Nephroblastoma Therapeutics Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Nephroblastoma Therapeutics Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Nephroblastoma Therapeutics Market Size and Share Forecast Outlook 2025 to 2035
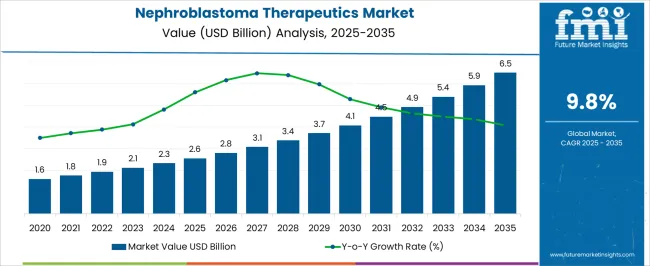
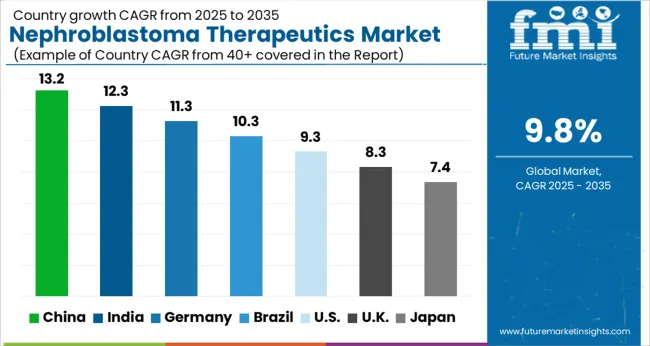
The Nephroblastoma Therapeutics Market is estimated to be valued at USD 2.6 billion in 2025 and is projected to reach USD 6.5 billion by 2035, registering a compound annual growth rate (CAGR) of 9.8% over the forecast period.
Quick Stats for Nephroblastoma Therapeutics Market
- Nephroblastoma Therapeutics Market Industry Value (2025): USD 2.6 billion
- Nephroblastoma Therapeutics Market Forecast Value (2035): USD 6.5 billion
- Nephroblastoma Therapeutics Market Forecast CAGR: 9.8%
- Leading Segment in Nephroblastoma Therapeutics Market in 2025: Favorable Histology (57.3%)
- Key Growth Region in Nephroblastoma Therapeutics Market: North America, Asia-Pacific, Europe
- Top Key Players in Nephroblastoma Therapeutics Market: Merck & Co., Inc., Recordati Rare Diseases, Pfizer Inc., Sun Pharmaceutical Industries Ltd., Cipla Inc., Actiza Pharmaceutical Private Limited, Teva Pharmaceutical Industries Ltd., Alvogen, Accord Healthcare Ireland Ltd., Amneal Pharmaceuticals LLC
| Metric | Value |
|---|---|
| Nephroblastoma Therapeutics Market Estimated Value in (2025 E) | USD 2.6 billion |
| Nephroblastoma Therapeutics Market Forecast Value in (2035 F) | USD 6.5 billion |
| Forecast CAGR (2025 to 2035) | 9.8% |
Rationale for Segmental Growth in the Nephroblastoma Therapeutics Market
The nephroblastoma therapeutics market is experiencing steady progress driven by advancements in oncology research, improved treatment protocols, and the integration of multidisciplinary approaches in pediatric oncology care. Growing emphasis on early diagnosis, rising survival rates through combined therapies, and expanding access to oncology drugs in emerging regions are shaping the market dynamics.
Pharmaceutical companies are channeling investments into developing more targeted and less toxic drug regimens to improve quality of life for young patients. Supportive regulatory frameworks and increasing collaborations between hospitals, research institutes, and cancer foundations are also facilitating accelerated availability of innovative treatments.
The market outlook remains positive as efforts continue to focus on reducing relapse rates, optimizing chemotherapy combinations, and expanding distribution networks to ensure accessibility of nephroblastoma therapies worldwide.
