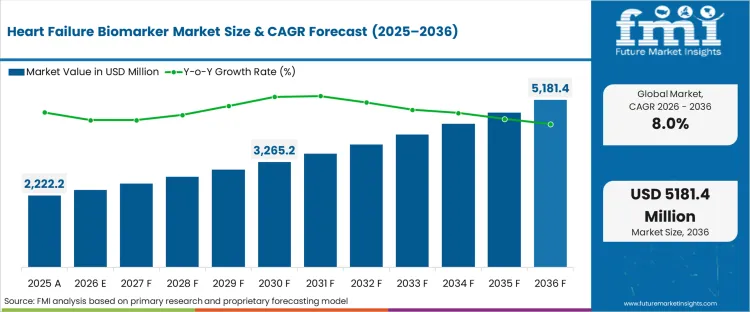
In 2026, the heart failure biomarker market was valued at USD 2,400.0 million. Based on Future Market Insights analysis, demand for heart failure biomarkers is estimated to grow to USD 5,200.0 million by 2036. FMI projects a CAGR of 8.0% during the forecast period.
Absolute dollar growth of USD 2,800.0 million over the decade signals structural expansion rather than a routine assay refresh. As per FMI, growth is expected to be supported by wider BNP/NT-proBNP use in emergency triage and inpatient management, while reimbursement controls and lab capacity constraints are expected to keep adoption paced across lower-resource settings. "We are extremely pleased that the FDA has granted Fast Track designation for VS-041, This designation reinforces the potential of VS-041 to address a serious unmet medical need with a novel mechanism of action that targets the underlying pathophysiology of HFpEF. It also supports the opportunity to establish a precision medicine paradigm in diagnosis and treatment of HFpEF by utilizing an emerging biomarker. We look forward to working closely with the FDA as we advance VS-041 through clinical development."[1]
Governmental regulatory approvals and adherence to national laboratory standards are also shaping purchasing decisions, as buyers are focusing on assays that have fulfilled government-approved performance standards. Purchasing decisions are also being shaped by standardization of cardiology and emergency department algorithms, as buyers are focusing on the ability of assays to be comparable across platforms, consistent turnaround times, and reagent standardization to prevent disruptions in high-volume testing. Multi-year reagent contracts are being utilized to standardize per-test costs, while device location strategies are being aligned with central lab capacity and point-of-care needs.
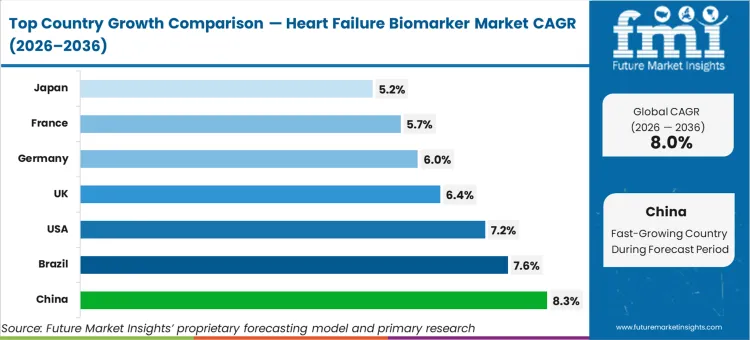
China (8.3% CAGR, supported by rising cardiometabolic burden and expanded hospital testing) and Brazil (7.6% CAGR, driven by growth in emergency and hospital diagnostics) are expected to lead growth. The U.S. (7.2% CAGR) is expected to expand through protocolised testing and readmission management focus. Mature markets including the UK (6.4%), Germany (6.0%), France (5.7%), and Japan (5.2%) are expected to contribute more through replacement and guideline-driven utilisation, constrained by budget governance and platform verification workload.
