Biosimilar monoclonal antibody comparability test platforms market crossed a valuation of USD 0.8 billion in 2025 and is projected to reach USD 0.9 billion in 2026. By 2036, the market is forecast to expand to USD 2.3 billion at a 10% CAGR. Based on Future Market Insights analysis, the market is expected to create an incremental opportunity of USD 1.4 billion during 2026 to 2036. Demand is growing because developers need clearer analytical evidence, more consistent batch-to-batch comparison, and stronger support for regulatory filings.
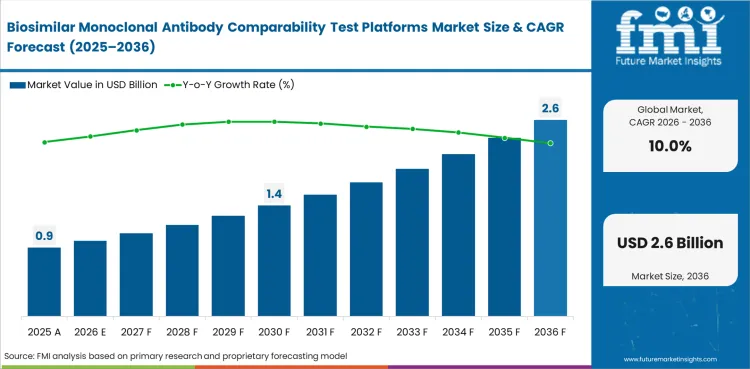
| Parameter | Details |
|---|---|
| Market value (2026) | USD 0.9 billion |
| Forecast value (2036) | USD 2.3 billion |
| CAGR (2026 to 2036) | 10% |
| Estimated market value (2025) | USD 0.8 billion |
| Incremental opportunity | USD 1.4 billion |
| Leading product type | Instruments (36% of product type segment) |
| Leading workflow stage | Analytical Similarity (44% of workflow stage segment) |
| Leading technology | Chromatography (27% of technology segment) |
| Leading assay type | Structural Assays (31% of assay type segment) |
| Leading molecule class | IgG1 mAbs (42% of molecule class segment) |
| Leading deployment model | In-house Labs (61% of deployment model segment) |
| Leading end user | Biosimilar Developers (49% of end user segment) |
| Key players | Danaher Corporation, Waters Corporation, Thermo Fisher Scientific, Agilent Technologies, Sartorius AG, Shimadzu Corporation, Charles River Laboratories |
Source: Future Market Insights, 2026.
Analytical comparison matters more in biosimilar monoclonal antibody development because regulators expect clear analytical evidence. Early test results help teams identify molecular differences before later development stage begin. Platform choice depends on method sensitivity, result consistency, software support, and how clearly the data can support comparability claims. Adoption improves when developers aligns each platform to routine characterization, validation, and filing needs.
India is expected to rise at 11.4% CAGR by 2036 because biosimilar development activity and regulatory progress are supporting more analytical platform use. South Korea is projected to grow at 11.1% CAGR as biologics manufacturing strength and biosimilar focus continue supporting demand. Demand in China is anticipated to expand at 10.7% CAGR through 2036, supported by rising domestic biologics development and broader analytical investment. Germany is likely to register 9.6% CAGR, while the United States is set to rise at 9.3% CAGR and the United Kingdom at 8.9% CAGR as established development environments continue adding more advanced comparability tools. Japan is expected to expand at 8.5% CAGR through 2036.
The biosimilar monoclonal antibody comparability test platforms market includes instruments, software, reagents, standards, and related testing systems used to compare a biosimilar monoclonal antibody with its reference product across structural, functional, and stability parameters. The scope is limited to platforms used for analytical similarity work, method development, validation support, and comparability review. This makes the market narrower than the broader monoclonal antibodies, space because it focuses on tools used to establish similarity.
The market includes chromatography systems, mass spectrometry platforms, electrophoresis tools, spectroscopy systems, bioassay platforms, data analysis software, reference standards, and supporting reagents used in biosimilar monoclonal antibody comparison. It also use across analytical similarity studies, method qualification, validation work, and stability-linked comparison. The focus stays on platforms directly used in biosimilar monoclonal antibody comparability programs.
The scope excludes full clinical trial services, biologic manufacturing equipment, fill-finish systems, upstream bioprocess hardware, and general laboratory tools that do not have a direct role in biosimilar monoclonal antibody comparison. Routine plant monitoring tools and broader control systems are excluded because they do not establish product similarity in the way analytical comparability.
