Rapid Microbiology Testing Market Size and Share Forecast Outlook From 2025 to 2035
The rapid microbiology testing market is experiencing robust expansion driven by increasing demand for faster diagnostic outcomes, stringent regulatory requirements for product safety, and growing adoption across clinical and industrial applications. Market growth is supported by advancements in molecular diagnostics, automation, and data integration technologies that enhance accuracy and turnaround time.
Rising incidences of infectious diseases and heightened focus on early detection are fueling the need for reliable testing solutions. The current landscape is characterized by continuous investment in research and development aimed at improving assay sensitivity and operational efficiency.
Future growth is expected to be influenced by expanding healthcare infrastructure in emerging markets and broader implementation of quality assurance frameworks across industries such as pharmaceuticals, food safety, and environmental monitoring The overall growth rationale is grounded in the market’s capacity to deliver precise, rapid, and scalable testing methods that support both patient care and compliance requirements, ensuring consistent adoption and sustained revenue expansion over the forecast period.

Quick Stats for Rapid Microbiology Testing Market
- Rapid Microbiology Testing Market Industry Value (2025): USD 5.5 billion
- Rapid Microbiology Testing Market Forecast Value (2035): USD 13.4 billion
- Rapid Microbiology Testing Market Forecast CAGR: 9.4%
- Leading Segment in Rapid Microbiology Testing Market in 2025: Reagents and Kits (46.8%)
- Key Growth Region in Rapid Microbiology Testing Market: North America, Asia-Pacific, Europe
- Top Key Players in Rapid Microbiology Testing Market: BioMerieux SA (France), Abbott. (USA), BD (USA), Bruker (USA), Charles River (USA), Danaher (USA), Don Whitley Scientific Limited (USA), Merck KGaA, Neogen Corporation (USA)

| Metric |
Value |
| Rapid Microbiology Testing Market Estimated Value in (2025 E) |
USD 5.5 billion |
| Rapid Microbiology Testing Market Forecast Value in (2035 F) |
USD 13.4 billion |
| Forecast CAGR (2025 to 2035) |
9.4% |
Segmental Analysis
The market is segmented by Product, Method, Applications, and End Users and region. By Product, the market is divided into Reagents and Kits, Instruments, and Consumables. In terms of Method, the market is classified into Nucleic Acid-Based Rapid Microbiology Testing, Growth-Based Rapid Microbiology Testing, Cellular component-Based Rapid Microbiology Testing, and Viability-Based Rapid Microbiology Testing.
Based on Applications, the market is segmented into Clinical Disease Diagnostics, Food & Beverage Testing, Pharmaceutical & Biological Drug Testing, Environmental Testing, Cosmetics And Personal Care Products Testing, Research Applications, and Other Applications.
By End Users, the market is divided into Laboratories And Hospitals, Food And Beverage Companies, Pharmaceutical and Biotechnology Companies, Contract Research Organizations, and Other End-Users. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Product Segment
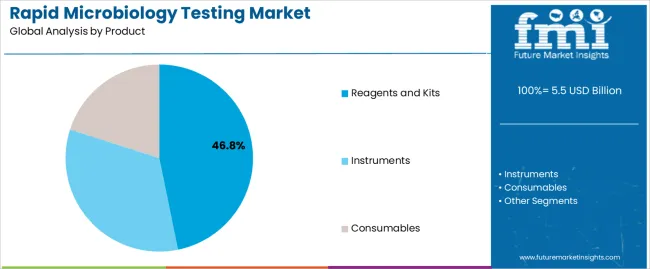
The reagents and kits segment, holding 46.80% of the product category, has established dominance due to its essential role in facilitating rapid, accurate, and cost-effective testing processes. Continuous improvements in reagent formulation and kit design have enhanced sensitivity, reduced error rates, and optimized user convenience.
Demand has been supported by increased testing volumes in hospitals, diagnostic centers, and pharmaceutical manufacturing facilities. The segment benefits from recurring consumable purchases that ensure stable revenue generation for market players.
