Antibiotics API Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
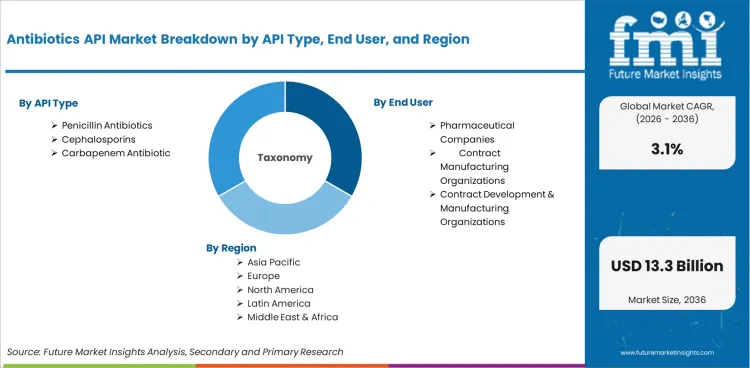
The Antibiotics API Market is segmented by API Type (Penicillin Antibiotics, Cephalosporins, and Carbapenem Antibiotics), End User (Pharmaceutical Companies, Contract Manufacturing Organizations, and Contract Development & Manufacturing Organizations), Manufacturing Route (Fermentation, Semi‑Synthetic, and Synthetic), Supply Model (Merchant Supply, Captive Production, and Toll Manufacturing), Therapy Role (Essential Medicine APIs, Reserve Therapy APIs, and Specialty Therapy APIs), Production Scale (Large Volume, Mid Volume, and Low Volume APIs), and Region. The forecast period spans 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Antibiotics API Market Size, Market Forecast and Outlook By FMI
Summary of the Antibiotics API Market
- Demand and Growth Drivers
- Steady anti-infective drug production supports recurring purchase of bulk antibiotic ingredients.
- Essential medicine supply programs keep approved antibiotic APIs in routine formulation plans.
- Rising outsourcing across regulated pharma production adds fresh volume for specialist API suppliers.
- Product and Segment View
- Penicillin antibiotics are expected to lead API type since large routine volumes are focused on older access molecules.
- Pharmaceutical companies are projected to lead end user demand as they convert the largest share of antibiotic APIs into finished dosage forms.
- Fermentation based APIs are expected to lead manufacturing route because penicillin and part of cephalosporin output still rely on large biological processes.
- Geography and Competitive Outlook
- India is expected to outpace the global average as domestic manufacturing depth and export-linked formulation output support stronger API demand.
- Canada and Spain stay above the global rate as regulated sourcing and stable formulation activity support steady orders.
- Sandoz International GmbH and Aurobindo Pharma Limited hold strong positions. Centrient Pharmaceuticals and CordenPharma International add supply depth across regulated anti-infective lines.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Analyst for Healthcare at FMI, states, "Demand for antibiotics APIs is propelled by process consistency and supply assurance. Pharmaceutical companies place the largest bulk orders because finished drug production depends on reliable qualified supply. Suppliers that protect batch quality and regulatory files are likely to defend their position through the forecast period."
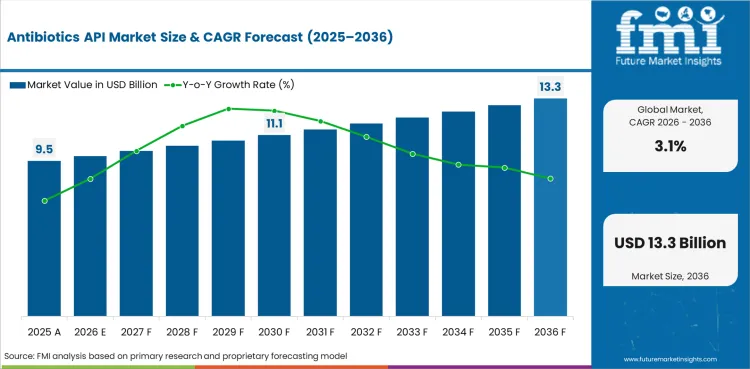
- Antibiotics API Market Value Analysis
- The antibiotics API sector is linked to essential medicine production and recurring formulation cycles.
- Bulk antibiotic inputs support hospital supply chains and retail prescription volumes across mature pharma systems.
- Fermentation scale and compliance records influence supplier selection across large antibiotic programs.
- Outsourcing activity adds another revenue stream for specialist API manufacturers that can support stable documentation.
- European antibiotic manufacturers need a more stable commercial setting to protect supply security across essential medicine lines.
Antibiotics API Market Definition
The antibiotics API industry covers bulk active pharmaceutical ingredients used in regulated anti-infective drug manufacturing. Scope includes penicillin antibiotics, cephalosporins, and carbapenem antibiotic APIs supplied to pharmaceutical companies CMOs and CDMOs.