Demand for biosimilar monoclonal antibody comparability test platforms is rising because developers need stronger analytical evidence earlier in development. Small differences in structure, glycan profile, charge pattern, or potency can create larger problems later if they are not identify early. These platforms help developments teams compare the biosimilar with the reference product in a more consistent way.
Analytical similarity is the main workflow area because comparability decisions start with direct evidence on product quality attributes. Development teams are giving more attention to systems that can support repeatable characterization, clean data output, and stronger method confidence across multiple batches. Chromatography, mass spectrometry, and related characterization tools continue to take an important role in biosimilar monoclonal antibody work.
Market growth is also supported by wider biosimilar development activity in countries adding new programs, manufacturing capacity, and regulatory support. Companies are looking at these platforms as part of a development and filing strategy that strengthen submission packages and supports similarity assessment. Demand is becoming more important in routine biosimilar development settings.
The biosimilar monoclonal antibody comparability test platforms market is segmented by product type, technology, assay type, workflow stage, molecule class, deployment model, and end user. By product type, the market is divided into instruments, software, reagents, standards, and services. In terms of technology, the market is classified into chromatography, mass spectrometry, electrophoresis, spectroscopy, and bioassays. Based on assay type, the market is segmented into structural assays, purity assays, glycan assays, potency assays, and stability assays. By workflow stage, the market is divided into analytical similarity, method development, method validation, stability testing, and release testing. In terms of molecule class, the market is classified into IgG1 mAbs, IgG4 mAbs, Fc fusions, fragments, and conjugates. Based on deployment model, the market is segmented into in-house labs, CRO labs, and hybrid labs. By end user, the market is divided into biosimilar developers, CDMOs, CROs, academic centers, and regulators.
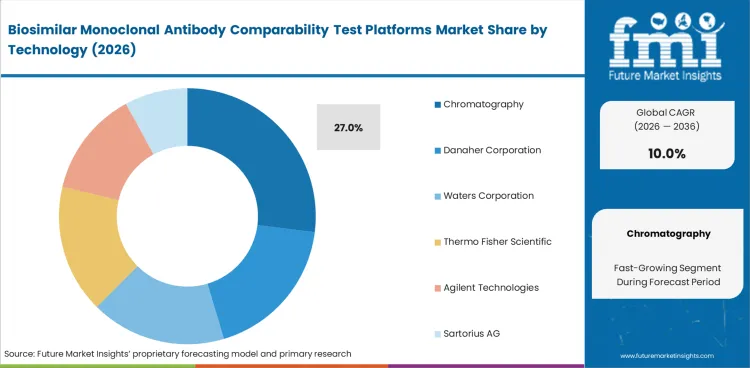
Chromatography leads this segment because biosimilar monoclonal antibody comparison requires clear separation of charge variants, size-related changes, purity differences, and glycan-linked variation. Chromatography is expected to account for 27% share in 2026. Mass spectrometry and electrophoresis remain important, though chromatography holds the broadest routine role because it is widely used, familiar, and dependable across biosimilar characterization workflows.
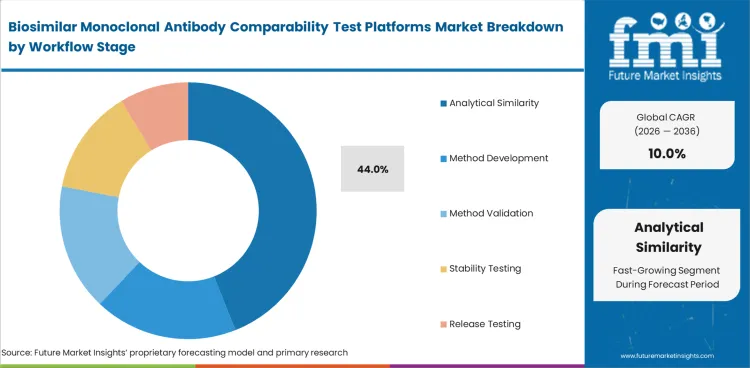
Analytical similarity leads this market because biosimilar assessment begins with direct comparison between the biosimilar and the reference product. Analytical Similarity is projected to hold 44% share in 2026. Developers give this stage the most attention because it supports similarity claims and later filing decisions. Method development and validation remain important, though they mainly support this core comparability work.
