EPO Biomarkers Market
This study analyzes the EPO biomarkers market with a focus on market size estimation, revenue forecasting, competitive benchmarking, demand patterns, growth catalysts, market restraints, biomarker type trends, indication outlook, and long-term strategic opportunities.
EPO Biomarkers Market Size, Market Forecast and Outlook By FMI
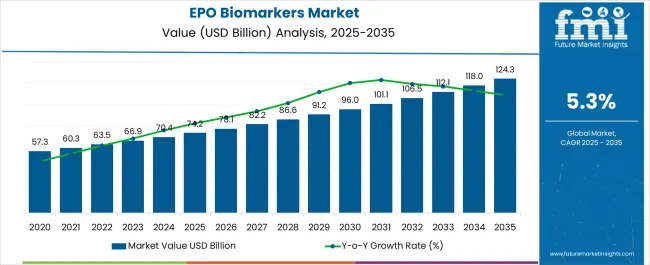
The epo biomarkers market was valued at USD 74.2 billion in 2025, projected to reach USD 78.13 billion in 2026, and is forecast to expand to USD 130.95 billion by 2036 at a 5.3% CAGR. chronic kidney disease burden requirements and evolving end-use specifications are accelerating adoption across global supply chains. What were once supplementary products have now become integral procurement priorities for buyers managing performance, compliance, and cost objectives simultaneously.
The absolute dollar increment from 2026 to 2036 reaches USD 52.82 billion, reflecting a steady accumulation anchored by chronic kidney disease prevalence, expanding oncology treatment volumes, and reimbursement-driven biosimilar adoption in cost-constrained public health systems. Feedstock biosafety requirements and pharmacovigilance compliance costs continue moderating margin expansion for new entrants, while originator companies leverage long-term hospital formulary agreements to sustain volume loyalty. Regulatory convergence between EMA biosimilar guidelines and WHO prequalification standards for erythropoietin is gradually reducing approval timelines in middle-income markets, widening the addressable base without proportionate cost inflation.
Summary of EPO Biomarkers Market
- Market Snapshot
- The epo biomarkers market is valued at USD 74.2 billion in 2025 and is projected to reach USD 130.95 billion by 2036.
- The industry is expected to grow at a 5.3% CAGR from 2026 to 2036, creating an incremental opportunity of USD 52.82 billion.
- The market operates as a specification-driven, compliance-sensitive category where performance differentiation and regulatory alignment define competitive positioning among leading suppliers.
- Demand and Growth Drivers
- Chronic Kidney Disease Burden is creating sustained procurement demand across end-use sectors requiring verified performance against established standards.
- Oncology Supportive Care Mandates is expanding the addressable volume base as infrastructure and industrial investment programs accelerate adoption in high-growth regions.
- Regulatory compliance requirements are reshaping formulation and sourcing decisions, driving buyers toward verified supplier relationships and qualified product grades.
- Among key countries, China leads at 7.2% CAGR, India leads at 6.6% CAGR, Germany leads at 6.1% CAGR, with mature markets including France at 5.6%, UK at 5%, USA at 4.5%, Brazil at 4%.
- Product and Segment View
- The market encompasses Recombinant human erythropoietin, Erythropoietin alfa, Erythropoietin beta and related product variants serving diverse application requirements.
- Recombinant human erythropoietin leads by Biomarker Type with 22.9% share in 2026, driven by established clinical and industrial adoption pathways.
- End stage renal disorder leads by Indication with 36.9% share, anchored by structural demand from the largest institutional procurement segment.
- Hospitals leads by End Users with 51.2% share, reflecting concentrated end-user procurement in primary institutional channels.
- The scope covers Recombinant human erythropoietin, Erythropoietin alfa, Erythropoietin beta, Erythropoietin zeta, Erythropoietin theta, Darbepoietin alfa across End stage renal disorder, Cancers, Rheumatoid arthritis, AIDS, Others, while excluding unrelated product categories and non-core applications.
- Geography and Competitive Outlook
- China and India are the fastest-growing markets, driven by expanding industrial investment and favourable regulatory frameworks for new product adoption.
