Trichomonas Rapid Tests Market
Trichomonas Rapid Tests Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Trichomonas Rapid Tests Market Size and Share Forecast Outlook 2025 to 2035
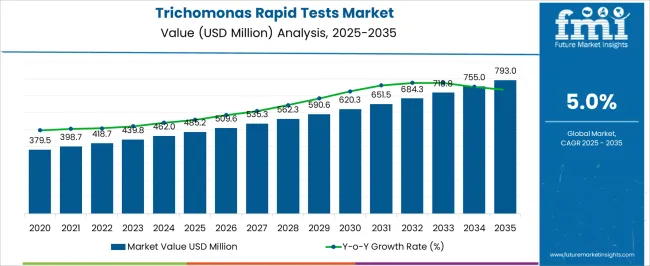
The Trichomonas Rapid Tests Market is estimated to be valued at USD 485.2 million in 2025 and is projected to reach USD 793.0 million by 2035, registering a compound annual growth rate (CAGR) of 5.0% over the forecast period.
Quick Stats for Trichomonas Rapid Tests Market
- Trichomonas Rapid Tests Market Industry Value (2025): USD 485.2 million
- Trichomonas Rapid Tests Market Forecast Value (2035): USD 793.0 million
- Trichomonas Rapid Tests Market Forecast CAGR: 5.0%
- Leading Segment in Trichomonas Rapid Tests Market in 2025: Cassette Rapid Test Kit (54.6%)
- Key Growth Region in Trichomonas Rapid Tests Market: North America, Asia-Pacific, Europe
- Top Key Players in Trichomonas Rapid Tests Market: Sekisui Diagnostics, Quidel Corporation, Roche Diagnostics, Home Health (UK) Ltd, Liming Bio, Orient Gene, Creative Diagnostics, Norgen Biotek Corp., Cepheid, BD, Cardinal Health, Jei Daniel Biotech Corp, Common Sense, nal von minden GmbH, Hologic, Inc., Abbott
| Metric | Value |
|---|---|
| Trichomonas Rapid Tests Market Estimated Value in (2025 E) | USD 485.2 million |
| Trichomonas Rapid Tests Market Forecast Value in (2035 F) | USD 793.0 million |
| Forecast CAGR (2025 to 2035) | 5.0% |
Rationale for Segmental Growth in the Trichomonas Rapid Tests Market
The trichomonas rapid tests market is experiencing steady growth due to the increasing prevalence of trichomoniasis, heightened awareness regarding sexually transmitted infections, and the demand for rapid and cost effective diagnostic solutions. The adoption of point of care testing is being encouraged by its ability to deliver accurate results within minutes, which supports timely treatment initiation and reduces transmission risks.
Advancements in assay design and sample handling techniques are further improving sensitivity and specificity, making these tests more reliable in both clinical and community settings. Public health initiatives and government screening programs are strengthening diagnostic coverage, particularly in high risk populations.
With rising emphasis on preventive healthcare and early detection, the market is positioned for continued expansion as accessibility and awareness continue to improve globally.
Segmental Analysis
Insights into the Cassette Rapid Test Kit Product Segment
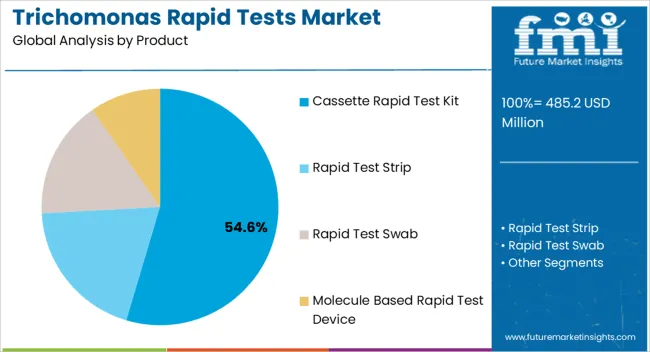
The cassette rapid test kit segment is projected to represent 54.60% of total revenue by 2025 within the product category, making it the dominant segment. Growth is driven by the simplicity of use, affordability, and minimal training requirements associated with cassette based kits.
Their compatibility with various healthcare settings, including clinics, hospitals, and outreach programs, further enhances adoption.
The ability to provide quick visual results without additional equipment strengthens their role as the preferred choice for frontline diagnostic applications.