Segmental Analysis
Insights into the Favorable Histology Type Segment
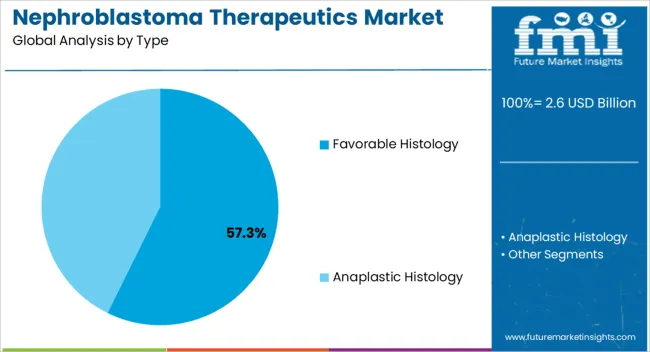
The favorable histology segment is projected to account for 57.30% of total market revenue by 2025 within the type category, making it the most prominent. This dominance is supported by the relatively better treatment outcomes and higher survival rates associated with favorable histology nephroblastoma compared to other subtypes.
The segment has benefited from standardized chemotherapy and surgical protocols that deliver consistent results, thereby reinforcing confidence among oncologists.
Continuous improvements in diagnostic imaging and pathology have also enhanced the identification and management of favorable histology cases, further supporting its leadership position.
Insights into the Dactinomycin Drug Type Segment
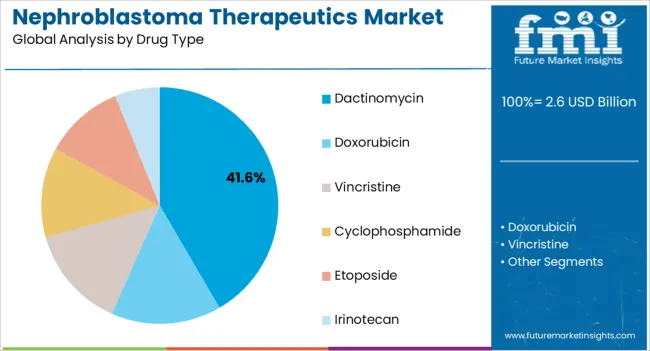
The dactinomycin segment is expected to contribute 41.60% of total revenue by 2025 within the drug type category, positioning it as the leading therapeutic option. Its widespread use in established treatment regimens for nephroblastoma, often in combination with other chemotherapeutic agents, has solidified its clinical importance.
The proven efficacy of dactinomycin in reducing tumor recurrence and improving long term survival outcomes has maintained its relevance in therapeutic protocols.
Additionally, its integration into global pediatric oncology guidelines has ensured consistent adoption across both developed and developing healthcare systems.
Insights into the Hospital Pharmacies Distribution Channel Segment
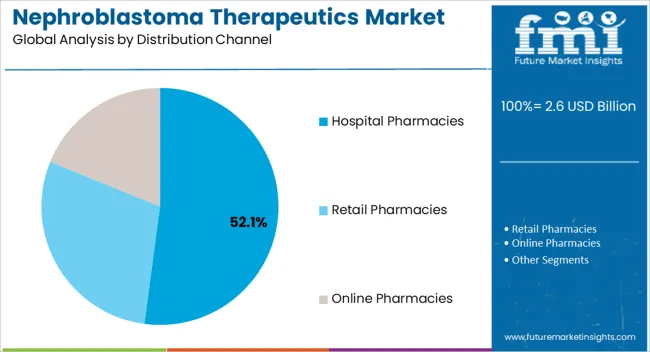
The hospital pharmacies segment is estimated to hold 52.10% of total revenue by 2025 within the distribution channel category, highlighting its dominance. This is primarily due to the critical nature of nephroblastoma treatment, which requires close supervision by oncology specialists and hospital based administration of chemotherapeutic agents.
Hospital pharmacies provide streamlined access to essential drugs, support adherence to treatment schedules, and ensure safe handling of potent oncology medications.
The segment’s prominence is further reinforced by the concentration of pediatric oncology expertise and supportive care infrastructure within hospital settings, making hospital pharmacies the central hub for nephroblastoma therapeutics distribution.
