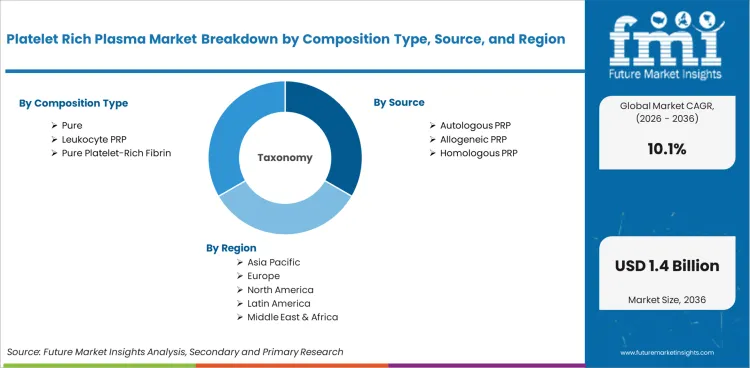
Platelet Rich Plasma Market
The Platelet Rich Plasma Market is segmented by Composition Type, Source, Application, End User and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
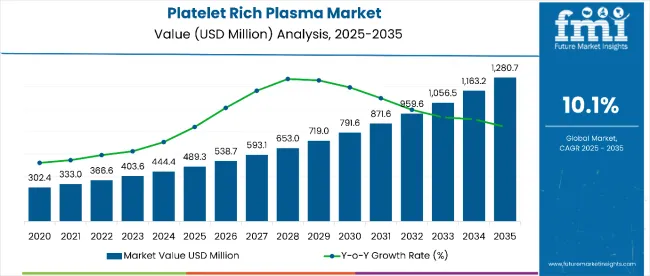
Platelet Rich Plasma Market Size, Market Forecast and Outlook By FMI
The platelet rich plasma market was valued at USD 0.5 billion in 2025. Industry value is projected to reach USD 0.5 billion by 2026 and advance at a CAGR of 10.1% in the assessment period to reach USD 1.4 billion by 2036. Pure platelet-rich plasma is projected to account for 42.0% of composition type demand in 2026. Autologous PRP is expected to hold 86.0% share.
Summary of the Platelet Rich Plasma Market
- Demand and Growth Drivers
- Orthopedic procedure demand is likely to support platelet rich plasma use as clinicians seek same-day biologic options for joint pain and tendon repair.
- Aesthetic clinics are expected to widen PRP use in hair restoration and skin repair programs due to repeat-visit treatment models.
- Wound care applications are entering wider use as diabetic and venous wound cases need products with clear documentation and payer-aligned use.
- Product and Segment View
- Pure platelet-rich plasma is likely to lead the composition type segment as clinics prefer preparations with lower leukocyte burden for routine use.
- Autologous PRP is expected to dominate source demand because same-patient preparation reduces donor screening and storage needs.
- Orthopedics is likely to stay the leading application area as PRP use is closely tied to knee care and tendon injury treatment.
- Geography and Competitive Outlook
- India is likely to record the stronger growth as private orthopedic and dermatology clinics expand regenerative procedure offerings.
- China is expected to gain volume from urban hospitals and aesthetics clinics that are adding PRP to specialist care pathways.
- Companies with closed preparation systems and clear regulatory positioning are likely to gain stronger account access during the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI says, “I see platelet rich plasma moving from informal use toward protocol-based specialist care. Orthopedic surgeons want better preparation consistency before they use PRP across tendon and joint cases. Dermatology clinics want systems that support repeat visits without adding handling risk. Wound care centers need products that can match coverage rules and documentation needs.”
- Platelet Rich Plasma Market Value Analysis
- Platelet rich plasma creates value through same-day biologic preparation that helps clinics add regenerative care without complex product inventory.
- Pure platelet-rich plasma holds commercial value as clinicians prefer cleaner preparation profiles for routine orthopedic and aesthetic procedures.
- Orthopedic use supports repeat procedure demand because joint pain and tendon repair often need follow-up care over time.
- Specialty clinics stay central to spending as PRP preparation fits outpatient treatment rooms and physician-led service models.
- Supplier advantage depends on closed-system handling, platelet yield consistency, and simple staff training.
Clinical services face a proof problem because PRP varies by device output, platelet dose and use route. Orthopedic suppliers gain a clearer entry point through cleared graft handling devices. OrthoAscent’s 2024 FG-001 summary lists a 60 mL capacity, 7 mL final output and two-spin processing for PRP used with autograft or allograft bone. Product selection becomes a regulatory and workflow decision for surgeons and distributors.