Antibiotics API Market Inclusions
Industry scope includes global and regional market sizing from 2026 to 2036. The study covers API type, end user, manufacturing route, supply model, therapy role, production scale, and country-level CAGR analysis.
Antibiotics API Market Exclusions
Finished dosage forms, excipients, packaging materials, and delivery devices are outside scope. Non-antibiotic pharmaceutical ingredients and hospital administration services are excluded.
Antibiotics API Market Research Methodology
- Primary Research: FMI analysts reviewed inputs from pharmaceutical companies, CMOs, CDMOs, distributors, and supplier-side commercial teams active in antibiotic API trade.
- Desk Research: The study combined regulatory publications, company materials, anti-infective manufacturing references, and public sourcing documents.
- Market Sizing and Forecasting: Baseline values were built from the supplied market inputs. FMI then aligned them with molecule demand, end use mix, and country growth patterns.
- Data Validation and Update Cycle: Forecasts were checked against segment shares, company presence, and country growth patterns across the antibiotics API industry.
Why is the Antibiotics API Market Growing?
- Recurring anti-infective formulation impacts bulk antibiotic ingredient demand growth.
- Essential medicine supply programs support approved API volumes across mature pharma systems.
- Outsourced production adds fresh volume for suppliers with reliable fermentation capacity and documentation.
Large, finished dose programs account for most antibiotic API consumption. Pharmaceutical companies are projected to hold 75.9% of end user demand in 2026 as they plan volumes and hold the main regulatory files for commercial supply. Merchant suppliers benefit when approved anti-infective lines require dependable third-party sourcing.
Essential medicine production provides another layer of support. Essential medicine APIs are expected to represent 68.0% of therapy role demand in 2026 because older access antibiotics account for the largest prescription volumes in routine care. This pattern supports recurring orders across mature anti-infective supply programs.
Market Segmentation Analysis
- Within API type, penicillin antibiotics are expected to account for 50.5% of demand in 2026 because routine oral and injectable therapies generate the largest bulk volumes.
- Pharmaceutical companies hold a projected 75.9% share of end user demand in 2026 as finished dose manufacturing is focused on branded and generic drug makers.
- Fermentation based APIs are expected to account for 61.0% of manufacturing route demand in 2026 since core beta lactam production relies on large fermentation trains.
- Essential medicine APIs are anticipated to represent 68.0% of therapy role demand in 2026 as mature access molecules are important to recurring hospital and retail supply.
Antibiotics API Market Analysis by API Type
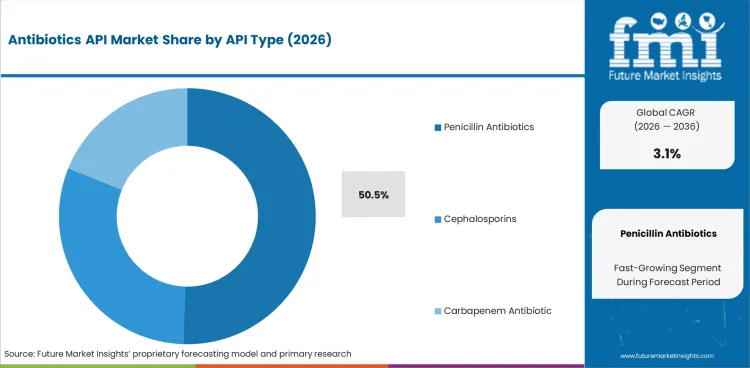
- Penicillin antibiotics are expected to account for 50.5% of API type demand in 2026 since amoxicillin and related molecules support the largest routine bulk volumes across anti-infective manufacturing.
- High prescription volume and established process familiarity pushes penicillin demand ahead of cephalosporins and carbapenem antibiotic lines.
- CDC and WHO guidance keeps access-group antibiotics central to routine use. That position supports penicillin-linked API demand.
Insights into the Carbapenem Antibiotic API Type Segment
- Carbapenem antibiotic APIs are expected to rise faster through 2036 because hospital use against resistant infections keeps this class in tighter clinical focus.
- Smaller base volumes leave room for faster percentage expansion. Total demand still remains below penicillin output.
- Process control and sterile formulation support are important for suppliers serving this higher value antibiotic class.
Antibiotics API Market Analysis by End User
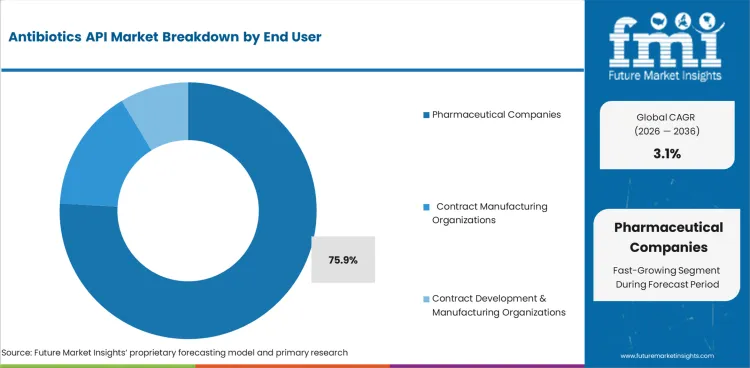
- Pharmaceutical companies are projected to contribute 75.9% of end user demand in 2026 as they convert most antibiotic APIs into finished tablets or capsules and injectables at commercial scale.
