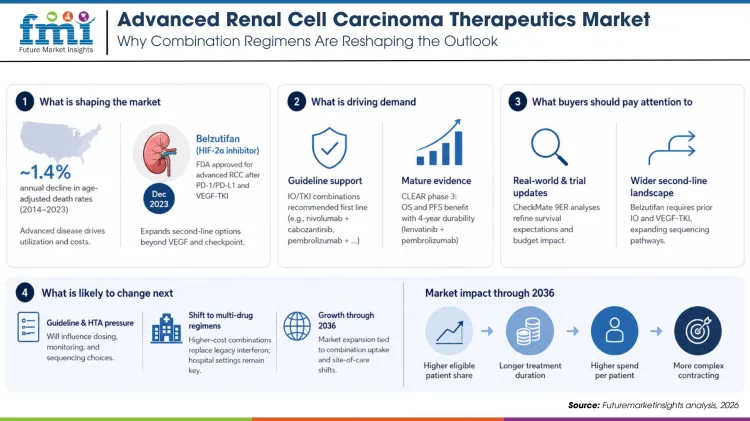
Kidney and renal pelvis cancers account for a sizeable USA burden, with SEER tracking rising prevalence and detailed stage and survival trends used in reimbursement and planning. Age-adjusted death rates have fallen about 1.4% per year from 2014 to 2023. Advanced disease still drives drug utilization and costs.
New mechanisms have reached market. The FDA approved belzutifan, a first-in-class HIF-2α inhibitor, on December 14, 2023 for adults with advanced RCC previously treated with a PD-1/PD-L1 inhibitor and a VEGF-TKI, expanding options beyond VEGF and checkpoint targets. This shifts second-line competition and informs value-based contracting around prior-therapy requirements.
USA and global guidelines recommend checkpoint and VEGF-TKI combinations in the first line for advanced RCC, including nivolumab plus cabozantinib and pembrolizumab plus. These endorsements increase eligible patient share and extend treatment durations compared with older monotherapies.
Mature readouts sustain the case for combinations. The phase 3 CLEAR program reported superior overall survival and progression-free survival for lenvatinib plus pembrolizumab versus sunitinib, with durability seen at four-year follow-up in peer-reviewed and conference updates. Such data support premium pricing and formulary priority.
Real-world and trial updates continue to refine sequence value. Analyses from CheckMate 9ER presented at ASCO 2024 examined survival states over four years for nivolumab plus cabozantinib versus sunitinib, informing expectations for. Payers should map these survival partitions to budget impact and stop-rules.
Second-line diversity is widening. The belzutifan label requires prior PD-1/PD-L1 and VEGF-TKI exposure, encouraging earlier IO/TKI use and creating a defined belzutifan-addressable cohort. NCCN and NCI PDQ sequencing notes frequent movement to VEGF-TKIs after IO-based regimens, which interacts with HIF-2α uptake and combination trials.
Guideline refreshes and cost-effectiveness work will pressure dosing, monitoring, and sequencing choices. Continuing CLEAR follow-ups and methodologic reviews show how dose intensity and risk mix affect outcomes, a factor that procurement teams should test in outcomes-based. Market growth through 2036 will track combination penetration and outpatient infusion shifts.
The FMI outlook points to a move away from legacy interferon use and toward higher-cost multi-drug regimens, while hospital oncology settings still anchor administration. Buyers should model site-of-care shifts as ambulatory centers take more volume.
For deeper sizing, forecasts, and regional growth rates across 2026-2036, see Advance Renal Cell Carcinoma Therapeutics Market on Future Market Insights.
This report covers the coronary heart disease diagnostic imaging market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, imaging pathway shifts, supply-side developments, and strategic growth opportunities.
This report covers the cell therapy cryopreservation and thaw performance test platforms market through analysis of market size, market share, revenue forecast, platform segmentation, assay readout trends, cell type demand, preservation format usage, automation level adoption, end-user demand, workflow stage requirements, regional outlook, technology developments, handling standardization needs, method transfer practices, comparability testing, supply chain dynamics, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
The Gene Editing Off-Target Effect Detection Test Systems Market is segmented by Detection Approach (Cell-based Assays, Biochemical Assays, In Silico Tools, Orthogonal Validation), Product Format (Services, Assay Kits, Software, Instruments, Reagents), Workflow Stage (Site Nomination, Site Confirmation, Structural Follow-up, Release Support), Editing Modality (CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, Other Editors), Readout Technology (Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, Long-read Sequencing), End User (Biopharma Companies, Academic Laboratories, CROs, CDMOs, Hospitals), Application (IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, Comparability Studies), Deployment (Outsourced Testing, In-house Testing, Hybrid Models), and Region. Forecast for 2026 to 2036.
This report covers the at-home blood collection kits and devices market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, self-collection workflow trends, regulatory developments, and strategic growth opportunities.
The Biosimilar Monoclonal Antibody Comparability Test Platforms Market is segmented by Product Type (Instruments, Software, Reagents, Standards, Services), Technology (Chromatography, Mass Spectrometry, Electrophoresis, Spectroscopy, Bioassays), Assay Type (Structural Assays, Purity Assays, Glycan Assays, Potency Assays, Stability Assays), Workflow Stage (Analytical Similarity, Method Development, Method Validation, Stability Testing, Release Testing), Molecule Class (IgG1 mAbs, IgG4 mAbs, Fc Fusions, Fragments, Conjugates), Deployment Model (In-house Labs, CRO Labs, Hybrid Labs), End User (Biosimilar Developers, CDMOs, CROs, Academic Centers, Regulators), and Region. Forecast for 2026 to 2036.