Antimicrobials Market
Antimicrobials Market Analysis Size, Share, and Forecast Outlook 2026 to 2036
Antimicrobials Market Forecast and Outlook 2026 to 2036
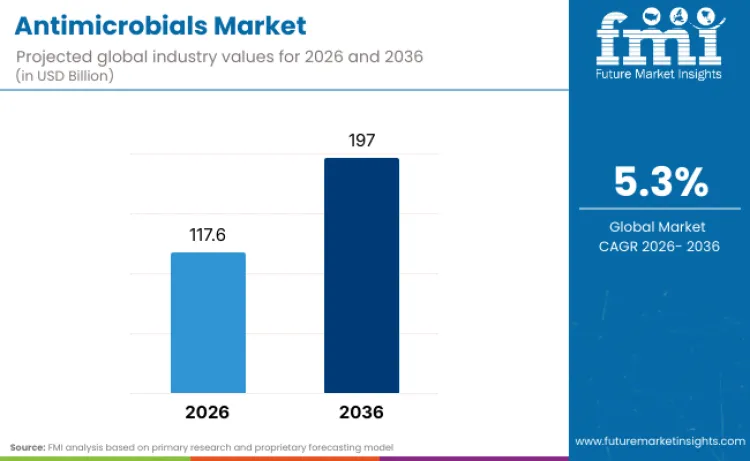
The global antimicrobials market is projected to reach USD 117.6 billion in 2026 and expand to USD 197.0 billion by 2036, registering a 5.3% CAGR over the forecast period. As per FMI opinion, this trajectory reflects a structural shift in infectious disease management away from broad-spectrum legacy antibiotics toward targeted, resistance-aware therapies designed around pathogen profiles and antimicrobial stewardship realities. Product development is increasingly focused on narrower-spectrum agents, combination strategies, and advanced delivery platforms, including sustained-release and targeted carriers, to improve tissue exposure while reducing resistance-selection pressure in routine care pathways, in line with CDC stewardship frameworks.
“GSK’s momentum continues with another quarter of strong performance, supporting upgraded guidance for 2025, and positioning us well for 2026 and achieving our longer-term growth outlooks. Together we have delivered a step-change in operating performance, new prospects for growth and a clear pathway for scale, patient impact and sustained shareholder value.”-Emma Walmsley, CEO, GSK
This executive positioning is reinforced by GSK’s infectious-disease execution, where Blujepa (gepotidacin) achieved USA FDA approval in 2025 for uncomplicated urinary tract infections and later for uncomplicated urogenital gonorrhoea, signalling that first-in-class antibiotics are re-entering standard-of-care pathways under mounting resistance pressure. This aligns with the WHO’s 2024 bacterial priority pathogens list, which explicitly identifies resistant threats as urgent public-health risks. As a result, the antimicrobials market is increasingly defined by precision therapies that can demonstrate resistance-aware efficacy while fitting real-world stewardship constraints, with delivery technologies emerging as critical enablers of sustained exposure and controlled dosing.
Quick Stats for Antimicrobials Market
- Antimicrobials Industry Value (2026): USD 117.6 Billion
- Antimicrobials Industry Forecast Value (2036): USD 197.0 Billion
- Antimicrobials Industry Forecast CAGR: 5.3%
- Antimicrobials Industry Leading Type: Antibacterial Agents (45.3%)
- Antimicrobials Industry Key Growth Regions: United States, China, Germany, Japan, United Kingdom
- Antimicrobials Industry Key Players: Pfizer Inc, Merck & Co Inc, Johnson & Johnson, Novartis AG, Bayer AG
Antimicrobials Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 117.6 billion |
| Market Forecast Value (2036) | USD 197.0 billion |
| Forecast CAGR (2026-2036) | 5.3% |
Source: Future Market Insights-analysis driven by proprietary forecasting models and primary research
Key Shift in the Antimicrobials Market During 2026 to 2036
Prior to 2026, antimicrobial development prioritised broad-spectrum pathogen elimination over resistance control. This approach generated predictable limitations, including accelerated resistance emergence, microbiome disruption, and declining real-world efficacy against evolved pathogens. Clinical confidence weakened as legacy antibiotics failed to deliver durable outcomes, and few development programmes were aligned with antimicrobial stewardship requirements or resistance-informed prescribing logic.