Insights into the Vaginal or Urethra Swab Sample Type Segment
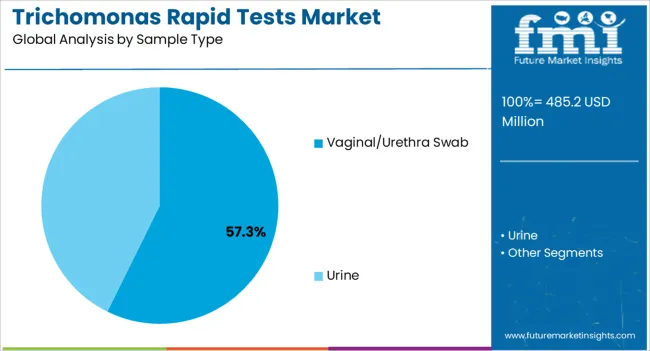
The vaginal or urethra swab segment is expected to account for 57.30% of total market revenue by 2025 within the sample type category, positioning it as the leading segment. Its dominance is attributed to the high accuracy and reliability of swab based collection methods in detecting trichomonas infections.
Ease of sample handling and consistency in results have supported its preference across clinical diagnostics.
The widespread use of swab samples in both hospital and outpatient settings ensures dependable detection rates, reinforcing their leadership in the sample type category.
Insights into the Hospitals End User Segment
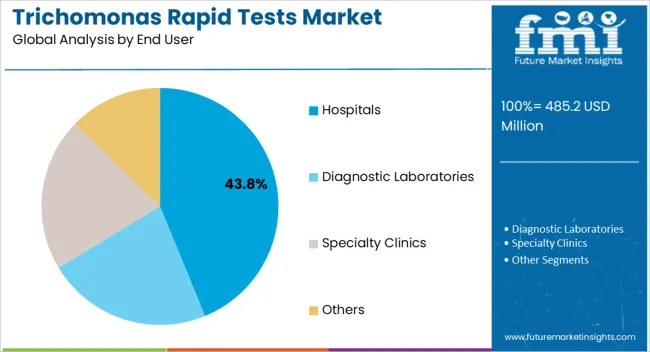
The hospitals segment is anticipated to hold 43.80% of total market revenue by 2025 within the end user category, making it the leading segment. This share is supported by the concentration of patient visits, availability of trained personnel, and the integration of rapid testing into routine diagnostic workflows.
Hospitals also serve as primary centers for disease screening and treatment, which amplifies their reliance on rapid diagnostic kits.
Their established infrastructure for infection control and laboratory testing further reinforces their role as the dominant end user in the trichomonas rapid tests market.
Sales Analysis of the Trichomonas Rapid Tests Market (2020 to 2024) vs Outlook for 2025 to 2035
The global market recorded a historic CAGR of 4.2% in the last 5 years from 2020 to 2025.
The global Trichomonas rapid test market contributes around 11.0% to the USD 485.2 Billion global Sexually Transmitted Disease (STI) diagnostics market in 2025.
Growing demand in healthcare settings is a prominent market driver for the trichomonas rapid tests market. Clinics, hospitals, and diagnostic laboratories all play an important role in the diagnosis and treatment of trichomoniasis.
To begin, healthcare providers understand the significance of early and accurate trichomoniasis diagnosis. Rapid testing yields speedy answers, allowing healthcare providers to make treatment decisions in real-time. These tests' ease and efficiency make them essential tools in busy healthcare environments, allowing for timely patient management.
Subsequently, the usage of rapid tests in healthcare settings corresponds to the larger trend of point-of-care testing (POCT). POCT stands for point-of-care testing, which is performed at or near the site of patient care, providing quick results and removing the need to transmit samples to a centralized laboratory.
Trichomonas rapid tests are ideal for POCT because they provide quick and accurate findings without requiring complex laboratory equipment or lengthy sample processing. This allows healthcare providers to diagnose and commence therapy during the patient's visit, improving overall healthcare efficiency.
Likewise, the rising prevalence of trichomoniasis and the resulting need for efficient management have increased the demand for quick diagnosis in healthcare settings. Trichomoniasis is a common sexually transmitted infection that, if left untreated, can have serious health repercussions, particularly in women. Healthcare practitioners to enable early discovery, effective treatment, and complication prevention are rapidly using rapid tests in diagnostic protocols.
