The demand for biological indicator vials in South Korea is expected to reach USD 8.5 million by 2036, growing at a CAGR of 4.4% from USD 5.5 million in 2026. This category holds strategic importance within the broader healthcare and pharmaceutical industries, where precise sterilization validation is essential to ensure product safety and regulatory compliance.
Key factors driving this market include stringent regulatory requirements and the ongoing necessity for compliance with evolving standards. These elements create a steady demand for high-quality biological indicators, emphasizing the importance of not only meeting but exceeding industry expectations. As companies navigate these challenges, the emphasis on maintaining rigorous quality control while adapting to regulatory changes becomes increasingly essential.
Suppliers face strategic trade-offs as they strive to balance the costs associated with high-quality production against the need for competitive pricing. To thrive in this dynamic landscape, businesses must prioritize innovation and efficiency, focusing on streamlining manufacturing processes and managing supply chain dependencies. As the regulatory landscape continues to develop, organizations that are agile and proactive will be better positioned to maintain their competitive edge and secure their growth in the market.
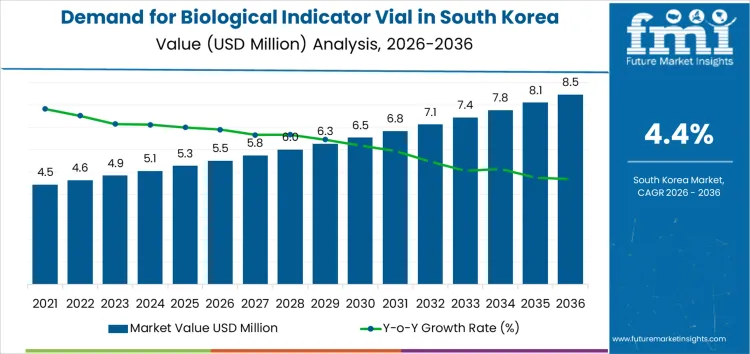
What is the Growth Forecast for the Biological Indicator Vial Market in South Korea through 2036?
The biological indicator vial market in South Korea is projected to grow steadily from 2026 to 2036. Starting at USD 5.5 million in 2026, the market is expected to rise to USD 5.8 million in 2027. By 2028, the market value will reach USD 6.0 million, followed by USD 6.3 million in 2029. The market will continue its steady upward trajectory, reaching USD 6.5 million in 2030 and USD 6.8 million in 2031.
In the subsequent years, demand for biological indicator vials will continue to rise, with the market projected to reach USD 7.1 million in 2032 and USD 7.4 million in 2033. By 2034, the market value will reach USD 7.8 million, growing to USD 8.1 million in 2035. By 2036, the market is projected to reach USD 8.5 million. The compound annual growth rate (CAGR) analysis shows steady growth at a rate of 4.4%, driven by the increasing need for sterilization validation and rising demand for biological indicator vials in various industries, especially in healthcare and pharmaceuticals.
| Metric | Value |
|---|---|
| Industry Sales Value (2026) | USD 5.5 million |
| Industry Forecast Value (2036) | USD 8.5 million |
| Industry Forecast CAGR (2026 to 2036) | 4.4% |
Demand for biological indicator vials in South Korea has strengthened as healthcare facilities, pharmaceutical manufacturers, and medical device producers prioritise sterility assurance and regulatory compliance. Biological indicator vials are used to validate sterilisation processes such as autoclaving, dry heat, and ethylene oxide cycles, providing direct evidence that microbial spores are inactivated under specific conditions. Hospitals, particularly those with surgical and intensive care units, incorporate routine steriliser validation into quality management systems to reduce the risk of nosocomial infections. South Korea’s regulatory environment, which aligns with international standards for sterilisation validation (including guidelines from the Ministry of Food and Drug Safety), drives consistent utilisation of biological indicators in clinical and industrial settings. Procurement teams assess products based on reproducibility, clear pass/fail results, and compatibility with established sterilisation equipment, ensuring reliable performance in audit and accreditation processes.
