The biological indicator vial market in the United Kingdom is projected to grow from USD 18.4 million in 2026 to USD 29.5 million by 2036, at a CAGR of 4.8%. Cost structures in this market will be closely tied to manufacturing precision, with stringent quality standards required for biological indicator production. Suppliers must manage high production costs, especially as they scale operations to meet growing demand. Pricing control will be key, as companies that can maintain competitive yet profitable pricing will be better positioned to secure contracts with major healthcare providers.
However, margin concentration will increase as larger players with established production facilities and stronger relationships with regulatory bodies dominate the space. Smaller firms may struggle to maintain profitability without achieving economies of scale. The companies that capture value will not only innovate in product development but also optimize their cost structures, ensuring long-term sustainability and strong positioning in a competitive market.
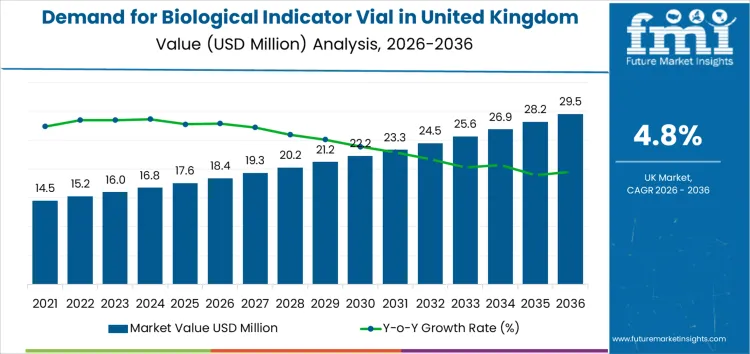
Demand for biological indicator vials in the United Kingdom is expected to grow steadily through the forecast period, supported by expanding sterilization validation requirements across healthcare, biotechnology, and pharmaceutical operations. As hospitals, medical device manufacturers, and life science facilities increase focus on infection prevention and quality assurance, biological indicators remain essential for verifying sterilization effectiveness and ensuring safe processing of critical instruments and products. Growth is also reinforced by the rising volume of sterilization cycles in clinical and laboratory settings.
This outlook reflects the stable and predictable nature of demand in regulated environments where sterilization validation is mandatory. Continued expansion of the UK’s healthcare services and life sciences manufacturing base will strengthen consumption, particularly as facilities adopt more advanced sterilization methods and upgrade compliance workflows. Stricter regulatory oversight, growing use of sterile medical products, and increasing emphasis on traceable documentation will further support adoption. Overall, biological indicator vials will maintain consistent long-term demand, driven more by regulatory compliance, patient safety standards, and industry quality systems than by short-term fluctuations in healthcare spending.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 18.4 million |
| Industry Forecast Value (2036) | USD 29.5 million |
| Industry Forecast CAGR (2026 to 2036) | 4.8% |
Demand for biological indicator vials in the United Kingdom has increased as healthcare facilities, sterilisation service providers, and life science laboratories responded to heightened expectations for sterilisation assurance and regulatory compliance. Historically, biological indicators were used primarily in central sterile services departments (CSSDs) to validate sterilisation cycles for surgical instruments and critical medical devices. Over time, the range of applications expanded into pharmaceutical manufacturing, tissue banks, and research institutions where validated sterilisation is essential for product safety and process control.
Infection control protocols within NHS trusts and private healthcare networks emphasise rigorous monitoring of sterilisation performance, which drives routine use of biological indicator vials in steam, ethylene oxide, and dry heat sterilisation cycles. Sterilisation assurance officers and quality managers in these environments value biological indicator vials for their ability to detect the presence of resistant spores and confirm effective microbial inactivation, which complements physical and chemical indicator systems.