Also, the adoption of trichomonas rapid tests in healthcare settings improves patient satisfaction. Rapid tests lessen waiting periods and anxiety associated with delayed diagnosis due to their speedy response time. Patients can receive rapid results and begin therapy as soon as possible, resulting in better overall patient experiences.
Ultimately, the increased demand for Trichomonas rapid tests in healthcare settings can be ascribed to the emphasis on early diagnosis, the adoption of POCT, the rising burden of trichomoniasis, and the emphasis on improving patient satisfaction and outcomes. These reasons are driving the market expansion for Trichomonas rapid testing in healthcare settings.
Opportunities Supporting the Growth of Trichomonas Rapid Tests Across Healthcare Settings
Diagnostic technology advances have had a profound impact on the global market for Trichomonas rapid testing. These developments have transformed the accuracy, sensitivity, and speed of trichomoniasis diagnostic testing. Nucleic acid amplification techniques, for example, have increased the detection capacities of Trichomonas tests, allowing for early and reliable diagnosis.
- According to the article Rapid and point-of-care tests for the diagnosis of Trichomonas vaginalis in women and men published by Sexually transmitted infections Journal on 10th December 2020, OSOM trichomonas rapid test is an easy to perform and CLIA-waived rapid test, with sensitivities of 83%-86% compared to nucleic acid amplification tests (NAATs) which is performed within 15 minutes.
AmpliVue and Solana tests are molecularly amplified and can be completed in less than an hour. Solana has a sensitivity of 100% for vaginal swabs and 90.7% for NAATs. GeneXpert TV is a moderately complex test that requires a small platform and can be performed in less than one hour, with excellent sensitivity and specificity for both men and women.
Further, many companies have been making efforts to reduce the cost of the device by collaborating with regional institutions and research laboratories. They are providing funds to grade their kits and devices capabilities and to back their technological advancements with scientific data seeking for various government and clinical approvals.
Likewise, the introduction of point-of-care devices and portable testing platforms has increased the accessibility and convenience of fast testing. These technical improvements have not only improved the performance of rapid tests, but have also contributed to their wider usage in a variety of healthcare settings, allowing for faster diagnosis and treatment of Trichomoniasis, which propels the market growth exponentially.
Restraining Factors Impacting the Demand for Trichomonas Rapid Tests Worldwide
The cost factor is a limitation for the Trichomonas rapid testing market. The cost of Trichomonas rapid tests can have an impact on their widespread usage, particularly in locations with low healthcare resources or no insurance coverage.
The expense of producing, delivering, and acquiring these tests may provide difficulties for both healthcare practitioners and patients. High-test fees may restrict access, particularly in low-income groups where affordability is a main concern.
Likewise, healthcare organizations and facilities may need to use their resources wisely, taking into account the cost-effectiveness of various diagnostic options. To limit the influence of cost concern as a market restraint, the availability of cost-effective Trichomonas rapid tests and the deployment of pricing mechanisms that ensure affordability is critical.
Analysis of Top Countries Enhancing Access to Trichomonas Rapid Testing Solutions
Why is the USA Market Booming?
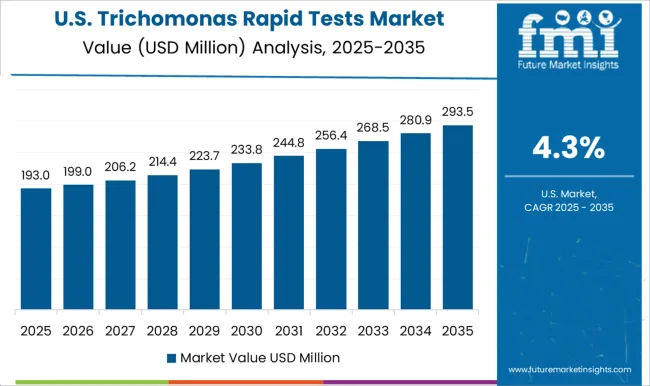
The USA dominates the global market with a market share of over 34.0% in 2025 and is projected to continue experiencing high growth throughout the forecast period.
The rising prevalence of trichomoniasis in the United States is a key market driver for trichomonas rapid tests. Trichomoniasis, a sexually transmitted infection caused by Trichomonas vaginalis, is on the rise, fueling demand for quick diagnostic testing.