- Direct control over regulatory files, batch release, and finished dose planning keeps pharmaceutical manufacturers ahead of CMOs and CDMOs.
- Approved API partners support long supply programs and keep volume concentration high with large formulators.
Insights into the Contract Development & Manufacturing Organizations Segment
- CDMOs are expected to expand faster through 2036 because more pharmaceutical companies seek external development and scale-up support for selected antibiotic lines.
- Flexible project intake and formulation linked services support faster growth from a smaller base.
- External development demand aligns with broader contract manufacturing activity in small-molecule pharmaceuticals.
Antibiotics API Market Analysis by Manufacturing Route
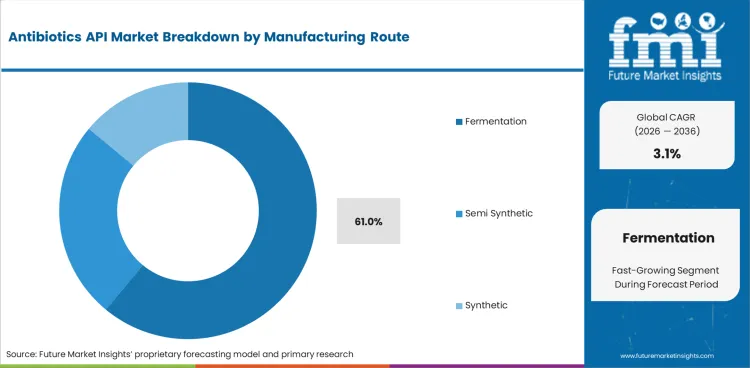
- Fermentation based APIs are expected to represent 61.0% of manufacturing route demand in 2026 as penicillin and part of cephalosporin production still rely on large biological fermentation assets.
- Large tank economics and long process experience keep fermentation at the center of cost-sensitive beta lactam output.
- Suppliers with steady yield control and clean batch records hold an advantage in high-volume antibiotic programs.
Insights into the Synthetic Manufacturing Route Segment
- Synthetic APIs are likely to advance faster through 2036 because selected reserve molecules and higher process control needs rely more on controlled chemical synthesis.
- This route supports tighter impurity control for specialized antibiotic lines that do not scale well through fermentation.
- Process development demand is expected to expand across specialized antibiotic APIs from this smaller base.
Antibiotics API Market Analysis by Supply Model
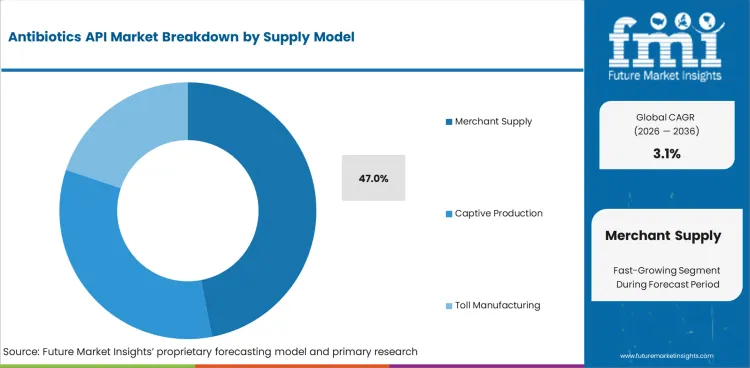
- Merchant supply is expected to account for 47.0% of supply model demand in 2026 because many finished dose manufacturers still buy antibiotic APIs from specialist producers rather than produce every molecule in house.
- Approved third-party sources help manufacturers protect cost control and continuity across large molecule portfolios.
- This pattern reflects the balance between captive production and outsourced API supply in small-molecule manufacturing.
Insights into the Toll Manufacturing Segment
- Toll manufacturing is expected to grow faster through 2036 as smaller formulators seek batch flexibility without carrying full site cost.
- Project-based production supports short campaigns and selected regional supply requirements.
- Contract demand stays stronger for molecules with uneven order patterns and tighter capital discipline.