Wound care extends PRP beyond injection rooms because FDA’s 2025 SkinDisc record covers point-of-care PRP and PPP from a patient’s blood for diabetic ulcers and pressure ulcers. Wound care coverage gives clinics a dressing-based revenue case tied to debrided wound management. Orthopedic evidence has become dose sensitive. Bensa et al. reviewed 18 trials with 1,995 patients and found high-platelet PRP delivered pain relief beyond MCID at 3, 6 and 12 months. Manufacturers that document platelet recovery and output consistency will have a clearer value case with physicians and hospitals.
Platelet Rich Plasma Market Definition
Platelet rich plasma market scope includes platelet-concentrated biologic preparations and related systems used in regenerative care. Products include pure platelet-rich plasma, leukocyte PRP, pure platelet-rich fibrin, and leukocyte PRF.
Platelet Rich Plasma Market Inclusions
Market scope includes PRP preparation kits, centrifuge-compatible systems, autologous PRP products, allogeneic PRP products, PRF formats, orthopedic use, aesthetics use, dental use, sports medicine use, wound healing use, hospitals, specialty clinics, ambulatory centers, trauma centers, research institutes, and all covered regions. Stakeholders include PRP kit manufacturers, biologic preparation system suppliers, orthopedic surgeons, dermatologists, wound care specialists, dental clinicians, sports medicine physicians, hospital purchasing teams, specialty clinic operators, and regenerative medicine distributors.
Platelet Rich Plasma Market Exclusions
Scope excludes standalone centrifuges not sold for PRP workflows, stem cell treatments, bone graft materials, viscosupplements, standard wound dressings, unrelated plasma products, and non-platelet biologic injectables.
Platelet Rich Plasma Market Research Methodology
- Primary Research: FMI analysts reviewed inputs from orthopedic surgeons, dermatologists, wound care clinicians, sports medicine physicians, and dental procedure specialists. Interviews covered preparation choice, patient payment behavior, clinic workflow, and supplier selection criteria.
- Desk Research: Desk research used regulatory references, clinical guidance, official company product data, FDA clearance records, and society-level procedure evidence. Review covered product scope, source type, preparation quality, and clinical use limits.
- Market Sizing and Forecasting: Forecast model combined PRP procedure volumes, kit pricing, country-level clinic penetration, and application-level use rates. Forecasts were checked against specialty care expansion and product availability across covered countries.
- Data Validation: Validation compared composition type demand, autologous use, and clinical channel patterns across primary interviews and public evidence. Company portfolios were checked before final taxonomy and competitive placement were set.
Why is the Platelet Rich Plasma Market Growing?
- Orthopedic specialists are using PRP in structured care pathways for knee pain and tendon repair support.
- Aesthetic clinics are adding PRP to hair restoration and skin rejuvenation protocols that often involve repeat visits.
- Chronic wound programs create demand for PRP products that match documentation and coverage needs.
- Closed preparation systems help clinics reduce handling risk during office-based procedures.
- Clinical evidence requirements are pushing suppliers to offer better preparation consistency and clearer protocol support.
Orthopedic demand faces more evidence-focused review as PRP moves through specialist review. Clinicians want biologic options that can support joint pain care and tendon repair without adding workflow burden. Hospitals and orthopedic clinics are placing higher weight on repeatable preparation quality. Product selection is likely to depend on platelet yield, ease of use, and clear patient selection guidance. Suppliers that can support training and procedure consistency will have better access to clinical accounts.
Aesthetic care gives PRP a separate growth path through repeat treatment models. Dermatology and hair restoration clinics can prepare PRP during the same visit and use it as part of physician-led services. Product use fits in outpatient rooms because it needs limited storage and fits short procedure windows. Clinics prefer systems that reduce staff steps and keep preparation quality steady. Compact PRP systems and closed kits gain a practical value case.