In the upcoming years, demand for biological indicator vials in South Korea is expected to grow as both clinical and manufacturing sectors expand quality assurance infrastructure and adopt advanced sterilisation protocols. Pharmaceutical and biotech facilities engaged in sterile drug and vaccine production require frequent validation testing throughout manufacturing and release processes, which increases usage volumes of biological indicators. Suppliers with robust quality systems, traceability, and third-party certification are positioned to meet the exacting requirements of industrial customers and hospital quality departments. Value for healthcare providers and manufacturers extends beyond product supply to include technical support, training on proper use, and integration with facility quality management systems. Emerging infection control priorities and heightened scrutiny of sterilisation validation during audits may also elevate demand for higher-performance indicators that offer shorter incubation times or enhanced detection sensitivity. Companies that align offerings with local regulatory expectations and provide reliable post-sales support can strengthen their presence in South Korea’s biological indicator market.
The demand for biological indicator vials in South Korea is driven by sterilization type and incubation time. The key sterilization types include steam sterilization, which accounts for 54% of the market, and ethylene oxide sterilization. Incubation time segments include 24 to 32 hours, which holds the largest share at 47.4%, followed by shorter and longer incubation times. The growth in demand is driven by sectors that require sterilization validation, such as healthcare, pharmaceuticals, and medical device manufacturing.
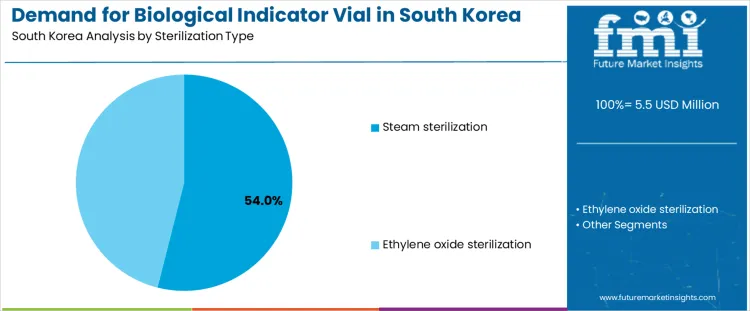
Sterilization type plays a significant role in shaping the demand for biological indicator vials in South Korea, with steam sterilization accounting for 54% of the market share. Steam sterilization, also known as autoclaving, is the most common method used in healthcare and medical device manufacturing for sterilizing equipment and materials. The effectiveness of biological indicators in confirming the sterility of materials after autoclaving has made them essential in ensuring compliance with stringent health and safety regulations. Biological indicator vials used for steam sterilization typically contain microorganisms that are resistant to the sterilization process, providing a clear and reliable way to confirm that sterilization conditions have been met. Ethylene oxide sterilization, while less common than steam sterilization, is also important for sterilizing heat-sensitive materials and medical devices that cannot tolerate high temperatures. The use of biological indicator vials for ethylene oxide sterilization ensures that the sterilization process is effective and that materials are safe for use. As South Korea’s medical, pharmaceutical, and healthcare sectors continue to grow, the demand for biological indicator vials is expected to increase, driven by the need for reliable sterilization validation in these industries.
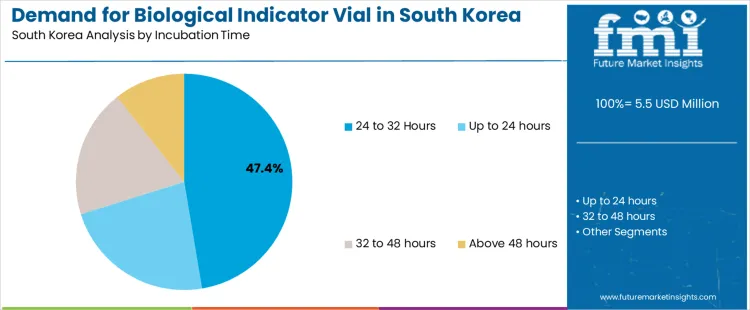
Incubation time is a key factor influencing the demand for biological indicator vials in South Korea, with the 24 to 32-hour incubation time segment holding the largest share at 47.4%. This incubation period is optimal for many sterilization processes, allowing sufficient time for microorganisms in the biological indicator vials to grow and show whether the sterilization was effective. Biological indicator vials with this incubation time are commonly used in healthcare settings, including hospitals and pharmaceutical companies, where ensuring sterility is critical for patient safety and regulatory compliance. The demand for vials with shorter incubation times (up to 24 hours) is driven by the need for quick verification of sterilization, particularly in fast-paced environments where rapid turnaround is necessary. These vials are often used in settings where the sterilization process needs to be verified quickly, such as in surgical units or dental offices. Vials with longer incubation times (32 to 48 hours and above 48 hours) are typically used in more specialized applications, where longer observation periods are needed to ensure the complete effectiveness of the sterilization process. These longer incubation periods are important in industries that require highly controlled sterilization, such as in the production of medical devices or pharmaceuticals, where ensuring that all microbial life is eradicated is a top priority. As demand for more precise and dependable sterilization processes continues to rise across South Korea’s healthcare and pharmaceutical industries, the demand for biological indicator vials will grow, with variations in incubation time addressing different industry needs.