Antibiotics API Market Analysis by Therapy Role
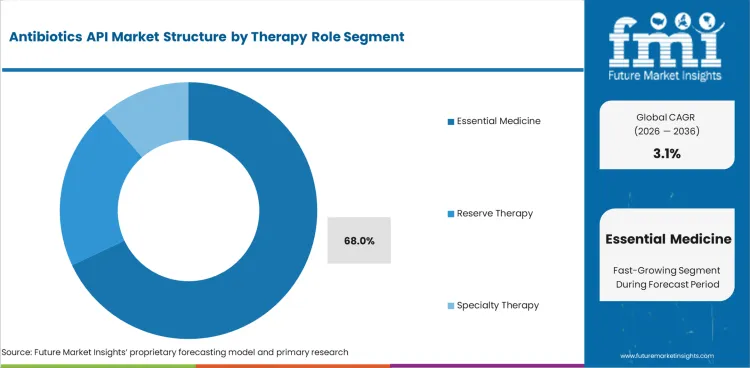
- Essential medicine APIs are expected to represent 68.0% of therapy role demand in 2026 due to high use access antibiotics accounting for the largest share of regular bacterial treatment programs.
- Routine hospital purchasing and retail prescription demand keep older essential molecules ahead of reserve and specialty lines. This pattern supports steady volume flow across adjacent antibiotic formulations, especially where proven molecules remain central to routine treatment.
Insights into the Reserve Therapy APIs Segment
- Reserve therapy APIs are likely to expand faster through 2036 because antimicrobial resistance keeps selected last line antibiotics in sharper clinical focus.
- Suppliers serving this space need tighter documentation and closer clinical supply planning.
Antibiotics API Market Analysis by Production Scale
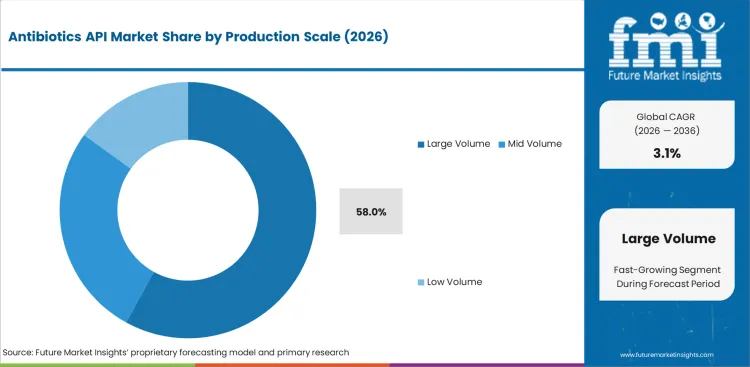
- Large volume APIs are projected to contribute 58.0% of production scale demand in 2026 as older penicillin and cephalosporin molecules go through long campaign manufacturing for routine global supply.
- Long cycle production lowers unit cost and supports large formulation demand across tablets, capsules, and injectables.
- Scale efficiency keeps these molecules tied to cost control and stable solvent use across pharmaceutical manufacturing.
Insights into the Mid-Volume APIs Segment
- Mid-volume APIs are expected to rise faster through 2036 because many newer or region-specific antibiotic lines need enough scale for steady supply without full commodity-style capacity.
- This band suits flexible scheduling and mixed product portfolios at specialist plants.
- Demand in this layer is shaped by selective outsourcing and smaller clinical supply programs.
Antibiotics API Market Drivers and Restraints and Opportunities
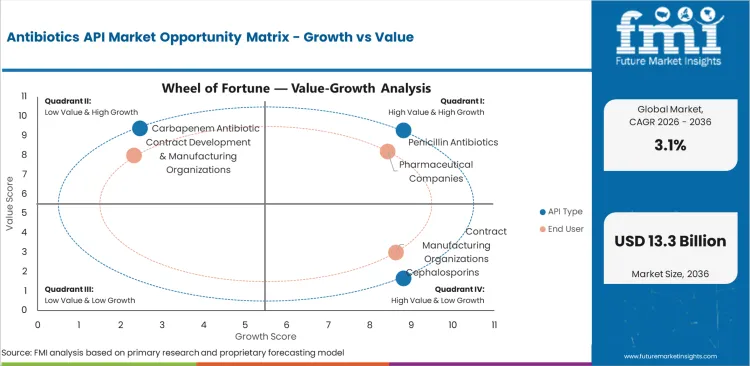
- Essential medicine supply pushes the baseline demand to remain steady.
- Environmental controls and price pressure limit margin growth.
- Outsourced manufacturing influences new order flow for approved API suppliers.
Demand in antibiotics APIs aligns with recurring anti-infective production. Penicillin antibiotics lead API type and pharmaceutical companies lead end user demand, which keeps the market grounded in large-volume mature supply programs. The category reflects demand from established drug manufacturing platforms rather than short-cycle buying.
Essential Medicine Supply
Routine hospital use and retail pharmacy demand keep antibiotic production active across common access molecules. Manufacturers need approved bulk ingredients on a steady basis because many older antibiotics still move through large prescription volumes each year. Supply continuity matters because any break in API availability can delay formulation output and disrupt medicine access.
Compliance Cost Pressure
Antibiotic API plants face steady cost pressure from waste treatment controls and documentation work linked to regulated manufacturing. Environmental handling requires added validation work, which raises plant operating expense and slows release procedures. This burden is stronger for older molecules because price competition remains firm even as compliance tasks add cost.
