The antibiotic zone reader market is valued at USD 260.0 million in 2026 and is projected to reach USD 610 million by 2036 at a CAGR of 8.9%. Value behavior reflects increasing procedural dependence on standardized antimicrobial susceptibility measurement within regulated laboratory environments. Zone readers convert diffusion assay outcomes into quantifiable digital records that reduce operator variability across shifts and sites. Clinical laboratories rely on these systems where treatment decisions depend on narrow interpretive thresholds defined by testing guidelines. Capital allocation favors platforms that deliver measurement consistency traceability and integration with laboratory information systems. Spending intensity aligns with testing volume audit exposure and need for defensible results rather than discretionary automation.
Adoption patterns are constrained by validation workload cost sensitivity and plate level variability inherent in diffusion testing. Readers must demonstrate equivalence with manual measurement under accreditation requirements which lengthens deployment timelines. Performance depends on agar depth disc placement and colony morphology requiring operator oversight despite automation. Budget limitations in low throughput laboratories restrict replacement of manual methods. Procurement prioritizes compatibility with existing media formats software reporting standards and maintenance support. Growth remains tied to laboratory capacity expansion regulatory scrutiny and infection surveillance intensity rather than research driven experimentation globally.
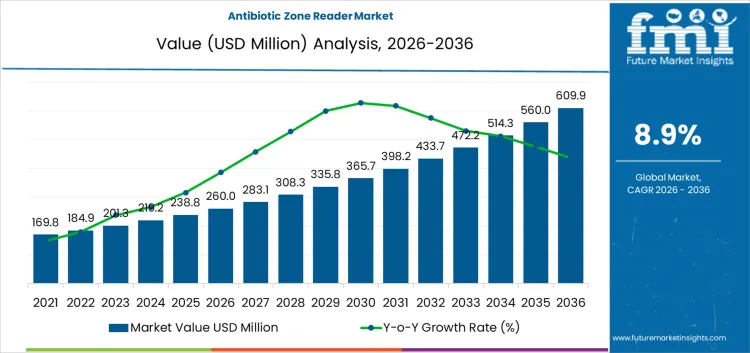
| Metric | Value |
|---|---|
| Market Value (2026) | USD 260.0 million |
| Market Forecast Value (2036) | USD 610.0 million |
| Forecast CAGR 2026 to 2036 | 8.9% |
Demand for antibiotic zone readers is increasing as clinical microbiology laboratories and healthcare diagnostics units require precise measurement of inhibition zones to determine antimicrobial susceptibility profiles. Manual measurement methods are labor intensive and subject to interoperator variation, and antibiotic zone readers provide digital imaging and standardized analysis that support consistent interpretation of assay results. Microbiology professionals specify these systems to accelerate turnaround time for susceptibility reports that guide therapeutic decision making in infection management. Procurement teams evaluate resolution, software analytics, workflow integration, and compliance with laboratory quality systems to ensure devices align with throughput expectations and regulatory documentation requirements. Adoption is reinforced by emphasis on documented results that support audit trails and traceability for sterility testing in pharmaceutical quality control and environmental monitoring programs.
Healthcare facilities are reinforcing infection control practices and antimicrobial stewardship initiatives that depend on reliable susceptibility data to limit spread of resistant organisms. Research laboratories engaged in new antibiotic development require accurate zone measurement to validate compound activity across diverse bacterial strains. Advances in digital imaging sensors and pattern recognition software improve differentiation of overlapping zones and support integration with laboratory information management systems. Technical staff benefit from customizable protocols, instrument calibration routines, and vendor support that reduce cycle times and support operational consistency. These application driven needs are contributing to sustained uptake of antibiotic zone readers across clinical and research environments.
Demand for antibiotic zone readers is shaped by antimicrobial susceptibility testing requirements, diagnostic accuracy expectations, and compliance with laboratory standards. Clinical and quality control laboratories require consistent measurement of inhibition zones to guide treatment decisions and verify antibiotic potency. Adoption aligns with rising antimicrobial resistance monitoring and standardized testing protocols. System selection prioritizes measurement precision, documentation reliability, and workflow efficiency. Segment classification reflects differentiation by testing methodology, system automation level, and laboratory end use. Structure highlights how testing approach, reader capability, and operational setting influence specification priorities and utilization intensity across microbiology laboratories.