Demand for biological indicator vials in South Korea is driven by the stringent requirements for sterilization assurance across healthcare, pharmaceutical, medical device, and laboratory environments. Biological indicator vials serve as critical tools for validating sterilization cycles in autoclaves, ethylene oxide (EO), and dry heat systems. Hospitals, surgical centers, and diagnostic laboratories rely on these vials to confirm microbial inactivation and maintain sterilization compliance. Regulatory frameworks, including Korean Good Manufacturing Practice (KGMP) and ISO 11138 standards, shape procurement specifications and quality expectations. Distribution channels comprising medical supply distributors, direct institutional procurement, and e-commerce platforms influence access and inventory flow for suppliers and manufacturers.
What are the Drivers of Demand for Biological Indicator Vials in South Korea?
A principal driver is the emphasis on patient safety and infection prevention in clinical settings, which mandates robust sterilization validation protocols. Growth in surgical procedures, increased use of reusable medical instruments, and expanding laboratory testing volumes all generate recurring need for biological indicators. National health accreditation programs and quality assurance audits require documented evidence of sterilization efficacy, boosting demand for biological indicator vials that provide reliable performance and traceability. Pharmaceutical and biotech production facilities also procure these vials to support sterilization validation for aseptic processing lines. Supplier capabilities in delivering accredited, lot-traceable products with technical support strengthen procurement confidence among institutional buyers.
Restraints include budgetary pressures in smaller hospitals and clinics, where tight operating margins can limit purchase frequency or volume for biological indicator products. Per-use cost considerations, especially for high-throughput sterilization environments, may prompt healthcare facilities to balance frequency of biological testing with operational budgets. Training requirements for correct use and interpretation of biological indicators can slow adoption where technical expertise is limited. Variability in sterilization equipment types and cycle parameters requires buyers to match specific indicator strains and formats to their sterilizers, complicating inventory management. Suppliers must also navigate regulatory clearance processes, which can affect time to market for new biological indicator vials.
An emerging trend is the integration of rapid readout biological indicators that shorten verification times compared with traditional culture-based methods, supporting faster turnaround in high-volume sterilization environments. Adoption of digital sterilization monitoring and electronic record-keeping systems encourages use of vials with compatible traceability features, such as barcodes and data integration capabilities. Demand is increasing for indicator formats tailored to advanced sterilization technologies, including low-temperature plasma and vaporized hydrogen peroxide systems. Collaborative programs between suppliers and hospital infection control teams enhance education on best practices and strengthen long-term procurement relationships. Manufacturers investing in locally compliant production and distribution networks improve responsiveness to regional quality standards and institutional buying patterns.
The demand for biological indicator vials in South Korea is growing across various regions, driven by the increasing need for sterilization monitoring in industries such as healthcare, pharmaceuticals, and biotechnology. South Gyeongsang, with a projected CAGR of 4.6%, leads the market, supported by its advanced medical and pharmaceutical infrastructure. North Jeolla, with a growth rate of 3.5%, shows moderate demand, reflecting ongoing industrial development and the rising adoption of sterilization monitoring systems. South Jeolla, with a projected CAGR of 4.2%, is seeing steady demand as the region’s biotechnology and pharmaceutical sectors expand. Jeju, with the highest growth rate of 5.2%, stands out for its emerging healthcare services and growing focus on medical device quality assurance. These regional trends provide significant opportunities for suppliers of biological indicator vials to tap into a growing market that is increasingly focused on safety, compliance, and sterilization effectiveness.
