocols. Middle-class expansion guarantees continuous volume growth across mass-market supplement categories.
- India: In India, the landscape is forecast to advance at 6.0% CAGR through 2036, with growth anchored in USV acquiring a 79% stake in D2C brand Wellbeing Nutrition that converts into measurable budget allocation. [7] Financial backing propels regional distribution networks. Institutional quality controls elevate domestic safety standards.
FMI's report includes distinct commercial modeling across developing consumer bases. Bangladesh and Sri Lanka represent untapped volume potential for affordable preventive nutrition formats. Formulators investigate localized botanical profiles to develop unique algae based supplement derivatives suitable for tropical climates.
Latin America Multi Nutritional Supplement Market Analysis
Latin American regulatory agencies modernize oversight frameworks to align with international food safety standards. Upgraded labeling requirements force formulators to transparently declare active ingredient concentrations and potential allergens. Government bodies seek to reduce rising public healthcare costs by promoting proactive nutritional supplementation. Manufacturers scale specialized production lines to accommodate localized ingredient preferences. Improved extraction techniques harness indigenous botanical compounds for export toward premium Western markets.
- Brazil: Brazil uptake is anticipated to climb at 5.3% CAGR over the forecast period as typical dietary magnesium absorption remains limited to 30% to 40%, helping suppliers align positioning with local buying triggers. [8] Targeted mineral supplementation offsets dietary shortfalls. Upgraded manufacturing protocols ensure consistent active compound delivery.
FMI's report includes detailed analysis for the market in Brazil, Mexico, Chile, Argentina, Peru and Rest of Latin America. Emerging economies across the continent transition toward regulated health food sectors. Domestic formulators expand portfolios to include pet dietary supplements to capture affluent urban households.
Competitive Aligners for Market Players
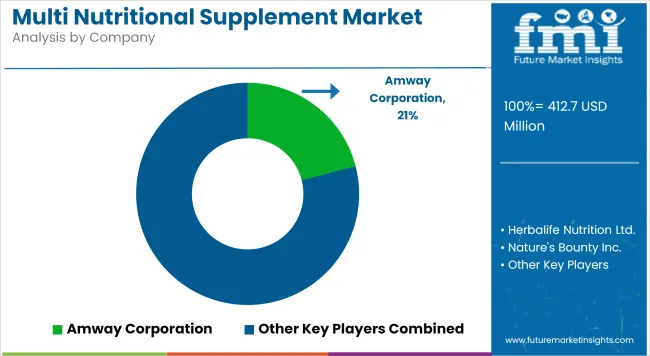
Market structure remains fragmented globally, yet practical competition is concentrated among a limited set of suppliers capable of meeting volume and compliance expectations. Regional capacity expansion dictates pricing leverage across wholesale ingredient networks. Producers maintaining captive active compound extraction facilities absorb raw material price volatility more effectively and sustain supply continuity during logistical disruptions.
The primary competitive variable centers on regulatory survivability, not peak margin capture. Suppliers operating without vertical integration depend on spot contracts, increasing exposure to margin compression. Formulators must constantly evaluate ingredient integrity to avoid catastrophic border rejections or retail recalls.
Regulatory compliance further narrows the competitive field. The ability to supply consistent, biologically active grades at scale requires formulation stability, advanced testing infrastructure, and process control discipline. Customer concentration reinforces buyer leverage, as large retail groups multi-source finished products to prevent supplier dependency.
Recent Developments:
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In November 2025, Probi expanded its portfolio into specialized sports and active nutrition arenas. [9]
- In June 2025, Amway India launched Nutrilite Triple Protect as a plant-based supplement blend. [10]
Key Players in Multi Nutritional Supplement
- Nestlé Health Science
- dsm-firmenich
- Vitaquest
- Solabia
- Dr. Reddy’s
- Sabinsa
- Probi
- Amway
- GNC
- Myprotein
- HealthKart
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 437.1 billion (2026) to USD 773.7 billion (2036), at a CAGR of 5.9% |
| Market Definition | Concentrated nutrient sources manufactured to supplement nutritional intake beyond standard dietary consumption via diverse dose formats. |
| Product Type Segmentation | Sports Nutrition, Dietary Supplements, Functional Foods |
| Consumer Group Segmentation | Adults, Pediatric, Geriatric |
| Application Coverage | Immune Support, Performance Enhancement, Metabolic Balance, Cognitive Function, Joint Mobility |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40 plus countries |
| Key Companies Profiled | Nestlé Health Science, dsm-firmenich, Vitaquest, Solabia, Dr. Reddy’s, Sabinsa, Probi, Amway, GNC, Myprotein, HealthKart |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with formulation scientists, supported by trade data benchmarking and plant level capacity verification. |
Multi Nutritional Supplement Market Analysis by Segments
Product Type:
- Sports Nutrition
- Dietary Supplements
- Functional Foods
Consumer Group:
- Adults
- Pediatric
- Geriatric
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] GlobeNewswire. (2026). Vitaquest International LLC Opens World-Class Probiotics Suite. Corporate Press Release, February 2026.
- [2] World Health Organization. (2025). Global hypertension prevalence and unawareness rates. Fact Sheet, September 2025.
- [3] Centers for Disease Control and Prevention. (2024). Adult obesity prevalence statistics. Data Brief, September 2024.
- [4] UK Government. (2025). National Diet and Nutrition Survey 2019-2023. Official Statistics Report, June 2025.
- [5] RSG Group. (2025). Exclusive partnership announced with ESN for targeted sports nutrition. Corporate Announcement, April 2025.