According to a study published by Sex Transm Di, on 1st April 2024, Trichomonas vaginalis infections affected 379.5 million individuals aged 15-59, with 470,000 men and 2.1 million women, and incident infections were 6.9 million in 2020.
Changes in sexual practices, a lack of understanding, and a lack of prophylactic measures all contribute to the increased prevalence of trichomoniasis. The demand for Trichomonas rapid tests is driven by the necessity for early detection and treatment to avoid problems and slow transmission rates.
The increasing frequency of trichomoniasis in the United States emphasizes the need for improved diagnostic tools to combat this condition, which also propels the market growth significantly.
What Makes the Germany a Highly Lucrative Market for Trichomonas Rapid Tests Market?

Germany holds about 8.1% of the global market share in 2025.
The growing emphasis on preventative healthcare in Germany is boosting the demand for Trichomonas rapid testing. There is a rising emphasis on early detection and screening for sexually transmitted diseases (STIs), including trichomoniasis, as part of a proactive approach to healthcare.
Preventive healthcare programs and efforts encourage high-risk people to get regular testing, raising awareness about the need of early diagnosis and treatment.
As healthcare practitioners and individuals prioritize proactive methods to identify and manage trichomoniasis to prevent its spread and related health consequences, the demand for Trichomonas rapid tests is increasing in Germany.
What Makes China, an Emerging Market for Trichomonas Rapid Test?
China holds about 9.6% share in the global trichomonas rapid test market in 2025 and is projected to increase during the forecast period.
There is an increasing emphasis on women's healthcare in China, which is a significant market driver for the Trichomonas rapid tests. Women's particular health requirements and challenges, including the risk of trichomoniasis, are becoming better recognized.
This increased attention on women's healthcare has resulted in a higher emphasis on early detection and treatment of illnesses such as trichomoniasis to avoid problems. Healthcare practitioners and organizations are prioritizing screening and diagnostic interventions geared exclusively toward women.
Trichomonas quick tests provide a practical and efficient solution for timely diagnosis in women's healthcare settings, contributing to their rising usage and demand in China.
Trichomonas Rapid Tests Market Insights by Test Type and Application Segment
Which Product Segment Accounts for High Market Share Holding?
Rapid test strip hold a revenue share of 38.1% in 2025, and the same trend is being followed over the forecasted period.
Due to their simplicity, accessibility of use, and quick results, rapid test strips dominate the product category globally. Rapid test strips are a popular choice among healthcare practitioners and patients because they provide a convenient and cost-effective alternative for Trichomonas identification.
Which Sample Type Occupies a Dominant Share of the Market?
Vaginal/urethra swab holds a market share of 79.5% in 2025, within the forecasted period.
Due to its direct sampling method from the site of infection, convenience, ease of collection, and accurate detection of Trichomonas vaginalis, the vaginal/urethra swab are the most sought after sample segment globally.
Which End Users Occupies a Dominant Share of the Market?
Hospitals hold a market share of 36.7% in 2025, within the forecasted period.
Given their comprehensive healthcare facilities, availability of qualified employees, and capacity to provide immediate testing and treatment for patients, hospitals dominate the end user segment globally.
Competitive Landscape of the Trichomonas Rapid Tests Industry – Key Companies and Strategies
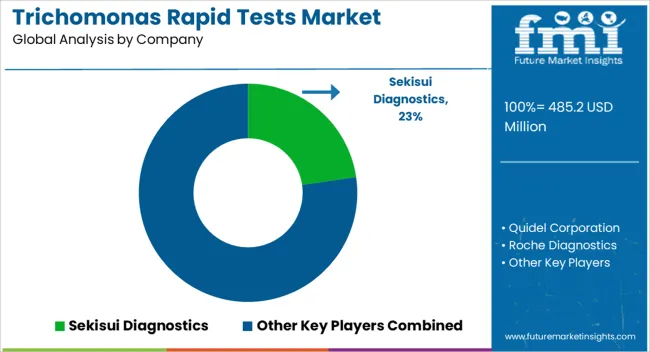
Advancements in the diagnostic technology for the trichomonas rapid tests and growing focus on geographical expansion by the acquisition of local market players are key strategies undertaken by leading firms. These strategies help companies to gain high revenue and expand their sales footprint in emerging countries
- On 6th March 2025, Cardinal Health and Signify Health collaborated to provide in-home clinical and medication management services, reducing costs and addressing care gaps for 2.3 million members nationwide.