Outsourcing Expansion
CMOs and CDMOs add another demand stream for antibiotic APIs as drug manufacturers move selected production batches to external partners. This shift supports API orders from third-party production networks that need qualified ingredient supply for contracted formulation work. Outsourced manufacturing is rising in selected programs because companies want more flexibility in batch planning and plant utilization.
Analysis of Antibiotics API Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 5.7% |
| Canada | 3.8% |
| Brazil | 3.8% |
| Spain | 3.6% |
| United States | 2.7% |
| Germany | 1.2% |
| China | 0.9% |
Source: Future Market Insights analysis based on the provided study inputs for 2026.
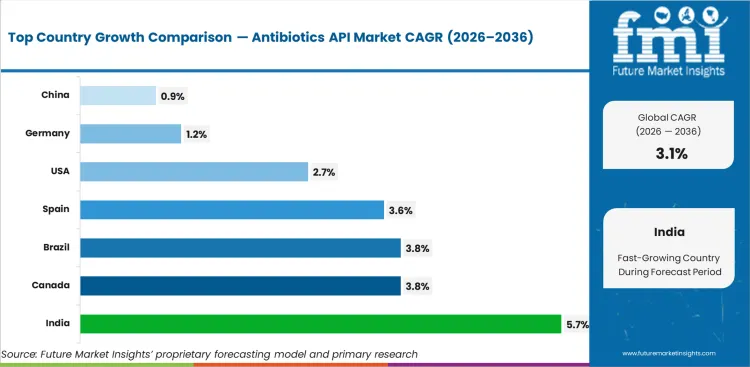
Antibiotics API Market CAGR Analysis by Country
- India is anticipated to lead country‑level growth at a 5.7% CAGR through 2036. This is expected due to the country’s strong generic antibiotic manufacturing base, export‑linked formulation output, and cost‑competitive large‑scale API production capabilities.
- Canada is projected to record a 3.8% CAGR, supported by steady formulation output and continued reliance on qualified, regulated antibiotic API sourcing.
- Brazil is expected to expand at a 3.8% CAGR, reflecting stable domestic antibiotic formulation demand and gradual strengthening of regulated sourcing practices.
The global antibiotics API market is projected to grow at a 3.1% CAGR from 2026 to 2036, supported by recurring essential medicine production and stable formulation demand across mature pharmaceutical systems. The study covers more than 40 countries, with the primary growth markets summarized below
Demand Outlook for Antibiotics API Market in the United States
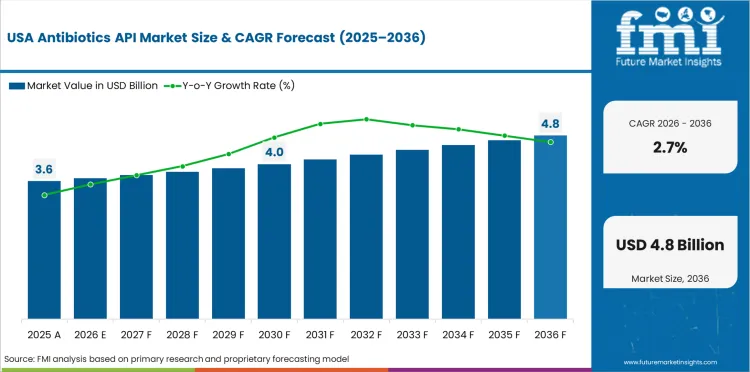
Demand for antibiotics APIs in the United States is expected to grow at a 2.7% CAGR through 2036 because essential antibiotic production supports regulated drug manufacturing. Generic drug manufacturers rely on steady bulk ingredient supply for common anti-infective formulations sold through hospital and retail channels. Mature manufacturing patterns keep growth measured as the country prioritizes supply continuity over major capacity expansion.
- Essential antibiotic production supports steady API demand.
- Generic drug output sustains steady bulk ingredient sourcing.
- Mature manufacturing patterns keep growth measured.
Sales Analysis of Antibiotics API Market in China
Sales in China are projected to record a 0.9% CAGR through 2036 because pricing pressure and environmental control costs limit value expansion in antibiotic API manufacturing. The country maintains a large production base and steady plant utilization across established molecules. Domestic formulation demand and export activity continue to support output, even though the value outlook remains slow.
- Large production base leads to a steady supply output.
- Environmental control costs influence low value expansion.
- Mature capacity limits faster growth.
Opportunity Analysis of Antibiotics API Market in India
Demand in India is expected to expand at a 5.7% CAGR through 2036 because the country has a strong antibiotic production base and rising need for regulated formulation supply. Drug manufacturers in India serve both domestic demand and export-linked output across common anti-infective molecules. Manufacturing scale and cost competitiveness help the country attract a larger share of global antibiotic API activity.
- Strong production base propels a faster expansion.
- Export-linked output lifts recurring API demand.
- Cost competitiveness supports national growth leadership.