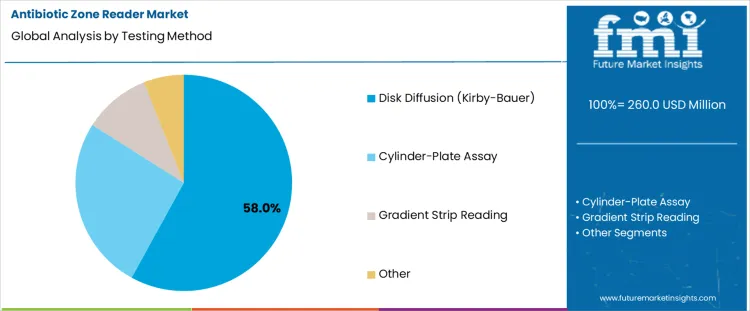
Disk diffusion testing holds 58.0%, representing the largest share among testing methods due to widespread standardization and clinical acceptance. The Kirby-Bauer method is routinely applied in hospital and reference laboratories for antimicrobial susceptibility assessment. Clear interpretive guidelines support consistent result comparison across facilities. Simplicity of setup and low consumable complexity sustain broad adoption. Cylinder-plate assays support antibiotic potency evaluation within quality control environments. Gradient strip reading enables minimum inhibitory concentration estimation with lower testing volume. Testing method segmentation reflects dominance of disk diffusion where regulatory alignment, procedural familiarity, and reproducibility remain essential.
Key Points
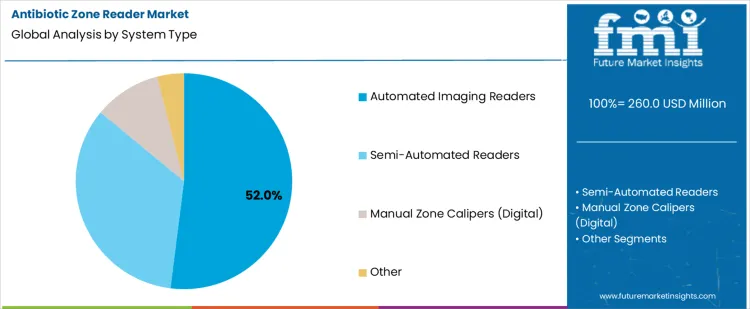
Automated imaging readers hold 52.0%, representing the largest share among system types due to measurement consistency and documentation capability. Automated systems capture high-resolution images and calculate inhibition zones with reduced operator variability. Digital archiving supports audit readiness and traceability requirements. Integration with laboratory information systems improves reporting efficiency. Semi-automated readers balance cost and accuracy through assisted measurement workflows. Manual digital calipers support low-throughput settings with greater operator dependence. System type segmentation reflects preference for automation where accuracy, repeatability, and data integrity are operational priorities.
Key Points
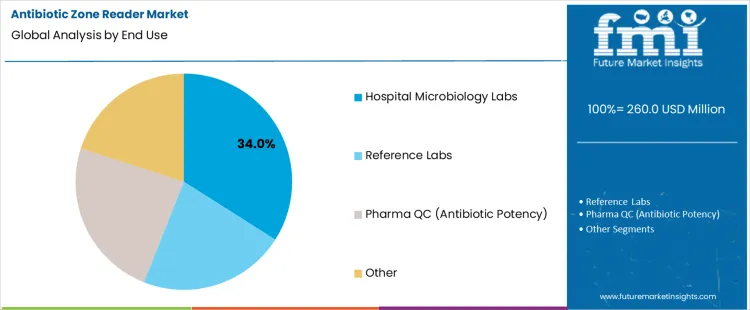
Hospital microbiology labs hold 34.0%, representing the largest share among end-use settings due to routine diagnostic testing volume. These laboratories perform daily susceptibility assessments to support clinical treatment decisions. Rapid and reliable zone measurement directly influences antimicrobial selection accuracy. Integration with diagnostic workflows supports timely reporting. Reference laboratories manage confirmatory testing with lower aggregate volume. Pharmaceutical quality control applies zone readers for antibiotic potency validation. Other users address surveillance and research needs. End-use segmentation reflects concentration of demand where diagnostic frequency and clinical decision support requirements remain highest.
Key Points
Demand for antibiotic zone readers reflects laboratory requirement for objective measurement of inhibition zones in antimicrobial susceptibility testing. Adoption concentrates within clinical microbiology laboratories, hospital diagnostics, pharmaceutical quality control, and public health surveillance units. Global scope aligns with standardized susceptibility testing protocols and rising antimicrobial resistance monitoring. Usage centers on optical and image-based readers integrated with disk diffusion workflows to ensure measurement consistency and auditability.
Clinical laboratories require precise zone diameter measurement to classify bacterial susceptibility according to established interpretive criteria. Demand increases as manual ruler-based readings introduce operator variability that affects categorical results. Automated zone readers provide consistent edge detection and calibrated measurement, supporting reproducible outcomes across technicians and shifts. Integration with laboratory information systems improves result traceability and reporting speed. Public health programs rely on standardized readings to track resistance trends across sites. Pharmaceutical laboratories adopt automated readers to support batch release testing and method validation. High sample volumes benefit from faster plate throughput without compromising accuracy. Adoption reflects operational dependence on consistency in susceptibility interpretation rather than workflow automation alone.