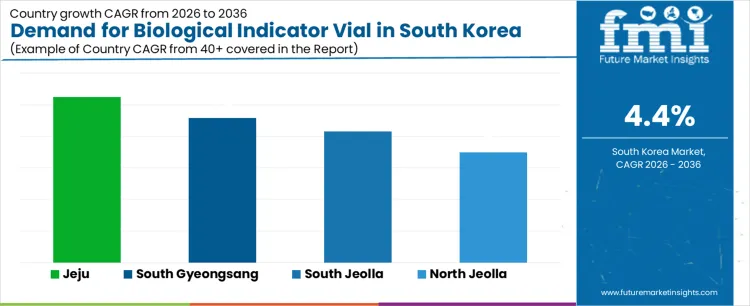
| Region | CAGR (2026 to 2036) |
|---|---|
| South Gyeongsang | 4.6% |
| North Jeolla | 3.5% |
| South Jeolla | 4.2% |
| Jeju | 5.2% |
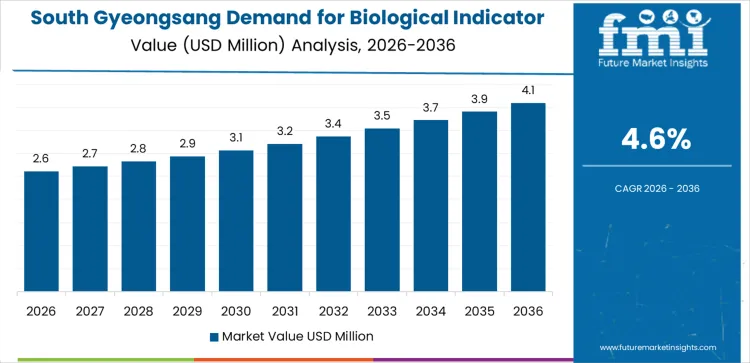
South Gyeongsang, with a projected CAGR of 4.6%, offers a promising market for manufacturers of biological indicator vials. The region's well-established pharmaceutical, biotechnology, and healthcare sectors create a strong demand for reliable sterilization monitoring systems. Companies should focus on offering high-quality, precision biological indicator vials that comply with local and international sterilization standards. By building relationships with hospitals, pharmaceutical manufacturers, and medical device suppliers in South Gyeongsang, manufacturers can position themselves as trusted partners in sterilization control. As the region continues to expand its medical and pharmaceutical industries, demand for biological indicator vials will likely rise, offering growth opportunities for suppliers.
North Jeolla, with a CAGR of 3.5%, is experiencing moderate demand for biological indicator vials. The region’s pharmaceutical and medical industries are growing, particularly as the adoption of sterilization monitoring systems increases in local manufacturing facilities. As North Jeolla continues to develop its industrial and healthcare infrastructure, there will be an increased need for effective sterilization solutions. Manufacturers should focus on providing cost-effective, easy-to-use biological indicator vials that meet the needs of both small-scale and large-scale producers in the region. Partnerships with regional healthcare facilities and pharmaceutical manufacturers could help drive demand and establish a foothold in this growing market.
South Jeolla, with a projected CAGR of 4.2%, shows steady demand growth for biological indicator vials, driven by the expanding pharmaceutical and biotechnology industries. As these sectors increase their focus on product safety and regulatory compliance, the demand for sterilization monitoring tools, including biological indicator vials, is expected to rise. Manufacturers targeting South Jeolla should offer high-quality, reliable biological indicator vials designed for both small and large-scale sterilization processes. Providing educational resources and technical support for local healthcare providers and manufacturers will also help drive market growth. As the region continues to invest in healthcare and biotechnology, the demand for sterilization solutions is expected to increase, creating opportunities for suppliers.