- North America and Europe represent mature, high-value demand bases sustained by replacement cycle procurement and performance upgrade requirements.
- Competition is shaped by product qualification barriers, regulatory compliance track records, and supply chain integration capabilities.
- Key companies active in this market include 3Sbio Group, Amgen Inc., Biocon, Bio-Rad Laboratories Inc., Eve Technologies, F. Hoffmann-La Roche Ltd, Merck KGaA, Pfizer Inc., Siemens Healthineers AG, Kyowa Kirin Co. Ltd, GenScript, Bioagilytix Labs.
EPO Biomarkers Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 78.13 billion |
| Industry Value (2036) | USD 130.95 billion |
| CAGR (2026-2036) | 5.3% |
| Source | Future Market Insights, 2026 |
Source: FMI analysis based on primary research and proprietary forecasting model
Regional growth trajectories diverge across the forecast period. China advances at a 7.2% CAGR, India advances at a 6.6% CAGR, Germany advances at a 6.1% CAGR as expanding industrial and institutional procurement activity drives new deployment volumes. France tracks at 5.6%, UK tracks at 5%, USA tracks at 4.5%, Brazil tracks at 4%, reflecting mature market infrastructure and replacement-cycle demand patterns. The full country CAGR breakdown spans: China at 7.2%, India at 6.6%, Germany at 6.1%, France at 5.6%, UK at 5%, USA at 4.5%, Brazil at 4%.
EPO Biomarkers Market Definition
EPO biomarkers encompass a suite of diagnostic and therapeutic molecular indicators used to monitor erythropoietin activity, assess anemia management outcomes, and evaluate EPO receptor signalling pathways in clinical and research settings. These biomarkers span recombinant erythropoietin formulations, biosimilar variants, and darbepoetin analogs deployed across hematology, nephrology, and oncology care pathways. Laboratories and hospital systems use EPO biomarker panels to guide dosing decisions, detect therapeutic non-response, and satisfy pharmacovigilance requirements imposed by national drug regulatory authorities.
EPO Biomarkers Market Inclusions
The report scope covers global and regional market sizing for EPO biomarker products including recombinant erythropoietin formulations, biosimilar EPO variants, and darbepoetin alfa analogs. Forecast data spans 2026 to 2036 with segmentation by biomarker type, clinical indication, end-user category, and geography. Pricing trend analysis, reimbursement coverage assessments, and competitive landscape mapping are included within the analytical boundary.
EPO Biomarkers Market Exclusions
The scope excludes non-EPO hematopoietic growth factors, iron deficiency diagnostic panels not linked to erythropoietin management, and downstream consumables such as reagent kits used independently of EPO formulation monitoring. Contract manufacturing services for EPO active pharmaceutical ingredients and gene therapy platforms targeting erythropoietin pathways fall outside the analytical perimeter.
EPO Biomarkers Market Research Methodology
- Primary Research: FMI analysts conducted structured interviews with procurement leads, product managers, and technical specialists across the epo biomarkers value chain to capture first-hand market intelligence on pricing trends, product qualification cycles, and demand drivers not available through public data sources.
- Desk Research: Secondary data collection incorporated regulatory filings, government procurement records, trade association publications, peer-reviewed technical literature, and official company disclosures including annual reports, earnings call transcripts, and verified press releases from allowable institutional sources.
- Market-Sizing and Forecasting: Market values are derived through a hybrid bottom-up and top-down methodology combining supply-side production and shipment data with demand-side end-use consumption analysis. Regional forecasts apply segment-specific growth rate inputs validated against macroeconomic and industrial activity benchmarks.
- Data Validation and Update Cycle: All primary interview findings are cross-validated against secondary sources. Quantitative projections are stress-tested against scenario analysis incorporating regulatory, macroeconomic, and supply chain risk inputs. Model outputs are updated on a defined review cycle aligned with material market developments.
Why is the EPO Biomarkers Market Growing?