In-Depth Analysis of Antibiotics API Market in Germany
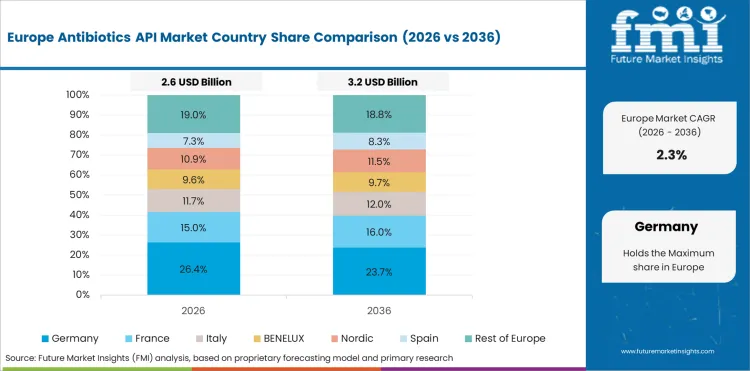
Germany is likely to post a 1.2% CAGR through 2036 because mature antibiotic production and strict cost control keep expansion modest. Essential medicine supply remains important, but the country is not adding capacity at the pace seen in lower-cost manufacturing hubs. High compliance needs keep producers focused on continuity and quality control.
- Mature production keeps expansion modest.
- Compliance leads to a strict quality control.
- Limited fresh capacity holds growth at a low pace.
In-Depth Analysis of Antibiotics API Market in Japan
Japan is forecast to grow at a 1.5% CAGR through 2036 because stable antibiotic demand supports recurring API need across established pharmaceutical production. Domestic manufacturers continue to source approved ingredients for essential anti-infective lines used in routine care. Growth remains moderate because the market is mature and output gains are expected to stay limited.
- Stable antibiotic demand is led by recurring API need.
- Approved ingredient sourcing sustains in routine production.
- Mature market conditions keep growth moderate.
Antibiotics API Market Analysis in France
Demand in France is expected to register a 3.1% CAGR through 2036 because recurring formulation demand supports regular API sourcing across antibiotic manufacturing programs. Domestic production and supply security concerns sustain interest in dependable anti-infective ingredient availability. Standard molecule demand remains steady across hospital and retail medicine channels.
- Recurring formulation demand influences regular sourcing.
- Supply security is led by approved API use.
- Standard molecule demand keeps growth steady.
Antibiotics API Market Analysis in Canada
Canada is projected to record a 3.8% CAGR through 2036 because steady formulation output supports demand for qualified antibiotic API supply. Canadian pharmaceutical manufacturing continues to rely on dependable bulk ingredient access for routine anti-infective drug production. Growth remains above the global average because the country maintains a sound balance between manufacturing demand and supply continuity.
- Steady formulation output supports API demand.
- Qualified supply access stays important in manufacturing.
- Balanced production needs sustain growth above the global average.
Competitive Position and Strategic Direction
- Competition centers on manufacturing reliability, documentation strength, and continuity of supply across approved antibiotic programs.
- No single supplier holds every molecule line and competition stays sharper in older high-volume antibiotics.
- Entry barriers remain moderate because fermentation assets, waste control systems, and regulatory files require time and capital.
Sandoz International GmbH, Aurobindo Pharma Limited, and Centrient Pharmaceuticals hold strong positions in large anti-infective supply programs. Their standing is supported by established manufacturing records and ongoing work with regulated formulation companies.
Fresenius Kabi, CordenPharma International, ACS DOBFAR SPA, The United Laboratories International Holdings Limited, and DAEWOONG PHARMACEUTICAL CO., LTD are some regional and product-specific players in the sector.
Key Companies in the Antibiotics API Market
The antibiotics API industry includes established antibiotic ingredient manufacturers and formulation-linked suppliers. Regional anti-infective producers remain active as well.
- Global Leaders: Sandoz International GmbH, Aurobindo Pharma Limited, and Centrient Pharmaceuticals hold strong positions through scale and long product experience.
- Regional Specialists: Fresenius Kabi, CordenPharma International, and ACS DOBFAR SPA maintain strong positions through regulated supply programs and European manufacturing depth.
- Focused Asian Suppliers: The United Laboratories International Holdings Limited and DAEWOONG PHARMACEUTICAL CO., LTD. compete through selected anti-infective lines and regional manufacturing strength.
Competitive Benchmarking. Antibiotics API Market
| Company | Product Breadth | Manufacturing Depth | Regulatory Support | Footprint |
|---|---|---|---|---|
| Sandoz International GmbH | High | Strong | Strong | Global |
| Aurobindo Pharma Limited | High | Strong | Strong | Global |
| Centrient Pharmaceuticals | Moderate | Strong | Strong | Europe |
| Fresenius Kabi | Moderate | Moderate | Strong | Global |
| CordenPharma International | Moderate | Moderate | Strong | Europe |
| ACS DOBFAR SPA | Focused | Moderate | Strong | Europe |
| Regional Suppliers | Low | Low | Moderate | Local |
Source. Future Market Insights analysis based on the provided study inputs for 2026.
