Thermodilution catheters remain relevant because certain critical-care decisions still demand invasive hemodynamic precision. In those settings, the debate is not whether the technology is old. It is whether the clinical context still justifies its use.
That makes this a selective but durable market. Demand is concentrated in the highest-acuity cardiac and shock settings, where protocol discipline, training, and risk management matter as much as simple procedure volume.
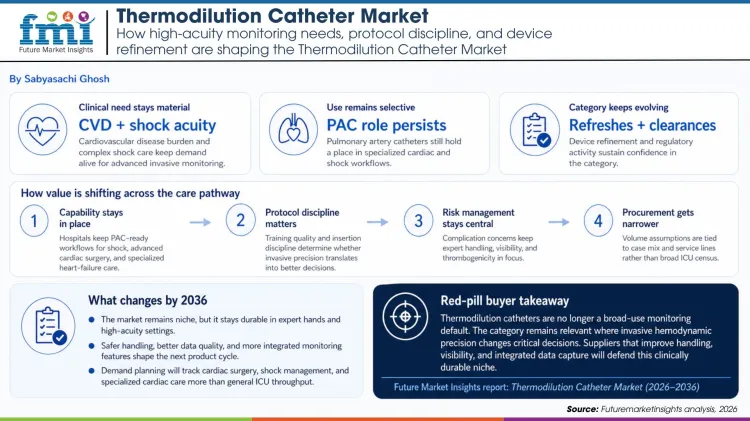
The global cardiovascular disease burden remains immense, and that keeps pressure on hospitals to maintain tools for complex perioperative and shock management. Thermodilution-based monitoring persists because certain patient groups still require more detailed hemodynamic assessment than noninvasive methods can reliably provide.
Usage is no longer broad by default, but it is still meaningful where acuity is highest. Contemporary CICU data and recent reviews show that PAC use remains concentrated in shock and specialized cardiac populations, which supports ongoing demand even as routine use stays limited.
Guidelines such as the Surviving Sepsis Campaign do not prescribe thermodilution in every case, but they leave room for advanced monitoring where individualized resuscitation and hemodynamic precision are necessary. Hospitals that manage complex cardiac and shock populations therefore keep PAC-ready workflows in place as a hedge against poor decision-making in unstable cases.
The supplier side also remains active. FDA clearances and device refreshes show that manufacturers still see commercial value in improving catheter performance, visibility, and data capture. That ongoing refinement helps prevent the category from slipping into pure legacy status.
| Market dimension | 2015-2025 | 2026-2036 |
|---|---|---|
| Clinical role | Broader use had already narrowed toward higher-acuity cases. | Use remains selective but stays important in shock and complex cardiac care. |
| Buyer rationale | Hospitals bought largely to preserve capability in specialized units. | Procurement focuses more tightly on case mix, protocol fit, and training quality. |
| Technology narrative | The category was often framed as mature and controversial. | Iterative improvements and smarter integration support continued use. |
| Risk lens | Complication concerns constrained broader adoption. | Risk management stays central, with more emphasis on expert handling. |
| Commercial outlook | Demand depended on specialized service lines. | That dependence remains, but high-acuity monitoring needs keep the niche durable. |
Pulmonary artery injury and other adverse events remain a serious consideration, which is why buyers cannot evaluate these catheters as simple disposable volume. Training, insertion protocols, thrombogenicity, and visibility features all affect whether the device improves care or introduces avoidable risk.
Commercial planning should be equally selective. The right demand lens is advanced cardiac surgery, shock management, and specialized heart-failure care, not broad ICU census numbers. In this market, a realistic case-mix view is essential to good contracting.
Recent product activity suggests the category will evolve through refinement rather than radical reinvention. Buyers should expect continued work on materials, torque control, and integrated sensing that improves workflow and confidence without changing the core invasive logic of the device.
That should keep the market stable rather than explosive. It remains indication-specific, but the indications that support it are serious and persistent. For deeper segment detail and the 2026-2036 outlook, see the Future Market Insights report: Thermodilution Catheter Market (2026 - 2036) - https://www.futuremarketinsights.com/reports/thermodilution-catheter-market
Growth in the Thermodilution Catheter Market is being supported by stronger end-user demand, operational efficiency needs, regulatory pressure, and wider adoption across relevant commercial and industrial applications.
High upfront costs, validation requirements, supply chain constraints, pricing pressure, and slower adoption among cost-sensitive buyers can restrict expansion in the Thermodilution Catheter Market.
Demand typically comes from manufacturers, service providers, healthcare or industrial operators, distributors, and specialized buyers that need reliable performance, compliance, and cost efficiency.
Regulations are pushing suppliers toward safer materials, better documentation, stronger quality controls, and products that help customers meet environmental, safety, or performance standards.
Companies should track raw material costs, technology upgrades, customer purchasing cycles, regional policy changes, and competitive moves that can alter pricing and adoption rates.
This report covers the coronary heart disease diagnostic imaging market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, imaging pathway shifts, supply-side developments, and strategic growth opportunities.
This report covers the mRNA vaccine formulation stability and stress test systems market through analysis of market size, market share, revenue forecast, system pricing benchmarks, company positioning, competitive landscape, demand outlook, growth drivers, restraints, technology trends, formulation testing requirements, assay integration, analytical method continuity, end-user adoption, supply chain developments, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
This report covers the cell therapy cryopreservation and thaw performance test platforms market through analysis of market size, market share, revenue forecast, platform segmentation, assay readout trends, cell type demand, preservation format usage, automation level adoption, end-user demand, workflow stage requirements, regional outlook, technology developments, handling standardization needs, method transfer practices, comparability testing, supply chain dynamics, regulatory and quality expectations, investment opportunities, profitability outlook, and strategic growth opportunities during 2026 to 2036.
The Gene Editing Off-Target Effect Detection Test Systems Market is segmented by Detection Approach (Cell-based Assays, Biochemical Assays, In Silico Tools, Orthogonal Validation), Product Format (Services, Assay Kits, Software, Instruments, Reagents), Workflow Stage (Site Nomination, Site Confirmation, Structural Follow-up, Release Support), Editing Modality (CRISPR-Cas9, Base Editing, Prime Editing, Cas12 Systems, Other Editors), Readout Technology (Amplicon NGS, Short-read WGS, Single-cell DNA, ChIP-seq Workflows, Long-read Sequencing), End User (Biopharma Companies, Academic Laboratories, CROs, CDMOs, Hospitals), Application (IND-enabling Studies, Discovery Screening, Cell Therapy Quality Assessment, In vivo Profiling, Comparability Studies), Deployment (Outsourced Testing, In-house Testing, Hybrid Models), and Region. Forecast for 2026 to 2036.
This report covers the at-home blood collection kits and devices market through analysis of market size, revenue forecast, competitive landscape, demand outlook, growth drivers, restraints, self-collection workflow trends, regulatory developments, and strategic growth opportunities.