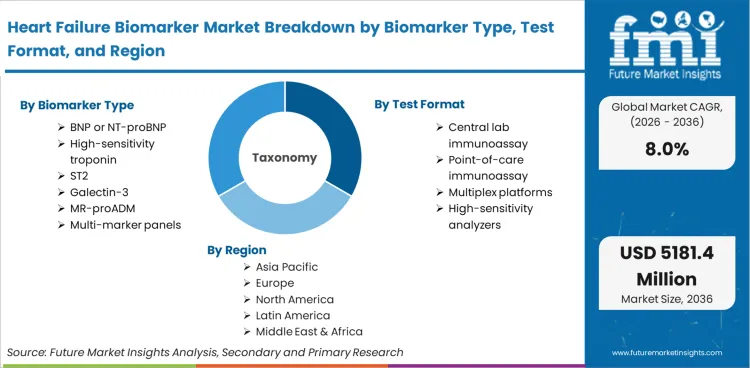
The market includes in vitro diagnostic tests for the detection and monitoring of biomarkers that support the diagnosis, triage, risk assessment, and monitoring of therapy in heart failure. The market includes BNP/NT-proBNP tests and other biomarkers such as high-sensitivity troponin, ST2, galectin-3, MR-proADM, and multi-biomarker panels, which are provided by immunoassay systems for central laboratory testing, point-of-care systems, and multiplex platforms. The tests are conducted using serum/plasma, whole blood, urine, and other types of samples in hospital, reference laboratory, and clinic settings, and selected home or remote monitoring settings. The market is driven by emergency triage requirements, pathway standardization, and the reduction of readmissions.
The report includes global and regional market sizes and a 10-year forecast for 2026 to 2036. Segment-level sizing is provided by biomarker type, test format, clinical use, sample, and end user, with country-level CAGR comparisons across major markets. Coverage includes assessment of protocol adoption in emergency and cardiology pathways, procurement dynamics in central lab versus point-of-care placements, and competitive positioning of leading assay and platform suppliers, as per FMI analysis.
The scope excludes imaging diagnostics such as echocardiography and cardiac MRI, as well as therapeutic drug products used to treat heart failure. It also omits hospital service revenue, procedure fees, and broader cardiometabolic lab testing not used for heart failure pathways. Consumer wellness monitoring devices and non-clinical screening tools are excluded. The focus remains on clinical biomarker assays and platforms aligned to the stated biomarker types, test formats, clinical uses, samples, and end users.
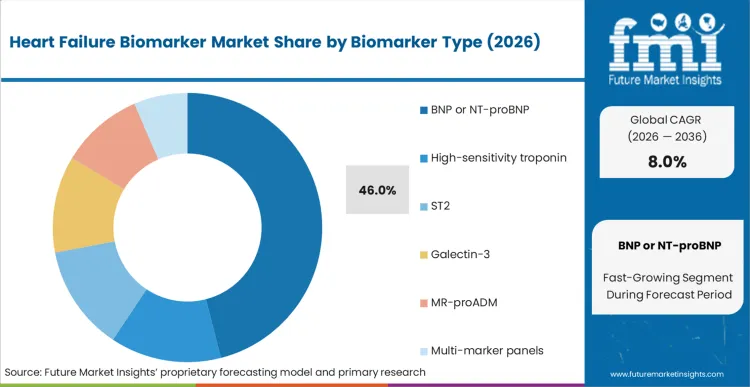
Based on FMI’s report, BNP/NT-proBNP is estimated to hold 46% share in 2026, followed by high-sensitivity troponin at 16% share. BNP/NT-proBNP leads because it is routinely embedded in diagnosis and triage pathways, with repeat use in monitoring and readmission management programs. High-sensitivity troponin follows due to overlap with acute care chest-pain pathways and broader cardiovascular workups, which sustains steady testing even when it is not exclusive to heart failure.
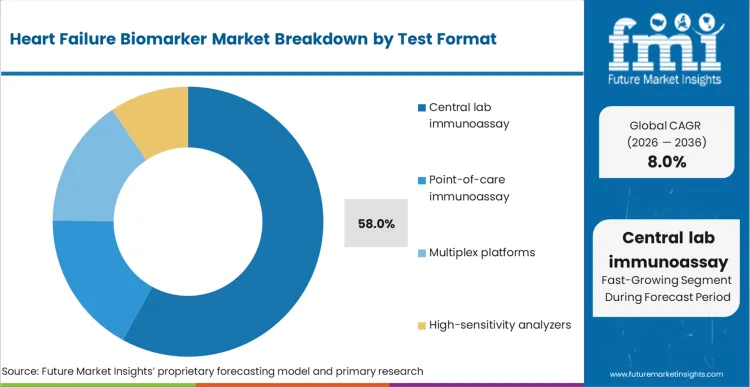
The market share of central lab immunoassay is 58% in 2026, followed by point-of-care immunoassay with 24% market share, according to the report by FMI. The dominance of central labs can be attributed to their higher volume, strict QC practices, and economies of scale, which are well-suited for hospital-wide ordering. Point-of-care immunoassay is driven by regions where rapid triage is emphasized, and results are required within short decision times.
