Heart Pump Device Market
The Heart Pump Device Market is segmented by Type (Implanted Heart Pump Devices and Extracorporeal Heart Pump Devices), Product (Left Ventricular Assist Devices (LVAD), Total Artificial Hearts (TAHs), Intra-aortic Balloon Pumps (IABPs), and Extracorporeal Membrane Oxygenation (ECMO)), End Use (Hospitals, Cardiac Centers, and Others) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Heart Pump Device Market Forecast and Outlook By FMI
In 2025, the heart pump device market was valued at USD 4.8 billion. Based on Future Market Insights’ analysis, demand for heart pump devices is estimated to grow to USD 5.5 billion in 2026 and USD 23.2 billion by 2036. FMI projects a CAGR of 15.4% during the forecast period. Growth is held up by widening eligibility for durable support in advanced heart failure and by ICU shock pathways that increasingly use percutaneous support, while costs, adverse-event management, and centre capability requirements keep uptake concentrated in specialized hospitals.
As Daniel Timms, Founder and Chief Technical Officer of BiVACOR, noted regarding the first-in-human total artificial heart milestone, “I’m incredibly proud to witness the successful first-in-human implant of our TAH. This achievement would not have been possible without the courage of our first patient and their family, the dedication of our team, and our expert collaborators at The Texas Heart Institute. Utilizing advanced MAGLEV technology, our TAH brings us one step closer to providing a desperately needed option for people with end-stage heart failure who require support while waiting for a heart transplant. I look forward to continuing the next phase of our clinical trial. [1]” The near-term implication is that the product roadmap is widening beyond LVAD iteration into full-replacement platforms, which raises the ceiling for future procedure volumes, but also increases the evidence burden for payers and guideline bodies.
Summary of Heart Pump Device Market
- Heart Pump Device Market Definition
- The market covers mechanical circulatory support devices, implantable and extracorporeal, used to sustain blood flow in advanced heart failure and cardiogenic shock, procured mainly by tertiary hospitals and specialist cardiac centres.
- Demand Drivers in the Market
- Heart failure prevalence and ageing demographics expand the eligible cohort and increase referral into advanced therapy pathways. [5]
- Transplant scarcity and prolonged waiting times push higher use of bridge strategies and destination therapy, sustaining durable LVAD demand. [6]
- Evidence-led shock protocols and cath-lab adoption of percutaneous support expand use in AMI-related cardiogenic shock episodes. [3]
- Key Segments Analyzed in the FMI Report
- Type: Implanted heart pump devices lead (68.7% in 2025) as long-duration therapy cohorts expand in transplant-constrained systems.
- Product: LVADs lead (69.7% in 2025), while total artificial hearts progress from clinical milestones toward early commercial pathways.
- Geography: North America and Western Europe set reimbursement and protocol benchmarks, while East Asia and South Asia & Pacific add incremental patient volumes through tertiary-care expansion and transplant ecosystem buildout. [6]
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Senior Analyst for Medical Devices at Future Market Insights, opines, ‘In the updated version of the Heart Pump Device Market Report for 2026 to 2036, CXOs will find that adoption is governed by coverage rules, centre accreditation, and complication economics more than device miniaturisation. My analysis tracks how haemocompatibility event control, field-action readiness, and centre enablement programmes convert into contracting leverage and share gains.’
- Strategic Implications/Executive Takeaways
- Prioritise payer-facing evidence packages linked to readmissions, adverse-event rates, and total episode cost because reimbursement sets the utilisation ceiling. [6]
- Invest in centre enablement, proctoring, and troubleshooting infrastructure to expand implant-capable footprints in high-growth countries without diluting outcomes.
- Build rapid field-action playbooks and dual-source critical components to protect hospital confidence when safety notices or kit issues arise. [4]
- Methodology
- Hybrid procedure-led top down and bottom up modelling validated through primary interviews with cardiac surgeons, heart failure cardiologists, perfusion teams, and hospital procurement leaders.