Market Segmentation Analysis
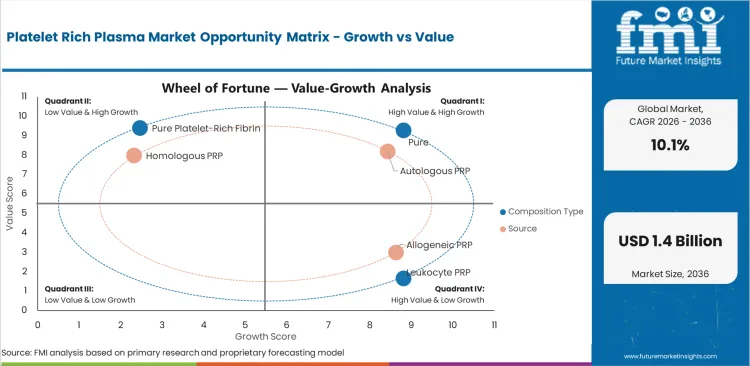
- Pure platelet-rich plasma is projected to hold 42.0% of composition type demand in 2026 as lower leukocyte content supports routine protocols.
- Autologous PRP is expected to represent 86.0% of source demand in 2026 with same-patient preparation.
- Orthopedics is estimated to account for 34.0% of application demand in 2026 as knee and tendon cases drive use.
- Specialty clinics are projected to contribute 38.0% of end user demand in 2026 as office-based procedures expand.
Market segmentation covers composition type, source, application, end user, and region. Composition type includes pure platelet-rich plasma, leukocyte PRP, pure platelet-rich fibrin, and leukocyte PRF. Source includes autologous PRP, allogeneic PRP, and homologous PRP. Application includes orthopedics, aesthetics, dentistry, sports medicine, and wound healing. End user includes hospitals, specialty clinics, ambulatory centers, trauma centers, and research institutes.
Insights into Pure Platelet-Rich Plasma in the Platelet Rich Plasma Market by Composition Type
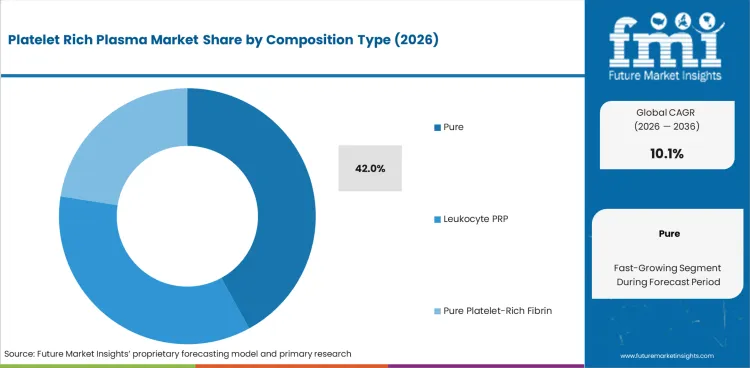
- Pure platelet-rich plasma is projected at 42.0% by 2026 as clinics prefer lower leukocyte content for repeat orthopedic and aesthetic procedures.
- Leukocyte PRP keeps selective use in sports medicine when clinicians need a more inflammatory preparation for specific soft tissue cases.
Insights into Autologous PRP in the Platelet Rich Plasma Market by Source
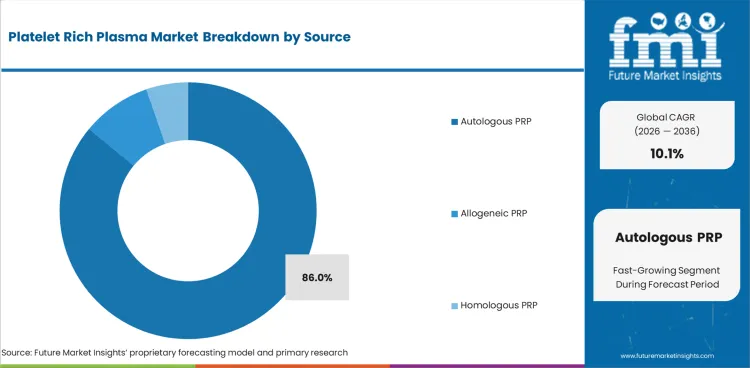
- Autologous PRP is expected to reach 86.0% in 2026 since same-patient preparation reduces donor handling and storage complexity.
- Allogeneic PRP stays more limited in the forecast period as donor screening and inventory control add steps for office-based clinics.