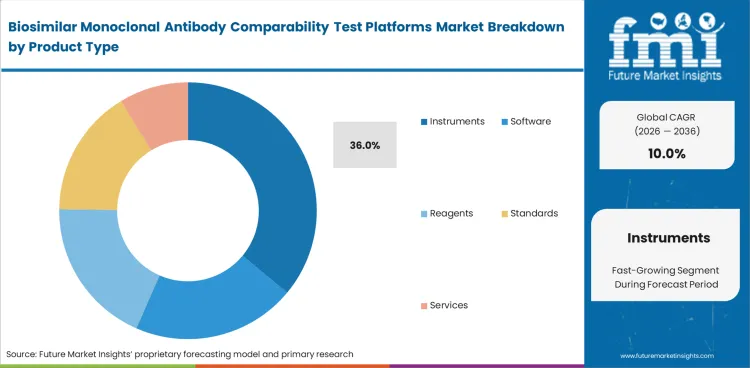
Instruments lead this segment because comparability programs depend first on the systems that generate the main analytical evidence. Instruments are likely to account for 36% share in 2026. Biosimilar developers need reliable tools for separation, detection, and characterization across many monoclonal antibody attributes. Software, reagents, and standards remain important, though spending usually begins with core laboratory platforms.
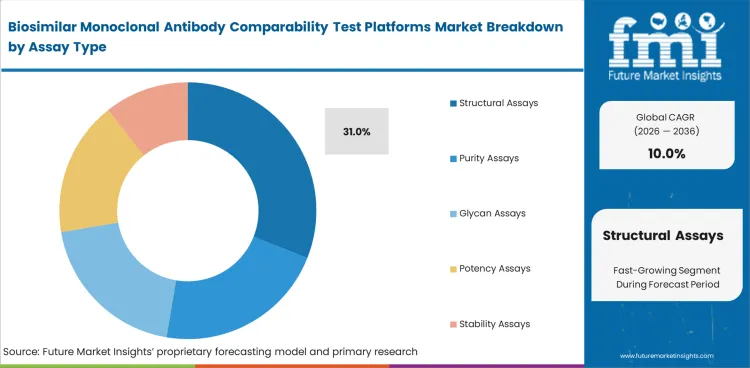
Structural assays lead because similarity studies usually begin with close review of the biosimilar’s physical and chemical profile. Structural Assays are projected to account for 31% share in 2026. Developers need early clarity on molecular composition, higher-order structure, and visible analytical differences before moving further. This first layer of evidence helps interpret later purity, potency, glycan, and stability findings.
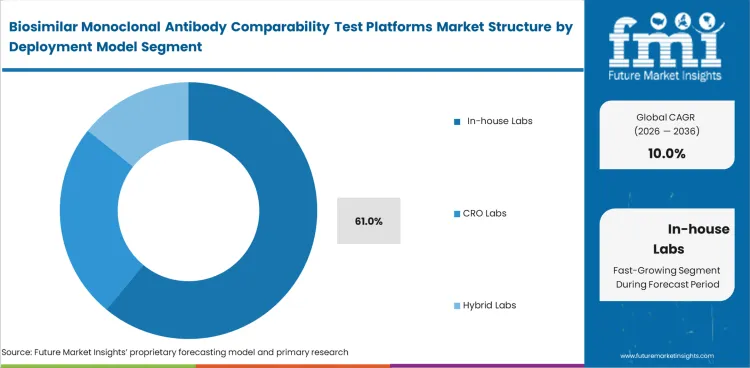
In-house labs are expected to account for 61% share in 2026 because many companies prefer to keep critical comparability work within their own scientific and quality groups. Internal control remains important in biosimilar development, especially in data review and submission planning. CRO labs support many programs, though sponsors often keep the most sensitive analytical work within their own organizations.
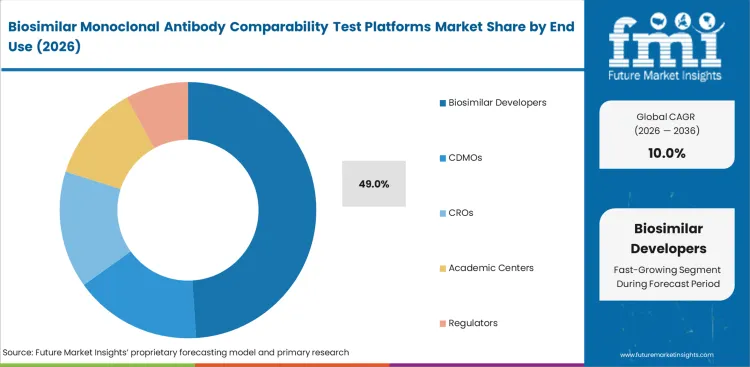
Biosimilar developers are anticipated to hold 49% market share in 2026 because they decide the comparability strategy and carry responsibility for proving similarity to regulators. CDMOs and CROs provide useful support, but the largest share of demand still comes from companies that own the biosimilar program. Platform needs continue rising as more sponsor-led biosimilar development programs move forward.