- Desk research anchored in regulator communications, payer coverage rules, and national transplant reporting, with triangulation against clinical pathway and centre capability indicators. [4]
- All assumptions stress-tested against verifiable disclosures and published clinical evidence, with updates triggered by safety notices, coverage revisions, and major product or trial milestones. [3]
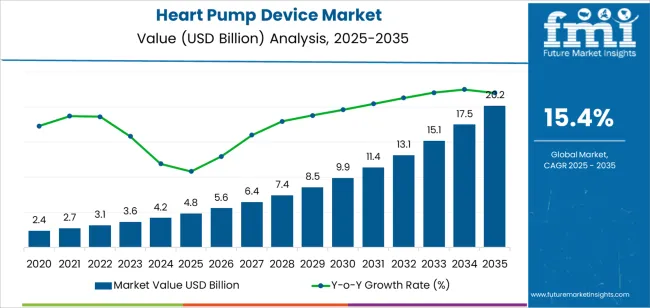
| Metric | Value |
|---|---|
| Estimated market value (2026) | USD 5.5 billion |
| Forecast market value (2036) | USD 23.2 billion |
| CAGR (2026 to 2036) | 15.4% |
India (17.8% CAGR) and China (17.2% CAGR) are projected to accelerate through tertiary-care expansion and rising advanced heart failure case finding, while South Korea (16.9% CAGR) advances via reimbursement-linked centre buildout. The United Kingdom (15.8% CAGR) and Japan (14.6% CAGR) expand through structured transplant and MCS programmes with measured patient selection. Mature markets such as the United States (13.5%), Germany (13.9%), and France (13.2%) generate a mix of replacement and pathway broadening, with growth constrained by donor availability, complication management costs, and strict centre criteria.
Heart Pump Device Market Definition
The heart pump device market covers mechanical circulatory support devices used to assist or replace cardiac pumping function in severe heart failure or acute cardiogenic shock. It includes implantable pumps such as LVADs and total artificial hearts, plus extracorporeal systems used in hospitals such as ECMO and balloon pumps. Buyers are mainly hospitals and specialist cardiac centres that use these devices for bridge-to-transplant, bridge-to-recovery, or long-term therapy when transplantation is not available or not suitable.
Market inclusions
This report covers global and regional market sizing for 2026 to 2036, with historical anchoring for 2025. It includes segmentation by type, product, and end use, plus region and selected country-level forecasts. The scope includes adoption drivers tied to reimbursement, transplant constraints, and clinical pathway design, alongside competitive positioning, pipeline signals, and selected regulatory and safety events relevant to device utilisation.
Market exclusions
This scope excludes pharmaceuticals for heart failure, remote monitoring software sold as standalone platforms, and general ICU consumables not specific to mechanical circulatory support. It also excludes downstream rehabilitation services, home-care nursing services, and non-device transplant infrastructure spending. Paediatric-only devices are not modelled as a separate market unless sold into the same commercial portfolio and purchasing channel as adult systems.
Heart Pump Device Market Research Methodology
- Primary research: FMI conducts interviews with cardiac surgeons, heart failure cardiologists, perfusionists, hospital procurement leads, and device OEM product managers to map procedure volumes, pathway changes, and purchasing criteria.
- Desk research: FMI reviews regulator communications, payer coverage rules, guideline statements, device safety notices, company disclosures, and national transplant activity reporting to validate adoption constraints and utilisation patterns. [4]
- Market-sizing and forecasting: FMI uses a hybrid model combining bottom-up procedure-based estimates (implant and support episodes) with top-down hospital spending and device shipment triangulation, then applies country-level adoption curves linked to centre availability and reimbursement.
- Data validation and update cycle: Forecast outputs are cross-checked against historical procedure signals, payer rule changes, major safety events, and disclosed portfolio updates, then refreshed when new regulator notices, national transplant reporting, or company evidence changes the addressable cohort.
