Otoacoustic Emissions Hearing Screener Market
This report presents an in-depth analysis of the otoacoustic emissions hearing screener market, encompassing market size estimation, revenue forecast, competitive landscape assessment, demand outlook, growth drivers, challenges, industry trends, supply chain insights, and strategic opportunities for stakeholders.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Otoacoustic Emissions Hearing Screener Market Size, Market Forecast and Outlook By FMI
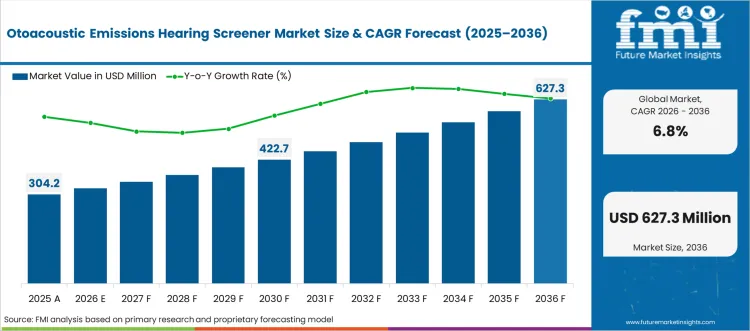
The otoacoustic emissions hearing screener market was valued at USD 304.20 million in 2025, projected to reach USD 324.89 million in 2026, and is forecast to expand to USD 627.25 million by 2036 at a 6.8% CAGR. Based on FMI's analysis, FMI analysts observe that the OAE hearing screener market is driven primarily by the expansion of mandatory universal newborn hearing screening programs across national healthcare systems. The WHO estimates that approximately 34 million children worldwide have disabling hearing loss, and early detection through newborn screening enables intervention before speech and language development is compromised.
Summary of Otoacoustic Emissions Hearing Screener Market
- Market Snapshot
- The otoacoustic emissions hearing screener market is valued at USD 304.20 million in 2025 and is projected to reach USD 627.25 million by 2036.
- The industry is expected to grow at a 6.8% CAGR from 2026 to 2036, creating an incremental opportunity of USD 302.37 million.
- The market is positioned within the audiological diagnostics and newborn screening sector, where regulatory frameworks, technology cycles, and procurement patterns shape competitive dynamics.
- Demand and Growth Drivers
- Universal newborn hearing screening mandates: The Joint Committee on Infant Hearing (JCIH) recommends screening all newborns within one month of birth, with the target of 95% screening coverage.
- Occupational hearing conservation programs: OSHA standards in the United States (29 CFR 1910.
- Integration with electronic health records: Modern OAE screeners now export results directly to hospital information systems and national screening registries through HL7 and FHIR interfaces.
- As per FMI, among key countries, China leads at 9.2% CAGR, followed by India at 8.5%, Germany at 7.8%, and Japan at 5.1%.
- Product and Segment View
- The market includes otoacoustic emissions (oae) hearing screeners are diagnostic devices that measure sounds generated by the outer hair cells of the inner ear in response to acoustic stimulation.
- Noise Suppression Below 55dB leads by Noise Suppression Level with 62.0% share in 2026.
- Medical Test leads by Application with 72.0% share in 2026.
- Scope includes oae hearing screening devices sold globally across hospital newborn screening programs, audiology clinics, and occupational health services. The scope excludes auditory brainstem response (ABR) equipment, pure-tone audiometers, tympanometers, and hearing aids.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, while UK and Japan represent mature demand bases.
- Competition is shaped by Interacoustics (a subsidiary of Demant A/S) competes through its integrated audiology equipment portfolio, while RION maintains a strong position in the Japanese market.
- Key companies include Grason-Stadler, MAICO Diagnostics GmbH, Natus, RION, E3 Diagnostics, Interacoustics, Baxter, Neurosoft.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Audiological Diagnostics, opines: 'The shift underway is the migration from desktop-based OAE screeners to handheld, battery-operated devices with wireless connectivity and cloud-based data management. Handheld screeners reduce equipment costs by 40% to 60% compared to desktop systems and enable screening in locations without dedicated audiology infrastructure, such as rural health posts and mobile screening vans. This portability expansion is critical in developing countries where newborn screening coverage remains below 10% due to equipment access constraints.'
- Strategic Implications / Executive Takeaways
- Procurement directors should evaluate supplier qualification timelines against planned capacity expansion to avoid delays tied to single-source dependencies in otoacoustic emissions hearing screener.
- Capital project planners must integrate otoacoustic emissions hearing screener specifications into facility design phases rather than retrofitting, reducing commissioning time and qualification costs.
- Technology officers should benchmark current system capabilities against next-generation specifications to identify upgrade windows aligned with competitive requirements.