The EPO biomarkers market is experiencing steady growth, supported by the increasing role of biomarkers in enhancing diagnostic accuracy, patient monitoring, and treatment optimization for chronic conditions. Erythropoietin biomarkers are widely utilized to assess anemia and related disorders, particularly in patients undergoing renal replacement therapies. The growing prevalence of chronic kidney disease and end stage renal disorders is reinforcing the need for early detection and efficient management, thereby driving demand for EPO biomarkers.
Technological advancements in molecular diagnostics, coupled with rising investments in precision medicine, are expanding the application scope of these biomarkers across diverse clinical settings. Regulatory encouragement for biomarker-based testing in personalized treatment regimens is also accelerating market adoption.
In addition, the emphasis on reducing hospitalization costs through timely disease management is influencing healthcare providers to integrate biomarker testing into routine clinical practice As healthcare systems worldwide shift toward outcome-driven models, the market for EPO biomarkers is poised to benefit from increasing clinical validation, growing patient awareness, and wider adoption in hospital and research environments.
Segmental Analysis
The epo biomarkers market is segmented by biomarker type, indication, end users, and geographic regions. By biomarker type, epo biomarkers market is divided into Recombinant human erythropoietin, Erythropoietin alfa, Erythropoietin beta, Erythropoietin zeta, Erythropoietin theta, and Darbepoietin alfa. In terms of indication, epo biomarkers market is classified into End stage renal disorder, Cancers, Rheumatoid arthritis, AIDS, and Others. Based on end users, epo biomarkers market is segmented into Hospitals, Diagnostic centers, and Ambulatory care and surgical centers. Regionally, the epo biomarkers industry is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Recombinant Human Erythropoietin Biomarker Type Segment
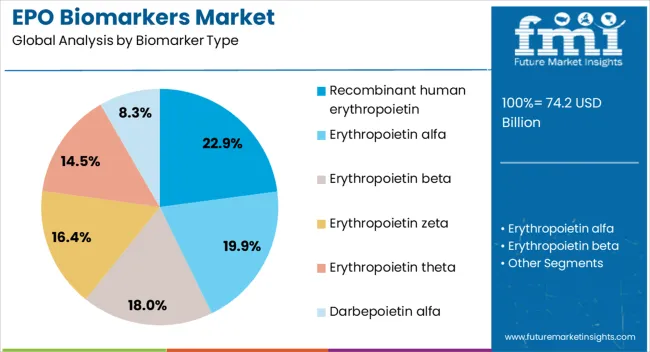
The recombinant human erythropoietin biomarker type segment is projected to hold 22.9% of the EPO biomarkers market revenue share in 2026, positioning it as the leading biomarker type. This dominance is being driven by its widespread use in detecting and monitoring anemia linked with chronic kidney disease and other hematological conditions. The biomarker enables accurate assessment of erythropoietin activity, supporting physicians in determining appropriate therapeutic strategies and dosage adjustments.
Its clinical relevance is further strengthened by its ability to provide reliable insights into the effectiveness of erythropoietin-stimulating agents used in patient care. The segment’s growth is supported by ongoing research and development initiatives that are expanding its utility across oncology and hematology.
Increasing awareness among clinicians about the role of recombinant biomarkers in guiding treatment decisions is further reinforcing adoption As personalized medicine becomes more integrated into routine healthcare delivery, the reliance on recombinant human erythropoietin biomarkers for precise disease evaluation is anticipated to strengthen their leading position in the global market.
Insights into the End Stage Renal Disorder Indication Segment
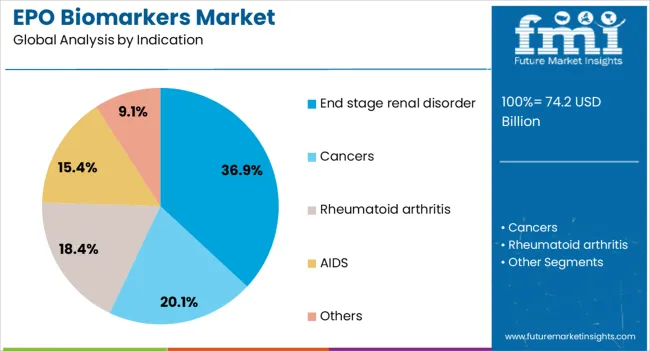
The end stage renal disorder indication segment is expected to account for 36.9% of the EPO biomarkers market revenue share in 2026, making it the leading indication. This dominance is being reinforced by the high global prevalence of advanced kidney diseases requiring dialysis and transplantation. EPO biomarkers are critical in assessing anemia severity in these patients, ensuring timely intervention and guiding therapeutic approaches.