The biosimilar monoclonal antibody comparability test platforms market is expanding steadily as developers place more weight on analytical evidence that supports similarity claims with less uncertainty. Adoption depends on whether a platform can deliver repeatable output, support method confidence, and remains useful across multiple stages of comparability work. Activity in the biosimilars and follow-on biologics market continues to shape demand for these tools. Growth remains firm, though platform selection is still influenced by validation burden, workflow complexity, and the challenge of turning large volumes of analytical data into conclusions that hold up during review.
Demand is rising because biosimilar developers need clearer evidence that a candidate product remains close to the reference molecule across structural and functional attributes. Small difference in glycan profile, charge pattern, aggregation level, or potency can raise bigger questions later when they are not understood early. This is increasing investment in platforms that improve visibility at the analytical stage.
Broader adoption still faces limits because developers need more than sensitive instruments and wider assay coverage. They need validated methods, dependable reference standards, clean cross-batch comparison, and a clear path for explaining results that do not align neatly across assays. Similar buying appears in the analytical laboratory service market, where data quality must also supports defensible review, smoother documentation, and confidence in final decisions.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.4% |
| South Korea | 11.1% |
| China | 10.7% |
| Germany | 9.6% |
| United States | 9.3% |
| United Kingdom | 8.9% |
| Japan | 8.5% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research.
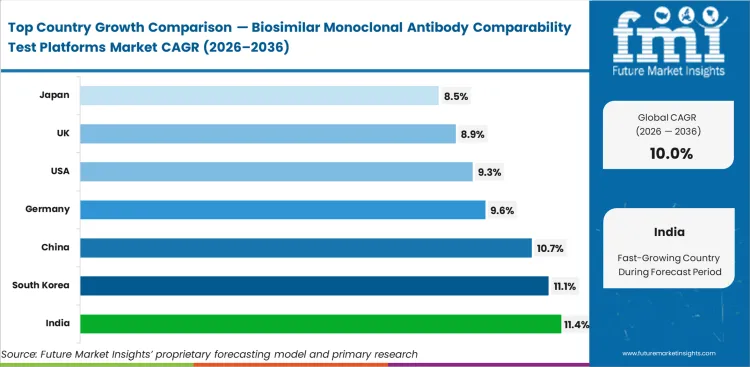
India is projected to expand at an 11.4% CAGR through 2036. Biosimilar development is growing across domestic and export-focused programs, which is increasing demand for platforms that support stronger head-to-head comparison with reference biologics. Companies want analytical systems that help build cleaner comparability packages without stretching timelines too far. This patterns also reflects growth in the large molecule bioanalytical testing services market.
South Korea is expected to grow at an 11.1% CAGR by 2036. Strong biologics manufacturing capability and a well-established biosimilar base continue to support demand for advanced comparability tools. Developers want platforms that improve similarity assessment without adding unnecessary complexity across analytical workflows. This gives an advantage to suppliers with stronger software, method support, and application depth.
China is projected to record 10.7% CAGR from 2026 to 2036. A growing biologics development base gives the market greater capacity to absorb advanced analytical systems across biosimilar monoclonal antibody programs. Adoption is supported by rising domestic research and manufacturing investment, though manufacturers still place strong emphasis on platform reliability, technical support, and result consistency.
Germany is projected to expand at 9.6% CAGR through 2036. Strong scientific infrastructure and a clear preference for methods that hold up under detailed technical review support this market. Developers and laboratory groups assess platforms on data clarity, reproducibility, and service depth rather than on instrument capability alone. This keeps demand steady for suppliers that can support well-documented, method-driven use across biosimilar characterization programs.
The United States is expected to grow at a 9.3% CAGR from 2026 to 2036. A large biologics and biosimilar development base supports continued demand for comparability platforms across sponsor laboratories, CROs, and selected CDMO-linked settings. Adoption moves forward through careful scientific and regulatory review, though the installed analytical foundation is strong enough to absorb new technologies without a full reset.
The United Kingdom is projected to register an 8.9% CAGR through 2036. Demand comes from research-led and development-led environments where method quality, scientific credibility, and workflow clarity carry more expansion. Organizations invest in comparability platforms with a long-term view, especially when those systems support better interpretation across structural and functional testing.