Insights into Orthopedics in the Platelet Rich Plasma Market by Application
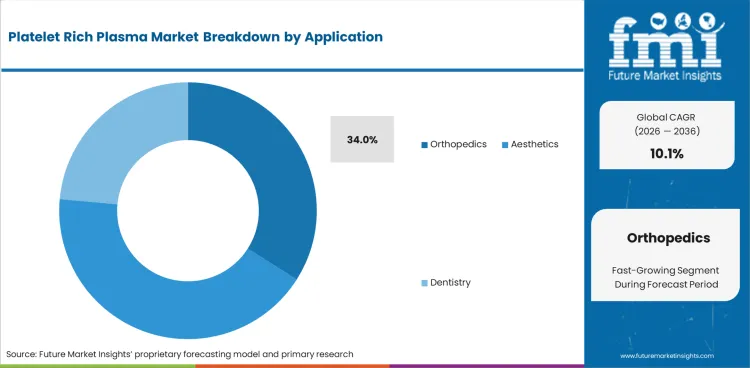
- By the base forecast year, orthopedics is estimated at 34.0% as knee care and tendon repair form the main clinical use base.
- Sports medicine overlaps with orthopedic care because active patients often need same-day biologic preparation after imaging review.
Insights into Specialty Clinics in the Platelet Rich Plasma Market by End User
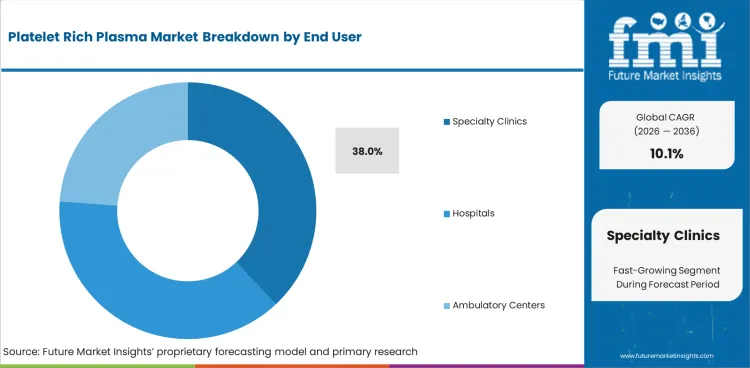
- Specialty clinics are projected at 38.0% during 2026 as PRP procedures fit outpatient regenerative care and dermatology rooms.
- Hospitals keep a defined role in wound care and orthopedic surgery as documentation and payer review often need formal clinical support.
Platelet Rich Plasma Market Drivers, Restraints, and Opportunities
- Orthopedic use creates demand for PRP systems that offer repeatable platelet preparation and clear procedure support.
- Aesthetic clinics prefer compact PRP systems that reduce handling steps during same-day skin and hair procedures.
- Wound care creates demand for PRP systems that support clean handling and patient-specific preparation.
- Reimbursement limits restrain wider PRP use when procedures fall outside clearly covered wound care pathways.
- FDA-cleared devices give suppliers clearer access through point-of-care systems and clear use indications.
Preparation Consistency Need
PRP adoption depends on preparation quality because clinicians compare systems by platelet yield and handling control. Arthrex, Inc. lists ACP, ACP Max and Angel as platelet-rich plasma processing systems for different procedure needs. Zimmer Biomet Inc. describes its GPS III Platelet Concentration System as a restorative therapy product that processes PRP from whole blood. Arthrex and Zimmer Biomet data show why supplier capability matters in clinical accounts. Clinics need systems that reduce variation between procedures and make staff training easier.
Point-of-Care Device Access
Point-of-care access matters as PRP procedures often occur inside outpatient treatment rooms. FDA’s 2025 biological device approvals list includes PRP-related systems intended for safe and rapid preparation from a patient’s own blood. FDA listings support the role of regulated device pathways in clinic confidence. Physicians need products with clear use scope before routine adoption. Suppliers that match device claims with clinic workflows can reduce review friction and improve account access.
Wound Care Product Path
Wound care gives PRP suppliers a more specific route when products address patient-specific preparation and chronic wound handling. In September 2025, Smith+Nephew launched the CENTRIO Platelet-Rich-Plasma System in the United States. Smith+Nephew describes CENTRIO as a biodynamic hematogel derived from a patient’s own platelets and plasma. CENTRIO launch shows how PRP suppliers connect product design with wound care workflow needs. Clinics will value products that simplify preparation and support consistent bedside use.
Analysis of Platelet Rich Plasma Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| United States | 7.0% |
| Canada | 8.2% |
| China | 11.4% |
| India | 12.6% |
| Germany | 7.8% |
| France | 9.4% |
| United Kingdom | 9.0% |
Source: FMI analysis based on primary research and proprietary forecasting model.