Looking ahead, demand for biological indicator vials in the United Kingdom is expected to continue rising as regulatory frameworks and quality standards evolve and as facility managers prioritise patient safety and operational efficiency. Providers of sterilisation services and contract manufacturers supporting medical device and pharmaceutical clients will require reliable indicator solutions that integrate with standard operating procedures and audit documentation. Procurement teams will assess offerings based on indicator performance, expiry stability, and ease of integration with automated steriliser tracking systems. Suppliers that provide comprehensive training on indicator interpretation and compliance documentation will support buyers tasked with demonstrating adherence to ISO and MHRA expectations. Expansion of complex healthcare services, increasing volumes of reusable medical equipment, and focus on validated sterilisation outcomes position biological indicator vials as essential tools for quality assurance and risk mitigation across UK clinical and manufacturing operations.
Demand for biological indicator vials in the United Kingdom is influenced by sterilization type and incubation time. Steam sterilization leads the sterilization type segment with 51.3%, while the 24 to 32 hours incubation time dominates the incubation time segment with 45%. Other sterilization types such as ethylene oxide sterilization, and incubation times such as up to 24 hours, 32 to 48 hours, and above 48 hours, also contribute to the market. These segments highlight the growing use of biological indicators for ensuring effective sterilization processes in healthcare, pharmaceutical, and manufacturing industries.
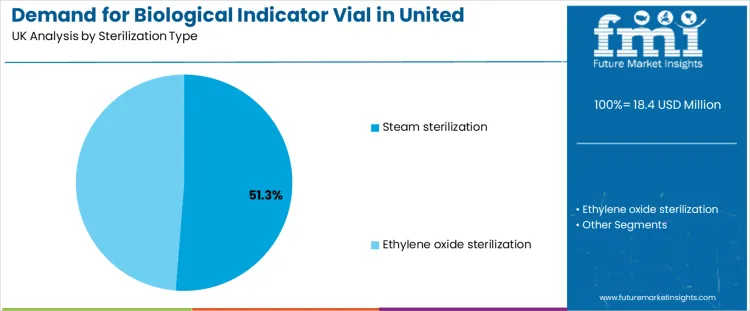
The sterilization type segment for biological indicator vials in the United Kingdom is primarily led by steam sterilization, which accounts for 51.3% of the market share. Steam sterilization, also known as autoclaving, is one of the most common sterilization methods used across healthcare settings, laboratories, and pharmaceutical manufacturing. It is highly effective in killing microorganisms using high-pressure steam at elevated temperatures, making it a preferred method for sterilizing equipment, tools, and materials that can withstand heat. Biological indicator vials are used to monitor and validate the effectiveness of the steam sterilization process, ensuring that the sterilization cycle has been adequately completed.
The demand for biological indicator vials in steam sterilization is driven by its widespread application, particularly in hospitals, medical device manufacturing, and pharmaceutical production. Ethylene oxide sterilization follows as another important segment, particularly for materials that cannot withstand the high temperatures used in steam sterilization. Ethylene oxide sterilization is commonly used for sterilizing heat-sensitive medical equipment, plastics, and devices. Biological indicator vials are essential for validating the effectiveness of this sterilization method, ensuring that ethylene oxide has properly penetrated and sterilized the materials. Although steam sterilization remains the dominant method, the growth of more specialized sterilization methods, such as ethylene oxide, continues to drive demand for biological indicator vials across a range of industries.
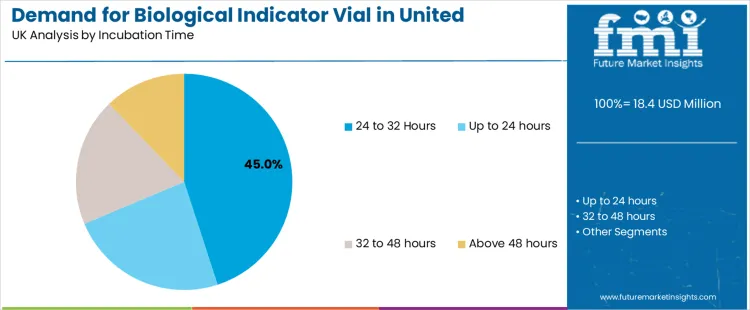
The incubation time segment for biological indicator vials in the United Kingdom is predominantly led by the 24 to 32 hours incubation time, which accounts for 45% of the market share. This incubation time range is commonly used to assess the sterility of items after sterilization processes, particularly for biological indicators that require a longer period to confirm the inactivation of microbial life. This duration provides enough time for any surviving microorganisms to grow and be detected, ensuring the sterilization process has been effective. The 24 to 32 hours range is typically used in industries such as healthcare, pharmaceuticals, and laboratories where precise sterilization validation is critical.