By 2026, the segment transitions into what FMI defines as the “Resistance-Aware Development Phase.” Progress in pathogen genomics, molecular diagnostics, and combination-therapy engineering enables next-generation antimicrobials to outperform traditional agents across key clinical measures, including pathogen specificity, resistance suppression, shortened treatment courses, and improved safety and tolerability. Development focus shifts from empirical coverage to targeted efficacy supported by diagnostic confirmation.
What are the Various Segments of the Antimicrobials Market?
The antimicrobials market is segmented by type into antibacterial, antifungal, antiviral, and others; by origin into synthetic, natural, semi-synthetic, and others; by route of administration into oral, topical, parenteral, and others; by distribution channel into hospital pharmacy, retail pharmacy, online pharmacy, and others; and by region into Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, rest of Europe), North America (USA, Canada, Mexico), Latin America (Brazil, Chile, rest of Latin America), and Middle East & Africa (Kingdom of Saudi Arabia, other GCC countries, Turkey, South Africa, other African Union, rest of Middle East & Africa).
Which Type Holds the Largest Share in Antimicrobials?
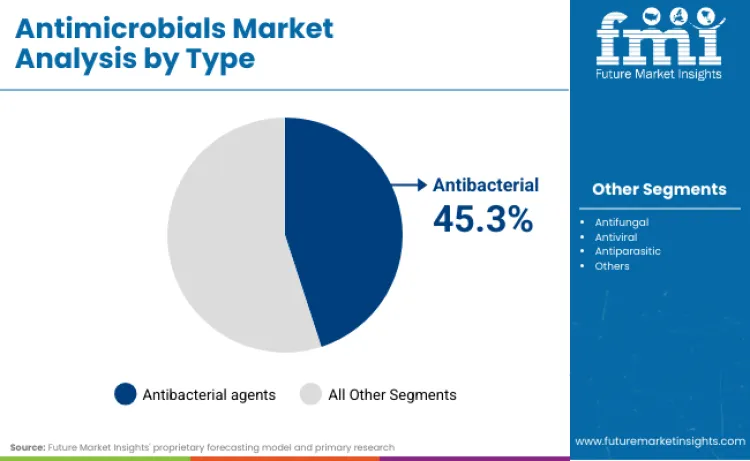
Antibacterial agents hold the largest share of the antimicrobials market at 45.3% because they are the first-line workhorses for high-risk infections managed across hospitals and the community, where speed, reliability, and guideline-backed use matter most. This dominance is reinforced by how major players describe their priorities and scientific focus. Pfizer highlights its hospital footprint directly: "Between these business units, your Company collectively addresses 15 therapy areas with a portfolio of over 150 products that include therapeutics and vaccines, including Hospitals." GSK signals momentum in infectious diseases development: "GSK's late-stage R&D pipeline achieved 13 positive phase III clinical trial readouts in 2024 across Respiratory, Immunology & Inflammation, Oncology, HIV and Infectious Diseases-a record for the company."
Merck & Co., Inc. reinforces broad prevention-and-treatment ambition: "As a leading biopharmaceutical company, we are at the forefront of scientific research, working tirelessly to provide innovative health solutions to advance the prevention and treatment of diseases in both humans and animals." Astellas Pharma frames high-value innovation: "Our Philosophy and VISION. On the forefront of healthcare change to turn innovative science into VALUE for patients." AstraZeneca underscores the burden rationale: "Our therapy areas: Vaccines & Immune Therapies, ~8 million deaths are associated with serious bacterial infections."
How Do Origin Categories Influence Antimicrobial Development and Selection?