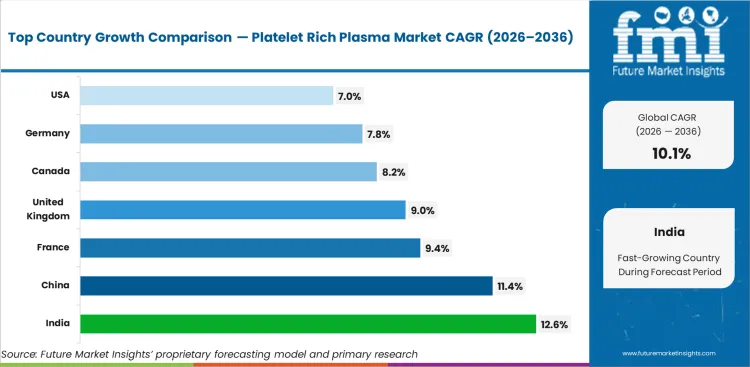
Platelet Rich Plasma Market CAGR Analysis by Country
- United States is projected to record a 7.0% CAGR through 2036 as payer limits keep non-wound PRP use selective.
- Canada is expected to expand at 8.2% CAGR through 2036 since private specialty clinics support gradual regenerative care use.
- China is forecast to grow at 11.4% CAGR through 2036 as urban hospitals add orthopedic and aesthetics procedure capacity.
- India is expected to advance at 12.6% CAGR through 2036 as private specialty clinics and self-pay procedures expand.
- Germany is projected to rise at 7.8% CAGR through 2036 as orthopedic use stays evidence-led and clinic protocols remain cautious.
- France is forecast at 9.4% CAGR through 2036 as outpatient regenerative care gains specialist familiarity.
- United Kingdom is expected to post 9.0% CAGR through 2036 as private clinics support PRP use despite evidence scrutiny.
Demand Outlook for Platelet Rich Plasma Market in the United States
United States demand is projected to grow at 7.0% CAGR through 2036. Non-wound PRP injections face payer scrutiny, so clinical use stays selective in many orthopedic settings. Aesthetic clinics continue to use PRP in hair and skin procedures because treatment can be prepared during the same visit. Suppliers need clear device positioning and practical training support to protect access in physician offices.
- CMS coverage rules create a clearer route for PRP use in chronic wound care.
- Orthopedic specialists use PRP more carefully when evidence review affects treatment choice.
- FDA device review keeps point-of-care preparation central for clinic confidence.
Sales Analysis of Platelet Rich Plasma Market in Canada
Canada’s platelet rich plasma market depends on specialty clinic use and private-pay care pathways. Canada is expected to expand at 8.2% CAGR in the forecast period as orthopedic and aesthetics procedures stay concentrated in private clinics. Office-based care supports PRP use because clinics can prepare small volumes during patient visits. Supplier success depends on training support and compact preparation systems.
- Ontario specialty clinics create demand through orthopedic and dermatology service access.
- British Columbia supports PRP use through sports medicine clinics in Vancouver.
- Quebec clinics need clear documentation and French-language training support.
Sales Analysis of Platelet Rich Plasma Market in China
China demand reflects hospital expansion and private aesthetics demand in large cities. Urban hospitals in Shanghai and Beijing support PRP use across orthopedic and dermatology settings. China is forecast to grow at 11.4% CAGR through 2036 as specialist procedure access widens. Suppliers with local registration support and physician training can progress faster through hospital review.
- Shanghai private hospitals support PRP use in aesthetics and hair restoration.
- Beijing tertiary hospitals influence orthopedic procedure standards and product review.
- Guangdong private clinics support office-based aesthetic treatment demand.
Opportunity Analysis of Platelet Rich Plasma Market in India
India has the stronger outlook due to private specialty clinics and self-pay regenerative procedures. India is expected to advance at 12.6% CAGR through 2036. Dermatology and orthopedic clinics in large cities are adding PRP to routine service menus. Same-patient preparation suits price-sensitive settings because it reduces storage and donor handling needs.
- Delhi NCR supports PRP demand through hair restoration and skin repair clinics.
- Bengaluru sports medicine clinics use PRP in musculoskeletal care for active patients.
- Mumbai private hospitals support higher-quality PRP systems for orthopedic use.