Segmental Analysis
Heart Pump Device Market Analysis by Type
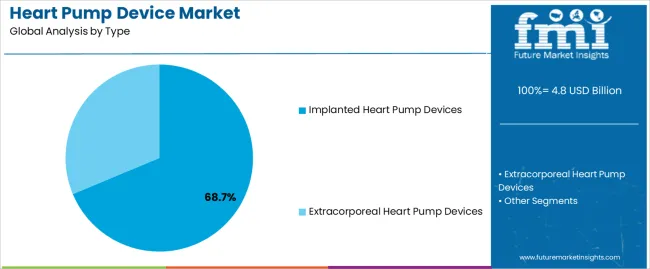
Based on FMI’s heart pump device market report, consumption of implanted heart pump devices is estimated to hold 68.7% share in 2025. Implantable systems dominate because they support long-duration therapy outside the ICU and fit destination-therapy pathways when transplant supply is limited, making them central to advanced heart failure programmes.
- Label shift: Abbott reported an FDA-approved labelling update supporting an aspirin-free regimen for HeartMate 3 patients, a practical signal aimed at bleeding-risk management that can influence centre preference and protocol design. [2]
- Safety discipline: The FDA classified a HeartMate 3 LVAS implant kit recall as Class I due to risks tied to blood leakage or air entering the system, reinforcing procurement focus on kit integrity and field-action readiness. [4]
- Category expansion: BiVACOR’s first-in-human total artificial heart milestone raises the strategic ceiling for implanted solutions by extending beyond assist to full replacement, expanding long-run addressable cohorts if outcomes scale. [1]
Heart Pump Device Market Analysis by Product
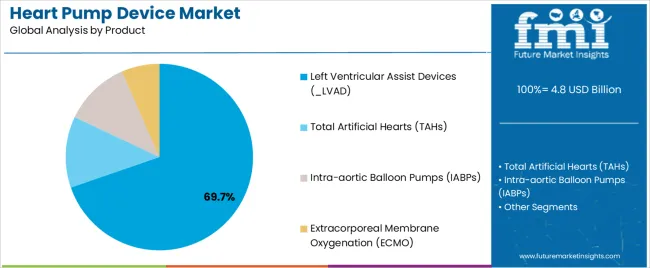
Based on FMI’s heart pump device market report, consumption of left ventricular assist devices (LVAD) is estimated to hold 69.7% share in 2025. LVADs lead because left-sided failure is the most common mechanical support indication, and LVAD pathways are supported by established centre experience and payer frameworks.
- Shock pathway evidence: In AMI-related cardiogenic shock, the DanGer Shock trial summary reports a mortality reduction with Impella support versus standard care, supporting wider cath-lab adoption of percutaneous support protocols in suitable patients. [3]
- Guideline and event focus: The 2024 ISHLT consensus on hemocompatibility-related events in durable VADs reflects how bleeding and thrombotic risk management is central to long-term LVAD outcomes and purchasing evaluation. [7]
- Transplant constraint effect: The UK’s national reporting on heart transplantation activity and waiting-list pressures reinforces the structural role of LVADs as bridge or long-term therapy when donor availability limits transplant throughput. [8]
Heart Pump Device Market Drivers, Restraints, Opportunities
Future Market Insights analysis indicates the market’s size is anchored in a high-severity clinical need: advanced heart failure cohorts that exceed transplant supply, plus acute shock episodes where rapid mechanical support can stabilise patients. Heart failure prevalence data in large markets supports the scale of the treatable pool, but conversion into device therapy depends on referral timing, centre capacity, and payer coverage rules. [5]
A clear transition is underway from “bridge-to-transplant only” positioning to broader lifetime-support pathways that include destination therapy and multi-year outpatient management. This shifts value from one-time device placement toward longitudinal service capability, remote follow-up, and complication management. At the same time, safety events and hemocompatibility risks keep decision-makers focused on outcomes and total cost of care rather than unit price.
- Coverage-gated adoption: CMS coverage for destination therapy ties utilisation to FDA indication and patient selection, which makes reimbursement rules a hard constraint on volumes in the United States. [6]
- Complication economics: Pump thrombosis, bleeding, and infection risks drive protocol intensity and raise episode costs, pushing hospitals to favour devices and pathways with measurable adverse-event reductions. [7]
- Centre concentration: Implant volumes track the distribution of advanced heart failure centres and transplant-linked networks, so capacity expansion in India and China can lift volumes faster than in saturated markets.
