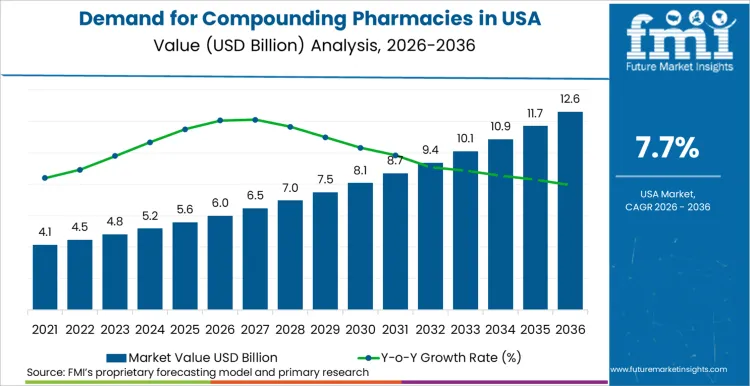
Compounding pharmacies industry in US is estimated to reach USD 6.0 billion in 2026. The sector is expecting a rise to USD 12.7 billion by 2036, progressing at a compound annual growth rate of 7.7%. US healthcare providers increasingly rely on compounded medications to mitigate persistent national drug shortages and address unique patient requirements not met by commercially available FDA-approved drugs. Personalized medicine trends drive this expansion as physicians seek specific dosages or allergen-free formulations for patients with sensitivities to mass-produced excipients.
Hospitals and clinics integrate these services to ensure continuity of care during supply chain disruptions affecting critical sterile injectables. Growing geriatric demographics requiring hormone replacement therapies and pain management solutions further solidify revenue trajectory for this essential healthcare domain.
Regulatory frameworks established by the Drug Quality and Security Act continue to shape operational landscapes for 503A and 503B facilities. FDA oversight ensures higher safety standards which builds trust among prescribers and health systems utilizing outsourcing facilities for bulk orders. 503B outsourcing facilities are witnessing substantial investment as hospitals offload complex sterile preparations to certified external partners to reduce internal compliance costs and liability.
Chronic disease prevalence necessitates tailored therapeutic regimens where standard dosages fail to achieve desired clinical outcomes. Insurance reimbursement policies are gradually evolving to cover specific compounded preparations thereby expanding patient access to these necessary customized treatments across various states.
Technological integration within compounding facilities involves advanced pharmacy automation systems to ensure precise ingredient measuring and sterility. Adoption of sterile prep pack workstations enhances workflow efficiency while minimizing human error during formulation of high-risk injectables. Facilities investing in unit dose packaging solutions improve medication safety and adherence for patients in long-term care settings. Digital platforms facilitate easier ordering processes which supports growth of epharmacy sector specializing in customized prescriptions. Innovation extends to oral controlled release drug delivery technology allowing compounders to create sophisticated dosage forms that improve therapeutic efficacy and patient convenience.
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 6.0 Billion |
| Industry Value (2036) | USD 12.7 Billion |
| CAGR (2026 to 2036) | 7.7% |
Key catalyst for growth is increasing incidence of drug shortages affecting US hospitals particularly for intravenous solution compounders and pain medications. Healthcare providers turn to compounding facilities to bridge these supply gaps ensuring patients receive critical treatments without interruption. Rising demand for compounded topical drugs in dermatology and pain management also stimulates activity. Patients seeking hormone replacement therapy often require dosages not available in standard commercial products driving business to specialized pharmacies. Veterinary needs for flavored medications or specific strengths for pets further diversify revenue streams. Shift towards super generics and value-added formulations encourages innovation in delivery methods.
US compounding landscape is segmented to address regulatory distinctions and specific patient needs. It is categorized by Pharmacy Type reflecting FDA designations. Product Type segment highlights various dosage forms produced. Application segment underscores diverse patient demographics served. Each segment plays vital role where 503A facilities focus on individual prescriptions and oral medications dominate due to ease of administration.