- [6] Solabia. (2026). Mibelle Biochemistry joins Solabia Group to enhance active ingredient availability. Corporate Press Release, February 2026.
- [7] Indian Retailer. (2026). USV acquires 79% stake in Wellbeing Nutrition to enter nutraceutical market. Industry Report, February 2026.
- [8] National Institutes of Health. (2026). Typical dietary magnesium absorption rates. Health Professional Fact Sheet, January 2026.
- [9] Probi. (2025). Probi expands portfolio into Sports & Active Nutrition. Press Release, November 2025.
- [10] Business Standard. (2025). Amway India launches Nutrilite Triple Protect plant-based supplement. Corporate News, June 2025.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market intelligence to enable structured strategic decision making across mature and emerging nutraceutical economies
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by validated capacity and trade benchmarks
- Growth opportunity mapping across sports nutrition, dietary supplements, and functional foods with emphasis on clean-label transitions
- Segment and regional revenue forecasts covering adult, pediatric, and geriatric consumer groups
- Competition strategy assessment including integration models, cost survivability positioning, and compliance capability benchmarking
- Product reformulation and ingredient compliance tracking including FDA, EFSA, and regional safety standard aligned grades
- Regulatory impact analysis covering health claim mandates, clinical validation thresholds, and export-driven specification upgrades
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational use
Frequently Asked Questions
How large is the demand for Multi Nutritional Supplement in the global market in 2026?
Demand for Multi Nutritional Supplement in the global market is estimated to be valued at USD 437.1 billion in 2026.
What will be the market size of Multi Nutritional Supplement in the global market by 2036?
Market size for Multi Nutritional Supplement is projected to reach USD 773.7 billion by 2036.
What is the expected demand growth for Multi Nutritional Supplement in the global market between 2026 and 2036?
Demand for Multi Nutritional Supplement in the global market is expected to grow at a CAGR of 5.9% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Sports nutrition is expected to be a prominent segment, capturing 38.2% of global share in 2026 due to escalating athletic performance standards.
How significant is the role of Adults in driving Multi Nutritional Supplement adoption in 2026?
Adult formulations represent a critical segment, projected to hold a substantial 41.5% share in 2026 as older demographics seek non-pharmaceutical management options.
What is driving demand in India?
Accelerated corporate consolidation and targeted acquisitions by established pharmaceutical entities are driving regional volume expansion.
What compliance standards or regulations are referenced for India?
Institutional quality controls and localized Ayurvedic processing guidelines are referenced as key compliance benchmarks.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 6.0% during 2026 to 2036.
Why is China described as a priority region in this report?
Growth is catalyzed by streamlined regulatory pathways that speed novel product introductions across vast consumer networks.
What type of demand dominates in China?
Adoption-led demand for premium imported brands possessing unblemished safety records dominates regional consumption.
What is the China growth outlook in this report?
China is projected to expand at a CAGR of 5.8% during 2026 to 2036.
Does the report cover Germany in its regional analysis?
Germany is included within Europe under the regional scope of analysis.
What are the sources referred to for analyzing Germany?
Corporate partnership announcements and fitness industry integration bulletins are cited as primary reference sources.
What is the main demand theme linked to Germany in its region coverage?
European demand is associated with aggressive fitness industry integration that increases the value of reliability and compliance.
Does the report cover Brazil in its regional analysis?
Brazil is included within Latin America under the regional coverage framework.
What is the main Brazil related demand theme in its region coverage?
Targeted mineral supplementation to offset local dietary absorption shortfalls is emphasized.
Which product formats or configurations are strategically important for North America supply chains?
Bio-active compounds and specialized botanical extracts are prioritized due to rigid regulatory enforcement and premium brand positioning.
What is Multi Nutritional Supplement and what is it mainly used for?
Multi nutritional supplements are concentrated nutrient sources manufactured to augment daily intake for immune support, performance enhancement, and metabolic balance.
What does Multi Nutritional Supplement mean in this report?
Multi nutritional supplement refers to global production, trade, and industrial consumption of formulated vitamins, minerals, and botanicals in diverse dose formats.
What is included in the scope of this Multi Nutritional Supplement report?
Scope covers product types such as sports nutrition and dietary supplements, consumed by adult, pediatric, and geriatric demographics.
What is excluded from the scope of this report?
Regulated pharmaceutical medications, unprocessed whole foods, and single-ingredient generic inpatient vitamins are excluded.
What does market forecast mean on this page?
Market forecast represents a model-based projection built on defined demographic shifts for strategic planning purposes.
How does FMI build and validate the Multi Nutritional Supplement forecast?
Forecast is developed using hybrid top-down and bottom-up modeling validated through trade data, capacity checks, and industry review.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Sports Nutrition
- Dietary Supplements
- Functional Foods
- Sports Nutrition
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Consumer Group
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Consumer Group, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Consumer Group, 2026 to 2036
- Adult
- Pediatric
- Geriatric
- Adult
- Y to o to Y Growth Trend Analysis By Consumer Group, 2021 to 2025
- Absolute $ Opportunity Analysis By Consumer Group, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Consumer Group
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Consumer Group
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Consumer Group
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Consumer Group
- Competition Analysis
- Competition Deep Dive
- Nestlé Health Science
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- dsm-firmenich
- Vitaquest
- Solabia
- Dr. Reddy’s
- Sabinsa
- Probi
- Amway
- Nestlé Health Science
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Consumer Group, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Consumer Group
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Consumer Group
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Consumer Group
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Consumer Group
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Consumer Group
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Consumer Group
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Consumer Group
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Consumer Group, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Consumer Group, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Consumer Group
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

