Advanced Therapy Medicinal Products Market
Advanced Therapy Medicinal Products Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Advanced Therapy Medicinal Products Market Size and Share Forecast Outlook 2025 to 2035
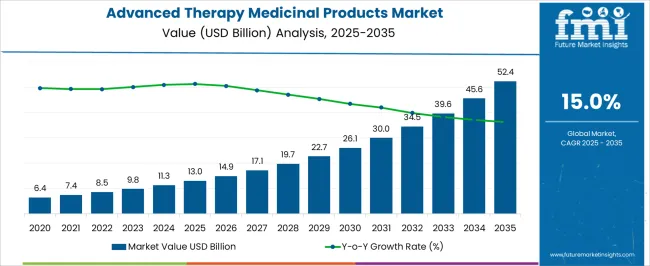
The Advanced Therapy Medicinal Products Market is estimated to be valued at USD 13.0 billion in 2025 and is projected to reach USD 52.4 billion by 2035, registering a compound annual growth rate (CAGR) of 15.0% over the forecast period.
Quick Stats for Advanced Therapy Medicinal Products Market
- Advanced Therapy Medicinal Products Market Industry Value (2025): USD 13.0 billion
- Advanced Therapy Medicinal Products Market Forecast Value (2035): USD 52.4 billion
- Advanced Therapy Medicinal Products Market Forecast CAGR: 15.0%
- Leading Segment in Advanced Therapy Medicinal Products Market in 2025: Cell Therapy (41.7%)
- Key Growth Region in Advanced Therapy Medicinal Products Market: North America, Asia-Pacific, Europe
- Top Key Players in Advanced Therapy Medicinal Products Market: Novartis AG, Spark Therapeutics, Inc., Bluebird Bio, Inc., UniQure N.V., Celgene Corporation, Gilead Lifesciences, Inc., Kolon TissueGene, Inc., JCR Pharmaceuticals Co., Ltd., MEDIPOST, Vericel Corporation, PHARMICELL Co., Ltd, Organogenesis Inc.
| Metric | Value |
|---|---|
| Advanced Therapy Medicinal Products Market Estimated Value in (2025 E) | USD 13.0 billion |
| Advanced Therapy Medicinal Products Market Forecast Value in (2035 F) | USD 52.4 billion |
| Forecast CAGR (2025 to 2035) | 15.0% |
Rationale for Segmental Growth in the Advanced Therapy Medicinal Products Market
The Advanced Therapy Medicinal Products market is gaining significant traction due to its transformative potential in treating diseases with limited or no therapeutic options. This market is being driven by continuous advancements in regenerative medicine, genetic engineering, and immunotherapy platforms. Regulatory support from leading health authorities and accelerated approval pathways have further encouraged innovation and commercialization of novel therapies.
Industry news and investor presentations have highlighted increased clinical trial activity, especially in the areas of oncology, hematology, and rare genetic disorders. Strategic collaborations between biotechnology firms and academic research centers have also been observed, enhancing development capabilities. The outlook for the market remains promising as demand for personalized and curative treatment options continues to rise globally.
As indicated in corporate press releases and healthcare innovation forums, long-term investment in manufacturing infrastructure and growing awareness among healthcare professionals are expected to pave the way for broader adoption These factors are collectively contributing to a favorable environment for sustained growth in the market.
Segmental Analysis
The market is segmented by Therapy Type and region. By Therapy Type, the market is divided into Cell Therapy, CAR-T Therapy, Gene Therapy, and Tissue Engineered Product. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Cell Therapy Segment
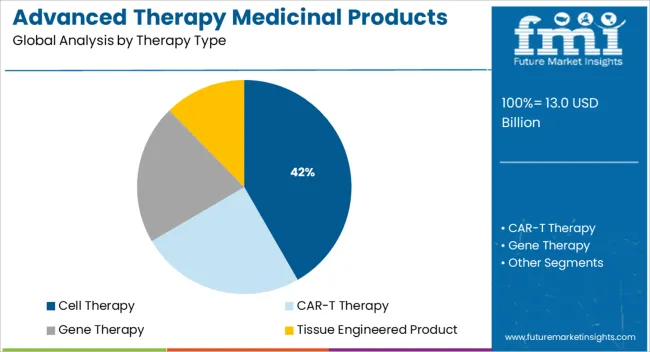
The cell therapy segment is projected to account for 41.7% of the Advanced Therapy Medicinal Products market revenue share in 2025, establishing it as the leading therapy type segment. This position has been achieved due to its proven efficacy in addressing chronic and life-threatening conditions such as cancer and autoimmune diseases. Cell therapies have gained momentum as they utilize a patient’s own or donor cells, modified or expanded ex vivo, to initiate targeted therapeutic effects.