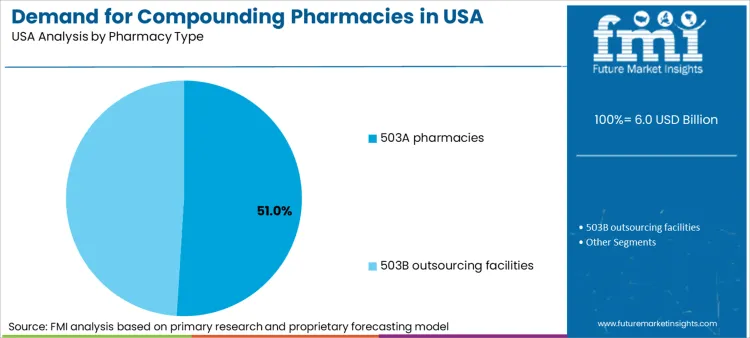
503A pharmacies command a 51.0% share of US sector value. These traditional compounding pharmacies operate under state board regulations and focus on fulfilling patient-specific prescriptions. Dominance stems from their deep integration into local communities and strong relationships with individual prescribers. Patients requiring unique capsule tablet counter prepared dosages for allergies or specific strengths rely on these local providers. Flexibility to adjust formulations rapidly for individual needs keeps 503A facilities at forefront of personalized care.
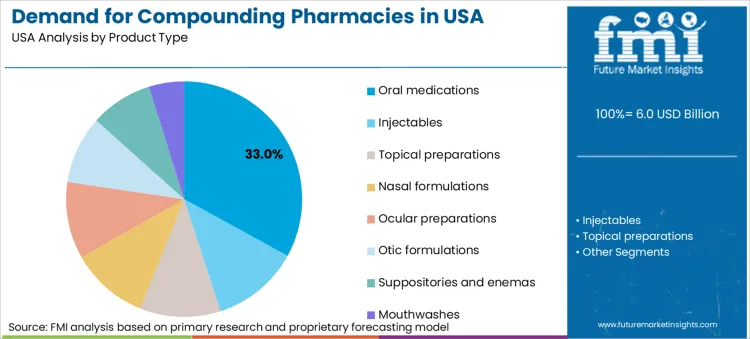
Oral medications account for 33.0% of product volume. Preference for non-invasive delivery methods drives high consumption of compounded capsules, liquids, and lozenges. Pediatric and geriatric populations often struggle with swallowing standard tablets necessitating liquid formulations or flavored suspensions. Compounding allows for exclusion of dyes or preservatives found in commercial oral drugs making them suitable for sensitive patients. Versatility of oral forms for treating conditions ranging from hormone imbalance to pain management secures leading position.
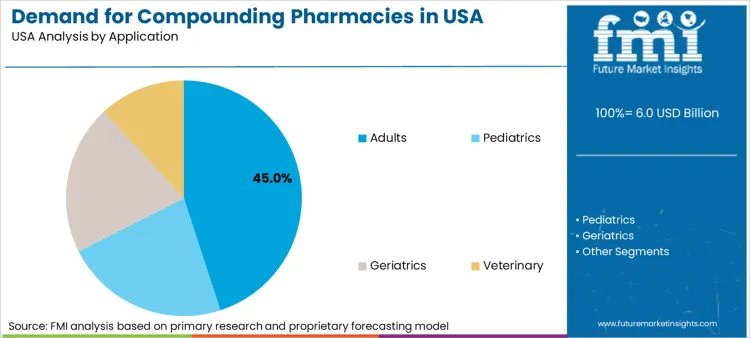
Adults represent dominant application segment with 45.0% share. High prevalence of chronic conditions such as arthritis, hormone deficiencies, and dermatological issues among US adult population fuels demand. Hormone Replacement Therapy (HRT) is significant driver as many adults seek bio-identical hormones tailored to specific physiological needs. Sports medicine also contributes as athletes require pharmaceutical grade magnesium sulfate or topical pain relief formulations. Ability to customize dosages for weight management programs further anchors this demographic's dominance.