Regional Analysis
The market is analysed across North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and the Middle East & Africa. Regional forecasts reflect differences in transplant availability, centre capability, payer rules, and the maturity of cardiogenic shock pathways, with the full report providing market attractiveness analysis by country and segment.
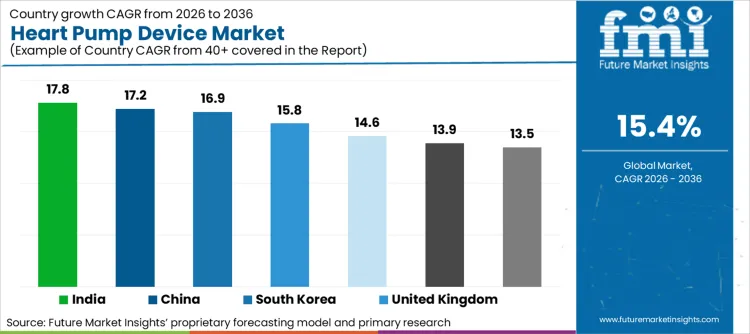
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 17.8% |
| China | 17.2% |
| South Korea | 16.9% |
| United Kingdom | 15.8% |
| Japan | 14.6% |
| Germany | 13.9% |
| United States | 13.5% |
| France | 13.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America functions as the evidence and reimbursement rule-setter for durable LVAD utilisation, with clinical pathways shaped by major academic centres and payer criteria. Abbott (HeartMate portfolio) and Johnson & Johnson’s Abiomed (Impella) are central reference suppliers, and hospital buyers place heavy weight on outcomes evidence, complication profiles, and regulator communications. [2]
- United States: Demand for heart pump devices in the United States is projected to rise at 13.5% CAGR through 2036. Coverage rules are a primary gate: CMS destination therapy coverage ties reimbursement to FDA-approved indications and defined patient selection, which pushes hospitals to standardise evaluation workflows and centre qualification. [6] Clinical adoption in acute shock also tracks evidence-led protocols, with DanGer Shock results supporting broader consideration of percutaneous support in AMI-related cardiogenic shock for selected patients. [3] Safety and quality systems remain procurement-critical, as illustrated by FDA recall communications affecting implant kits, which can drive immediate inventory checks and supplier scrutiny across implanting centres. [4]
- Canada: Demand for heart pump devices in Canada is projected to rise at 13.1% CAGR through 2036. Utilisation is shaped by the concentration of advanced heart failure and transplant programmes in a limited number of high-volume centres, which tends to centralise procurement and standardise protocols. Canadian hospitals typically align device selection to international evidence and guideline-based haemocompatibility management approaches, since long-term outcomes and complication reduction drive total episode cost and ICU utilisation. [7] Donor constraints remain a background driver for bridge strategies, so devices that reduce adverse events and support outpatient stability carry procurement weight. Canada’s purchasing model often emphasises value-based evaluation, where safety notices and regulator communications can accelerate internal reviews and drive conservative stocking and training practices.
FMI's analysis of heart pump device market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find centre concentration mapping, payer-rule impact assessment, and segment attractiveness by type and product for advanced heart failure and shock pathways.