The use of these biomarkers provides clinicians with valuable insights into the adequacy of treatment regimens, particularly in relation to erythropoietin therapy and iron supplementation. Rising incidence of diabetes and hypertension, which are major contributors to renal failure, is further expanding the patient pool for biomarker testing.
Healthcare systems are increasingly emphasizing early detection and management of anemia in renal patients to improve quality of life and reduce hospital readmissions As nephrology practices integrate biomarker-based approaches into standard protocols, the end stage renal disorder segment is expected to maintain its leadership, supported by consistent clinical demand and expanding global dialysis populations.
Insights into the Hospitals End User Segment
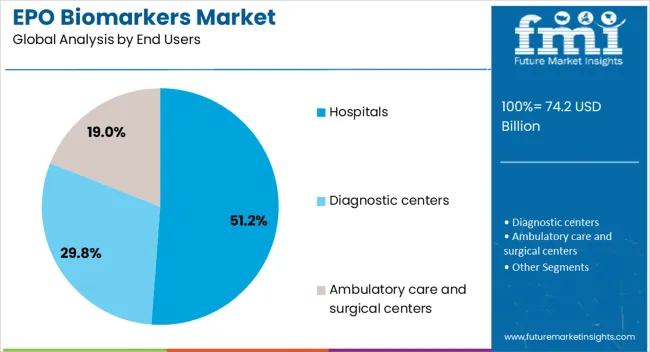
The hospitals end user segment is projected to capture 51.2% of the EPO biomarkers market revenue share in 2026, positioning it as the largest end-use setting. Its leadership is being supported by the concentration of advanced diagnostic facilities and the availability of trained professionals capable of performing biomarker-based tests.
Hospitals are the primary point of care for patients with anemia, renal disorders, and oncology-related complications, which increases their reliance on biomarker testing for accurate diagnosis and treatment planning. The segment’s growth is also being fueled by the integration of biomarker-based testing into comprehensive patient management pathways, supported by electronic health records and clinical decision support systems.
Strong investment in diagnostic infrastructure, coupled with regulatory frameworks that prioritize biomarker validation for clinical use, is further reinforcing adoption in hospitals With the rising burden of chronic diseases requiring long-term management, hospitals are expected to continue as the dominant end-user segment, driven by their ability to provide timely, large-scale, and high-quality diagnostic services to diverse patient populations.
Market Overview
Accuracy to hold the key for Deeper Market Penetration
As EPO biomarkers can be used to identify and diagnose several types of cancers, thus, their use has grown over the past few years. Comparing EPO biomarkers to other types of biomarkers like CA125, CA15-3, PSA, and others, it is known that they are more sensitive, specific, and accurate.
Therefore, it is anticipated that during the forecast period, the demand for more accurate and reliable diagnostics and therapeutics is likely to increase along with the number of oncology patients.
The high potential for revenue in the EPO biomarkers market draws potential investors. Increased awareness of the advantages of early diagnosis, an increase in the frequency of chronic diseases, a boom in the biotechnology and pharmaceutical industries, and technological advancements in biomarker detection leads to the higher adoption of EPO biomarkers.
Due to the increasing launch and adoption of Erythropoietin Alfa in cancer patients who suffer from anemia, the EPO biomarkers market is anticipated to experience significant growth over the course of the forecast period.
For instance, in November 2026, Pfizer introduced Retacrit, an Epoetin Alfa biosimilar, at a price that was 33% lower than that of the brand-name medications in the United States to treat anemia brought on by chemotherapy and chronic kidney disease.
EPO Biomarkers Market: Drivers & Restraints
The prime drivers for the market include growing incidence of cancers and renal diseases. Changes in lifestyle and improper eating habits contribute to pancreatic and endocrine cancers, which often lead to severe anaemia, where external erythropoietin infusion is required. Other drivers for the EPO biomarker market is technological advancement in devising proprietary testing methods apart from tried and tested ones.