Future Outlook for Platelet Rich Plasma Market in Germany
Germany is projected to rise at 7.8% CAGR across the forecast timeframe. Specialist adoption is cautious because orthopedic and dermatology physicians place strong weight on clinical review. Demand depends more on product reliability than rapid clinic expansion. Suppliers must support documentation and staff training before larger clinic groups standardize PRP systems.
- Bavaria orthopedic centers support careful use of biologic treatments.
- Berlin aesthetic clinics use PRP within physician-led hair and skin care.
- North Rhine-Westphalia outpatient networks can support specialist procedure growth.
Demand Outlook for Platelet Rich Plasma Market in France
France combines specialist acceptance with careful clinical oversight in outpatient care. France is forecast at 9.4% CAGR between 2026 and 2036. Regenerative procedures are entering orthopedic and aesthetics clinics. Paris and Lyon support premium dermatology services that use autologous PRP in hair and skin applications.
- Paris dermatology clinics create steady PRP demand in hair restoration.
- Lyon orthopedic centers support musculoskeletal PRP use under specialist protocols.
- French outpatient clinics favor systems that simplify preparation and handling.
In-depth Analysis of Platelet Rich Plasma Market in the United Kingdom
United Kingdom industry shows steady demand through private clinics and evidence-conscious specialists. London is a large clinic hub for hair restoration and skin rejuvenation. PRP use continues in aesthetics and musculoskeletal care, with the market expected to post 9.0% CAGR by 2036. Suppliers need clear claims and training materials that match clinician scrutiny.
- London aesthetic clinics support PRP use in hair and skin treatment programs.
- Manchester sports medicine providers create demand for tendon care protocols.
- NHS evidence standards influence wider musculoskeletal use across private clinics.
Competitive Landscape and Strategic Positioning
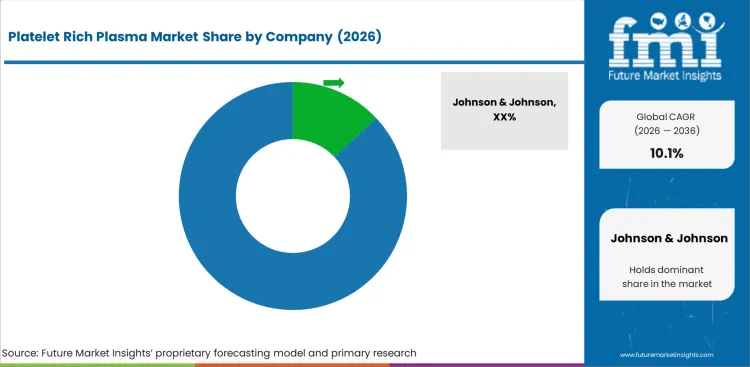
- Supplier base is moderately fragmented with global medical device suppliers and PRP-focused specialists.
- Large companies compete through orthopedic access, product breadth and hospital relationships.
- Specialist firms compete through closed-system preparation and clinician training.
- Entry barriers include FDA clearance, protocol evidence and specialty account access.
- Pricing pressure is high because many procedures are self-pay outside covered wound indications.
Competition is shaped by point-of-care speed and preparation consistency. Globus Medical positions the Harvest SmartPrep 3 System as an autologous biologic centrifuge platform for PRP and related cellular biologics. Globus Medical states that processing can take less than 15 minutes. Faster processing gives clinics a practical workflow advantage during outpatient regenerative procedures. Suppliers that reduce chair-time can compete better in specialty clinics.
Specialist suppliers compete through preparation control rather than broad device portfolios. Regen Lab SA states that RegenPRP uses a closed system and separator gel for reproducible leukocyte-reduced PRP. Regen Lab reports platelet recovery above 80.0%. Recovery data help physicians compare system output more clearly during supplier selection. Products with measurable recovery claims can gain preference in accounts that value repeatable preparation.
Key Companies in the Platelet Rich Plasma Market
Competitive base includes orthopedic companies, regenerative specialists, and PRP preparation system companies.
- Global Leaders: Johnson & Johnson, Zimmer Biomet Inc., Arthrex, Inc., and Terumo Corporation compete through medical device reach and procedural access.
- PRP Specialists: EmCyte Corporation, Regen Lab SA, and Dr. PRP America LLC compete through focused PRP preparation systems and clinical support.
- Regenerative Adjacent Players: CollPlant Biotechnologies Ltd., Nuo Therapeutics, Apex Biologix, and Celling Biosciences compete through wound care or regenerative procedure support.