Nephroblastoma Therapeutics Market Historical Period 2020 to 2025 Demand Analysis Vs. Forecast 2025 to 2035
The global nephroblastoma therapeutics market grew at a CAGR of 8.88% from 2020 to 2025, as per Future Market Insights, a provider of market research and competitive intelligence.
Although most patients with a histological diagnosis of Wilms tumor do well with current treatment, approximately 10% of patients have histopathological features that are associated with a worse prognosis, and in some types, with a high incidence of relapse and death.
Nowadays, improved risk stratification has divided the population of patients into small subgroups, creating the challenge of designing and executing clinical trials that are sufficiently powered to demonstrate the desired outcomes. The fruits of stepped-up international collaboration are beginning to be realized, with the discovery of new genes, biological markers, and therapeutic targets. The benefits of COG-SIOP collaboration will hopefully translate into the application of evidence-based diagnostic and therapeutic approaches in low-income countries.
Prominent Growth Drivers Influencing Nephroblastoma Therapeutics Market
Increase in approvals of regulatory bodies
Key players in the market are focused on receiving approvals from regulatory bodies for drugs used to treat nephroblastoma is expected to drive the market growth over the forecast period.
Frequent regulatory approvals for new nephroblastoma treatments are expected to propel the growth of the global nephroblastoma treatment market during the forecast period. Market players are focusing on approvals from regulatory authorities for new drugs.
Focusing on the growth of R&D activities
Moreover, market players are focusing on R&D activities for new treatments, which is also expected to augment the growth of the global nephroblastoma treatment market during the forecast period. For instance, in August 2025, Children’s Oncology Group studying the combination of chemotherapy and surgery effects on young patients suffering from Wilms tumor revealed to be in Phase 3 trial and that the study is likely to be completed by end of the year 2025.
What Could be the Key Restraint for the Nephroblastoma Therapeutics Market?
The high cost of treatment leads acts as a restraining factor
The major factor that hinders the growth of the global nephroblastoma therapeutics market includes the high cost of nephroblastoma. According to an article published by the American Society of Clinical Oncology, the cost for the full course of treatment was determined between USD 1490 and USD 2093 for a patient suffering from nephroblastoma.
Region-Wise Analysis of Nephroblastoma Therapeutics Market
What Is the Outlook of The North American Nephroblastoma Therapeutics Market?
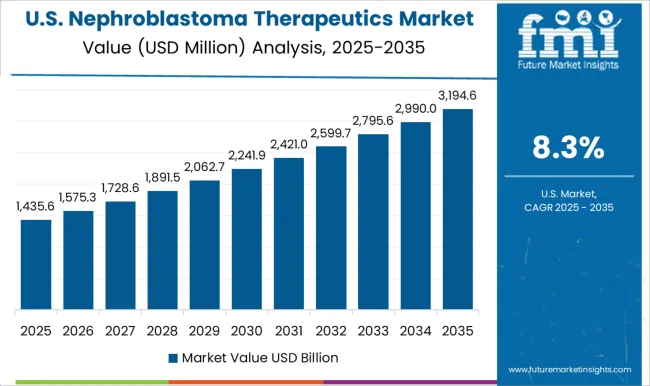
Favorable reimbursement policies offering moneymaking opportunities in the United States
The United States is known for its healthcare infrastructure and favorable reimbursement scenario as both of them are expected to push the demand for nephroblastoma treatment shortly. A rise in the incidence of different types of tumors, rapid adoption of novel cancer treatments, and an increase in investment in medical research and development are also expected to favor market growth in the future.
What Is the Outlook of The Asia Pacific Nephroblastoma Therapeutics Market?
Increasing investments in healthcare infrastructural development will favor market growth in Japan
Demand for nephroblastoma treatment is expected to be primarily driven by an increasing focus on healthcare infrastructure development and a rise in investments in medical R&D, the growing awareness regarding nephroblastoma, an increase in spending patterns on health and fitness, and the growing availability of novel cancer treatments with a rise in the number of cases are the factors that will augment the development of the market over the next ten years.