Western Europe Market Analysis
Western Europe acts as a structured adopter region where guideline alignment, transplant capacity, and national service configuration shape the pace of LVAD and short-term support growth. Abbott’s LVAD franchise and J&J Abiomed’s Impella platform are widely referenced in clinical pathways, while national transplant bodies influence referral timing and therapy selection. [2]
- United Kingdom: Demand for heart pump devices in the United Kingdom is projected to rise at 15.8% CAGR through 2036. National reporting indicates persistent transplant system pressure, and the 2024/2025 heart transplantation report provides the planning backbone for capacity and pathway decisions across specialist centres. [8] This environment supports LVAD use as bridge and as longer-duration therapy where eligible cohorts exceed donor throughput, with purchasing decisions centred on complication management and service capability rather than device hardware alone. ICU and perioperative standards are also shaped by UK-focused clinical practice resources for LVAD patient management, reinforcing the role of trained multidisciplinary teams and standardised response to bleeding, infection, and device alarms. [7]
- Germany: Demand for heart pump devices in Germany is projected to rise at 13.9% CAGR through 2036. Germany’s advanced heart failure ecosystem is closely tied to European guideline positioning for long-term mechanical support in selected refractory patients, which supports a stable base of LVAD utilisation in high-volume cardiac centres. [9] A notable demand signal comes from ongoing registry-driven practice and published analyses of LVAD therapy trends, which shape hospital protocol choices and training priorities. [9] Procurement emphasis often sits on haemocompatibility risk reduction, complication control, and service readiness, aligning with international consensus focus on durable VAD adverse-event management. [7]
FMI's analysis of heart pump device market in Western Europe consists of country-wise assessment that includes the United Kingdom and Germany. Readers can find transplant-linked pathway modelling, reimbursement and service configuration impacts, and product-level attractiveness for LVAD and percutaneous support systems.
East Asia Market Analysis
East Asia is a capacity-build and localisation region, combining rising advanced heart failure detection with domestic device programmes and growing clinical trial footprints. Local manufacturers in China and Japan’s structured MCS ecosystem influence adoption, while international suppliers remain influential in high-acuity segments.
- China: Demand for heart pump devices in China is projected to rise at 17.2% CAGR through 2036. A key mechanism is domestic capability building: China’s regulator has previously issued approvals for domestically developed implantable “artificial heart” systems, signalling local industrial commitment to LVAD platforms. [10] Clinical scale signals also come from published experience with domestically used LVAD systems, including reports of post-approval utilisation and multi-centre trial activity, which supports broader physician familiarity and training depth. [11] As domestic platforms mature, hospitals can reduce dependence on imported systems for some cohorts, which can improve affordability and expand eligible patient conversion, particularly in large tertiary centres. [3]
- Japan: Demand for heart pump devices in Japan is projected to rise at 14.6% CAGR through 2036. Japan’s destination therapy pathway has been building with structured patient selection and registry-style learning, and published discussion notes the recording of destination-therapy LVAD cases as the pathway expands. [12] This supports durable implanted device demand in a system where transplant waiting times and donor constraints are relevant decision factors. Care delivery is strongly protocol-led, so haemocompatibility management and adverse-event prevention weigh heavily in centre evaluations, consistent with the focus of 2024 consensus work on durable VAD event management. [7]
FMI's analysis of heart pump device market in East Asia consists of country-wise assessment that includes China and Japan. Readers can find domestic versus imported platform positioning, centre capability mapping, and product adoption curves for LVAD and ICU-based support pathways.
South Asia & Pacific Market Analysis
South Asia & Pacific is the highest incremental volume opportunity region, driven by the expansion of tertiary cardiac care, a rising transplant base, and the creation of structured organ donation and allocation programmes. International suppliers shape high-end care, while local programmes improve conversion of end-stage heart failure patients into advanced therapy pathways. [13]
- India: Demand for heart pump devices in India is projected to rise at 17.8% CAGR through 2036. The adoption mechanism is capacity and system building: national reporting shows growth in deceased donation and deceased transplants between 2016 and 2024, which supports broader transplant-linked cardiac care ecosystems and raises the relevance of bridge-to-transplant strategies. [13] Professional reporting also documents heart transplant activity counts in recent years, reinforcing the expansion of advanced heart failure treatment capacity that often co-develops with mechanical support capability. [14] As tertiary centres expand, LVAD and ECMO utilisation can rise through better referral networks, more perfusion capacity, and improved perioperative protocols.
- South Korea: Demand for heart pump devices in South Korea is projected to rise at 16.9% CAGR through 2036. A strong adoption driver is the maturation of transplant and mechanical support practice: published reporting from the Korean Organ Transplant Registry reflects a growing transplant base and the rising utilisation of mechanical support within advanced cardiac care. [15] This increases the clinical workforce familiarity with MCS management and can raise LVAD candidacy identification. South Korea’s high-tech hospital ecosystem supports protocol-led expansion, with an emphasis on adverse-event management and long-run haemocompatibility risk control aligned with international consensus work. [7]
FMI's analysis of heart pump device market in South Asia & Pacific consists of country-wise assessment that includes India and South Korea. Readers can find transplant-system-linked forecasting, hospital capacity mapping, and segment attractiveness for implantable versus extracorporeal support.