Integration of ready-to-use reagent kits with automated systems has streamlined laboratory workflows, reducing turnaround times and supporting compliance with quality standards Ongoing product innovation, coupled with expanding distribution networks and heightened awareness regarding infection control, is expected to sustain the segment’s leadership and drive continued growth across diverse application domains.
Insights into the Method Segment
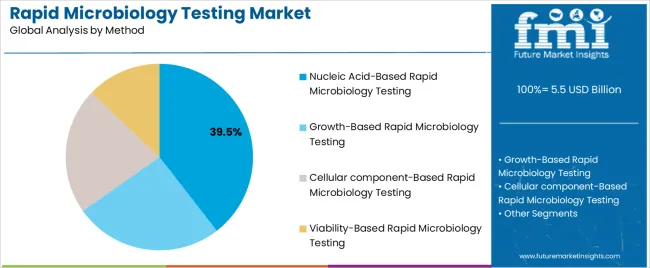
The nucleic acid-based rapid microbiology testing segment, accounting for 39.50% of the method category, has gained prominence due to its superior precision, rapid detection capability, and ability to identify pathogens at the genetic level. Adoption has been fueled by technological advancements in polymerase chain reaction (PCR), quantitative nucleic acid amplification, and next-generation sequencing platforms.
The method’s high sensitivity and specificity have made it a preferred choice for critical diagnostic applications where early and accurate detection is essential. Increased investment in molecular testing infrastructure and ongoing cost reductions in reagent production are further enhancing accessibility.
Regulatory support for molecular diagnostics and the integration of digital data management systems are strengthening operational reliability The segment’s continued evolution is expected to sustain its market share, supporting broader clinical and industrial applications.
Insights into the Applications Segment
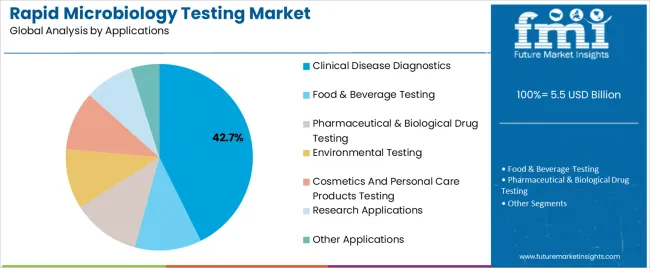
The clinical disease diagnostics segment, representing 42.70% of the applications category, has maintained its leadership owing to the increasing global burden of infectious diseases and the necessity for rapid pathogen identification. Its growth has been driven by the demand for early diagnosis, patient management efficiency, and infection control in hospital settings.
Integration of rapid microbiology testing into routine clinical workflows has enhanced treatment precision and reduced hospitalization durations. The segment has also benefited from government initiatives promoting disease surveillance and advanced diagnostic capabilities.
Ongoing technological developments, combined with training programs for laboratory professionals, have improved test reliability and operational proficiency Expanding healthcare access in developing regions and continued public health investments are expected to reinforce the segment’s dominance and ensure long-term growth momentum in the global market.
Top Dynamics and Industry Trends Shaping the Rapid Microbiology Testing Market
- Growing consumer awareness leads to stricter food safety regulations, necessitating rapid microbiology testing to ensure food product safety and quality assurance.
- The increasing prevalence of infectious diseases necessitates rapid diagnostics for prompt treatment and outbreak control, presenting significant public health challenges.
- Regulatory bodies are imposing stringent standards on pharmaceuticals, medical devices, and food products, which drives the adoption of rapid microbiology testing methods.
- Technological advancements such as automation, miniaturization, PCR, and next-generation sequencing enhance the speed, accuracy, and efficiency of microbiology testing.
- Rapid microbiology testing methods prove to be cost-effective, facilitate global food supply chain management, and play a crucial role in outbreak control.
- Government initiatives and pharmaceutical manufacturing benefit from rapid testing, enabling faster product release, reduced cycle times, and enhanced process efficiency.
- Rising awareness among consumers, industry stakeholders, and healthcare professionals fuels the adoption of microbiology testing for quality assurance and risk management strategies.