Category-Wise Insights into Nephroblastoma Therapeutics Industry
Which Type of drug is used in the treatment of Nephroblastoma Therapeutics Market?
Dactinomycin is said to hold a dominant share of the market
According to the reports of FMI, of 13 children treated with dactinomycin in addition to nephrectomy and postoperative radiotherapy, 92% (12 of 13) are living and free of tumors. It was concluded that the survival of children with nephroblastoma is markedly prolonged by the administration of dactinomycin at the time of diagnosis. The beneficial effect of dactinomycin was attributed to the ‘cidal’ action of this drug on small or poorly established metastases.
Which distribution channel segment is expected to gain traction from 2025 to 2035?
The demand for hospital pharmacies will be more during the forecast period.
According to a report by Future Market Insights, the hospital pharmacies would grow within the anticipated time frame. This market sector is expected to hold a greater share of the worldwide market from 2025 to 2035.
Due to the growing number of people visiting hospitals for advice on treatment related to nephroblastoma, the hospital pharmacy is anticipated to dominate the worldwide nephroblastoma therapeutics market. As a result, individuals more pertain towards their health and well-being. Thus, the majority of consumers are expected to only buy medications from hospital pharmacies.
Start-up Scenario in the Nephroblastoma Therapeutics Landscape
The Soonicorns - soon to be Unicorns - are the handful of highly valued startups that have successfully grown out of their nascency to attract valuations of over a few hundred million. Watch out for these companies in the coming years as they take on the journey to becoming elite Unicorns.
Penn Medicine’s Abramson Cancer Center published early data from the first USA clinical trial of CRISPR-edited immune cells, showing that genetically editing the T cells of human cancer patients and infusing the cells back into the patients appeared safe and feasible. Investigators reported infusing three participants - two with multiple myeloma and one with sarcoma.
How Competitive is the Nephroblastoma Therapeutics Market?
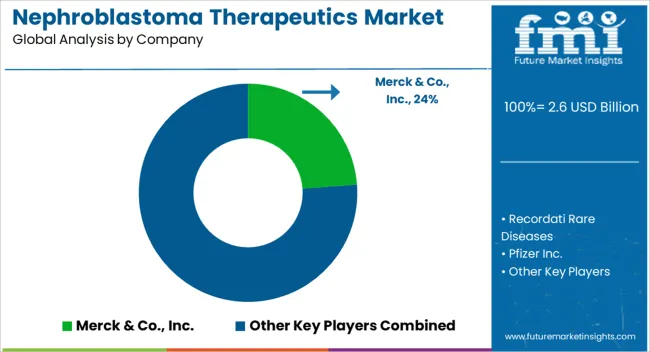
Some of the prominent players in the global market for Nephroblastoma Therapeutics Market treatment are
- Merck & Co., Inc.
- Recordati Rare Diseases
- Pfizer Inc.
- Sun Pharmaceutical Industries Ltd.
- Cipla Inc.
- Actiza Pharmaceutical Private Limited
- Teva Pharmaceutical Industries Ltd.
- Alvogen
- Accord Healthcare Ireland Ltd.
- Amneal Pharmaceuticals LLC.
Some of the important developments of the key players in the market are
- In October 2020, Y-mAbs Therapeutics, In. announced a clinical update on naxitamab for the treatment of nephroblastoma at the International Society of Pediatric Oncology Annual Congress held in France.
- In June 2025, Omblastys (Omburtamab) was granted a priority review for a Biologics License Application by the Food and Drug Administration of the United States. The drug was intended to use for the treatment affected by leptomeningeal metastasis caused by nephroblastoma.