Key Developments in the Antibiotics API Market
- In December 2025, Delius Pharmaceuticals secured CDSCO approval to import ceftriaxone sodium API from Guangxi Kelun Pharmaceutical. The approval supports supply security planning.
- In December 2025, Biocon Limited signed an out-licensing agreement with Ajanta Pharma Ltd. to market liraglutide in 26 countries. The deal reflects continued activity in complex API-linked supply programs.
Key Players in the Antibiotics API Market
Major Companies
- Sandoz International GmbH
- Fresenius Kabi
- Aurobindo Pharma Limited
- DAEWOONG PHARMACEUTICAL CO., LTD.
- The United Laboratories International Holdings Limited
- Centrient Pharmaceuticals
- CordenPharma International
- ACS DOBFAR SPA
Regional Participants
- Other regional antibiotic API suppliers
Report Scope and Coverage
| Item | Value |
|---|---|
| Quantitative Units | USD 9.5 billion in 2026 to USD 12.9 billion by 2036 at a 3.1% CAGR |
| Market Definition | Bulk active pharmaceutical ingredients used in regulated anti-infective drug manufacturing. |
| API Type | Penicillin Antibiotics Cephalosporins Carbapenem Antibiotic |
| End User | Pharmaceutical Companies Contract Manufacturing Organizations Contract Development & Manufacturing Organizations |
| Manufacturing Route | Fermentation Semi Synthetic Synthetic |
| Supply Model | Merchant Supply Captive Production Toll Manufacturing |
| Therapy Role | Essential Medicine APIs Reserve Therapy APIs Specialty Therapy APIs |
| Production Scale | Large Volume APIs Mid Volume APIs Low Volume APIs |
| Regions Covered | North America Latin America East Asia South Asia and Pacific Western Europe Eastern Europe Middle East and Africa |
| Countries Covered | United States Canada Brazil Germany United Kingdom France Italy Spain India China Japan and 40 plus countries |
| Key Companies Profiled | Sandoz International GmbH Fresenius Kabi Aurobindo Pharma Limited DAEWOONG PHARMACEUTICAL CO., LTD. The United Laboratories International Holdings Limited Centrient Pharmaceuticals CordenPharma International ACS DOBFAR SPA |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modelling validated through primary interviews and demand triangulation across antibiotic production volumes and API mix |
Source. Future Market Insights analysis based on the provided study inputs for 2026.
Antibiotics API Market by Segments
API Type
- Penicillin Antibiotics
- Cephalosporins
- Carbapenem Antibiotic
End User
- Pharmaceutical Companies
- Contract Manufacturing Organizations
- Contract Development & Manufacturing Organizations
Manufacturing Route
- Fermentation
- Semi Synthetic
- Synthetic
Supply Model
- Merchant Supply
- Captive Production
- Toll Manufacturing
Therapy Role
- Essential Medicine APIs
- Reserve Therapy APIs
- Specialty Therapy APIs
Production Scale
- Large Volume APIs
- Mid Volume APIs
- Low Volume APIs
Region
- North America
- Latin America
- East Asia
- South Asia and Pacific
- Western Europe
- Eastern Europe
- Middle East and Africa
Research Sources and Bibliography
- Centrient Pharmaceuticals (2025). Centrient Pharmaceuticals supports European Critical Medicines Alliance recommendations in Strategic Report.
- Centers for Disease Control and Prevention (2026). Antibiotic Use and Stewardship in the United States 2025 Update Progress and Opportunities
- AMR Industry Alliance (2024). Equitable and Responsible Access Roadmap.
- Delius Pharma (2025). Delius Pharma gets regulatory approval to import Ceftriaxone API from China
- Biocon Limited (2025). Biocon Limited signs out licensing agreement with Ajanta to market liraglutide in 26 countries.
The bibliography is provided for reader reference.
This Report Answers
- What size is the antibiotics API market in 2026 and how large can it become by 2036?
- How fast is antibiotics API demand expected to rise between 2026 and 2036?
- Which API type is expected to lead 2026 demand in the antibiotics API market?
- Which end user group accounts for the largest antibiotics API demand in 2026?
- Why do pharmaceutical companies retain the largest share of antibiotics API purchases?
- How do fermentation assets and compliance records influence antibiotics API supplier choice?
- Which countries are expected to post the fastest growth through 2036 in antibiotics API demand?
- What role does outsourced production play in antibiotics API volume planning?
- Which companies stay most active in regulated antibiotics API supply?
- How is the antibiotics API market defined and what does the forecast include?
Frequently Asked Questions
How large is the antibiotics API market in 2026?
The antibiotics API market is estimated at USD 9.5 billion in 2026 based on FMI analysis of bulk anti-infective ingredient demand.
What will the antibiotics API market size be by 2036?