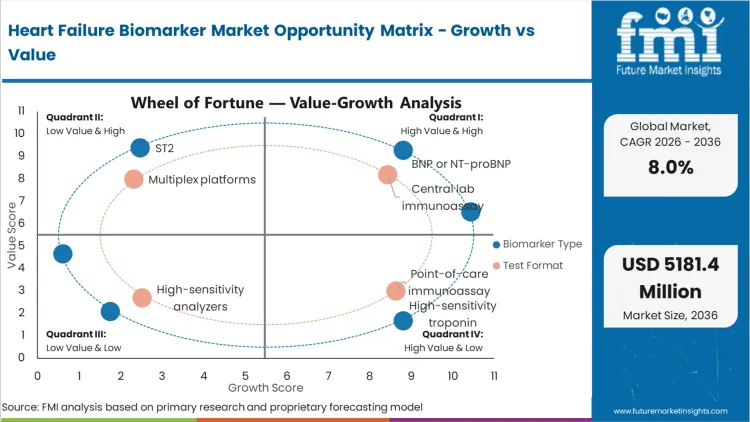
Future Market Insights analysis that historical patterns point at a protocol-driven diagnostics category where demand is anchored in emergency triage, inpatient decision-making, and chronic heart failure monitoring. Current demand is being shaped by hospital readmission reduction focus and the need to act on biomarker results within defined clinical decision windows, as per FMI.
While growth is being restrained by reimbursement discipline, assay standardization requirements, and variability in point-of-care deployment readiness, value expansion is being supported by rising test frequency in monitoring pathways and broader adoption of structured risk stratification programs. Based on FMI’s report, purchasing decisions are increasingly being filtered by analyzer compatibility, turnaround reliability, and supply continuity rather than by test price alone.
Based on the regional analysis, heart failure biomarker market is segmented into North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East & Africa across 40+ countries. Regional performance is interpreted through ED triage protocols, central lab immunoassay adoption, and penetration of BNP or NT-proBNP testing in hospitals and reference laboratories, as per FMI. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR |
|---|---|
| United States | 7.2% |
| Germany | 6.0% |
| United Kingdom | 6.4% |
| France | 5.7% |
| Japan | 5.2% |
| China | 8.3% |
| Brazil | 7.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
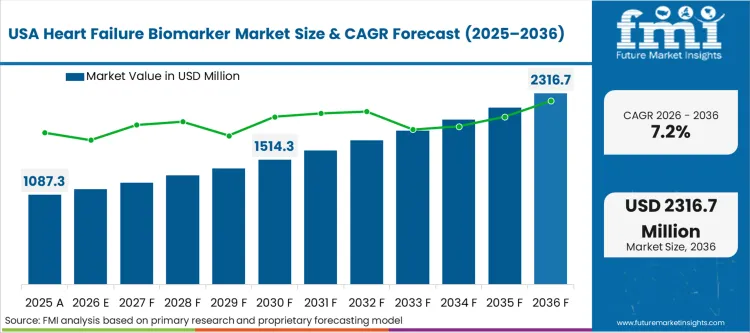
North America is shaped by ED driven diagnosis pathways and high central lab testing penetration, where BNP or NT-proBNP remains the default biomarker for diagnosis and triage, and where risk stratification demand supports repeat testing. Roche Diagnostics holds a strong position in central lab immunoassay workflows with broad hospital access. Abbott is widely adopted through integrated immunoassay platforms and emergency care channel reach. Siemens Healthineers competes through core lab presence and high-sensitivity assay ecosystems. FMI analysts note that hospitals remain the dominant end users, with reference labs supporting overflow and outpatient monitoring.