Incubation times up to 24 hours also represent a significant segment, as they are used for faster turnaround processes in environments where quick results are needed. Shorter incubation times are often preferred in high-volume settings, such as hospitals and outpatient clinics, where rapid confirmation of sterilization effectiveness is necessary for continued operations. Longer incubation times, such as 32 to 48 hours and above 48 hours, are used in more specialized applications, particularly in research and certain industrial settings, where confirmation of sterility is required over an extended period for more complex sterilization processes. As industries continue to prioritize sterilization quality and compliance, the demand for biological indicator vials across various incubation times will continue to grow, with the 24 to 32 hours range remaining the most widely used.
Demand for biological indicator vials in the United Kingdom is driven by stringent sterilisation assurance requirements across healthcare, pharmaceutical manufacturing, medical device production, and laboratory environments. Biological indicator vials are used to validate sterilisation processes by confirming that specific sterilisation conditions effectively inactivate resistant microbial spores. Procurement activity reflects compliance with regulatory standards, quality management systems, and audit readiness. Volume demand corresponds to steriliser throughput, frequency of validation cycles, and adoption of risk based quality frameworks by hospital sterilisation departments, contract sterilisation facilities, and industrial users.
Several factors are supporting demand for biological indicator vials in the United Kingdom. Regulatory mandates from bodies such as the Medicines and Healthcare products Regulatory Agency (MHRA) and Medical Devices Regulations require routine biological validation of sterilisation processes for critical medical equipment and pharmaceuticals. Healthcare providers, including NHS trusts and private surgical facilities, rely on biological indicators to ensure safe reprocessing of reusable instruments. Pharmaceutical and biotechnology manufacturers integrate biological indicator vials into batch release protocols to demonstrate compliance with aseptic processing standards. Quality assurance expectations and the need to minimise risk of contamination in end products further reinforce repeat procurement of validated indicator systems.
Despite clear necessity, several restraints influence biological indicator vial demand in the United Kingdom. Cost pressures in public and private healthcare budgets can tighten purchases, especially for facilities with high frequency of sterilisation validations. Competition from alternative rapid sterility assessment methods, such as enzyme based indicators or automated microbiological systems, may temper growth where speed of results is prioritised. Variability in sterilisation technologies (e.g., steam, ethylene oxide, hydrogen peroxide plasma) requires multiple indicator types, increasing complexity in inventory management. Supply chain disruption, including lead times for specialised indicator spores and manufacturing constraints, can affect availability and planning for validation programmes.
Key trends shaping biological indicator vial demand in the United Kingdom include the shift toward rapid biological indicators and accelerated testing technologies that shorten cycle validation time and improve operational efficiency. Suppliers are expanding portfolios to include integrated indicator systems with traceable documentation and digital result capture compatible with quality management software. Increased focus on data integrity and audit readiness helps facilities meet regulatory inspection expectations with comprehensive records. Outsourced sterilisation service providers and contract sterilisation facilities are standardising biological indicator use to support multi client quality assurance. There is also heightened interest in indicators tailored for specific sterilisation modalities, as healthcare and industrial users seek fit for purpose validation solutions that align with evolving process technologies.