Scope of the Nephroblastoma Therapeutics Market Report
| Report Attributes | Details |
|---|---|
| Growth Rate | CAGR of 9.88% from 2025 to 2035 |
| Market value in 2025 | USD 2.6 billion |
| Market value in 2035 | USD 6.5 billion |
| Base Year for Estimation | 2025 |
| Historical Data | 2020 to 2025 |
| Forecast Period | 2025 to 2035 |
| Quantitative Units | USD Billion for Value and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, Trends, and Pricing Analysis |
| Segments Covered | Type, Drug type, Distribution channel, Region |
| Regions Covered | North America; Latin America; Europe; South Asia; East Asia; Oceania; Middle East & Africa |
| Key Countries Profiled | United States, Canada, Brazil, Mexico, Rest of Latin America, Germany, United Kingdom, France, Spain, Italy, Rest of Europe, India, Malaysia, Singapore, Thailand, Rest of South Asia, China, Japan, South Korea, Australia, New Zealand, GCC countries, South Africa, Israel |
| Key Companies Profiled | Merck & Co., Inc.; Recordati Rare Diseases; Pfizer Inc.; Sun Pharmaceutical Industries Ltd.; Cipla Inc.; Actiza Pharmaceutical Private Limited; Teva Pharmaceutical Industries Ltd.; Alvogen; Accord Healthcare Ireland Ltd.; Amneal Pharmaceuticals LLC. |
| Customization Scope | Available on Request |
Key Segments Profiled in the Nephroblastoma Therapeutics Market Report
Type:
- Favorable histology
- Anaplastic histology
Drug type:
- Dactinomycin
- Doxorubicin
- Vincristine
- Cyclophosphamide
- Etoposide
- Irinotecan
Distribution channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Region:
- North America
- Latin America
- Europe
- South Asia
- East Asia
- Oceania
- Middle East & Africa
Frequently Asked Questions
How big is the nephroblastoma therapeutics market in 2025?
The global nephroblastoma therapeutics market is estimated to be valued at USD 2.6 billion in 2025.
What will be the size of nephroblastoma therapeutics market in 2035?
The market size for the nephroblastoma therapeutics market is projected to reach USD 6.5 billion by 2035.
How much will be the nephroblastoma therapeutics market growth between 2025 and 2035?
The nephroblastoma therapeutics market is expected to grow at a 9.8% CAGR between 2025 and 2035.
What are the key product types in the nephroblastoma therapeutics market?
The key product types in nephroblastoma therapeutics market are favorable histology and anaplastic histology.
Which drug type segment to contribute significant share in the nephroblastoma therapeutics market in 2025?
In terms of drug type, dactinomycin segment to command 41.6% share in the nephroblastoma therapeutics market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2025 to 2035
- Favorable Histology
- Anaplastic Histology
- Y-o-Y Growth Trend Analysis By Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Drug Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Drug Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Drug Type, 2025 to 2035
- Dactinomycin
- Doxorubicin
- Vincristine
- Cyclophosphamide
- Etoposide
- Irinotecan
- Y-o-Y Growth Trend Analysis By Drug Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Drug Type, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2025 to 2035
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2020 to 2024
- Absolute $ Opportunity Analysis By Distribution Channel, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Drug Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Drug Type
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Drug Type
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Drug Type
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Merck & Co., Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Recordati Rare Diseases
- Pfizer Inc.
- Sun Pharmaceutical Industries Ltd.
- Cipla Inc.
- Actiza Pharmaceutical Private Limited
- Teva Pharmaceutical Industries Ltd.
- Alvogen
- Accord Healthcare Ireland Ltd.
- Amneal Pharmaceuticals LLC
- Merck & Co., Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Type , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 4: Global Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 5: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: North America Market Value (USD Million) Forecast by Type , 2020-2035
- Table 7: North America Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 8: North America Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 10: Latin America Market Value (USD Million) Forecast by Type , 2020-2035
- Table 11: Latin America Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 12: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: Western Europe Market Value (USD Million) Forecast by Type , 2020-2035
- Table 15: Western Europe Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 16: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Type , 2020-2035
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 22: East Asia Market Value (USD Million) Forecast by Type , 2020-2035
- Table 23: East Asia Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 24: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2020-2035
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Type , 2020-2035
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Drug Type, 2020-2035
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