503B outsourcing facilities hold significant 49.0% share. Created to address safety concerns regarding sterile compounding, these facilities operate under strict cGMP standards similar to pharmaceutical manufacturers. Hospitals increasingly rely on 503B partners for bulk drug compounding to maintain stock of critical injectables. Consolidation in hospital networks drives large-volume contracts with these facilities. Focus on parenteral compounding for operating rooms and intensive care units ensures steady growth for this regulated segment.
Significant trend involves tightening regulatory scrutiny from FDA and state boards. Implementation of USP <800> guidelines for handling hazardous drugs requires pharmacies to invest heavily in negative pressure rooms and protective equipment. While improving safety, these requirements increase operational costs for smaller independent pharmacies potentially driving consolidation or exit. Compliance with Drug Supply Chain Security Act (DSCSA) adds another layer of complexity regarding traceability. Facilities that can navigate these regulatory hurdles efficiently will gain competitive advantage over less capitalized peers.
Emerging dynamic is adoption of robotics and automation in compounding workflows. Automated systems reduce risk of human error in calculations and mixing which is critical for sterile preparations. Use of united states dispensing trays and automated filling machines ensures consistency and sterility. Technology integration also aids in documentation and record-keeping which is essential for regulatory audits. Investments in these technologies signal shift towards industrialization of compounding to meet higher volume demands while maintaining rigorous quality standards.
Opportunity lies in specializing in high-demand niche therapies such as ophthalmology or veterinary medicine. Shortages of commercial ophthalmic drugs create openings for sterile compounders to supply essential eye drops. Veterinary compounding is expanding as pet owners seek pharmacy and drug store franchises capable of creating palatable medications for animals. Developing expertise in these specific areas allows pharmacies to differentiate themselves and command higher margins compared to generalist compounders.
Geographic distribution of compounding services correlates strongly with population density and concentration of healthcare facilities. Southern states exhibit robust demand driven by large retiree population requiring chronic care management and hormone therapies. Midwest regions leverage strong manufacturing base to support 503B outsourcing facilities serving hospitals nationwide. Northeast states focus on specialized hospital-based compounding due to high density of academic medical centers. Western states prioritize wellness and integrative medicine driving demand for customized nutritional and dermatological formulations.
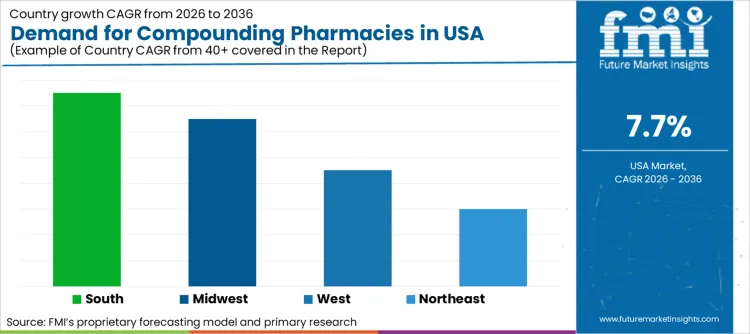
| Region | CAGR (2026 to 2036) |
|---|---|
| South | 8.1% |
| Midwest | 7.9% |
| West | 7.5% |
| Northeast | 7.2% |
Adoption of compounded medications in South is projected to expand at CAGR of 8.1%. States like Florida and Texas have large geriatric populations that heavily utilize custom medications for age-related conditions. Demand is driven by independent pharmacies catering to retirees needing hormone therapies and pain management solutions. Favorable business climates in these states also encourage establishment of new compounding facilities. High concentration of long-term care facilities further supports consistent volume for unit-dose compounded products.
Sales of bulk compounded drugs in Midwest are likely to reach CAGR of 7.9%. Central location makes this region ideal hub for 503B outsourcing facilities that need to ship sterile products nationwide within strict stability windows. Demand is fueled by large hospital systems in Illinois and Ohio outsourcing their sterile preparation needs. Investment in large-scale cGMP facilities drives regional growth as companies capitalize on lower operational costs compared to coastal regions.