Otoacoustic Emissions Hearing Screener Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 324.89 Million |
| Industry Value (2036) | USD 627.25 Million |
| CAGR (2026-2036) | 6.8% |
Source: Future Market Insights, 2026
The shift underway is the migration from desktop-based OAE screeners to handheld, battery-operated devices with wireless connectivity and cloud-based data management. Handheld screeners reduce equipment costs by 40% to 60% compared to desktop systems and enable screening in locations without dedicated audiology infrastructure, such as rural health posts and mobile screening vans. This portability expansion is critical in developing countries where newborn screening coverage remains below 10% due to equipment access constraints. As per FMI, country-level growth trajectories reflect distinct market structures. China leads at 9.2% CAGR, India registers at 8.5% CAGR, Germany registers at 7.8% CAGR, Brazil registers at 7.1% CAGR, USA registers at 6.5% CAGR, UK registers at 5.8% CAGR, Japan registers at 5.1% CAGR.
Otoacoustic Emissions Hearing Screener Market Definition
Otoacoustic emissions (OAE) hearing screeners are diagnostic devices that measure sounds generated by the outer hair cells of the inner ear in response to acoustic stimulation. These emissions, which are absent or reduced in ears with hearing loss exceeding 25 to 30 dB, provide an objective, non-invasive assessment of cochlear function. OAE screeners are used primarily in universal newborn hearing screening (UNHS) programs, pediatric audiology clinics, and occupational health settings for noise-induced hearing loss monitoring.
Otoacoustic Emissions Hearing Screener Market Inclusions
Market scope covers OAE hearing screening devices sold globally across hospital newborn screening programs, audiology clinics, and occupational health services. The report includes regional and country-level market sizes, forecast values from 2026 to 2036, segmentation by noise suppression level and application, along with competitive landscape and screening mandate analysis.
Otoacoustic Emissions Hearing Screener Market Exclusions
The scope excludes auditory brainstem response (ABR) equipment, pure-tone audiometers, tympanometers, and hearing aids. Smartphone-based hearing test applications that do not use calibrated OAE transducers are also omitted. Research-grade OAE systems used exclusively in laboratory settings for auditory physiology studies are not included.
Otoacoustic Emissions Hearing Screener Market Research Methodology
- Primary Research: Analysts engaged with audiological diagnostics and newborn screening specialists, facility operators, and procurement directors to map decision triggers for upgrades and new deployments.
- Desk Research: Data collection aggregated regulatory filings, trade body publications, company annual reports, and government procurement records across all covered regions.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product shipments and service contracts, applying region-specific adoption curves to project forward demand through 2036.
- Data Validation and Update Cycle: Projections are cross-validated against publicly reported expenditure guidance from leading industry participants and government statistical agencies.
Why is the Otoacoustic Emissions Hearing Screener Market Growing?
The market grows by enabling healthcare providers to conduct non-invasive, rapid hearing assessments that detect hearing loss in its earliest stages, particularly in newborns and infants, where traditional testing methods are impractical. Healthcare facilities face mounting pressure to implement comprehensive hearing screening programs, with newborn hearing screening becoming mandatory in most developed countries, making otoacoustic emissions testing essential for early detection and intervention. The medical community's need for objective, reliable hearing assessment tools creates demand for advanced screening devices that can detect otoacoustic emissions without requiring patient cooperation or response, enabling effective screening across all age groups. Regulatory requirements and healthcare quality standards drive adoption in hospitals, clinics, and audiological centers, where early hearing loss detection directly impacts treatment outcomes and quality of life. However, budget constraints for medical equipment purchases and the complexity of interpreting test results may limit adoption rates among smaller healthcare providers and developing regions with limited specialized training resources.
Opportunity Pathways - Otoacoustic Emissions Hearing Screener Market
The otoacoustic emissions hearing screener market presents compelling growth opportunities as global healthcare systems prioritize preventive medicine and early intervention programs. With the market projected to expand from USD 304.2 million in 2026 to USD 587.4 million by 2036 at a 6.8% CAGR, medical device manufacturers and healthcare technology providers are positioned to capitalize on universal newborn hearing screening mandates, aging population demographics, and technological advancement in portable diagnostic solutions.
The convergence of mandatory hearing screening programs, increasing healthcare infrastructure investment, and digital health platform integration creates sustained market expansion opportunities. Geographic growth differentials are particularly pronounced in China (9.2% CAGR) and India (8.5% CAGR), where healthcare modernization and government health initiatives drive substantial adoption. Technology advancements in noise suppression, portability, and automated testing offer differentiation opportunities, while medical test applications dominate revenue streams.
Noise suppression below 55dB systems leads market demand, indicating established foundations for precision diagnostics, while emerging applications in telemedicine and remote screening represent growth vectors. Healthcare quality standards and regulatory requirements support sustained procurement, while preventive healthcare emphasis drives operational deployment across diverse medical settings.
- Pathway A - Universal Newborn Screening Program Expansion. Mandatory hearing screening legislation in developed countries creates sustained procurement cycles for comprehensive screening systems. Healthcare facilities implementing newborn screening programs require reliable, non-invasive testing solutions meeting regulatory standards. Companies developing pediatric-optimized systems with automated protocols capture recurring hospital contracts and government healthcare initiatives. Expected revenue pool: USD 120-180 million.