Lucrative Opportunities Transforming the Rapid Microbiology Testing Market
- Rapid microbiology testing companies have the potential to expand into emerging markets where awareness of the importance of testing is growing and healthcare infrastructure is improving.
- Investing in research and development to create innovative technologies can open up new market opportunities and differentiate companies from their competitors.
- Customizing solutions for different industries, such as food and beverage, pharmaceuticals, healthcare, and environmental monitoring, can help capture niche markets and foster long-term partnerships.
- Integrating with digital health platforms can streamline data management and reporting processes, thereby enhancing healthcare providers' value proposition and improving patient outcomes.
- Strategic partnerships with other companies, research institutions, and regulatory agencies can facilitate knowledge sharing and accelerate product development cycles.
- Point-of-care testing solutions can be developed for various healthcare settings, while regulatory compliance solutions can assist companies in navigating complex requirements.
- Rapid microbiology testing can address public health challenges by developing diagnostic tests and surveillance systems.
- Expanding applications in food safety testing can aid in the detection of foodborne pathogens, spoilage organisms, and allergens.
Factors Restraining the Demand for Rapid Microbiology Testing Solutions
- Rapid microbiology testing methods are complex and require specialized equipment, trained personnel, and intricate data analysis procedures.
- Validation and standardization processes can be challenging, especially when dealing with complex sample matrices and diverse microbial populations.
- Meeting regulatory requirements can be time-consuming and expensive, particularly in highly regulated industries.
- Cost considerations may be higher than those of traditional culture-based methods, which can pose barriers to adoption.
- Sample preparation is critical, and optimizing protocols can be challenging. Inhibitors may interfere with certain methods, leading to false-positive or false-negative results.
- Sensitivity and specificity may be limited compared to traditional culture-based methods.
- Data analysis and interpretation can be challenging, and training personnel to effectively use these methods may require significant time and resources.
- Resistance to change and skepticism about the reliability of rapid microbiology testing methods may hinder their widespread adoption.
Rapid Microbiology Testing Industry Analysis by Top Investment Segments
Instrumentation Emerges as Market Frontrunner, Spearheading Product Categories
| Attributes |
Details |
| Product |
Instruments |
| CAGR of ( 2025 to 2035) |
9.1% |
- Instruments are essential for scientific and industrial applications due to their precision and accuracy in measurements.
- Instruments are utilized across various sectors such as healthcare, manufacturing, research, and environmental monitoring.
- Continuous advancements in instruments enhance their capabilities, thereby improving efficiency and reliability.
- Instruments are crucial for quality control as they provide precise data and measurements.
- Many industries require instruments that adhere to safety and quality assurance regulatory standards.
- Instruments play a crucial role in process optimization, enhancing productivity and cost-efficiency.
Strategic Dissection Elevates Clinical Disease Diagnosis to Premier Application Status
| Attributes |
Details |
| Top Application |
Clinical Disease Diagnosis |
| CAGR of ( 2025 to 2035) |
8.8% |
- This segment focuses on diagnosing patients' diseases through various diagnostic tests and procedures. Accurate diagnosis is vital for successfully implementing treatment and patient care.
- Early diagnosis is crucial for prompt intervention and preventive measures, leading to improved patient outcomes.
- Clinical disease diagnosis frequently involves the use of laboratory tests such as blood tests, imaging studies, and genetic testing.
- The segment is witnessing a surge in rapid testing methods for swift and precise diagnosis, particularly in urgent care facilities.
- The integration of technology enhances diagnostic capabilities, enabling more sophisticated and efficient disease diagnosis.
- Point-of-care testing devices enable rapid diagnosis at the bedside or in the clinic, facilitating prompt decision-making.
- The clinical disease diagnosis segment is experiencing substantial growth due to the rise in healthcare demands and advancements in diagnostic technologies.