Investor briefings and scientific communications have emphasized their increasing success rates in clinical trials and real-world outcomes. Moreover, the scalability of autologous and allogeneic platforms has been enhanced by technological advancements in cell culture, cryopreservation, and logistics.
Health system adoption has been further encouraged by supportive reimbursement frameworks and fast-track designations granted by global regulatory bodies These developments have established a strong foundation for the segment’s continued expansion, making it a central component in the commercial growth of the Advanced Therapy Medicinal Products market.
Comparison of 2020 to 2025 Revenue vs 2025 to 2035 Demand Forecast for Advanced Therapy Medicinal Products
As per the Advanced Therapy Medicinal Products Market research by Future Market Insights - a market research and competitive intelligence provider, historically, from 2020 to 2024, the market value of the Advanced Therapy Medicinal Products Market increased at around 11.7% CAGR.
The introduction of Advanced Therapy Medicinal Products has changed the pharmaceutical sector and the landscape of disease treatment. It has paved the way for innovative treatments for incurable diseases and cancers of various types.
Although the market for advanced therapeutic pharmaceutical products is still in its infancy, it is steadily rising. Traditional medication producers looking for a competitive edge are considering ATMPs as a profitable source of revenue in future healthcare systems. This field is now quite busy, with corporations investing heavily in ATMP clinical trials following the approval of authorized drugs.
Increased cases of Alzheimer4.4;s disease around the world, increased demand for advanced gene therapies, increased prevalence of neurodegenerative diseases, increased geriatric population, and surged genetic disorders are driving the growth of the advanced therapy medicinal products market. Owing to these factors, the market is projected to reach a valuation of USD 52.4 billion by 2035.
According to recent sources, there is a lot of development effort in the ATMP industry, which doesn4.4;t seem to match the small number of ATMPs now in the European market. Despite the fact that over 500 clinical trials using ATMPs were conducted between 2009 and 2020, only 19 market authorization applications were submitted to the European Medicines Agency.
The consolidated marketing permission was given to ten ATMPs. Three of these corporations later revoked the license, and one stopped marketing the product, all for commercial reasons. To appropriately appraise the potential of ATMPs, a thorough understanding of stakeholder difficulties encountered during development is required.
ATMP4.4;s high cost makes it unaffordable for both public and private payers, limiting patient access to therapy for life-threatening disorders and diseases. Due to ATMP4.4;s limited utilization in ordinary clinical treatment, the majority of the issues in this sector are connected to trial implementation.
Key Drivers Fueling Demand for Advanced Therapy Medicinal Products
Surged investment to fund clinical trials has boosted the market of cell therapy in recent years. The institution of operational guidelines to assist cell therapy as well as the approval of sophisticated medicines have also aided the category's dominance.
The clearance of gene treatments in recent years has considerably expedited clinical trials in this field. Furthermore, Covid-19 opened new avenues for major players to invest in T-cell research to check viral infection. The research community is examining the possibilities of innovative medicines for COVID-19 patients, which will help the market flourish.
The United States' robust pipeline of Advanced Therapy Medicinal Products has propagated market revenue. In addition, corporations in the United States are moving their attention from traditional medication research to Advanced Therapy Medicinal Products, which is propelling regional growth.