Restraints for the market include lack of availability of skilled labour and sufficient quality controlled testing laboratories in most countries in less developed regions. Insufficient R&D in identification of biomarkers due to adoption of cost cutting measures in European regions is expected to restrain proper quantitative assessment of drugs and their therapeutic efficacy in blood.
This could also hamper progress of ongoing clinical trials and proper stratification of patients.
EPO Biomarkers Market: Overview
EPO biomarker market is account to rise in value during the forecast period. Due to expansion in the product segment of EPO biomarkers the market share will be rising with a promising rate, also with the increase in demand for diagnosis and treatment of widely spread chronic disease the EPO market is expected to grow with a promising CAGR during the forecast period.
Biologic drug industry is investing on R&D and the key players are focusing on increasing their production capacity for EPO biomarkers.
EPO Biomarkers Market: Region-wise Outlook
Considering all potential geographic regions, EPO biomarkers market is segmented into seven key regions: North America, Latin America, Eastern Europe, Western Europe, Japan, Asia Pacific and Middle East & Africa.
North America is been dominating in the EPO biomarkers due to rise in acceptance for bio-similar weakly followed by Western and Eastern Europe, Asia Pacific has a dominating presence in EPO biomarker market and most of the key players are locally producing the EPO biomarkers, that is enhancing their market share and product reach. There is a good opportunity for Japan in EPO biomarker market.
The emerging regions in Asia Pacific, Middle East and Africa holds promising future for rise in EPO biomarker market, due to increase in awareness towards the chronic disease and increase in population simultaneously.
The report covers exhaustive analysis on
- Market Segments
- Market Dynamics
- Market Size
- Supply & Demand
- Current Trends/Issues/Challenges
- Competition & Companies involved
- Technology
- Value Chain
Regional analysis includes
- North America (USA, Canada)
- Latin America (Mexico, Brazil)
- Western Europe (Germany, Italy, UK, Spain, France, Rest of Western Europe)
- Eastern Europe (Russia)
- Asia Pacific (China, India, ASEAN, Australia & New Zealand)
- Japan
- Middle East and Africa (GCC, S. Africa)
The report is a compilation of first-hand information, qualitative and quantitative assessment by industry analysts, inputs from industry experts and industry participants across the value chain. The report provides in-depth analysis of parent market trends, macro-economic indicators and governing factors along with market attractiveness as per segments. The report also maps the qualitative impact of various market factors on market segments and geographies.
Analysis of EPO Biomarkers Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| China | 7.2% |
| India | 6.6% |
| Germany | 6.1% |
| France | 5.6% |
| UK | 5.0% |
| USA | 4.5% |
| Brazil | 4.0% |
Source: FMI analysis based on primary research and proprietary forecasting model
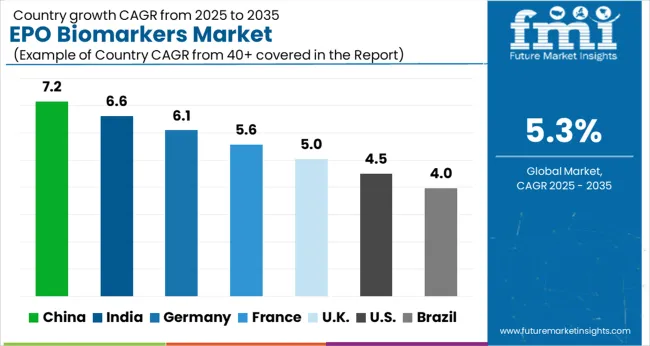
Country-wise Analysis
The EPO Biomarkers Market is expected to register a CAGR of 5.3% during the forecast period, exhibiting varied country level momentum. China leads with the highest CAGR of 7.2%, followed by India at 6.6%. Developed markets such as Germany, France, and the UK continue to expand steadily, while the USA is likely to grow at consistent rates. Brazil posts the lowest CAGR at 4.0%, yet still underscores a broadly positive trajectory for the global EPO Biomarkers Market. In 2026, Germany held a dominant revenue in the Western Europe market and is expected to grow with a CAGR of 6.1%. The USA EPO Biomarkers Market is estimated to be valued at USD 28.0 billion in 2026 and is anticipated to reach a valuation of USD 43.6 billion by 2036. Sales are projected to rise at a CAGR of 4.5% over the forecast period between 2026 and 2036. While Japan and South Korea markets are estimated to be valued at USD 3.8 billion and USD 2.3 billion respectively in 2026.