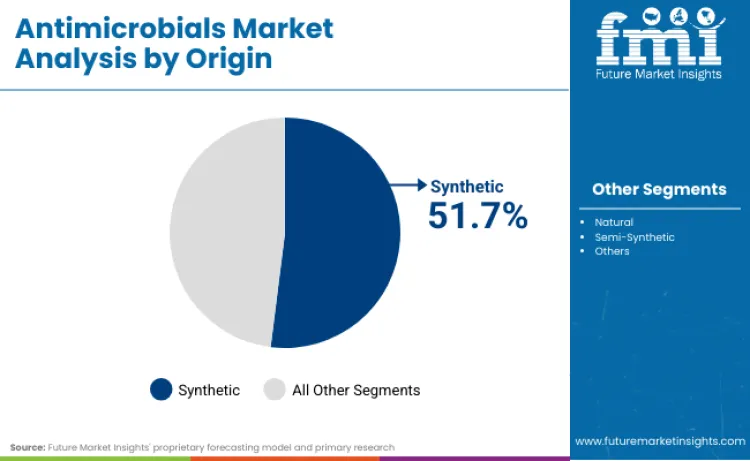
Synthetic antimicrobials represent 51.7% of the global antimicrobials market because developers prefer controllable, reproducible, and scalable pathways that reduce variability and support predictable performance in real-world use. Synthetic origin allows precise molecular engineering, tighter pharmacokinetic control, and resistance-risk optimisation, which fits stewardship-aligned deployment and hospital procurement requirements. As antimicrobial R&D shifts toward targeted mechanisms and combination strategies, synthetic platforms also offer clearer regulatory pathways and stronger intellectual property protection than purely natural alternatives. This preference is reinforced by how leading companies frame their innovation and responsibility agendas.
AbbVie states, "AbbVie's mission is to discover and deliver innovative medicines that solve serious health issues and address the medical challenges." Johnson & Johnson emphasizes science-led infectious disease ambition: "Leveraging strong foundations in science and technology to develop medicines and vaccines that can prevent, treat and, ultimately, cure some of the world’s deadliest infectious diseases." Teva Pharmaceutical Industries links scale to AMR accountability: "We're doing our part to address AMR by manufacturing our medicines responsibly and holding suppliers accountable."
How Do Administration Routes Affect Antimicrobial Prescribing Patterns?
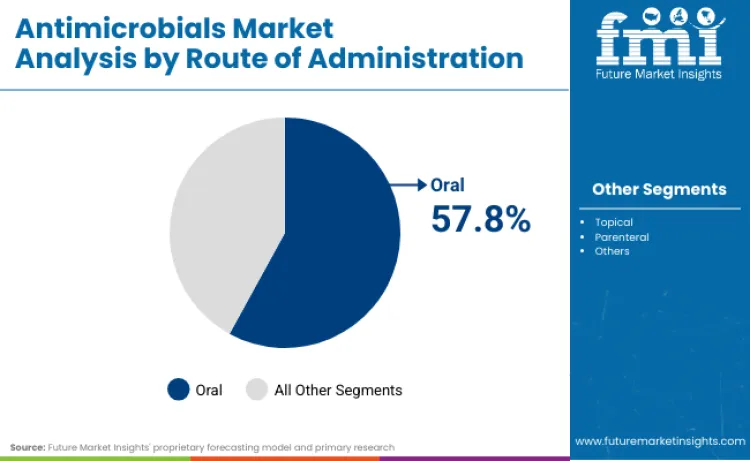
Oral antimicrobials account for 57.8% of total antimicrobial usage because modern care pathways prioritise outpatient treatment, adherence, and cost control over prolonged inpatient administration. Oral delivery enables early discharge, step-down therapy from IV regimens, and longer infection management outside hospitals, which aligns with payer efficiency and provider capacity constraints. This structural preference is reinforced by how major healthcare companies frame prevention, access, and global health commitments: "As Sanofi chases the miracles of science to improve people's lives, it continues to engage across the entire health spectrum from prevention with vaccines to wellness, treatment, patient support & capacity building."
"Efforts on four flagship programs in malaria, sickle cell disease, Chagas disease and leprosy, while also taking a coordinated approach to tackling other pressing global health challenges such as antimicrobial resistance." "Driven by its mission of 'Health for All, Hunger for None,' Bayer is reaffirming its commitment to addressing the urgent needs of millions of affected patients through the manufacturing and donation of essential medicines." At scale, Bristol Myers Squibb, Viatris, Teva Pharmaceutical Industries, Sandoz, and Cipla support broad oral access, while parenteral routes remain essential for severe infections.
What Structural Drivers Make Antimicrobials Win Share?
Antimicrobials gain (or lose) share when the market moves from “clinical need exists” to “systems make development and access economically rational.” One driver is R&D targeting discipline set by World Health Organization through the 2024 Bacterial Priority Pathogens List (BPPL), which prioritises antibiotic-resistant threats (notably Gram-negative pathogens) and acts as a signalling mechanism for funders, developers, and policy makers on where discovery and trials should concentrate. A second driver is reimbursement redesign via NHS England’s Antimicrobial Products Subscription Model (“Netflix model”), which pays for access through fixed-style contracts, reducing the commercial penalty of stewardship by delinking returns from sales volume.