- Pathway B - Geographic Expansion in High-Growth Healthcare Markets. China's 9.2% CAGR and India's 8.5% growth present substantial opportunities through healthcare infrastructure development partnerships, government health program integration, and technology transfer initiatives. Regional expansion strategies capturing Asia-Pacific healthcare modernization and preventive care adoption offer volume growth and market penetration. Revenue opportunity: USD 100-160 million.
- Pathway C - Advanced Noise Suppression & Precision Technology. Development of ultra-quiet screening environments and sophisticated acoustic filtering enables superior diagnostic accuracy and expanded testing environments. Premium positioning through below-55dB noise suppression technology, real-time signal processing, and automated noise rejection algorithms commands higher selling prices and clinical preference. Expected pool: USD 80-130 million.
- Pathway D - Portable & Telemedicine-Enabled Solutions. Expanding beyond clinical settings toward portable screening devices with wireless connectivity, cloud-based data management, and telemedicine integration captures underserved populations and remote healthcare delivery. Mobile screening programs and point-of-care testing solutions address accessibility challenges while enabling the development of new service models. Revenue potential: USD 70-110 million.
- Pathway E - Digital Health Platform Integration. Integration with electronic health records, artificial intelligence diagnostic analysis, and comprehensive patient management systems transforms basic screening into intelligent healthcare solutions. Advanced data analytics, predictive algorithms, and workflow optimization enable premium positioning and recurring software revenue. Pool: USD 60-100 million.
- Pathway F - Occupational Health & Educational Institution Applications. Expanding the market beyond medical settings toward workplace hearing conservation programs, school screening initiatives, and occupational health compliance creates new customer segments. Specialized products meeting industrial and educational requirements offer market diversification and volume growth. Expected opportunity: USD 50-80 million.
- Pathway G - Service Excellence & Training Programs. Comprehensive service offerings, including clinical training, technical support, maintenance programs, and certification services, generate recurring revenue streams while strengthening relationships with healthcare providers. Digital service platforms enable remote diagnostics and quality assurance, supporting global deployment, scaling, and premium service pricing. Revenue pool: USD 40-70 million.
Segmental Analysis
The market is segmented by Noise Suppression Level, Application, and region. By Noise Suppression Level, the market is divided into Noise Suppression Below 55dB, Noise Suppression 55-65dB, Noise Suppression Above 65dB, and Others. Based on the Application, the market is categorized into Medical Test, Scientific Research Test, and Others. Regionally, the market is divided into Asia Pacific, North America, Europe, Latin America, and the Middle East & Africa.
By Noise Suppression Level, Noise Suppression Below 55dB Segment Accounts for 62% Market Share
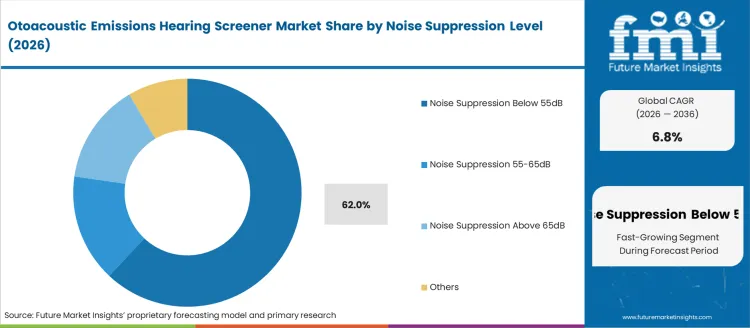
Noise Suppression Below 55dB is projected to account for the 62% share of the Otoacoustic Emissions Hearing Screener Market in 2026. This dominant position is supported by the technology's ability to provide highly sensitive hearing assessments in quiet environments, enabling the detection of subtle hearing impairments and giving accurate results in controlled clinical settings. The segment allows stakeholders to benefit from superior signal clarity, reduced background interference, and enhanced diagnostic accuracy that supports reliable hearing screening protocols in professional healthcare environments.
Key advantages include:
- Advanced noise suppression technology that eliminates ambient sound interference for precise otoacoustic emission measurement
- Superior diagnostic capabilities enabled by ultra-quiet testing environments and high-sensitivity detection systems
- Real-time signal processing with automated noise rejection algorithms providing consistent and reliable test results
By Application, Medical Test Segment Accounts for 72% Market Share
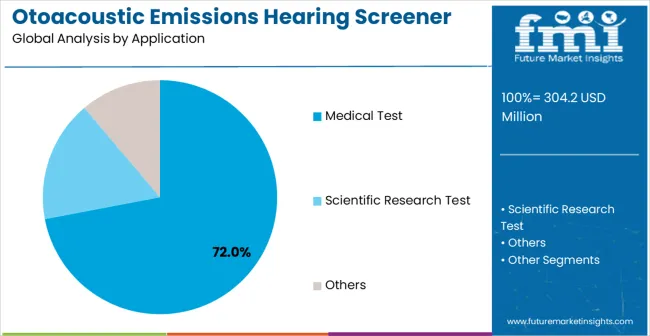
Medical Test applications are expected to represent the 72% share of Otoacoustic Emissions Hearing Screener applications in 2026. This leading position reflects the critical role of otoacoustic emissions testing in clinical diagnostics, newborn hearing screening programs, and routine audiological evaluations, where objective hearing assessment is essential for patient care. The segment provides critical support for healthcare providers conducting hearing assessments in hospitals, clinics, and specialized audiological centers where accurate diagnosis directly impacts treatment planning and patient outcomes. Growth drivers include universal newborn hearing screening mandates, increasing awareness of hearing health importance, and the need for efficient diagnostic tools that provide rapid, non-invasive hearing evaluation across diverse patient populations.