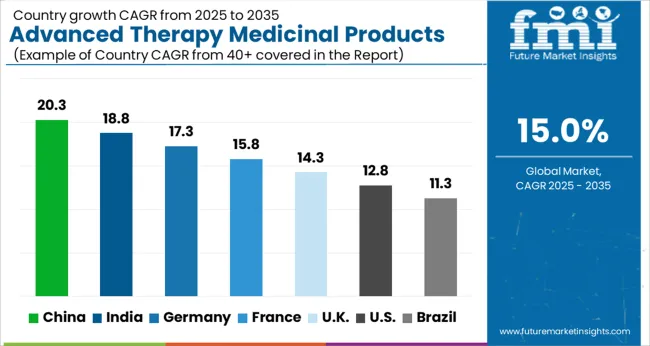
Due to the developing Advanced Therapy Medicinal Products landscape in emerging economies such as China, the market in the Asia Pacific is likely to grow at the highest rate. The rise of the regional market has been fueled by the expansion of China-based CDMOs.
Analysis of Regional Trends Shaping the Advanced Therapy Medicinal Products Industry
During the projected period, North America is expected to lead the global market for Advanced Therapy Medicinal Products with a CAGR of over 15%.
North America was the largest market for advanced therapy medicinal products with over 50% share in 2024. Investments in Research and Development and a favorable regulatory environment are the key dynamics for the large market for advanced therapy medicinal products in North America. In addition, the fast-growing number of clinical trials in this innovative therapeutic method in the United States contributes to regional growth.
Analysis of Top Countries Driving Advanced Therapy Medicinal Products Adoption
USA Advanced Therapy Medicinal Products Market Analysis
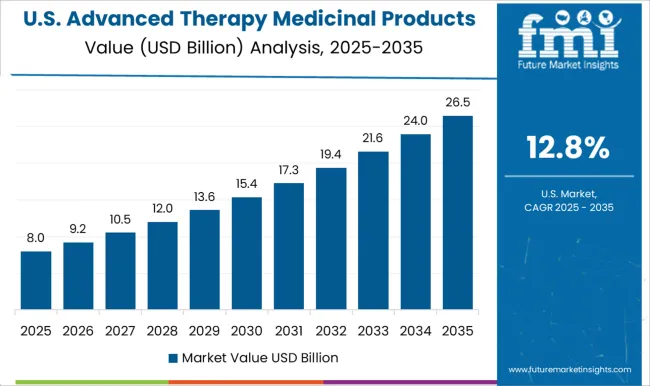
In March 2024, the United States accounted for roughly 60% of all ongoing gene therapy clinical trials worldwide, which is expected to lead to the advanced therapy medicinal products market at USD 52.4 Billion by 2035. The market in the US is expected to grow at a CAGR of 15.9%.
UK Advanced Therapy Medicinal Products Market Analysis
The market in the UK is projected to reach a valuation of USD 1.2 Billion by 2035. Growing at a CAGR of 15.9% from 2025 to 2035, the market in the country is expected to gross an absolute dollar opportunity of USD 13.2 Billion.
Japan Advanced Therapy Medicinal Products Market Analysis
In Japan, the market is expected to reach USD 1.4 Billion by 2035. With a CAGR of 15.3% during the forecast period, the market is projected to garner an absolute dollar opportunity of USD 1.1 Billion.
Advanced Therapy Medicinal Products Industry Analysis by Key Investment Segments
Which Component of Advanced Therapy Medicinal Products is expected to Score the Highest Growth in the Coming Years?
Tissue Engineered Product was the largest revenue-generating segment in the Advanced Therapy Medicinal Products Market in 2024, and it is expected to maintain its dominance during the forecast period, growing at a CAGR of 10.4%.
The incorporation of tissue-engineered procedures in treatments that need functional repair of any diseased or wounded area of the body can be linked to this dominance. The number of articles, research papers, translational products, and clinical trials, has increased significantly in the recent past.
Furthermore, the growth in revenue of tissue-engineered products is propelled by continual advancements in technology to encourage the tissue engineering process. For example, researchers at the University of Illinois in Chicago introduced a novel hydrogel-shaped-changing 4D material in February 2024. This material can change its shape in reaction to physiological stimuli.
On the other hand, the CAR-T treatment segment is gaining traction in the market and is predicted to grow at the quickest rate over the projection period. Novartis AG, for example, is concentrating on broadening the global reach of its CAR-T treatments. For instance, Health Sciences Authority recently authorized Kymriah as the first commercially licensed CAR-T therapy in Singapore.