Japan is expected to expand at 8.5% CAGR through 2036. Companies move carefully because new analytical platforms must improve similarity assessment without creating extra burden across established quality and development routines. Method confidence, stable performance, and use-case clarity stay central in system selection, which keeps adoption remains measured. This supports stronger preference for dependable long-use systems over quick shifts toward unfamiliar platforms.
The biosimilar monoclonal antibody comparability test platforms market includes analytical instrument companies, software providers, life science tool suppliers, and selected testing support providers serving biosimilar development environments that need strong similarity evidence across structural and functional attributes. Competition depends on more than raw analytical capability. Developers compare method fit, workflow continuity, software usability, documentation support, and application depth before a platform becomes part of regular comparability work.
Danaher Corporation, Waters Corporation, and Thermo Fisher Scientific remain well placed because their analytical reach, technical credibility, and broad biopharma presence support buyer confidence in regulated biosimilar programs. Agilent Technologies, Sartorius AG, and Shimadzu Corporation stay competitive through strong relevance in characterization workflows, while Charles River Laboratories remains important where buyers want added testing support around the core analytical package. Platform choice usually depends on how well the system fits day-to-day comparability work rather than on sensitivity claims.
Barriers to entry are moderate at the basic instrument level and higher where suppliers need to support repeatable use in regulated biosimilar development. A company may offer technically sound analysis, though wider commercial adoption depends on whether the platform supports validation, cross-batch consistency, software-backed interpretation, and dependable output under filing-oriented review conditions. This keeps competition focused on practical scientific fit as much as on hardware capability.
Key global companies leading the biosimilar monoclonal antibody comparability test platforms market include:
| Company | Analytical Capability | Validation Support | Workflow Fit | Service / Application Support |
|---|---|---|---|---|
| Danaher Corporation | High | High | Strong | Strong |
| Waters Corporation | High | High | Strong | Strong |
| Thermo Fisher Scientific | High | High | Strong | Strong |
| Agilent Technologies | High | High | Moderate | Strong |
| Sartorius AG | Medium | High | Moderate | Strong |
| Shimadzu Corporation | Medium | Medium | Moderate | Moderate |
| Charles River Laboratories | Medium | Strong | Moderate | Strong |
Source: Future Market Insights competitive analysis, 2026.
Recent Developments in Biosimilar Monoclonal Antibody Comparability Test Platforms Market
Major Global Players
Emerging Players / Specialized Participants

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.8 billion (2025) to USD 2.3 billion (2036), at a 10% CAGR |
| Market Definition | The biosimilar monoclonal antibody comparability test platforms market covers instruments, software, reagents, standards, and related analytical systems used to compare a biosimilar monoclonal antibody with its reference product. These platforms support structural, functional, purity, glycan, stability, and similarity assessment across biosimilar development and regulatory review. |
| Segmentation |
|
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, South Korea, China, Germany, United States, United Kingdom, Japan, and 30+ countries |
| Key Companies Profiled | Danaher Corporation, Waters Corporation, Thermo Fisher Scientific, Agilent Technologies, Sartorius AG, Shimadzu Corporation, Charles River Laboratories, and other regional and specialized participants |
| Forecast Period | 2026 to 2036 |
| Approach | A blended bottom-up and top-down approach was used, supported by biosimilar development activity, analytical platform relevance across comparability workflows, supplier revenue direction, regulatory requirements, and demand modeling by technology, assay type, workflow stage, deployment model, and end user. Primary interviews and secondary industry data were used to validate market estimates and forecast assumptions. |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is Biosimilar Monoclonal Antibody Comparability Test Platforms Market in 2026?
Biosimilar Monoclonal Antibody Comparability Test Platforms Market is expected to reach USD 0.9 billion in 2026, showing clear commercial relevance in biosimilar development.
What will the market be valued at by 2036?
FMI estimates market valuation will reach USD 2.3 billion by 2036, reflecting broader use of analytical platforms in biosimilar monoclonal antibody comparison.
What CAGR is projected for the market during 2026 to 2036?
FMI projects a 10% CAGR from 2026 to 2036, indicating steady expansion in this specialized analytical category.
Which Technology segment leads the market?
Chromatography leads Technology, with 27% share expected in 2026 because it remains widely used in routine comparability workflows.
Which Workflow Stage segment leads the market?
Analytical Similarity leads the category, with 44% share expected in 2026, as direct similarity evidence remains central to biosimilar assessment.
Which Product Type segment leads the market?
Instruments lead Product Type, with 36% share expected in 2026 due to their core role in generating primary analytical evidence.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.