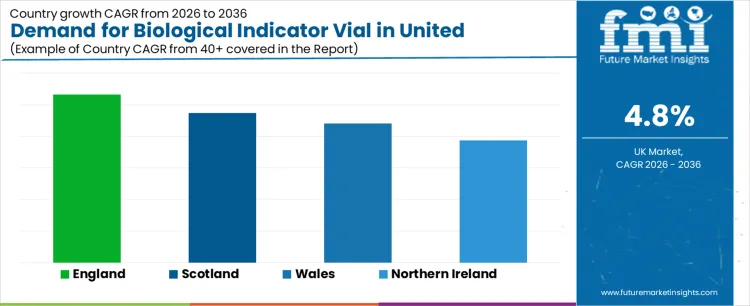
The demand for biological indicator vials in the United Kingdom varies by region, with England leading the market. This demand is largely driven by the country’s healthcare, pharmaceutical, and medical device industries, where biological indicator vials are crucial for sterilization validation and infection control. England’s large healthcare infrastructure, including hospitals, research institutions, and pharmaceutical manufacturing facilities, plays a key role in driving the market. Scotland, Wales, and Northern Ireland follow with steady demand, supported by the increasing need for sterilization monitoring in medical and laboratory environments. As the healthcare sector focuses on improving patient safety, ensuring sterile environments, and meeting regulatory standards, the demand for biological indicator vials is expected to rise across all regions, with England maintaining the highest market share.
| Country | CAGR (2026 to 2036) |
|---|---|
| England | 5.3% |
| Scotland | 4.7% |
| Wales | 4.4% |
| Northern Ireland | 3.9% |
The demand for biological indicator vials in England is projected to grow at a CAGR of 5.3% from 2026 to 2036. England’s robust healthcare infrastructure, including its extensive network of hospitals, clinics, pharmaceutical manufacturing facilities, and research centers, is a major driver of this demand. Biological indicator vials are essential in ensuring the effectiveness of sterilization processes used in medical devices, surgical instruments, and pharmaceuticals. The growing emphasis on infection control, patient safety, and adherence to stringent regulatory standards, such as those set by the UK Medicines and Healthcare products Regulatory Agency (MHRA) and the European Medicines Agency (EMA), is contributing to the market’s growth. As England’s healthcare system continues to evolve, the need for biological indicator vials to validate sterilization processes in various sectors-such as hospitals, laboratories, and pharmaceutical companies-will remain a critical component of infection control and quality assurance programs.
In Scotland, the demand for biological indicator vials is expected to grow at a CAGR of 4.7% from 2026 to 2036. The increasing demand for sterilization validation in healthcare, pharmaceutical manufacturing, and laboratory settings is driving the adoption of biological indicator vials. Scotland’s healthcare sector is expanding, with more hospitals and research institutions implementing stringent sterilization procedures to ensure patient safety and regulatory compliance. The growing focus on infection control and quality assurance in medical facilities, combined with increasing awareness about the importance of sterilization in maintaining safety standards, is contributing to the demand for biological indicators. Additionally, Scotland’s rising involvement in pharmaceutical and biotechnology research, where sterilization is critical, is expected to further fuel the market for biological indicator vials. As healthcare and research facilities continue to adopt advanced sterilization methods, demand for these vials will grow steadily.
The demand for biological indicator vials in Wales is projected to grow at a CAGR of 4.4% through 2036. The healthcare and pharmaceutical sectors in Wales are increasingly adopting biological indicators to validate sterilization processes in hospitals, clinics, and pharmaceutical manufacturing facilities. The growing recognition of the importance of infection control and the adoption of stringent sterilization practices to meet health and safety regulations are key factors driving this demand. The rising number of surgeries and medical treatments in Wales, combined with the increasing focus on patient safety, is fueling the need for sterilization monitoring tools like biological indicator vials. Additionally, Wales’ commitment to improving its healthcare system, along with a growing emphasis on research and development in biotechnology, is expected to contribute to continued market growth. As Wales continues to strengthen its healthcare and pharmaceutical industries, the demand for these vials will grow consistently.