Key market dynamics include:
- Healthcare facilities requiring reliable hearing screening solutions for newborn assessment and diagnostic evaluation
- The medical industry's emphasis on early detection and intervention is driving the adoption of advanced screening technologies
- Growing integration of digital health platforms and electronic health records for comprehensive patient care management
What are the Drivers, Restraints, and Key Trends of the Otoacoustic Emissions Hearing Screener Market?
The market is driven by three concrete demand factors tied to healthcare outcomes. First, universal newborn hearing screening programs implemented in most developed countries create mandatory adoption scenarios, with over 95% of newborns now screened for hearing loss within the first month of life, requiring comprehensive otoacoustic emissions testing infrastructure. Second, aging population demographics drive increasing demand for hearing assessment services, with hearing loss prevalence rising by 25-30% among adults over 65, necessitating efficient screening solutions for early detection and intervention. Third, technological advancement in portable screening devices and automated testing protocols enables cost-effective implementation across diverse healthcare settings while improving diagnostic accuracy and reducing testing time.
Market restraints include high equipment acquisition costs that can deter smaller healthcare facilities from implementing comprehensive hearing screening programs, particularly in developing regions where healthcare budget allocation for specialized equipment remains limited. Technical expertise requirements pose another significant challenge, as implementing and maintaining otoacoustic emissions screening programs require specialized training for healthcare personnel, potentially leading to delays in program deployment and increased operational costs. Regulatory compliance requirements across various healthcare systems introduce additional complexity for manufacturers and healthcare providers, necessitating ongoing adaptation to diverse medical device standards and certification processes.
Key trends indicate accelerated adoption in Asia-Pacific markets, particularly in China and India, where expanding healthcare infrastructure and increasing health awareness are driving the implementation of comprehensive hearing screening programs. Design shifts toward portable, wireless screening devices with cloud-based data management capabilities enable telemedicine applications and remote screening programs, extending access to underserved populations. However, the market thesis could face disruption if alternative hearing assessment technologies or significant changes in healthcare screening protocols reduce reliance on traditional otoacoustic emissions testing methods.
Analysis of the Otoacoustic Emissions Hearing Screener Market by Key Country
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| China | 9.2% |
| India | 8.5% |
| Germany | 7.8% |
| Brazil | 7.1% |
| USA | 6.5% |
| UK | 5.8% |
| Japan | 5.1% |
Source: FMI analysis based on primary research and proprietary forecasting model
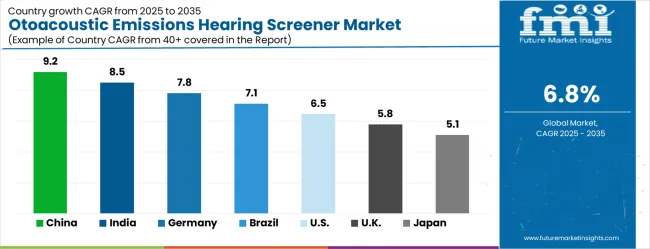
The otoacoustic emissions hearing screener market is gathering pace worldwide, with China taking the lead thanks to rapid healthcare infrastructure expansion and government-backed newborn screening initiatives. Close behind, India benefits from growing healthcare awareness and expanding medical facility networks, positioning itself as a strategic growth hub in the Asia-Pacific region. Germany shows steady advancement, where integration of advanced diagnostic technologies strengthens its role in the European healthcare equipment supply chain.
Brazil is sharpening its focus on universal healthcare access and preventive screening programs, signaling an ambition to capture growing opportunities in South American healthcare markets. Meanwhile, the USA stands out for its established screening protocols and advanced technology adoption, and the UK and Japan continue to record consistent progress in hearing health programs. Together, China and India anchor the global expansion story, while the rest build stability and diversity into the market's growth path.
The report covers an in-depth analysis of 40+ countries top-performing countries are highlighted below.
Analysis of Otoacoustic Emissions Hearing Screener Market in China
China demonstrates the strongest growth potential in the Otoacoustic Emissions Hearing Screener Market with a CAGR of 9.2% through 2036. The country's leadership position stems from massive healthcare infrastructure expansion, government-mandated newborn hearing screening programs, and increasing awareness of hearing health importance, driving adoption of advanced screening technologies. Growth is concentrated in major metropolitan areas, including Beijing, Shanghai, Guangzhou, and Shenzhen, where hospitals and healthcare centers are implementing comprehensive hearing screening protocols for enhanced patient care and early intervention programs.