Competitive Aligners for Market Players
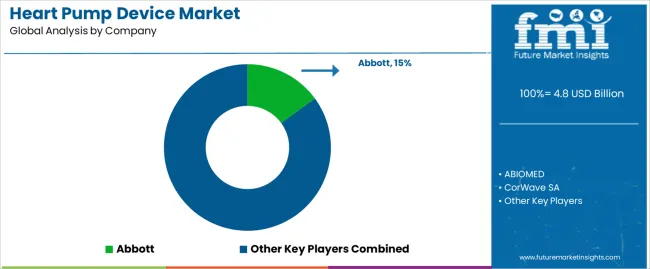
The competitive structure is moderately concentrated in core sub-segments. Durable implanted LVAD demand is led by established platforms with deep clinical evidence and centre familiarity, while acute shock support is influenced by cath-lab workflow integration and outcomes evidence. Competitive advantage is shaped less by list price and more by evidence depth, complication management performance, and the ability to train and support centres at scale. [2]
Companies with structural advantages tend to combine three assets: (1) long-running clinical datasets that satisfy payer and guideline scrutiny, (2) field support infrastructure that reduces downtime and improves adverse-event response, and (3) manufacturing and supply resilience that lowers the operational risk to implanting centres. Safety notices raise the value of strong field-action capability, since hospitals interpret response speed and transparency as part of the product’s risk profile. [4]
Buyer behaviour is increasingly pathway-driven. High-volume hospitals and transplant centres use formal value analysis committees, adverse-event registries, and protocol standardisation to reduce variability. This shifts leverage toward buyers on service terms and post-implant support requirements, while suppliers retain pricing power where switching costs are high due to training, clinical practice standardisation, and installed-base management.
Recent Developments:
- In May 2025, St Vincent’s Hospital Sydney (St Vincent’s clinicians) performed the world’s first-in-human implant of the CorWave Left Ventricular Assist Device (LVAD) (implanted on 28 May) as a bridge-to-transplant option [16].
- In July 2025, King Faisal Specialist Hospital & Research Centre (KFSHRC) performed the world’s first robotic-assisted BiVAD-HMIII implantation (two artificial pumps for biventricular support) for an advanced heart-failure patient [17].
- In April 2025, CoreMedTech (Core Medical) secured >$100 million Series D financing and stated its Corheart 6 LVAD had exceeded 700 cumulative implant surgeries [18].
Key Players in Heart Pump Device Market
- Abbott Laboratories
- Johnson & Johnson (Abiomed, Inc.)
- LivaNova PLC
- Berlin Heart GmbH
- SynCardia Systems, LLC
- CorWave SA
- Jarvik Heart, Inc.
- BiVACOR Inc.