FMI’s report includes a detailed analysis of the growth in the North American region, along with a country-wise assessment that includes the USA, Canada and Mexico. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the North America region.
Latin America is driven by expansion of hospital diagnostics capacity and higher adoption of standard cardiac biomarker panels, with growth shaped by public hospital budgets and private lab chain expansion. Roche Diagnostics and Abbott remain key participants through hospital and reference lab access, while Siemens Healthineers supports testing growth through immunoassay platform placements. FMI opines that uptake is accelerating in larger urban centres where ED load and cardiology workups are rising.
The report consists of a detailed analysis for the market in Brazil, Argentina and Rest of Latin America. Readers can find detailed information about several factors, such as the pricing analysis and regional trends, which are impacting growth in the Latin America region.
The East Asia market is driven by the growing number of cases of cardiovascular diseases and the increasing capacity of hospital diagnostics, which is supported by the increasing use of BNP or NT-proBNP testing. Roche Diagnostics and Abbott continue to be major players in the market through their core lab placements and assays. The regional platform suppliers also affect purchases, which are driven by cost and geographic coverage. According to FMI analysts, centralized lab testing is currently the leading method, with multiplex platforms attracting attention for broader cardiac panels.
FMI’s report includes a detailed analysis of the growth in the East Asia region, along with a country-wise assessment that includes China, Japan and South Korea. Readers can also find regional trends, regulations, and market growth based on different segments and countries in the East Asia region.
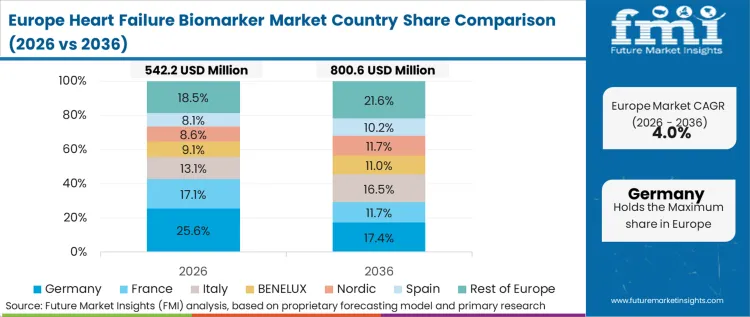
Western Europe is procurement-driven and guideline-driven, where adoption is influenced by ED pathways, national clinical guidelines, and central lab workflows. Roche Diagnostics maintains a strong base in core labs, Abbott competes through immunoassay platforms across hospitals, and Siemens Healthineers supports high-sensitivity analyzer ecosystems that interact with cardiac biomarker testing. According to FMI, the market is driven by the increasing use of risk stratification and the need for readmission reduction strategies in hospital settings.
FMI’s analysis of heart failure biomarker market in Western Europe consists of country-wise assessment that includes Germany, UK, France, Italy, Spain, BENELUX, Nordic Countries and Rest of Western Europe. Readers can know various regulations and latest trends in the regional market.
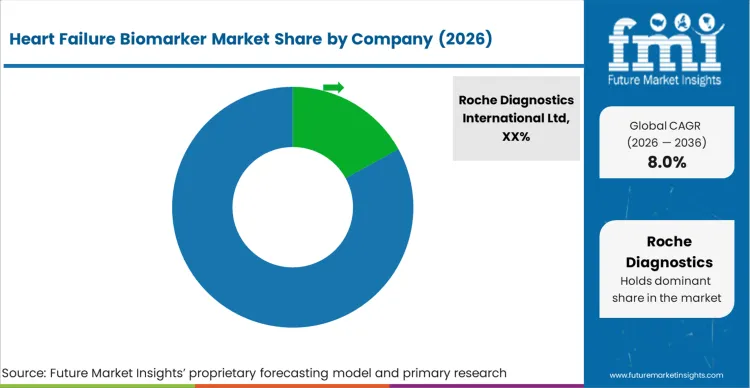
Market structure remains fragmented, yet practical competition is concentrated among a limited set of suppliers that can sustain assay quality consistency, maintain stable reagent supply, and support hospital and reference lab purchasing cycles with documented performance. Competition is shaped by clinical acceptance and repeat purchasing economics, since most volumes flow through central lab immunoassay workflows and are renewed through structured tenders. The primary competitive variable is confidence in result stability across lots and instruments, since the test is used in time-sensitive pathways such as emergency diagnosis support and inpatient risk management, as per FMI.