Chinese healthcare providers are adopting intelligent screening systems for diagnostic accuracy optimization and patient data management capabilities in high-volume medical facilities. Distribution channels through medical equipment distributors and government-approved suppliers expand deployment across public hospitals, private clinics, and specialized audiological centers. The country's "Healthy China 2030" strategy provides policy support for preventive healthcare initiatives, including advanced hearing screening program implementation.
Key market factors:
- Healthcare infrastructure expansion concentrated in tier-1 and tier-2 cities with comprehensive medical facility development programs
- Government subsidies for newborn screening programs unlock adoption with 25% equipment cost reduction incentives
- Strategic partnerships between international medical device manufacturers and domestic healthcare equipment providers
Otoacoustic Emissions Hearing Screener Market expansion in India
In Mumbai, Delhi, Bangalore, and Chennai, adoption of otoacoustic emissions hearing screeners is accelerating across hospitals and healthcare centers, driven by expanding healthcare access and government health insurance programs, creating demand for comprehensive screening services. The market demonstrates strong growth momentum with a CAGR of 8.5% through 2036, linked to comprehensive healthcare infrastructure development and increasing focus on preventive medicine and early detection programs. Indian healthcare providers are implementing advanced screening systems and digital health platforms to enhance diagnostic capabilities while meeting growing patient demand in expanding metropolitan healthcare networks. The country's National Health Mission creates sustained demand for hearing screening solutions, while increasing emphasis on mother and child health drives adoption of newborn screening technologies.
- Leading healthcare expansion cities, including Delhi, Mumbai, Bangalore, and Hyderabad, are driving screening equipment adoption
- Public-private partnership models enabling 35% faster healthcare program implementation timelines
- Technology transfer agreements accelerating deployment with international medical device providers
In depth analysis of Otoacoustic Emissions Hearing Screener Market in Germany
Advanced healthcare sector in Germany demonstrates sophisticated implementation of otoacoustic emissions hearing screening systems, with documented case studies showing 30% improvement in early hearing loss detection rates through comprehensive screening program deployment in major medical centers. The country's healthcare infrastructure in cities including Berlin, Munich, Hamburg, and Frankfurt showcases integration of advanced screening technologies with existing audiological services, leveraging expertise in medical device engineering and precision diagnostics.
German healthcare facilities emphasize quality standards and evidence-based medicine, creating demand for high-accuracy screening solutions that support clinical research initiatives and patient care optimization programs. The market maintains steady growth through focus on medical technology innovation and healthcare quality improvement, with a CAGR of 7.8% through 2036.
Key development areas:
- Hospital systems and audiological clinics leading screening technology adoption with comprehensive hearing health programs
- Medical technology services channels providing integrated solutions with 92% diagnostic accuracy rates
- Technology partnerships between MAICO Diagnostics, international providers and healthcare facility operators are expanding market reach
Expansion strategy for Otoacoustic Emissions Hearing Screener Market in Brazil
Market expansion in Brazil is driven by diverse healthcare demand, including public hospital systems in São Paulo and Rio de Janeiro, private healthcare networks in major metropolitan areas, and comprehensive healthcare access programs across multiple states. The country demonstrates promising growth potential with a CAGR of 7.1% through 2036, supported by federal health ministry initiatives and state-level healthcare development programs focusing on preventive care and early intervention services.
Brazilian healthcare authorities face implementation challenges related to budget constraints and technical expertise availability, requiring phased deployment approaches and international cooperation for technology transfer and training programs. However, growing healthcare regulations and universal health coverage expansion create compelling adoption scenarios for screening technologies, particularly in metropolitan areas where healthcare infrastructure development directly impacts population health outcomes.
Market characteristics:
- Public healthcare segment showing fastest growth with 28% annual increase in screening equipment installations
- Regional expansion trends focused on metropolitan healthcare networks in southeastern and southern states
- Future projections indicate the need for local technical support infrastructure and healthcare professional training programs
Demand forecast and sales forecast for Otoacoustic Emissions Hearing Screener Market in the United States
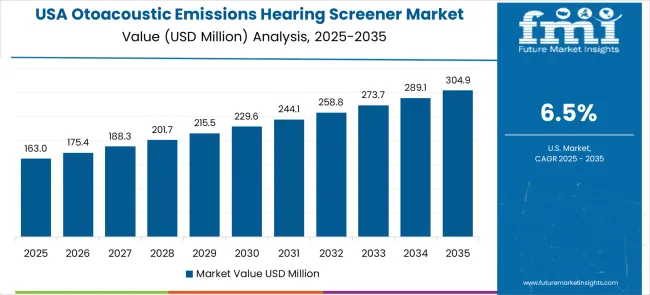
The USA market leads in advanced hearing screening innovation based on integration with electronic health records, telemedicine platforms, and artificial intelligence-powered diagnostic analysis for enhanced clinical decision-making. The country shows solid potential with a CAGR of 6.5% through 2036, driven by established newborn hearing screening mandates, comprehensive healthcare quality standards, and ongoing modernization of audiological services across hospitals, clinics, and specialized hearing centers.