Key Players in the EPO Biomarkers Market
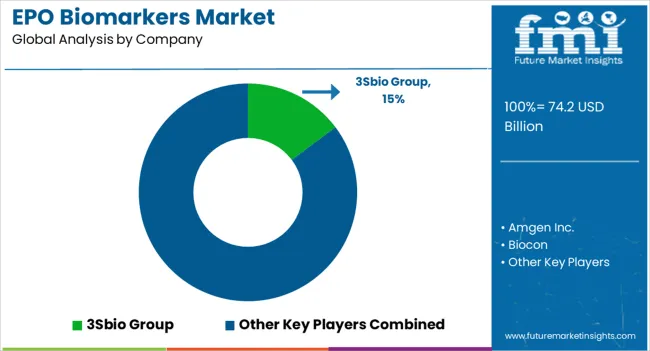
- 3Sbio Group
- Amgen Inc.
- Biocon
- Bio-Rad Laboratories Inc.
- Eve Technologies
- F. Hoffmann-La Roche Ltd
- Merck KGaA
- Pfizer Inc.
- Siemens Healthineers AG
- Kyowa Kirin Co. Ltd
- GenScript
- Bioagilytix Labs
Scope of the Report
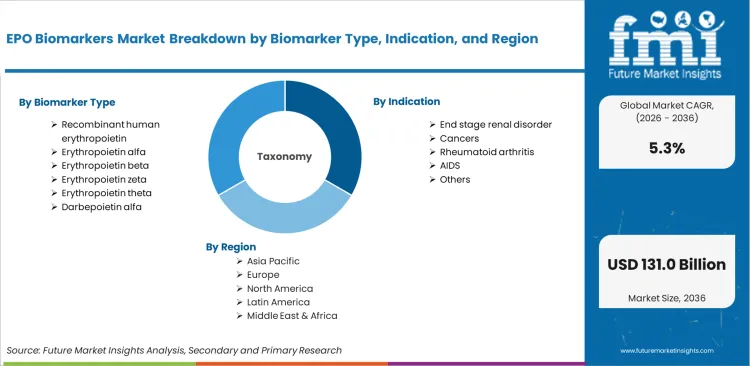
| Metric | Value |
|---|---|
| Quantitative Units | USD 78.13 billion to USD 130.95 billion, at a CAGR of 5.3% |
| Market Definition | EPO biomarkers encompass a suite of diagnostic and therapeutic molecular indicators used to monitor erythropoietin activity, assess anemia management outcomes, and evaluate EPO receptor signalling pat... |
| Biomarker Type Segmentation | Recombinant human erythropoietin, Erythropoietin alfa, Erythropoietin beta, Erythropoietin zeta, Erythropoietin theta, Darbepoietin alfa |
| Indication Segmentation | End stage renal disorder, Cancers, Rheumatoid arthritis, AIDS, Others |
| End Users Segmentation | Hospitals, Diagnostic centers, Ambulatory care and surgical centers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | 3Sbio Group, Amgen Inc., Biocon, Bio-Rad Laboratories Inc., Eve Technologies, F. Hoffmann-La Roche Ltd, Merck KGaA, Pfizer Inc., Siemens Healthineers AG, Kyowa Kirin Co. Ltd, GenScript, Bioagilytix Labs |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down forecasting model validated against primary research and public institutional data sources |
EPO Biomarkers Market by Segments
Biomarker Type:
- Recombinant human erythropoietin
- Erythropoietin alfa
- Erythropoietin beta
- Erythropoietin zeta
- Erythropoietin theta
- Darbepoietin alfa
Indication:
- End stage renal disorder
- Cancers
- Rheumatoid arthritis
- AIDS
- Others
End Users:
- Hospitals
- Diagnostic centers
- Ambulatory care and surgical centers
Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. World Health Organization. (2025). Model List of Essential Medicines, 23rd edition. WHO Press.