- On 8 September 2025, Orient Gene Biotech's Rapid COVID-19 Antigen Self-Test and Drug of Abuse Urine Test Cup receive UK CTDA and FDA certifications.
Similarly, recent developments related to companies in Trichomonas rapid tests market have been tracked by the team at Future Market Insights, which are available in the full report.
Scope of the Report Based on Trichomonas Rapid Tests Market Framework and Coverage
| Attributes | Details |
|---|---|
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2020 to 2025 |
| Market Analysis | USD Million for Value, Units for Volume |
| Key Regions Covered | North America; Latin America; Europe; South Asia; East Asia; Oceania; and Middle East & Africa |
| Key Countries Covered | USA, Canada, Brazil, Mexico, Argentina, UK, Germany, Italy, Russia, Spain, France, BENELUX, India, Thailand, Indonesia, Malaysia, Japan, China, South Korea, Australia, New Zealand, Türkiye, GCC Countries, South Africa, and Northern Africa |
| Key Market Segments Covered | Product, Sample, End User and Region |
| Key Companies Profiled | Sekisui Diagnostics; Quidel Corporation; Roche Diagnostics; Home Health (UK) Ltd; Liming Bio; Orient Gene; Creative Diagnostics; Norgen Biotek Corp.; Cepheid, BD; cardinal Health; Jei Daniel Biotech Corp; Common Sense; nal von minden GmbH; Hologic, Inc.; Abbott |
| Pricing | Available upon Request |
Key Market Segments Covered in Trichomonas Rapid Tests Industry Research by Product Type, Sample Type, and End User
By Product:
- Cassette Rapid Test Kit
- Rapid Test Strip
- Rapid Test Swab
- Molecule Based Rapid Test Device
By Sample Type:
- Vaginal/Urethra Swab
- Urine
By End User:
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Others
By Region:
- North America
- Latin America
- Europe
- South Asia
- East Asia
- Oceania
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the trichomonas rapid tests market in 2025?
The global trichomonas rapid tests market is estimated to be valued at USD 485.2 million in 2025.
What will be the size of trichomonas rapid tests market in 2035?
The market size for the trichomonas rapid tests market is projected to reach USD 793.0 million by 2035.
How much will be the trichomonas rapid tests market growth between 2025 and 2035?
The trichomonas rapid tests market is expected to grow at a 5.0% CAGR between 2025 and 2035.
What are the key product types in the trichomonas rapid tests market?
The key product types in trichomonas rapid tests market are cassette rapid test kit, rapid test strip, rapid test swab and molecule based rapid test device.
Which sample type segment to contribute significant share in the trichomonas rapid tests market in 2025?
In terms of sample type, vaginal/urethra swab segment to command 57.3% share in the trichomonas rapid tests market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Mn) Analysis, 2020-2024
- Current and Future Market Size Value (USD Mn) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Trichomonas Rapid Tests Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By Product , 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By Product , 2025-2035
- Cassette Rapid Test Kit
- Rapid Test Strip
- Rapid Test Swab
- Molecule Based Rapid Test Device
- Y-o-Y Growth Trend Analysis By Product , 2020-2024
- Absolute $ Opportunity Analysis By Product , 2025-2035
- Global Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Sample Type
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By Sample Type, 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By Sample Type, 2025-2035
- Vaginal/Urethra Swab
- Urine
- Y-o-Y Growth Trend Analysis By Sample Type, 2020-2024
- Absolute $ Opportunity Analysis By Sample Type, 2025-2035
- Global Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By End User, 2025-2035
- Hospitals
- Diagnostic Laboratories
- Specialty Clinics
- Others
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Mn) Analysis By Region, 2020-2024
- Current Market Size Value (USD Mn) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- Latin America Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- Western Europe Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- Eastern Europe Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- East Asia Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- South Asia and Pacific Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- Middle East & Africa Trichomonas Rapid Tests Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Sample Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Sample Type
- By End User
- Key Takeaways
- Key Countries Trichomonas Rapid Tests Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Product
- By Sample Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Sample Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Sekisui Diagnostics
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Quidel Corporation
- Roche Diagnostics
- Home Health (UK) Ltd
- Liming Bio
- Orient Gene
- Creative Diagnostics
- Norgen Biotek Corp.