Zone readers require validation against reference methods to demonstrate measurement equivalence, extending implementation timelines. Demand sensitivity rises where laboratory budgets favor manual methods for low test volumes. Performance depends on agar depth, disk placement accuracy, and colony growth characteristics, affecting edge detection reliability. Irregular inhibition zones challenge algorithm accuracy and require operator review. Training remains necessary to manage exceptions and verify results. Equipment cost and maintenance affect procurement in decentralized laboratories. Software updates and calibration introduce ongoing support requirements. Regulatory documentation expectations increase administrative effort, constraining rapid scaling across fragmented laboratory networks.
Demand for antibiotic zone readers is expanding globally due to rising antimicrobial resistance surveillance, standardized susceptibility testing, and laboratory automation requirements. Clinical microbiology, pharmaceutical quality control, and academic research drive adoption where repeatable measurement and auditability are required. Transition from manual calipers to digital imaging improves throughput and reduces operator variability. Growth rates in India at 10.8%, Indonesia at 9.7%, Vietnam at 9.3%, Saudi Arabia at 8.9%, and UAE at 8.5% indicate sustained expansion supported by laboratory capacity growth, regulatory scrutiny, and infection control programs across healthcare and public health systems.
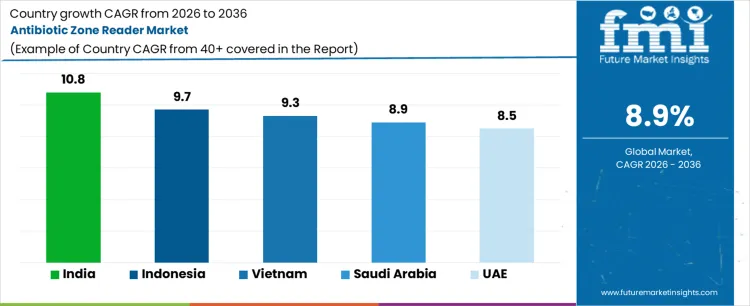
| Country | CAGR (%) |
|---|---|
| India | 10.8% |
| Indonesia | 9.7% |
| Vietnam | 9.3% |
| Saudi Arabia | 8.9% |
| UAE | 8.5% |
India is growing at a CAGR of 10.8%, supported by expanding clinical microbiology networks and antimicrobial resistance monitoring. High patient volumes increase routine susceptibility testing across hospitals and diagnostic chains. National surveillance initiatives emphasize standardized reporting and traceable results. Pharmaceutical manufacturing and contract research laboratories require validated zone measurement for quality control. Academic institutions expand microbiology programs, adding instructional laboratories. Demand growth reflects testing volume escalation and compliance-driven accuracy requirements rather than discretionary laboratory upgrades.
Antibiotic zone reader demand in Indonesia is expanding at a CAGR of 9.7%, driven by infection control priorities and laboratory modernization. Public hospitals increase susceptibility testing to guide antibiotic stewardship. Tropical disease prevalence elevates microbiological workload. Automation reduces analyst training burden and improves result consistency across dispersed facilities. Government investment supports upgrading diagnostic equipment. Demand growth reflects public health needs and laboratory standardization rather than expansion of pharmaceutical research activity.
Vietnam is growing at a CAGR of 9.3%, shaped by healthcare system expansion and export-oriented pharmaceutical testing. Hospitals adopt standardized susceptibility testing protocols to improve treatment outcomes. Pharmaceutical packaging and formulation facilities require validated microbiological quality checks. Automation supports growing laboratory networks with limited specialist availability. University laboratories increase applied microbiology research and training. Demand growth reflects healthcare expansion and compliance alignment rather than replacement of legacy automated systems.
Antibiotic zone reader demand in Saudi Arabia is expanding at a CAGR of 8.9%, supported by hospital capacity expansion and infection surveillance initiatives. Centralized laboratories implement digital measurement to standardize results across regions. High healthcare investment supports procurement of automated microbiology equipment. Pharmaceutical and food safety laboratories contribute additional demand. Emphasis on data integrity and reporting accuracy influences adoption. Growth reflects institutional investment and standardization rather than increases in routine diagnostic volumes alone.