Jeju, with the highest projected CAGR of 5.2%, is witnessing significant growth in the demand for biological indicator vials. This growth is driven by the region's expanding healthcare sector, particularly in medical tourism and quality assurance services. As Jeju continues to develop as a hub for healthcare and advanced medical services, there is increasing demand for sterilization solutions in hospitals, clinics, and medical device manufacturers. Suppliers targeting Jeju should focus on offering biological indicator vials that meet both local regulatory standards and international quality assurance requirements. By aligning products with the growing emphasis on safety and quality assurance in Jeju’s healthcare sector, manufacturers can capitalize on the region's robust demand for sterilization solutions. The increase in medical tourism also presents a unique opportunity for suppliers of biological indicator vials to expand their market reach.
Demand for biological indicator vials in South Korea is shaped by stringent sterilisation quality requirements in healthcare, pharmaceutical manufacturing, and laboratory testing environments. Hospitals, contract sterilisation providers, and life sciences firms specify biological indicators to verify sterilisation cycles for medical devices, surgical instruments, and critical materials. Procurement criteria commonly include documented spore resistance characteristics, clear performance validation data, and compatibility with steam, ethylene oxide, and other sterilisation modalities used in facility operations. Quality assurance teams assess indicator heat resistance profiles, incubation period parameters, and traceability features that support audit readiness and regulatory compliance. Facility buyers in biotechnology and pharmaceutical sectors also consider ease of integration into existing sterilisation workflows, consistency of indicator production batches, and availability of rapid readout options that shorten cycle verification timelines. Demand reflects both routine compliance testing and heightened emphasis on patient safety outcomes, driving specification refinement that prioritises reproducibility and documented supplier quality management systems.
Competition among key suppliers in the South Korean biological indicator vial segment centres on product specification clarity, technical support resources, and service reliability. VWR Corporation is recognised as a leading supplier, with product literature detailing spore strains, resistance values, and incubation procedures that inform technical evaluations by quality assurance teams. STERIS plc. materials present biological indicators accompanied by performance charts, recommended usage guidelines, and documented compliance references relevant to hospital sterile processing departments and pharma validation groups. Mesa Labs documentation emphasises analytical consistency, storage condition parameters, and batch traceability that support procurement teams evaluating long-term supplier reliability. 3M Company brochures describe indicator vials with specification tables on resistance performance and handling instructions that assist sterilisation engineers and laboratory managers. Siltex Australia (c) product information outlines biological indicators with focus on spore viability data, packaging integrity, and service support options that inform distributor relationships and customer training programmes. Brochure content from these companies typically includes test condition parameters, compliance standards, and performance thresholds that help South Korean healthcare facilities, contract sterilizers, and regulated manufacturers compare offerings against operational, regulatory, and quality assurance requirements.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD Million |
| Sterilization Type | Steam Sterilization, Ethylene Oxide Sterilization |
| Incubation Time | 24 to 32 Hours, Up to 24 Hours, 32 to 48 Hours, Above 48 Hours |
| End Use | Food & Beverages, Pharmaceutical, Cosmetics |
| Companies | VWR Corporation, STERIS plc., Mesa Labs, 3M Company, Siltex Australia (c) |
| Region Covered | South Korea |
| Provinces Covered | South Gyeongsang, North Jeolla, South Jeolla, Jeju |
| Additional Attributes | Dollar sales by sterilization type, incubation time, and end use. Demand is driven by strict compliance requirements in pharmaceutical and food sectors, with steam and EO-based sterilization indicators essential for validating sterilization cycles in high-precision environments. |
How big is the demand for biological indicator vial in south korea in 2026?
The demand for biological indicator vial in south korea is estimated to be valued at USD 5.5 million in 2026.
What will be the size of biological indicator vial in south korea in 2036?
The market size for the biological indicator vial in south korea is projected to reach USD 8.5 million by 2036.
How much will be the demand for biological indicator vial in south korea growth between 2026 and 2036?
The demand for biological indicator vial in south korea is expected to grow at a 4.4% CAGR between 2026 and 2036.
What are the key product types in the biological indicator vial in south korea?
The key product types in biological indicator vial in south korea are steam sterilization and ethylene oxide sterilization.
Which incubation time segment is expected to contribute significant share in the biological indicator vial in south korea in 2026?
In terms of incubation time, 24 to 32 hours segment is expected to command 47.4% share in the biological indicator vial in south korea in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.