- Medtronic plc
- Terumo Corporation
- Getinge AB
Scope of Report
| Metric | Value |
|---|---|
| Quantitative units | USD 4.8 billion (2025) to USD 23.2 billion (2036), at a CAGR of 15.4% |
| Market definition | The heart pump device market comprises mechanical circulatory support devices used to assist or replace cardiac pumping function in advanced heart failure and acute cardiogenic shock, including implantable systems such as LVADs and total artificial hearts, and extracorporeal systems such as ECMO and intra-aortic balloon pumps, primarily purchased by hospitals and specialist cardiac centres for bridge-to-transplant, bridge-to-recovery, and destination therapy pathways. |
| Type segmentation | Implanted Heart Pump Devices, Extracorporeal Heart Pump Devices |
| Product segmentation | Left Ventricular Assist Devices (LVAD), Total Artificial Hearts (TAHs), Intra-aortic Balloon Pumps (IABPs), Extracorporeal Membrane Oxygenation (ECMO) |
| End use segmentation | Hospitals, Cardiac Centers, Others |
| Regions covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Indonesia, Australia and 30 plus countries |
| Key companies profiled | Abbott Laboratories, Johnson & Johnson (Abiomed, Inc.), LivaNova PLC, Berlin Heart GmbH, SynCardia Systems, LLC, CorWave SA, Jarvik Heart, Inc., BiVACOR Inc. |
| Forecast period | 2026 to 2036 |
| Base year | 2025 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with cardiac surgeons, advanced heart failure cardiologists, hospital procurement leaders, and device OEM stakeholders, supported by regulator communications, payer coverage documentation, national transplant reporting, and triangulation against procedure volume and centre capability indicators. |
Heart Pump Device Market Analysis by Segments
Type:
- Implanted Heart Pump Devices
- Extracorporeal Heart Pump Devices
Product:
- Left Ventricular Assist Devices (LVAD)
- Total Artificial Hearts (TAHs)
- Intra-aortic Balloon Pumps (IABPs)
- Extracorporeal Membrane Oxygenation (ECMO)
End use:
- Hospitals
- Cardiac Centers
- Others
Region:
- North America (United States, Canada)
- Latin America
- Western Europe (United Kingdom, Germany, France, Italy, Spain)
- Eastern Europe
- East Asia (China, Japan, South Korea)
- South Asia & Pacific (India, Indonesia, Australia)
- Middle East & Africa
Bibliography
- [1] BiVACOR, Inc. (2024, July 25). The Texas Heart Institute implants BiVACOR total artificial heart.
- [2] Abbott. (2024, August). FDA approves aspirin-free labeling update for HeartMate 3.
- [3] American College of Cardiology. (2024, April). DanGer Shock: Routine mechanical circulatory support in AMI-related cardiogenic shock.
- [4] USA Food and Drug Administration. (2024, May). Abbott recalls HeartMate 3 left ventricular assist system (LVAS) implant kit for risk of blood leakage or air entering system.
- [5] Elsevier. (2025). Heart failure prevalence reflecting approximately 6.2 million adults in the United States (review summary). ScienceDirect.
- [6] Centers for Medicare & Medicaid Services. (2026). National coverage determination (NCD) for ventricular assist devices (20.9.1).
- [7] International Society for Heart and Lung Transplantation. (2024). 2024 consensus document on hemocompatibility-related events in durable ventricular assist devices (VADs).
- [8] NHS Blood and Transplant. (2025). Annual report on heart transplantation 2024/2025.
- [9] European Society of Cardiology. (2024). National trends and guideline positioning for LVAD therapy (conference supplement). European Heart Journal.
- [10] National Medical Products Administration. (2022, July). China approves domestically developed “artificial heart”.
- [11] Clinical experience and post-approval use of HeartCon-type LVAD in China. (2025). PubMed Central.
- [12] Destination therapy LVAD pathway and recorded DT-LVAD cases in Japan. (2025). PubMed Central.
- [13] National Organ & Tissue Transplant Organisation, Ministry of Health & Family Welfare. (2024). Organ donation and transplantation data (2013-2024).
- [14] Annual report of the Indian Society for Heart and Lung Transplantation (heart transplant counts referenced). (2024). Journal of Patient-Centered Cardiology.
- [15] Third official adult heart transplant report (Korean Organ Transplant Registry analysis). (2024). Korean Circulation Journal.
- [16] St Vincent’s Private Hospital. (n.d.). World first as St Vincent’s clinicians implant revolutionary new heart device.
- [17] King Faisal Specialist Hospital & Research Centre. (2025, July 16). KFSHRC performs world’s first robotic-assisted BiVAD implantation.
- [18] 36Kr. (2025, April 23). “Core Medical” secures over $100 million in Series D financing, completes over 700 artificial heart implant surgeries in total
This Report Addresses
- Market size and 10-year forecast for 2026 to 2036 with a 2025 baseline anchor
- Growth opportunity mapping by type, product, and end use to support portfolio prioritisation
- Country and regional forecasting tied to centre capability, transplant constraints, and coverage rules
- Reimbursement and policy impact assessment, with emphasis on destination therapy coverage rules
- Competitive structure review covering evidence depth, training capability, and field-action readiness
- Regulatory and safety-event tracking relevant to procurement and adoption risk management
- Strategic analysis of pathway shifts from bridge-to-transplant toward lifetime therapy cohorts
- Buyer behaviour analysis on value analysis committees, protocol standardisation, and switching costs
Frequently Asked Questions
How large is the heart pump device market in 2026?
