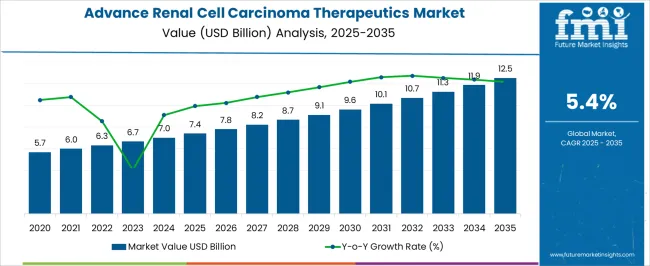
The advance renal cell carcinoma therapeutics market was valued at USD 7.40 billion in 2025, projected to reach USD 7.80 billion in 2026, and is forecast to expand to USD 13.20 billion by 2036 at a 5.4% CAGR. FMI opines that the sequential approval of combination immunotherapy and targeted therapy regimens is expanding the addressable treatment population while extending treatment duration per patient. Tyrosine kinase inhibitors and immune checkpoint combinations have established themselves as standard first-line protocols, shifting prescribing patterns away from single-agent interferon therapy toward higher-cost multi-drug regimens.
As per FMI, the treatment landscape continues to evolve as clinical trial readouts validate new sequencing strategies for second-line and subsequent therapy lines. Hospital oncology departments remain the primary prescribing and administration channel, though ambulatory surgical centers and multispecialty oncology clinics are absorbing an increasing share of infusion volume as health systems shift cancer care delivery toward outpatient settings. The expansion of biomarker-guided treatment selection is enabling more precise matching of patients to specific therapeutic mechanisms, improving response rates while concentrating drug spending on validated clinical subpopulations.
China leads with a 7.3% CAGR, driven by expanding oncology treatment access through national reimbursement list updates and increasing tyrosine kinase inhibitor adoption. India follows at 6.8%, supported by growing oncology infrastructure and improving access to targeted therapy regimens. Germany advances at 6.2%, anchored by comprehensive cancer care pathways and rapid adoption of newly approved combination protocols. Brazil records a 5.7% pace. The United States maintains a 5.1% trajectory, reflecting mature prescribing patterns with ongoing treatment line expansion. The United Kingdom grows at 4.6%, while Japan records a 4.0% rate, supported by established oncology care frameworks.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 7.80 billion |
| Industry Value (2036) | USD 13.20 billion |
| CAGR (2026 to 2036) | 5.4% |
Source: Future Market Insights, 2026
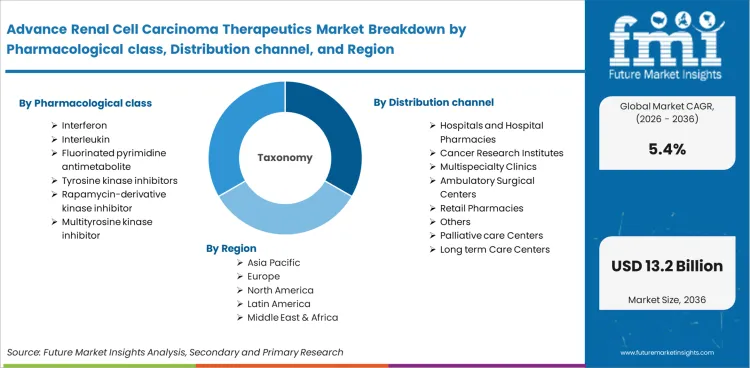
Advanced renal cell carcinoma therapeutics comprise pharmacological agents used in the systemic treatment of metastatic and locally advanced kidney cancers that have progressed beyond surgical management. The therapeutic scope includes interferons, interleukins, fluorinated pyrimidine antimetabolites, tyrosine kinase inhibitors, rapamycin-derivative kinase inhibitors, and multityrosine kinase inhibitors administered through hospital oncology departments, cancer research institutes, and specialty clinics as part of first-line, second-line, and subsequent treatment protocols.
Market scope covers interferon-based immunotherapy, interleukin-based treatment, fluorinated pyrimidine antimetabolite chemotherapy, tyrosine kinase inhibitors, rapamycin-derivative kinase inhibitors, and multityrosine kinase inhibitors used in advanced renal cell carcinoma treatment. The scope encompasses drug revenues across hospitals, cancer research institutes, multispecialty clinics, ambulatory surgical centers, retail pharmacies, palliative care centers, and long-term care centers.
Early-stage renal cell carcinoma managed exclusively through surgical nephrectomy without systemic therapy is excluded. The scope omits diagnostic imaging and biopsy services, radiation therapy equipment revenues, and generic supportive care medications not classified as primary RCC therapeutic agents.
Increase in aging population is one of the major factors driving the revenue growth of the renal cell carcinoma market. Furthermore, renal cell carcinoma is a rare disease with a small patient-base constitutingall ages from infants to older adults.However, the disease is predominantly noted in age group of ~ 60-70 years, exposing a higher probability of occurrence.
Expanding research into orphan drugs is a factor projected to create lucrative revenue potential for drug makers of renal cell carcinoma.
Restraints include lack of technological advances in genome sequencing, stagnant number of diagnosed population base and low patient awareness levels among others.The disease pattern of renal cell carcinoma disease is also affected by the genomic alteration, changes in lifestyle etc. Other inevitable restraints include lack of product availability on specific diseases target indications apart from feeble pipeline.
Advance Renal Cell Carcinoma Therapeutics Market is projected to decline during the forecast period due slow results of clinical trials with lack of safety data. Indeed, the advance renal cell carcinoma therapeutics market would show a stagnant CAGR due to increase in population in developing countries.