Competitive Benchmarking: Platelet Rich Plasma Market
| Company | Preparation System Breadth | Clinical Protocol Support | Specialty Channel Access | Geographic Footprint |
|---|---|---|---|---|
| Johnson & Johnson | Medium | High | Strong | Global |
| Zimmer Biomet Inc. | High | High | Strong | Global |
| Arthrex, Inc. | High | High | Strong | Global |
| Terumo Corporation | Medium | High | Strong | Global |
| EmCyte Corporation | High | Medium | Moderate | North America |
| Regen Lab SA | High | High | Moderate | Europe, North America |
| Dr. PRP America LLC | Medium | Medium | Moderate | North America |
| CollPlant Biotechnologies Ltd. | Low | High | Moderate | Europe, Asia Pacific |
| Nuo Therapeutics | Medium | High | Moderate | United States |
| Apex Biologix | Medium | Medium | Moderate | North America |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on preparation system breadth, clinical protocol support, and specialty channel access.
Key Developments in Platelet Rich Plasma Market
- In May 2025, APEX Biologix entered an exclusive USA distribution agreement with Bioventus LLC for the XCELL PRP System across orthopedics and sports medicine.
- In April 2026, Royal Biologics announced FDA clearance for Fibrinet® PRF Wound Matrix for exuding cutaneous wound management.
Key Players in the Platelet Rich Plasma Market
Global Leaders
- Johnson & Johnson
- Zimmer Biomet Inc.
- Arthrex, Inc.
- Terumo Corporation
PRP Specialists
- EmCyte Corporation
- Regen Lab SA
- Dr. PRP America LLC
- Apex Biologix
Regenerative Adjacent Players
- CollPlant Biotechnologies Ltd.
- Nuo Therapeutics
- Celling Biosciences
- Royal Biologics
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 0.5 billion to USD 1.4 billion, at a CAGR of 10.1% |
| Market Definition | Platelet-concentrated biologic preparations and related systems used across regenerative care, orthopedic care, aesthetic care, dental procedures, sports medicine, and wound healing. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, China, India, Germany, France, United Kingdom, 30+ countries |
| Key Companies Profiled | Johnson & Johnson, Zimmer Biomet Inc., Arthrex, Inc., Terumo Corporation, EmCyte Corporation, Regen Lab SA, CollPlant Biotechnologies Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified procedure data and supplier portfolio checks. |
Platelet Rich Plasma Market by Segments
Platelet Rich Plasma Market Segmented by Composition Type
- Pure Platelet-Rich Plasma
- Leukocyte PRP
- Pure Platelet-Rich Fibrin
- Leukocyte PRF
Platelet Rich Plasma Market Segmented by Source
- Autologous PRP
- Allogeneic PRP
- Homologous PRP
Platelet Rich Plasma Market Segmented by Application
- Orthopedics
- Aesthetics
- Dentistry
- Sports Medicine
- Wound Healing
Platelet Rich Plasma Market Segmented by End User
- Hospitals
- Specialty Clinics
- Ambulatory Centers
- Trauma Centers
- Research Institutes
Platelet Rich Plasma Market by Region
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- APEX Biologix. (2025, June 2). APEX Biologix enters exclusive USA distribution agreement with Bioventus LLC for XCELL PRP® System.
- Royal Biologics. (2026, April 7). Royal Biologics announces FDA clearance for Fibrinet® PRF Wound Matrix for the management of cutaneous wounds.
- Arthrex, Inc. ( May 12, 2026). Platelet-rich plasma (PRP) processing systems.
- Zimmer Biomet Inc. ( May 12, 2026). GPS III Platelet Concentration System.
- USA Food & Drug Administration. (2026, January 16). 2025 biological device application approvals. FDA.
- Smith+Nephew. (2025, September 3). Smith+Nephew extends Advanced Wound Bioactives portfolio with US launch of CENTRIO Platelet-Rich-Plasma System.
- Globus Medical. ( May 12, 2026). Harvest™ SmartPrep™ 3 System.
- Regen Lab SA. ( May 12, 2026). RegenPRP™.
- Arthrex, Inc. (2025, June 30). Arthrex platelet-rich plasma processing solutions.
- Bensa, A., Previtali, D., Sangiorgio, A., Boffa, A., Salerno, M., & Filardo, G. (2025). PRP injections for the treatment of knee osteoarthritis: The improvement is clinically significant and influenced by platelet concentration: A meta-analysis of randomized controlled trials. The American Journal of Sports Medicine, 53(3), 745-754.