Analysis of Top Countries Certifying, Conducting, Distributing, and Using Rapid Microbiology Testing Technologies
| Countries |
Forecasted CAGR from 2025 to 2035 |
| United States |
9.7% |
| United Kingdom |
10.8% |
| China |
8.6% |
| Japan |
10.6% |
| South Korea |
11.7 % |
Commitment to Innovation and Regulatory Adaptations Define the United States Rapid Microbiology Testing Market
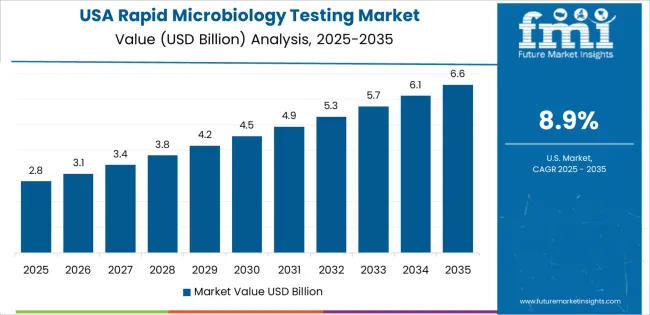
The rapid microbiology testing market in the United States is experiencing steady growth, with a CAGR of 9.7% from 2025 to 2035.
- The market is driven by innovative technologies, regulatory adaptations, and diverse industry applications.
- The United States market is a trailblazer in providing rapid microbiology testing solutions, influencing international standards and best practices.
- The market is characterized by a commitment to innovation, regulatory adaptations, and a diverse range of industries, including healthcare, pharmaceuticals, and food and beverage.
Resilient Regulatory Environment Spurs Competitiveness in United Kingdom's Rapid Microbiology Testing Market
The United Kingdom is experiencing a 10.8% CAGR in the rapid microbiology testing market, showcasing its agility in adopting advanced testing solutions.
- The regulatory landscape has created a resilient environment for rapid microbiology testing solutions, encouraging market competitiveness.
- Investment opportunities are emerging, fostering partnerships and collaborations that drive innovation and market growth.
- The United Kingdom is also promoting cross-industry collaborations, leveraging rapid microbiology testing solutions in healthcare, agriculture, and environmental sectors.
- The United Kingdom's commitment to quality and innovation has garnered international recognition, leading to global collaborations.
Government Backing and Regulatory Vigilance Propel China's Microbiology Market Forward
- China is a leading technological innovation hub in microbiology testing, with investments and advancements positioned at the forefront of cutting-edge solutions.
- Market growth in rapid microbiology testing extends its global influence, shaping industry standards and contributing to international collaborations.
- Government support and a proactive regulatory environment are crucial for propelling the market forward.
- China's market diversifies its applications, addressing challenges in healthcare, biotechnology, and food safety with tailored solutions.
Japan Sets High Quality Standards and Harmonizes Global Practices in Microbiology Testing
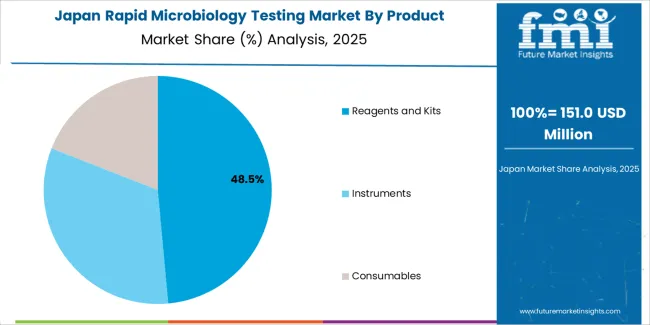
- Japan's market resurgence is driven by innovative technologies, global collaboration, and environmental considerations in microbiology testing.
- The country ensures high product quality standards and actively collaborates on a global scale, contributing to the harmonization of standards.
- It also incorporates environmental considerations into its testing practices, aligning market growth with sustainability goals.
- Strategic industry partnerships further support Japan's market growth, promoting knowledge exchange and collective advancements in microbiology testing.
Export-oriented Rapid Microbiology Testing Propels South Korea's Influence on Global Trade

- South Korea's innovation ecosystem is thriving, fostering breakthroughs in microbiology testing technologies and solidifying its position as a global market player.