While innovations like change in mode of action are being adapted; technical advancements in treatment procedures are expected to make the market shift significantly. The weak drug pipeline and lack of differentiation in drug therapy may hinder the progress of the advance renal cell carcinoma therapeutics market.
.webp)
Based on geographic regions, advance renal cell carcinoma therapeutics market is segmented into seven key regions: North America, Latin America, Eastern Europe, Western Europe, Japan, Asia Pacific and Middle East Africa. North America accounts for major revenue share in the global market of the disease, owing to an increasingly aging population and rising awareness towards innovative drug therapies.
The pattern is closely followed by markets in Western Eastern Europe and Asia Pacific regions. Lower acceptance of combination therapies followed by lack of facilities in disease diagnosis are factors that may have a negative impact on revenue generation from renal cell carcinoma therapeutics.
The advance renal cell carcinoma therapeutics market in Japan will have a constant growth due to the balance between acceptance of combinational therapies and rising disease incidence due to changes in lifestyle. Markets in Middle East and Africa pose lucrative revenue generation opportunities for players in the renal cell carcinoma therapeutics market. This is fuelled by increasing expenditure on healthcare from foreign countries in the region to cover a large untapped patient population base.
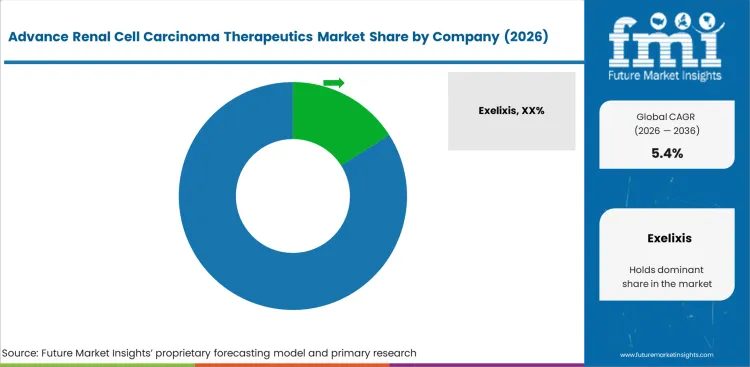
Key drug manufacturers targeting advance renal cell carcinoma therapeutics market include Exelixis, Bristol-Myers Squibb, Novartis, Pfizer, Eisai Co., Ltd, Genentech, GlaxoSmithKline, and Bayer among others.
The research report presents a comprehensive assessment of the market and contains thoughtful insights, facts, historical data, and statistically supported and industry-validated market data. It also contains projections using a suitable set of assumptions and methodologies. The research report provides analysis and information according to categories such as market segments, geographies, types, technology and applications.
| Metric | Value |
|---|---|
| Quantitative Units | USD 7.80 billion to USD 13.20 billion, at a CAGR of 5.4% |
| Market Definition | Advanced renal cell carcinoma therapeutics comprise pharmacological agents used in the systemic treatment of metastatic and locally advanced kidney cancers that have progressed beyond surgical management. The therapeutic scope includes interferons, interleukins, fluorinated pyrimidine antimetabolites, tyrosine kinase inhibitors, rapamycin-derivative kinase inhibitors, and multityrosine kinase inhibitors administered through hospital oncology departments, cancer research institutes, and specialty clinics as part of first-line, second-line, and subsequent treatment protocols. |
| Segmentation | Pharmacological Class: Interferon, Interleukin, Fluorinated Pyrimidine Antimetabolite, Tyrosine Kinase Inhibitors, Rapamycin-Derivative Kinase Inhibitor, Multityrosine Kinase Inhibitor; Distribution Channel: Hospitals and Hospital Pharmacies, Cancer Research Institutes, Multispecialty Clinics, Ambulatory Surgical Centers, Retail Pharmacies, Palliative Care Centers, Long Term Care Centers, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, Brazil, USA, UK, Japan, and 40 plus countries |
| Key Companies Profiled | Exelixis, Bristol-Myers Squibb, Novartis, Pfizer, Eisai Co., Ltd, Genentech, GlaxoSmithKline, Bayer |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with installed base metrics and project conversion rates, cross-validated against institutional expenditure guidance. |
Advance Renal Cell Carcinoma Therapeutics Market can be segmented based on drugs class and distribution channel.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for Advance Renal Cell Carcinoma Therapeutics in the global market in 2026?
Demand for Advance Renal Cell Carcinoma Therapeutics in the global market is estimated to be valued at USD 7.80 billion in 2026.
What will be the market size by 2036?
Market size is projected to reach USD 13.20 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 5.4% between 2026 and 2036.
Which Pharmacological Class is poised to lead in 2026?
Interferon accounts for 34.6% share in 2026, though combination targeted therapy regimens are progressively gaining prescribing share.
Which Distribution Channel leads in 2026?
Hospitals and Hospital Pharmacies represent 41.3% of distribution share as oncology infusion administration remains concentrated in hospital settings.
What is driving demand in China?
Expanding oncology treatment access through national reimbursement list updates and increasing tyrosine kinase inhibitor adoption are fueling demand at a 7.3% CAGR.
What is India's growth outlook?
India is projected to grow at a CAGR of 6.8% during 2026 to 2036.
What is included in the scope?
The market covers interferon, interleukin, fluorinated pyrimidine antimetabolite, tyrosine kinase inhibitor, rapamycin-derivative, and multityrosine kinase inhibitor revenues across hospital, clinic, and pharmacy distribution channels.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.