- Nguyen, L. V., & Nguyen, T. T. T. (2025, July 2). Effectiveness of autologous platelet-rich plasma for androgenetic alopecia: A double-center, non-controlled, randomized clinical study in Vietnam. Clinical, Cosmetic and Investigational Dermatology, 18, 1645-1656.
- OrthoAscent. (2024, September 5). FG-001 platelet rich plasma (PRP) device: 510(k) summary. USA Food and Drug Administration.
- Centers for Medicare & Medicaid Services. (2021, April 13). Blood-derived products for chronic non-healing wounds.
This Report Answers
- What is the current size and 2036 value of the platelet rich plasma market?
- How fast is the platelet rich plasma market expected to grow during the assessment period?
- Which composition type is projected to lead platelet rich plasma demand in 2026?
- Why does autologous PRP hold the leading source share in platelet rich plasma procedures?
- Which applications create the clearest growth path for platelet rich plasma suppliers?
- How do FDA clearances and CMS coverage rules influence platelet rich plasma adoption?
- Which countries are expected to record stronger growth in platelet rich plasma demand through 2036?
- Which companies hold the strongest platelet rich plasma system and channel positions?
- What products are included and excluded in the platelet rich plasma market scope?
- How does FMI estimate and validate the platelet rich plasma market forecast?
Frequently Asked Questions
What is the global market demand for platelet rich plasma in 2026?
In 2026, the global platelet rich plasma market is expected to be worth USD 0.5 billion based on FMI analysis.
How large will the platelet rich plasma market be by 2036?
By 2036, the platelet rich plasma market is projected to reach USD 1.4 billion as procedure use expands.
What CAGR is expected for the platelet rich plasma market?
Platelet rich plasma market is expected to grow at 10.1% CAGR during the forecast timeframe.
Which composition type is projected to lead platelet rich plasma demand in 2026?
Pure platelet-rich plasma is projected to account for 42.0% of composition type demand in 2026.
Which source segment is expected to lead platelet rich plasma demand in 2026?
Autologous PRP is expected to hold 86.0% of source demand in 2026 due to same-patient preparation.
Why is India the stronger-growth country for platelet rich plasma demand?
India is projected at 12.6% CAGR through 2036 as private clinics expand orthopedic and aesthetics procedures.
What does platelet rich plasma market mean in this report?
Market scope includes platelet-concentrated preparations and systems used in regenerative, orthopedic, aesthetic, dental, sports medicine, and wound care procedures.
How does FMI validate the platelet rich plasma market forecast?
FMI validates the forecast through primary interviews, procedure use checks, supplier portfolio review, and country-level adoption analysis.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Primary Modes
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Composition Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Composition Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Composition Type , 2026 to 2036
- Pure
- Leukocyte PRP
- Pure Platelet-Rich Fibrin
- Pure
- Y to o to Y Growth Trend Analysis By Composition Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Composition Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Source, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Source, 2026 to 2036
- Autologous PRP
- Allogeneic PRP
- Homologous PRP
- Autologous PRP
- Y to o to Y Growth Trend Analysis By Source, 2021 to 2025
- Absolute $ Opportunity Analysis By Source, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Orthopedics
- Aesthetics
- Dentistry
- Orthopedics
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Specialty Clinics
- Hospitals
- Ambulatory Centers
- Specialty Clinics
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Composition Type
- By Source
- By Application
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Composition Type
- By Source
- By Application
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Composition Type
- By Source
- By Application
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Composition Type
- By Source
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Johnson & Johnson
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Zimmer Biomet Inc.
- Arthrex, Inc.
- Terumo Corporation
- EmCyte Corporation
- Johnson & Johnson
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Composition Type , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Source, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Composition Type
- Figure 6: Global Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Source
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Composition Type
- Figure 29: North America Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Source
- Figure 32: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Application
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Composition Type
- Figure 42: Latin America Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Source
- Figure 45: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Application
- Figure 48: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by End User
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Composition Type
- Figure 55: Western Europe Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Source
- Figure 58: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Application
- Figure 61: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by End User
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Composition Type
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Source
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Application
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by End User
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Composition Type
- Figure 81: East Asia Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Source
- Figure 84: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Application
- Figure 87: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by End User
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Composition Type
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Source
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Composition Type , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Composition Type , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Composition Type
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Source, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Source, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Source
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