Companies with structural advantages tend to combine large installed bases in core lab immunoassay with broad assay menus and service coverage that keeps downtime low. Suppliers that can support both central lab systems and point-of-care formats are better positioned where hospitals want a single vendor across triage and routine monitoring. Control over calibrators, controls, and instrument specific assay tuning supports comparability across sites, which matters when networks standardise clinical protocols and quality dashboards. Firms with weaker service density or narrower menu coverage face higher exposure during rebids because hospitals tend to rationalise vendors when QC burden rises, based on FMI’s report.
Customer concentration reinforces buyer leverage. Hospitals and integrated systems commonly qualify more than one supplier, maintain approved alternates for high-volume markers, and use contract clauses tied to supply continuity and complaint closure timelines. Reference labs enforce similar discipline through volume tiers and performance scorecards, which keeps net pricing controlled in routine testing. Pricing power is therefore constrained for standard natriuretic peptide testing, while measured premiums are retained mainly where a platform offers workflow efficiency, rapid turnaround support, or integration that reduces revalidation effort across multi-site networks, Future Market Insights analysis.
Recent Developments:
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
| Metric | Value |
|---|---|
| Quantitative Units | USD 2,400.0 Million (2026) to USD 5,200.0 Million (2036), at a CAGR of 8.0% |
| Market Definition | The heart failure biomarker market comprises global production and trade of in vitro diagnostic tests and related consumables used to support diagnosis, triage, risk stratification, therapy monitoring, and prognosis assessment in heart failure pathways, where adoption is shaped by emergency care throughput, laboratory turnaround time requirements, and guideline aligned use of natriuretic peptides and related marker panels. |
| Biomarker Type Segmentation | BNP or NT-proBNP, High-sensitivity troponin, ST2, Galectin-3, MR-proADM, Multi-marker panels |
| Test Format Segmentation | Central lab immunoassay, Point-of-care immunoassay, Multiplex platforms, High-sensitivity analyzers |
| Clinical Use Coverage | Diagnosis or triage, Risk stratification, Therapy monitoring, Prognosis or readmission |
| Sample Coverage | Serum or plasma, Whole blood, Urine, Other |
| End User Coverage | Hospitals, Reference Laboratories, Diagnostic Laboratories, Specialty Clinics and Ambulatory Surgical Centers |
| Regions Covered | North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe, and Middle East & Africa. |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 40+ countries |
| Key Companies Profiled | Roche Diagnostics International Ltd, Abbott Laboratories, Siemens Healthineers AG, Beckman Coulter, Inc., bioMérieux SA, QuidelOrtho Corporation, Radiometer Medical ApS, Randox Laboratories Ltd, DiaSorin S.p.A., Thermo Fisher Scientific Inc. |
| Forecast period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with cardiology and emergency care stakeholders and diagnostics suppliers, supported by testing volume triangulation and platform placement benchmarking, as per FMI. |
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
How large is the demand for Heart Failure Biomarkers in the global market in 2026?
Demand for Heart Failure Biomarkers in the global market is estimated to be valued at USD 2,400.0 Mn in 2026, as per FMI.
What will be the market size of Heart Failure Biomarkers in the global market by 2036?
Market size for Heart Failure Biomarkers is projected to reach USD 5,200.0 Mn by 2036.
What is the expected demand growth for Heart failure biomarkers in the global market between 2026 and 2036?
Demand for Heart Failure Biomarkers in the global market is expected to grow at a CAGR of 8.0% between 2026 and 2036.
Which biomarker type is poised to lead global demand by 2026?
BNP or NT-proBNP is expected to be the dominant biomarker type, capturing 46% share in 2026.
Which test format is expected to account for the largest share in 2026?
Central lab immunoassay is expected to hold the highest share at 58% in 2026.
How significant is the hospital end user segment in 2026?
Hospitals are projected to account for 60% share of end user demand in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.