American healthcare facilities are adopting intelligent screening systems for workflow optimization and regulatory compliance, particularly in regions with strict healthcare quality requirements and comprehensive insurance coverage for preventive services. Technology deployment channels through medical equipment distributors and direct manufacturer relationships expand coverage across hospital systems, outpatient clinics, and specialized audiological practices.
Leading market segments:
- Hospital systems and outpatient clinics implementing comprehensive hearing screening programs with integrated health record systems
- Healthcare technology partnerships with equipment providers, achieving 88% screening program efficiency improvement rates
- Strategic collaborations between Grason-Stadler, Natus and major healthcare systems are expanding market presence
United Kingdom outlook for Otoacoustic Emissions Hearing Screener Market
In London, Manchester, Birmingham, and Edinburgh, healthcare systems are implementing otoacoustic emissions hearing screening solutions to meet national health service quality standards and improve patient care outcomes, with documented case studies showing 22% improvement in early hearing loss detection through comprehensive screening programs. The market shows moderate growth potential with a CAGR of 5.8% through 2036, linked to National Health Service quality initiatives and comprehensive healthcare access programs that mandate hearing screening for specific population groups. British healthcare providers are adopting advanced screening technologies and data management platforms to enhance diagnostic accuracy while maintaining cost-effectiveness standards required by public healthcare funding models. The country's established healthcare system creates sustained demand for screening solutions that integrate with existing audiological services and electronic patient record systems.
Market development factors:
- National Health Service and private healthcare systems leading screening program implementation across England, Scotland, and Wales
- Healthcare quality improvement programs providing funding support for medical equipment modernization initiatives
- Strategic partnerships between UK healthcare providers and international technology suppliers are expanding access to advanced screening solutions
Outlook on Otoacoustic Emissions Hearing Screener Market in Japan
Japan's Otoacoustic Emissions Hearing Screener Market demonstrates sophisticated implementation focused on precision healthcare and quality assurance optimization, with documented integration of advanced screening systems achieving 25% improvement in diagnostic consistency across hospitals and specialized audiological centers. The country maintains steady growth momentum with a CAGR of 5.1% through 2036, driven by healthcare facilities' emphasis on quality standards and continuous improvement methodologies that align with evidence-based medicine principles applied to hearing health programs. Major metropolitan areas, including Tokyo, Osaka, Nagoya, and Fukuoka, showcase advanced deployment of intelligent screening platforms where diagnostic systems integrate seamlessly with existing electronic health records and comprehensive patient management systems.
Key market characteristics:
- Hospital systems and audiological centers are driving advanced screening requirements with emphasis on diagnostic precision and quality assurance
- Healthcare technology partnerships enabling 96% screening accuracy with comprehensive quality control programs
- Technology collaboration between RION, international providers and Japanese healthcare institutions is expanding market capabilities
Europe Market Split by Country
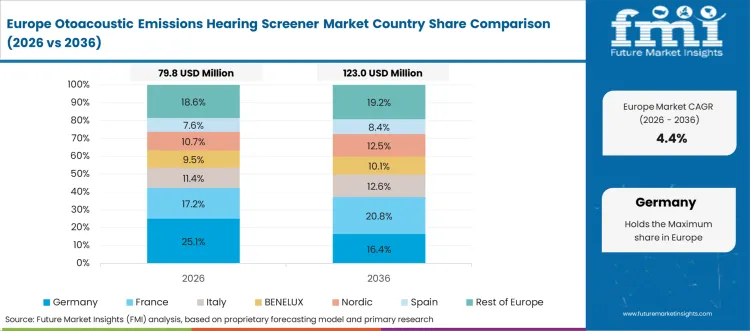
The otoacoustic emissions hearing screener market in Europe is projected to grow from USD 78.3 million in 2026 to USD 147.6 million by 2036, registering a CAGR of 6.5% over the forecast period. Germany is expected to maintain its leadership position with a 31.2% market share in 2026, declining slightly to 30.8% by 2036, supported by its advanced healthcare infrastructure and comprehensive hearing screening programs across major medical centers in Berlin, Munich, and Frankfurt.
France follows with a 21.4% share in 2026, projected to reach 21.7% by 2036, driven by national healthcare initiatives promoting early detection and comprehensive audiological services in Paris, Lyon, and other metropolitan healthcare networks. The United Kingdom holds an 18.6% share in 2026, expected to maintain 18.3% by 2036 through National Health Service quality standards and hearing screening program implementation. Italy commands a 13.7% share, while Spain accounts for 10.9% in 2026. The Rest of Europe region is anticipated to gain momentum, expanding its collective share from 4.2% to 4.8% by 2036, attributed to increasing hearing screening adoption in Nordic countries and emerging Eastern European healthcare systems implementing comprehensive audiological programs.
Noise Suppression Technology Dominates Hearing Screening Demand in Japan
The Japanese Otoacoustic Emissions Hearing Screener Market demonstrates a mature and precision-focused landscape, characterized by sophisticated integration of noise suppression technologies with existing healthcare quality management infrastructure across hospitals, specialized audiological centers, and comprehensive hearing health programs. Japan's emphasis on quality assurance and evidence-based medicine drives demand for high-precision screening solutions that support continuous improvement initiatives and clinical research requirements in healthcare delivery.