- 2. Organisation for Economic Co-operation and Development. (2025). Health at a Glance 2025: OECD Indicators. OECD Publishing.
- 3. European Medicines Agency. (2025). Guidelines on similar biological medicinal products. EMA/CHMP/BWP/247713/2012.
- 4. United States Food and Drug Administration. (2025). Guidance for Industry: Scientific Considerations in Demonstrating Biosimilarity to a Reference Product. FDA.
- 5. World Bank Group. (2025). World Development Indicators: Health, Nutrition and Population. World Bank.
- 6. International Energy Agency. (2025). World Energy Outlook 2025. IEA Publications.
- 7. United Nations Industrial Development Organization. (2025). Industrial Development Report 2025. UNIDO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for EPO Biomarkers in the global market in 2026?
Demand for epo biomarkers in the global market is estimated to be valued at USD 78.13 billion in 2026.
What will the market size of EPO Biomarkers be by 2036?
The epo biomarkers market is projected to reach USD 130.95 billion by 2036, reflecting sustained demand growth across primary end-use segments throughout the forecast period.
What is the expected CAGR for the EPO Biomarkers market between 2026 and 2036?
The epo biomarkers market is expected to grow at a CAGR of 5.3% between 2026 and 2036, generating an incremental opportunity of USD 52.82 billion over the forecast period.
Which Biomarker Type segment is poised to lead global sales in 2026?
Recombinant human erythropoietin accounts for 22.9% of biomarker type segment share in 2026, supported by established adoption patterns and strong institutional procurement demand.
Which Indication segment leads the EPO Biomarkers market?
End stage renal disorder holds 36.9% share by indication in 2026, reflecting its position as the primary end-use driver with the most concentrated procurement volumes.
Which country records the fastest growth in the EPO Biomarkers market?
China records the highest CAGR at 7.2% through 2036, driven by chronic kidney disease burden and expanding industrial procurement activity.
What does FMI forecast for the EPO Biomarkers market?
As per FMI analysis, the epo biomarkers market is forecast to grow from USD 78.13 billion in 2026 to USD 130.95 billion by 2036, driven by chronic kidney disease burden and oncology supportive care mandates across key regional markets.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Biomarker Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Biomarker Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Biomarker Type , 2026 to 2036
- Recombinant human erythropoietin
- Erythropoietin alfa
- Erythropoietin beta
- Erythropoietin zeta
- Erythropoietin theta
- Darbepoietin alfa
- Recombinant human erythropoietin
- Y to o to Y Growth Trend Analysis By Biomarker Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Biomarker Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- End stage renal disorder
- Cancers
- Rheumatoid arthritis
- AIDS
- Others
- End stage renal disorder
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users, 2026 to 2036
- Hospitals
- Diagnostic centers
- Ambulatory care and surgical centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End Users, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Biomarker Type
- By Indication
- By End Users
- By Country
- Market Attractiveness Analysis
- By Country
- By Biomarker Type
- By Indication
- By End Users
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Biomarker Type
- By Indication
- By End Users
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Biomarker Type
- By Indication
- By End Users
- Competition Analysis
- Competition Deep Dive
- 3Sbio Group
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Amgen Inc.
- Biocon
- Bio-Rad Laboratories Inc.
- Eve Technologies
- F. Hoffmann-La Roche Ltd
- Merck KGaA
- Pfizer Inc.
- Siemens Healthineers AG
- Kyowa Kirin Co. Ltd
- GenScript
- Bioagilytix Labs
- 3Sbio Group
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Biomarker Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Biomarker Type
- Figure 6: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Indication
- Figure 9: Global Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Users
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Biomarker Type
- Figure 26: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Indication
- Figure 29: North America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Users
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Biomarker Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Indication
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Biomarker Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Indication
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Biomarker Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Biomarker Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Indication
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Biomarker Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Biomarker Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Biomarker Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Biomarker Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