- The export-driven microbiology market contributes to global trade networks and influences international market dynamics.
- South Korea is at the forefront of digital transformation in microbiology, enhancing efficiency and accuracy.
- This rapid growth has regional implications, fostering local stability and contributing to global advancements.
Key Rapid Microbiology Testing Market Players and Concentration
The rapid microbiology testing market is shaped by technological advancements, regulatory compliance, strategic alliances, and market segmentation.
- Key players dominate the market with innovative products and extensive application areas.
- Competition is driven by technological advancements such as automated systems and molecular diagnostics, while regulatory frameworks shape market strategies.
- Market segmentation, based on application areas, testing methods, and geographic regions, defines competition.
- Companies must consider pricing strategies, customer preferences, and research investment to maintain competitiveness.
- Emerging trends like point-of-care testing and antimicrobial resistance foster competitive resilience.
Recent Development Rapid Microbiology Testing market
- In November 2025, IDEXX acquired Tecta-PDS, a Canada-based company specializing in automating water microbiology testing processes, thereby expanding its portfolio of solutions for laboratory-based and in-field applications.
- In October 2024, Merck KGAA and Mammoth Biosciences partnered to develop and commercialize a CRISPR-based diagnostic test for SARS-CoV-2, leveraging their strengths to create innovative diagnostic solutions.
- In September 2024, Bruker Corporation acquired Canopy Bioscience LLC, a United States-based company specializing in multi-omics and fluorescence-based imaging techniques, enhancing its market position and capabilities.
Leading Suppliers of Rapid Microbiology Testing Technologies
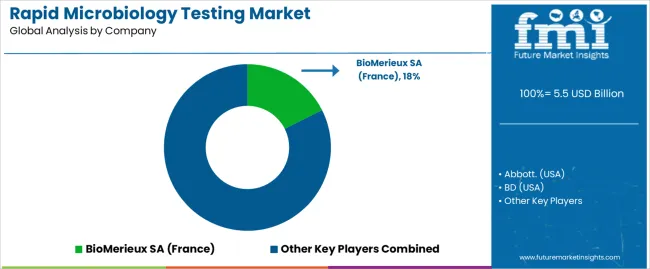
- Abbott. (USA)
- BD (USA)
- BioMerieux SA (France)
- Bruker (USA)
- Charles River (USA)
- Danaher (USA)
- Don Whitley Scientific Limited (USA)
- Merck KGaA
- Neogen Corporation (USA)
Top Segments Studied in the Rapid Microbiology Testing Market Report
By Product:
- Instruments
- Reagents and Kits
- Consumables
By Method:
- Growth-Based Rapid Microbiology Testing
- Cellular component-Based Rapid Microbiology Testing
- Nucleic Acid-Based Rapid Microbiology Testing
- Viability-Based Rapid Microbiology Testing
By Applications:
- Clinical Disease Diagnostics
- Food & Beverage Testing
- Pharmaceutical & Biological Drug Testing
- Environmental Testing
- Cosmetics and Personal Care Products Testing
- Research Applications
- Other Applications
By End Users:
- Laboratories and Hospitals
- Food and Beverage Companies
- Pharmaceutical and Biotechnology Companies
- Contract Research Organizations
- Other End-Users
By Region:
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the rapid microbiology testing market in 2025?
The global rapid microbiology testing market is estimated to be valued at USD 5.5 billion in 2025.
What will be the size of rapid microbiology testing market in 2035?
The market size for the rapid microbiology testing market is projected to reach USD 13.4 billion by 2035.
How much will be the rapid microbiology testing market growth between 2025 and 2035?
The rapid microbiology testing market is expected to grow at a 9.4% CAGR between 2025 and 2035.
What are the key product types in the rapid microbiology testing market?
The key product types in rapid microbiology testing market are reagents and kits, instruments and consumables.
Which method segment to contribute significant share in the rapid microbiology testing market in 2025?
In terms of method, nucleic acid-based rapid microbiology testing segment to command 39.5% share in the rapid microbiology testing market in 2025.