A third driver is regulatory pathway engineering. In the US, USA Food and Drug Administration explains how the GAIN Act supports incentives such as the QIDP designation for antibacterial/antifungal drugs targeting serious infections. In Europe, European Medicines Agency’s PRIME scheme offers enhanced early interaction and support to speed development for medicines addressing unmet need. Together, these are structural, not narrative, drivers: they shape what gets funded, how products get paid for, and how quickly they can reach patients.
What Factors Continue to Limit Antimicrobial Development
Antimicrobial progress slows where stewardship logic conflicts with commercial recovery. The most persistent constraint is late-stage financing risk. CARB-X identifies a funding gap where many antibacterial programmes stall after early development due to insufficient capital for Phase III trials and launch infrastructure. Stewardship practices further suppress uptake. High-value antimicrobials are deliberately restricted to preserve efficacy, reducing early sales velocity and weakening conventional return models. Manufacturing complexity also constrains scale.
Advanced formulations and specialised production increase cost of goods and complicate equitable global access across fragmented formularies. Policy uncertainty compounds these pressures. In the United States, the PASTEUR Act remains proposed legislation rather than an operational reimbursement mechanism, limiting long-term planning certainty for developers. Antimicrobial materials face parallel barriers. Durability validation, resistance-risk assessment, and extended healthcare procurement cycles delay adoption even when prevention benefits are established. These constraints persist despite clear clinical need, keeping development risk elevated relative to other therapeutic categories.
What Institutional Changes Are Driving Recent Momentum?
Recent momentum reflects institutional realignment rather than microbiological breakthrough. Delinked purchasing has moved into active deployment in the United Kingdom, where NHS England now operates national commercial pathways for antimicrobial subscription contracts. Early-stage funding continuity has strengthened. CARB-X continues active solicitations and secured renewed Wellcome funding for the 2026-2028 period, stabilising antibacterial and diagnostic pipelines. Diagnostic integration is also shifting prescribing behaviour.
Rapid susceptibility systems such as Sysmex’s PA-100 AST support earlier pathogen confirmation and faster efficacy assessment, reducing reliance on empiric broad-spectrum use. Global prioritisation has tightened. WHO’s updated priority pathogen framework clarifies investment focus while ongoing infection prevention reporting sustains institutional pressure to reduce healthcare-associated infections. These changes align incentives across therapeutics and prevention materials. Antimicrobials increasingly gain share through access guarantees, sustained pipeline financing, and precision-enabled deployment rather than through expanded prescribing volume.
How Is the Antimicrobials Market Evolving Globally?
Global demand for antimicrobials is intensifying as infectious threats evolve more rapidly than conventional drug development timelines. Healthcare systems increasingly depend on technologies that enable faster pathogen identification, resistance profiling, and therapy matching at the point of care. Market evolution is shifting away from broad-spectrum coverage toward precision-led approaches, where diagnostic-guided prescribing, resistance-targeted mechanisms, and combination therapies determine clinical success. Manufacturers are scaling portfolios that align antimicrobial action with pathogen specificity, treatment duration control, and stewardship requirements. This institutional integration improves access discipline and promotes appropriate utilization rather than volume expansion. As a result, the antimicrobials category is transitioning from reactive infection management toward system-level readiness, where effectiveness is measured by resistance containment, treatment reliability, and alignment with global public health priorities rather than prescription growth alone.