Competitive Outlook for the Advanced Therapy Medicinal Products Market
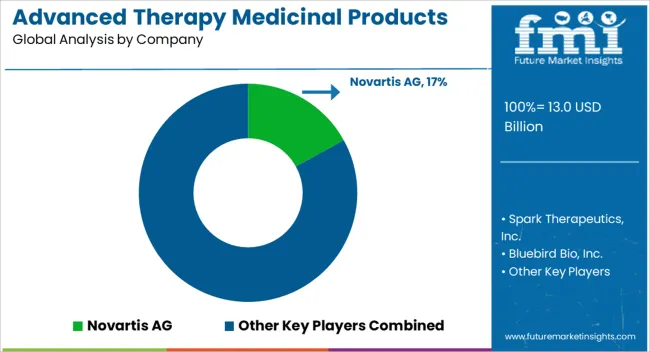
The Advanced Therapy Medicinal Products market is highly consolidated. Some of the key players in the market are Spark Therapeutics, Inc., Bluebird Bio, Inc., Novartis AG, UniQure N.V., Celgene Corporation, Gilead Lifesciences, Inc., Kolon TissueGene, Inc., JCR Pharmaceuticals Co., Ltd., MEDIPOST, Vericel Corporation, PHARMICELL Co., Ltd, and Organogenesis Inc.
Some of the recent developments of key Advanced Therapy Medicinal Products providers are as follows:
- In April 2025, the FDA broadened its recommendations for advanced biological products and offered further information to developers and manufacturers, with draught guidance for the development, manufacture, and control of chimeric antigen receptor (CAR) T cell products.
- In January 2024, FUJIFILM Diosynth Biotechnologies announced an investment of USD 40 million in a new process research and manufacturing facility for innovative medicines and viral vectors.
Similarly, recent developments related to companies in the Advanced Therapy Medicinal Products Market have been tracked by the team at Future Market Insights, which are available in the full report.
Top Segments Studied in the Advanced Therapy Medicinal Products Market Analysis
By Therapy Type:
- Cell Therapy
- Stem Cell Therapy
- Non-stem Cell Therapy
- CAR-T Therapy
- Gene Therapy
- Tissue Engineered Product
By Region:
- North America
- Europe
- Asia Pacific
- Middle East & Africa (MEA)
- Latin America
Frequently Asked Questions
How big is the advanced therapy medicinal products market in 2025?
The global advanced therapy medicinal products market is estimated to be valued at USD 13.0 billion in 2025.
What will be the size of advanced therapy medicinal products market in 2035?
The market size for the advanced therapy medicinal products market is projected to reach USD 52.4 billion by 2035.
How much will be the advanced therapy medicinal products market growth between 2025 and 2035?
The advanced therapy medicinal products market is expected to grow at a 15.0% CAGR between 2025 and 2035.
What are the key product types in the advanced therapy medicinal products market?
The key product types in advanced therapy medicinal products market are cell therapy, car-t therapy, gene therapy and tissue engineered product.
Which segment to contribute significant share in the advanced therapy medicinal products market in 2025?
In terms of , segment to command 0.0% share in the advanced therapy medicinal products market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Therapy Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Type , 2025 to 2035
- Cell Therapy
- CAR-T Therapy
- Gene Therapy
- Tissue Engineered Product
- Y-o-Y Growth Trend Analysis By Therapy Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Therapy Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Therapy Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Therapy Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Therapy Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Therapy Type
- Competition Analysis
- Competition Deep Dive
- Novartis AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Spark Therapeutics, Inc.
- Bluebird Bio, Inc.
- UniQure N.V.
- Celgene Corporation
- Gilead Lifesciences, Inc.
- Kolon TissueGene, Inc.
- JCR Pharmaceuticals Co., Ltd.
- MEDIPOST
- Vericel Corporation
- PHARMICELL Co., Ltd
- Organogenesis Inc.
- Novartis AG
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 3: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: North America Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 5: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: Latin America Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 7: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Western Europe Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 9: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 10: Eastern Europe Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 11: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 12: East Asia Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 13: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Type , 2020-2035
- Table 15: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 16: Middle East & Africa Market Value (USD Million) Forecast by Therapy Type , 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