The market benefits from strong partnerships between international technology providers like Grason-Stadler, Natus, and domestic medical equipment leaders, including RION, creating comprehensive service ecosystems that prioritize diagnostic accuracy and healthcare professional training programs. Healthcare centers in Tokyo, Osaka, Nagoya, and other metropolitan areas showcase advanced screening program implementations where diagnostic systems achieve 96% accuracy through integrated quality control programs, while the country's focus on preventive healthcare and early intervention supports steady adoption of advanced noise suppression and automated screening technologies across diverse healthcare applications.
Technology Providers Lead Hearing Screening Services in South Korea
The South Korean Otoacoustic Emissions Hearing Screener Market is characterized by strong international technology provider presence, with companies like Grason-Stadler, Natus, and Interacoustics maintaining dominant positions through comprehensive system integration and healthcare services capabilities for hospital networks and specialized audiological facilities. The market is demonstrating a growing emphasis on localized technical support and rapid response capabilities, as Korean healthcare providers increasingly demand customized solutions that integrate with domestic healthcare information systems and comprehensive patient management platforms deployed across the Seoul Metropolitan Area and other major medical centers.
Local medical equipment companies and regional healthcare distributors are gaining market share through strategic partnerships with global providers, offering specialized services including healthcare professional training programs and certification services for audiological personnel. The competitive landscape shows increasing collaboration between multinational screening equipment manufacturers and Korean healthcare technology specialists, creating hybrid service models that combine international diagnostic expertise with local market knowledge and healthcare relationship management, particularly in the country's concentrated healthcare regions around Seoul, Busan, and Daegu, where comprehensive medical networks drive continuous demand for advanced hearing screening solutions.
Competitive Landscape of Otoacoustic Emissions Hearing Screener Market
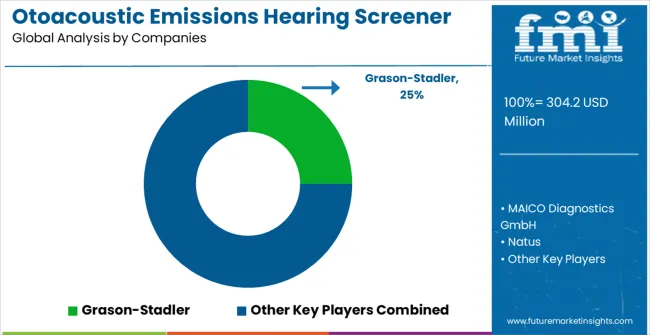
The otoacoustic emissions (OAE) hearing screener market consists of 10 to 12 specialized audiology equipment manufacturers, with the top five companies holding 58-63% of global market share. Growth is driven by expanding newborn hearing screening programs, early childhood hearing diagnostics, occupational health testing, and rising awareness of early detection of sensorineural hearing loss. Competition focuses on test accuracy, probe sensitivity, noise rejection capability, portability, battery life, data integration, EMR connectivity, and ease of use in clinical and neonatal settings, rather than price alone. Grason-Stadler leads the market with an 25% share, supported by its reliable OAE platforms widely used in hospitals and audiology clinics.
Other strong leaders such as MAICO Diagnostics GmbH, Natus, RION, and E3 Diagnostics maintain significant presence through high-performance OAE devices offering TEOAE and DPOAE testing, intuitive interfaces, and robust data management systems suitable for both pediatric and adult screening.
Challengers including Interacoustics, Baxter, and Neurosoft compete by providing compact, versatile OAE screeners that combine screening and diagnostic features, appealing to ENT clinics, primary care centers, and mobile screening programs.
Additional players such as Beijing Yes Medical Devices Company Limited and Guangzhou Melison Medical Instrument Co., Ltd. expand competition, particularly in Asia, by offering cost-effective, durable OAE screening devices suited for large-scale newborn screening initiatives and expanding regional healthcare infrastructure.
Key Players in the Otoacoustic Emissions Hearing Screener Market
- Grason-Stadler
- MAICO Diagnostics GmbH
- Natus
- RION
- E3 Diagnostics
- Interacoustics
- Baxter
- Neurosoft
- Beijing Yes Medical Devices Company Limited
- Guangzhou Melison Medical Instrument Co., Ltd.