In 2026, the heart pump device market is estimated at USD 5.5 billion, based on FMI’s forecast.
What will be the heart pump device market size by 2036?
FMI projects the market will reach USD 23.2 billion by 2036.
What is the expected CAGR for 2026 to 2036?
The market is projected to expand at a 15.4% CAGR from 2026 to 2036.
What was the market size in 2025?
In 2025, the market was valued at USD 4.8 billion.
What is the absolute dollar growth expected from 2026 to 2036?
Absolute growth is USD 17.7 billion, rising from USD 5.5 billion (2026) to USD 23.2 billion (2036).
Which type segment leads the market in the base year?
Implanted heart pump devices lead the type segment with an estimated 68.7% share in 2025
Which product segment leads the market in the base year?
Left Ventricular Assist Devices (LVAD) lead the product segment with an estimated 69.7% share in 2025.
What is the main buying centre for heart pump devices?
Hospitals remain the main buyers because implantation and post-operative management require specialised surgical and ICU infrastructure.
Why does the market expand faster than typical medtech categories?
The forecast reflects pathway broadening into destination therapy and shock protocols, which increases addressable procedures while raising average device value per patient episode.
What is the biggest adoption constraint in mature markets?
Mature markets face constraints from coverage rules, centre criteria, and complication management costs, which slow diffusion beyond specialised programmes. [6] [7]
How do safety notices affect procurement?
Regulator communications and recall classifications can trigger immediate inventory checks and supplier reassessment, influencing preference for OEMs with fast field-response systems. [4]
Do transplant constraints affect demand for LVADs?
Yes, transplant throughput and waiting-list pressure increase reliance on bridge and long-term mechanical support pathways, supporting durable LVAD utilisation. [8]
Is ECMO included in this market scope?
Yes, ECMO is included under the extracorporeal product segment as it is a core hospital-based mechanical circulatory support modality.
What is the forecasted market value used for planning in this report?
FMI uses USD 23.2 billion (2036) as the endpoint value for the 2026 to 2036 forecast.
What is the forecast starting point used for growth calculations?
The forecast starts at USD 5.5 billion in 2026, with 15.4% CAGR applied through 2036.
How sensitive is the forecast to reimbursement rules?
Reimbursement rules are a primary volume gate in major markets, especially destination therapy coverage, so rule changes can shift adoption curves materially. [6]
Which regions are expected to contribute the highest incremental volumes?
South Asia & Pacific and East Asia are expected to add significant incremental volume through tertiary-care expansion and improving transplant-linked ecosystems. [13]
What is the single most important number for commercial planning?
The most important planning number is the USD 23.2 billion forecast market value in 2036 at 15.4% CAGR.
How should OEMs win share in this market?
Share gains tend to come from protocol integration, complication reduction evidence, and centre enablement programmes that reduce hospital operational risk. [7]
Are the market numbers in this FAQ consistent with the forecast section?
Yes. All FAQ figures match the forecast anchor numbers: USD 4.8 billion (2025), USD 5.5 billion (2026), USD 23.2 billion (2036), CAGR 15.4%.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Implanted Heart Pump Devices
- Extracorporeal Heart Pump Devices
- Implanted Heart Pump Devices
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Left Ventricular Assist Devices (LVAD)
- Total Artificial Hearts (TAHs)
- Intra-aortic Balloon Pumps (IABPs)
- Extracorporeal Membrane Oxygenation (ECMO)
- Left Ventricular Assist Devices (LVAD)
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Product
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Johnson & Johnson
- LivaNova PLC
- Berlin Heart GmbH
- SynCardia Systems, LLC
- CorWave SA
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Product
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Product
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Product
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