United Arab Emirates is growing at a CAGR of 8.5%, driven by advanced healthcare facilities and regulatory emphasis on quality diagnostics. Hospitals prioritize rapid, standardized susceptibility testing to support antimicrobial stewardship. Medical laboratories serving international patients require audit-ready documentation. Academic and private research centers adopt digital readers for training and applied studies. Strong focus on healthcare quality standards sustains procurement. Growth reflects institutional quality benchmarks and laboratory automation rather than population-driven testing increases.
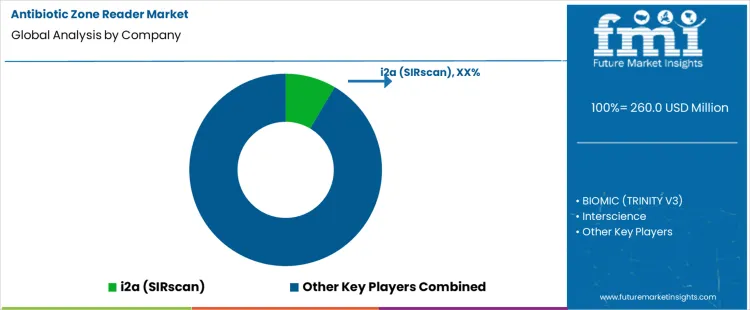
Demand for antibiotic zone readers is driven by clinical microbiology laboratories, hospitals, and diagnostic facilities requiring standardized antimicrobial susceptibility testing to guide appropriate therapy. These systems automate measurement of inhibition zones from disk diffusion assays, improving accuracy, reproducibility, and throughput compared with visual manual reading. Buyers evaluate imaging resolution, software analytics, integration with laboratory information systems, regulatory compliance, and ease of validation and quality control. Procurement teams prioritize suppliers with proven performance records, global service networks, training support, and compatibility with a wide range of culture media and plate formats. Trend in the global market reflects increased emphasis on antimicrobial resistance surveillance, workflow automation, and data traceability to support clinical decision-making and public health reporting.
i2a (SIRscan) holds leading positioning through specialized antibiotic zone readers and imaging software widely adopted by clinical and reference laboratories worldwide. bioMérieux supports demand with integrated susceptibility testing platforms and zone reading solutions aligned with global clinical microbiology workflows. BIOMIC (TRINITY V3) contributes automated zone readers and software analytics for routine and high-throughput testing environments. BD (Becton, Dickinson and Company) participates with imaging and digital analysis tools integrated into broader microbiology systems supporting culturing and susceptibility. Thermo Fisher Scientific supplies digital zone reading systems and laboratory automation technologies integrated with data management solutions. Competitive differentiation depends on imaging accuracy, software analytics sophistication, regulatory readiness, ease of integration, and ability to support diverse laboratory requirements.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Testing Method | Disk Diffusion (Kirby-Bauer); Cylinder-Plate Assay; Gradient Strip Reading; Other |
| System Type | Automated Imaging Readers; Semi-Automated Readers; Manual Zone Calipers (Digital); Other |
| End Use | Hospital Microbiology Labs; Reference Labs; Pharma QC (Antibiotic Potency); Other |
| Sales Channel | Direct OEM Sales; Clinical Lab Distributors; Microbiology Workflow Integrators; Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Indonesia, Vietnam, Saudi Arabia, UAE, and 40+ countries |
| Key Companies Profiled | i2a; BIOMIC; Interscience; bioMérieux; BD; Thermo Fisher Scientific; Bruker; Merlin Diagnostika; HiMedia Laboratories; Regional Suppliers |
| Additional Attributes | Dollar sales by testing method and system type; adoption trends for automated imaging readers to standardize antimicrobial susceptibility interpretation; zone measurement accuracy, repeatability, and edge-detection performance metrics; compatibility with EUCAST and CLSI guidelines and reporting formats; software capabilities for audit trails, LIS connectivity, and result archiving; throughput per shift and operator dependency considerations; maintenance, calibration, and lifecycle cost factors; compliance with laboratory accreditation and validation requirements influencing antibiotic zone reader selection. |
How big is the antibiotic zone reader market in 2026?
The global antibiotic zone reader market is estimated to be valued at USD 260.0 million in 2026.
What will be the size of antibiotic zone reader market in 2036?
The market size for the antibiotic zone reader market is projected to reach USD 609.9 million by 2036.
How much will be the antibiotic zone reader market growth between 2026 and 2036?
The antibiotic zone reader market is expected to grow at a 8.9% CAGR between 2026 and 2036.
What are the key product types in the antibiotic zone reader market?
The key product types in antibiotic zone reader market are disk diffusion (kirby-bauer), cylinder-plate assay, gradient strip reading and other.
Which system type segment to contribute significant share in the antibiotic zone reader market in 2026?
In terms of system type, automated imaging readers segment to command 52.0% share in the antibiotic zone reader market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.