The antibiotics API market is projected to reach USD 12.9 billion by 2036 as steady formulation demand keeps approved supplier intake active.
What is the expected CAGR for the antibiotics API market?
The antibiotics API market is forecast to advance at a 3.1% CAGR from 2026 to 2036 under steady essential medicine supply conditions.
Which API type leads the antibiotics API market in 2026?
Penicillin antibiotics lead the antibiotics API market with a projected 50.5% share in 2026 because routine prescription volumes still center on this class.
Which end user leads antibiotics API demand in 2026?
Pharmaceutical companies lead antibiotics API demand with an expected 75.9% share in 2026 due to their large commercial formulation volumes.
Why does India record the fastest rise in the antibiotics API market?
India is estimated to record the steady rise at an anticipated 5.7% CAGR by 2036 due to large generic antibiotic manufacturing and export linked formulation keeping bulk API intake strong.
What is included in the antibiotics API market definition?
The antibiotics API market includes bulk active ingredients used in regulated anti-infective drug manufacturing across pharmaceutical companies CMOs and CDMOs.
How was the antibiotics API market forecast prepared?
FMI built the forecast from supplied market inputs and aligned them with molecule demand end use mix segment shares and country growth patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By API Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By API Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By API Type , 2026 to 2036
- Penicillin Antibiotics
- Cephalosporins
- Carbapenem Antibiotic
- Penicillin Antibiotics
- Y to o to Y Growth Trend Analysis By API Type , 2021 to 2025
- Absolute $ Opportunity Analysis By API Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharmaceutical Companies
- Contract Manufacturing Organizations
- Contract Development & Manufacturing Organizations
- Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Manufacturing Route
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Manufacturing Route, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Manufacturing Route, 2026 to 2036
- Fermentation
- Semi Synthetic
- Synthetic
- Fermentation
- Y to o to Y Growth Trend Analysis By Manufacturing Route, 2021 to 2025
- Absolute $ Opportunity Analysis By Manufacturing Route, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Supply Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Supply Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Supply Model, 2026 to 2036
- Merchant Supply
- Captive Production
- Toll Manufacturing
- Merchant Supply
- Y to o to Y Growth Trend Analysis By Supply Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Supply Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Therapy Role
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Therapy Role, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Therapy Role, 2026 to 2036
- Essential Medicine
- Reserve Therapy
- Specialty Therapy
- Essential Medicine
- Y to o to Y Growth Trend Analysis By Therapy Role, 2021 to 2025
- Absolute $ Opportunity Analysis By Therapy Role, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Production Scale
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Production Scale, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Production Scale, 2026 to 2036
- Large Volume
- Mid Volume
- Low Volume
- Large Volume
- Y to o to Y Growth Trend Analysis By Production Scale, 2021 to 2025
- Absolute $ Opportunity Analysis By Production Scale, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- By Country
- Market Attractiveness Analysis
- By Country
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By API Type
- By End User
- By Manufacturing Route
- By Supply Model
- By Therapy Role
- By Production Scale
- Competition Analysis
- Competition Deep Dive
- Sandoz International GmbH
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Fresenius Kabi
- Aurobindo Pharma Limited
- DAEWOONG PHARMACEUTICAL CO., LTD.
- The Value (USD Million)ed Laboratories International Holdings Limited
- Centrient Pharmaceuticals
- CordenPharma International
- Sandoz International GmbH
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by API Type , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Manufacturing Route, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by Supply Model, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Therapy Role, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Production Scale, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by API Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Manufacturing Route
- Figure 12: Global Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Supply Model
- Figure 15: Global Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Therapy Role
- Figure 18: Global Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Production Scale
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by API Type
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Manufacturing Route
- Figure 41: North America Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Supply Model
- Figure 44: North America Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Therapy Role
- Figure 47: North America Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Production Scale
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by API Type
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Manufacturing Route
- Figure 60: Latin America Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Supply Model
- Figure 63: Latin America Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Therapy Role
- Figure 66: Latin America Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Production Scale
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by API Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Western Europe Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Manufacturing Route
- Figure 79: Western Europe Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Supply Model
- Figure 82: Western Europe Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Therapy Role
- Figure 85: Western Europe Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Production Scale
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by API Type
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by End User
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Manufacturing Route
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by Supply Model
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Therapy Role
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Production Scale
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by API Type
- Figure 111: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by End User
- Figure 114: East Asia Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Manufacturing Route
- Figure 117: East Asia Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by Supply Model
- Figure 120: East Asia Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Therapy Role
- Figure 123: East Asia Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Production Scale
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by API Type
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Manufacturing Route
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by Supply Model
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Therapy Role
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Production Scale
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by API Type , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by API Type , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by API Type
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Manufacturing Route, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Manufacturing Route, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Manufacturing Route
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by Supply Model, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by Supply Model, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by Supply Model
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Therapy Role, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Therapy Role, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Therapy Role
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Production Scale, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Production Scale, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Production Scale
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