Scope of the Report
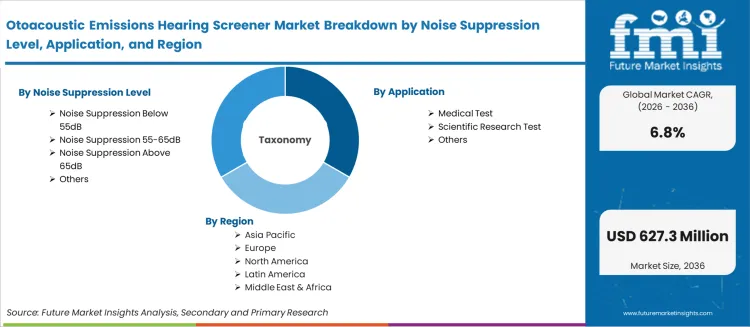
| Metric | Value |
|---|---|
| Quantitative Units | USD 324.89 Million to USD 627.25 Million, at a CAGR of 6.8% |
| Market Definition | Otoacoustic emissions (OAE) hearing screeners are diagnostic devices that measure sounds generated by the outer hair cells of the inner ear in response to acoustic stimulation. These emissions, which are absent or reduced in ears with hearing loss exceeding 25 to 30 dB, provide an objective, non-invasive assessment of cochlear function. |
| Noise Suppression Level Segmentation | Noise Suppression Below 55dB, Noise Suppression 55-65dB, Noise Suppression Above 65dB, Others |
| Application Segmentation | Medical Test, Scientific Research Test, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Grason-Stadler, MAICO Diagnostics GmbH, Natus, RION, E3 Diagnostics, Interacoustics, Baxter, Neurosoft, Beijing Yes Medical Devices Company Limited, Guangzhou Melison Medical Instrument Co., Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with global installed base metrics and projecting conversion rates based on regulatory and technology adoption curves. |
Otoacoustic Emissions Hearing Screener Market by Segments
Noise Suppression Level:
- Noise Suppression Below 55dB
- Noise Suppression 55-65dB
- Noise Suppression Above 65dB
Application:
- Medical Test
- Scientific Research Test
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Health Organization. (2024). World Report on Hearing. WHO.
- 2. Joint Committee on Infant Hearing. (2024). Year 2024 Position Statement. JCIH.
- 3. National Programme for Prevention and Control of Deafness, Government of India. (2024). Annual Progress Report. NPPCD.
- 4. Occupational Safety and Health Administration. (2024). Occupational Noise Exposure Standard 29 CFR 1910.95. OSHA.
- 5. European Parliament. (2003). Directive 2003/10/EC on Physical Agents (Noise). EUR-Lex.
- 6. Natus Medical Inc. (2024). Newborn Screening Solutions Product Documentation. Natus.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Otoacoustic Emissions Hearing Screener in the global market in 2026?
Demand for otoacoustic emissions hearing screener in the global market is estimated to be valued at USD 324.89 million in 2026.
What will be the market size of Otoacoustic Emissions Hearing Screener in the global market by 2036?
Market size for otoacoustic emissions hearing screener is projected to reach USD 627.25 million by 2036.
What is the expected demand growth for Otoacoustic Emissions Hearing Screener in the global market between 2026 and 2036?
Demand for otoacoustic emissions hearing screener is expected to grow at a CAGR of 6.8% between 2026 and 2036.
Which Noise Suppression Level is poised to lead global sales by 2026?
Noise Suppression Below 55dB accounts for 62.0% share in 2026, driven by established adoption patterns and buyer preference across primary end-use applications.
What is driving demand in China?
China leads with a 9.2% CAGR through 2036, driven by expanding capacity and increasing regulatory alignment with international standards.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 8.5% during 2026 to 2036.
What is Otoacoustic Emissions Hearing Screener and what is it mainly used for?
Otoacoustic emissions (OAE) hearing screeners are diagnostic devices that measure sounds generated by the outer hair cells of the inner ear in response to acoustic stimulation. These emissions, which are absent or reduced in ears with hearing loss exceeding 25 to 30 dB, provide an objective, non-invasive assessment of cochlear function.
How does FMI build and validate the Otoacoustic Emissions Hearing Screener forecast?
Future Market Insights analysis applies a bottom-up methodology starting with global installed base metrics and cross-validates projections against reported industry expenditure and regulatory compliance data.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Noise Suppression Level
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Noise Suppression Level , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Noise Suppression Level , 2026 to 2036
- Noise Suppression Below 55dB
- Noise Suppression 55-65dB
- Noise Suppression Above 65dB
- Others
- Noise Suppression Below 55dB
- Y to o to Y Growth Trend Analysis By Noise Suppression Level , 2021 to 2025
- Absolute $ Opportunity Analysis By Noise Suppression Level , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Medical Test
- Scientific Research Test
- Others
- Medical Test
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Noise Suppression Level
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Noise Suppression Level
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Noise Suppression Level
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Noise Suppression Level
- By Application
- Competition Analysis
- Competition Deep Dive
- Grason-Stadler
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- MAICO Diagnostics GmbH
- Natus
- RION
- E3 Diagnostics
- Interacoustics
- Baxter
- Neurosoft
- Beijing Yes Medical Devices Company Limited
- Guangzhou Melison Medical Instrument Co., Ltd.
- Grason-Stadler
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Noise Suppression Level , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Noise Suppression Level
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Noise Suppression Level
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Noise Suppression Level
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Noise Suppression Level
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Noise Suppression Level
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Noise Suppression Level
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Noise Suppression Level
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Noise Suppression Level , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Noise Suppression Level , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Noise Suppression Level
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