Northern Ireland’s demand for biological indicator vials is expected to grow at a CAGR of 3.9% from 2026 to 2036. Although Northern Ireland represents the smallest market for biological indicator vials in the United Kingdom, steady growth is expected, driven by the increasing adoption of sterilization practices in healthcare and pharmaceutical industries. With an expanding healthcare system focused on improving infection control and safety standards, the demand for biological indicators to monitor sterilization effectiveness will continue to rise. Northern Ireland’s healthcare sector is increasingly investing in advanced sterilization technologies, contributing to the growth of the market. As more medical and laboratory facilities in the region incorporate sterilization validation protocols to meet regulatory requirements, the demand for biological indicator vials is expected to expand steadily over the next decade.
Demand for biological indicator vials in the United Kingdom is defined by requirements from sterilization validation programmes in healthcare, pharmaceutical production, and laboratory facilities. Procurement teams, quality assurance divisions, and regulatory compliance units prioritise supplier offerings on documented resistance profiles, documented incubation conditions, and documented organism challenge data. VWR Corporation is positioned as a leading supplier with technical brochures that list documented spore strains, documented D value data, and recommended usage protocols. These documents are used by sterilisation engineers and QA professionals to compare supplier products during validation plan development and audit preparations.
STERIS plc. competes with documented biological indicators supported by specification sheets that detail documented organism load levels, documented temperature tolerance ranges, and documented compliance with HTM 01 01 and relevant ISO standards. Buyers in the United Kingdom reference these materials when aligning supplier choices with steriliser types and process cycles. Mesa Labs supplies documented products with literature that lists documented resistance studies, documented incubation instructions, and documented performance criteria that assist hospital sterile services and pharmaceutical QA teams in method selection. 3M Company provides biological indicator vials documented for specific sterilisation modalities, with technical documentation covering documented expiry data, documented storage conditions, and documented test result interpretation guidance. Siltex Australia (c) offers biological indicators with documented spore enumeration information and documented procedural notes that are referenced by users when integrating indicators into routine validation schedules.
Procurement decisions within NHS trusts, private healthcare networks, and contract sterilisation facilities incorporate documented performance data, documented service support options, and documented supply terms. Buyers assess documented shelf stability information and documented traceability features during tender evaluations. Logistics and inventory planners consider documented lead times and documented distribution reach when scheduling orders. Technical reviewers and compliance teams examine documented test criteria and documented acceptance limits to ensure that selected indicators align with internal validation protocols and regulatory expectations. Competitive activity in the United Kingdom biological indicator market is grounded in measurable technical specifications and documented support commitments that assist QA and procurement professionals in aligning supplier offerings with sterilisation validation objectives.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Sterilization Type | Steam Sterilization, Ethylene Oxide Sterilization |
| Incubation Time | 24 to 32 Hours, Up to 24 Hours, 32 to 48 Hours, Above 48 Hours |
| End Use | Food & Beverages, Pharmaceutical, Cosmetics |
| Companies | VWR Corporation, STERIS plc., Mesa Labs, 3M Company, Siltex Australia (c) |
| Regions Covered | United Kingdom |
| Countries Covered | England, Scotland, Wales, Northern Ireland |
| Additional Attributes | Dollar by sales by sterilization type, incubation time, end use, and region. Includes market trends towards regulatory compliance, sterilization efficiency, and the role of biological indicator vials in various industries. |
How big is the demand for biological indicator vial in United Kingdom in 2026?
The demand for biological indicator vial in United Kingdom is estimated to be valued at USD 18.4 million in 2026.
What will be the size of biological indicator vial in United Kingdom in 2036?
The market size for the biological indicator vial in United Kingdom is projected to reach USD 29.5 million by 2036.
How much will be the demand for biological indicator vial in United Kingdom growth between 2026 and 2036?
The demand for biological indicator vial in United Kingdom is expected to grow at a 4.8% CAGR between 2026 and 2036.
What are the key product types in the biological indicator vial in United Kingdom?
The key product types in biological indicator vial in United Kingdom are steam sterilization and ethylene oxide sterilization.
Which incubation time segment is expected to contribute significant share in the biological indicator vial in United Kingdom in 2026?
In terms of incubation time, 24 to 32 hours segment is expected to command 45.0% share in the biological indicator vial in United Kingdom in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.