Compounding sector in West is anticipated to rise at CAGR of 7.5%. California and adjacent states have strong culture of integrative and functional medicine. Demand is driven by patients seeking customized nutritional supplements, dermatological preparations, and anti-aging therapies. Tech-savvy consumers in this region are early adopters of online compounding platforms. Focus on personalized health and wellness sustains steady demand for non-sterile compounded products tailored to individual lifestyle goals.
Deployment of sterile compounding in Northeast is expected to increase at CAGR of 7.2%. High density of world-class hospitals and academic medical centers in New York and Massachusetts drives demand for high-acuity sterile preparations. Facilities focus on pediatric and oncology compounding to support complex treatment protocols found in these tertiary care centers. Stringent state regulations ensure high quality standards pushing growth towards larger, compliant 503B facilities that can meet hospital requirements.
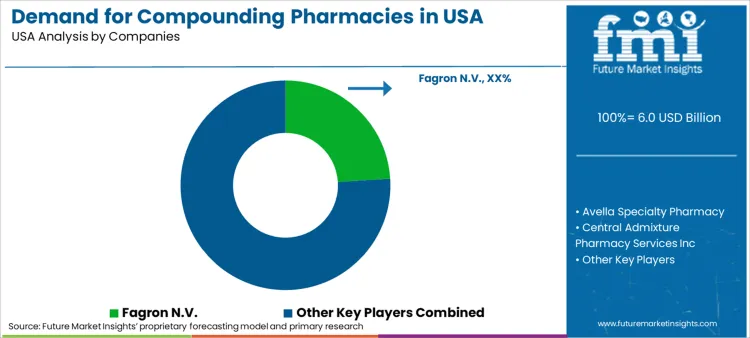
Competitive landscape is dominated by Fagron N.V. which commands robust 24.0% share. Strategy centers on vertical integration supplying both raw pharmaceutical ingredients and finished compounded preparations. Fagron strengthens position by acquiring regional facilities to expand geographic footprint and capacity. Avella Specialty Pharmacy competes by focusing on specialty medications and high-touch patient services particularly in oncology and ophthalmology. Major players invest heavily in quality control and sterility testing to differentiate themselves from smaller competitors and reassure hospital procurement teams.
Independent entities like Hoye's Pharmacy and Smith Caldwell Drug Store focus on deep community ties and specialized patient care. These players adapt by offering unique dosage forms not available from large outsourcers such as specific veterinary flavors or bio-identical hormone creams. Cantrell Drug Company and similar 503B facilities target hospital shortages providing essential backup during supply chain disruptions. Consolidation is active trend with larger entities acquiring successful independent labs to gain specialized expertise or customer lists.
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type | Oral medications, Injectables, Topical preparations, Nasal formulations, Ocular preparations, Otic formulations, Suppositories and enemas, Mouthwashes |
| Application | Adults, Pediatrics, Geriatrics, Veterinary |
| Pharmacy Type | 503A pharmacies, 503B outsourcing facilities |
| Regions Covered | Northeast, Midwest, South, West |
| Key Companies Profiled | Fagron N.V., Avella Specialty Pharmacy, Central Admixture Pharmacy Services Inc, Hoye's Pharmacy, and others. |
How big is the Compounding Pharmacies Industry in US in 2026?
The compounding pharmacies industry in US is estimated to be valued at USD 6.0 Billion in 2026.
What will be the size of Compounding Pharmacies in US in 2036?
The industry size for the Compounding Pharmacies in US is projected to reach USD 12.7 Billion by 2036.
How much will be the Compounding Pharmacies industry in US growth between 2026 and 2036?
The compounding pharmacies industry in US is expected to grow at a 7.7% CAGR between 2026 and 2036.
What are the key product types in the Compounding Pharmacies in US?
The key product types in Compounding Pharmacies in US are Oral medications, Injectables, Topical preparations, Nasal formulations and Ocular preparations.
Which end user segment is expected to contribute significant share in the Compounding Pharmacies in US in 2026?
In terms of end user, Adults segment is expected to command 45.0% share in the Compounding Pharmacies in US in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.