- Cepheid
- BD
- Cardinal Health
- Jei Daniel Biotech Corp
- Common Sense
- nal von minden GmbH
- Hologic, Inc.
- Abbott
- Sekisui Diagnostics
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Region, 2020-2035
- Table 2: Global Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 3: Global Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 4: Global Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 5: North America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 6: North America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 7: North America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 8: North America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 9: Latin America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 10: Latin America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 11: Latin America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 12: Latin America Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 13: Western Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 14: Western Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 15: Western Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 16: Western Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 17: Eastern Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 18: Eastern Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 19: Eastern Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 20: Eastern Europe Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 21: East Asia Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 22: East Asia Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 23: East Asia Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 24: East Asia Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 25: South Asia and Pacific Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 26: South Asia and Pacific Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 27: South Asia and Pacific Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 28: South Asia and Pacific Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
- Table 29: Middle East & Africa Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Country, 2020-2035
- Table 30: Middle East & Africa Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Product , 2020-2035
- Table 31: Middle East & Africa Trichomonas Rapid Tests Market Value (USD Mn) Forecast by Sample Type, 2020-2035
- Table 32: Middle East & Africa Trichomonas Rapid Tests Market Value (USD Mn) Forecast by End User, 2020-2035
List of Figures
- Figure 1: Global Trichomonas Rapid Tests Market Pricing Analysis
- Figure 2: Global Trichomonas Rapid Tests Market Value (USD Mn) Forecast 2020–2035
- Figure 3: Global Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 4: Global Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 5: Global Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 6: Global Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 7: Global Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 8: Global Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 9: Global Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: Global Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 11: Global Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 12: Global Trichomonas Rapid Tests Market Value (USD Mn) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: Global Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Region, 2025–2035
- Figure 14: Global Trichomonas Rapid Tests Market Attractiveness Analysis by Region
- Figure 15: North America Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 16: Latin America Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 17: Western Europe Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 18: Eastern Europe Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 19: East Asia Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 20: South Asia and Pacific Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 21: Middle East & Africa Trichomonas Rapid Tests Market Incremental Dollar Opportunity, 2025–2035
- Figure 22: North America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 23: North America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 24: North America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 25: North America Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 26: North America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 27: North America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 28: North America Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 29: North America Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 30: North America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 31: North America Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 32: Latin America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 33: Latin America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 34: Latin America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 35: Latin America Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 36: Latin America Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 37: Latin America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 38: Latin America Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 39: Latin America Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 40: Latin America Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 41: Latin America Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 42: Western Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 43: Western Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 44: Western Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 45: Western Europe Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 46: Western Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 47: Western Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 48: Western Europe Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 49: Western Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 50: Western Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 51: Western Europe Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 53: Eastern Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 54: Eastern Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 55: Eastern Europe Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 56: Eastern Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 57: Eastern Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 58: Eastern Europe Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 59: Eastern Europe Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 60: Eastern Europe Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 61: Eastern Europe Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 62: East Asia Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 63: East Asia Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 64: East Asia Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 65: East Asia Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 66: East Asia Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 67: East Asia Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 68: East Asia Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 69: East Asia Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 70: East Asia Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 71: East Asia Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 73: South Asia and Pacific Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 74: South Asia and Pacific Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 75: South Asia and Pacific Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 76: South Asia and Pacific Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 77: South Asia and Pacific Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 78: South Asia and Pacific Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 79: South Asia and Pacific Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 80: South Asia and Pacific Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 81: South Asia and Pacific Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Trichomonas Rapid Tests Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 83: Middle East & Africa Trichomonas Rapid Tests Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 84: Middle East & Africa Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Product , 2025–2035
- Figure 85: Middle East & Africa Trichomonas Rapid Tests Market Attractiveness Analysis by Product
- Figure 86: Middle East & Africa Trichomonas Rapid Tests Market Value Share and BPS Analysis by Sample Type, 2025 and 2035
- Figure 87: Middle East & Africa Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by Sample Type, 2025–2035
- Figure 88: Middle East & Africa Trichomonas Rapid Tests Market Attractiveness Analysis by Sample Type
- Figure 89: Middle East & Africa Trichomonas Rapid Tests Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 90: Middle East & Africa Trichomonas Rapid Tests Market Y-o-Y Growth Comparison by End User, 2025–2035
- Figure 91: Middle East & Africa Trichomonas Rapid Tests Market Attractiveness Analysis by End User
- Figure 92: Global Trichomonas Rapid Tests Market – Tier Structure Analysis
- Figure 93: Global Trichomonas Rapid Tests Market – Company Share Analysis