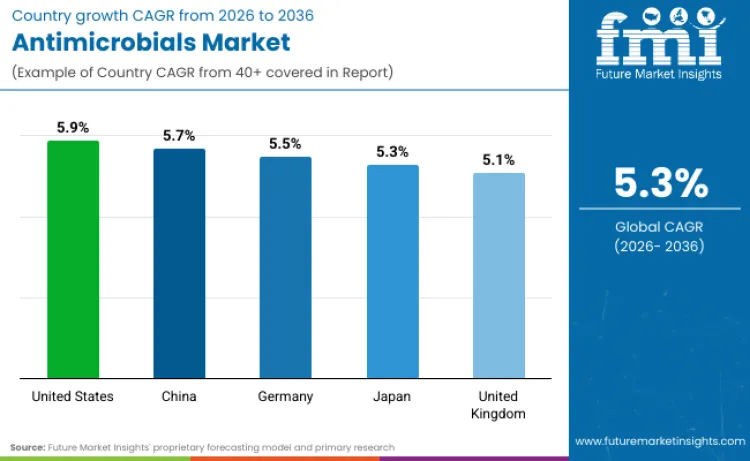
| Country | CAGR (2026-2036) |
|---|---|
| United States | 5.9% |
| China | 5.7% |
| Germany | 5.5% |
| Japan | 5.3% |
| United Kingdom | 5.1% |
Source: Future Market Insights' proprietary forecasting model and primary research
United Kingdom Antimicrobials Market Gaining Ground
The United Kingdom antimicrobial market is expanding at a 5.1% CAGR because the policy stack makes antimicrobial revenue a fixed cost for the NHS, not a variable sales target. According to NHS England, the UK has implemented a "subscription-style funding mechanism" where payments are made "regardless of the actual volume of sales." This creates a "compliance-efficient architecture" that rewards drug efficacy over sales volume. That shift matters because stewardship no longer destroys the business case: a drug can be conserved, used precisely, and still generate predictable returns. It reduces launch risk, supports supply continuity, and encourages investment in true novelty against resistant pathogens. For the NHS, the model reframes antibiotics as an availability service, aligning procurement with outcomes and resistance control in a single payment mechanism. It also improves planning in the UK, because hospitals can stock and deploy new agents for critical cases without budget shocks, while manufacturers receive signal strength that supports ongoing R&D and post-launch evidence generation.
United States Antimicrobials Market Accelerating Through Federal Contracting
The United States antimicrobial market is projected at a 5.9% CAGR because federal policy aims to stabilise a "broken" antibiotic market using long-horizon government purchasing. Under the PASTEUR Act (S.1355), the government seeks to enter into contracts that provide "fixed payments to the developer" for critical-need drugs. This turns antimicrobials into a "standardized routine purchase" for the government to ensure national biosecurity. Commercially, this reduces the penalty of stewardship, since returns are less tied to pushing volume and more tied to making products reliably available when outbreaks or resistant infections appear. It also creates clearer demand signals for manufacturers, supporting investment in novel mechanisms, modern trials, and supply resilience, while giving hospitals confidence that new options will not vanish after launch. For payers and providers, contracts can encourage faster formulary inclusion and step-down protocols, because the value is priced as readiness. The model also supports national stockpiling logic and coordinated stewardship guidelines across federal programmes for critical-care and outpatient settings.
China Antimicrobials Market Expanding Through Healthcare System Scale-Up
China’s antimicrobials market is expanding at a 5.7% CAGR because rising care capacity converts demand into real utilisation across both urban hospitals and grassroots clinics. State Council of the People's Republic of China reports that China has "established the world's largest healthcare service system," which includes a "15-minute medical service circle at the grassroots level." This ensures "mainstream visibility" and access for antimicrobial treatments in rural and urban sectors alike. As coverage and primary-care reach deepen, more infections are diagnosed earlier, prescriptions are written closer to the patient, and follow-up courses become easier to complete. That improves adherence and supports step-down transitions from inpatient to outpatient care, which raises overall oral antimicrobial throughput. At the same time, large-scale system build-out increases procurement consistency, giving manufacturers a clearer volume base and strengthening incentives to maintain steady supply and quality across provinces. It also tightens stewardship, because guidelines and diagnostics can be deployed more uniformly, reducing inappropriate use while protecting efficacy against resistant organisms at national scale.
Japan Antimicrobials Market Strengthened by Supply-Stability Revenue Guarantees
Japan’s antimicrobials market is growing at a 5.3% CAGR because the state is actively preventing supply fragility in an ageing, high-utilisation healthcare system. Ministry of Health, Labour and Welfare (Japan) has launched the "Antimicrobial Securement Support Program" to ensure the "stability of the supply" by providing "revenue guarantees" to manufacturers, mirroring the UK's high-trust model. This matters because essential antibiotics face low commercial returns, yet hospitals need them continuously for sepsis, pneumonia, and surgical prophylaxis. By underwriting continuity, the programme reduces shortage risk, supports domestic and diversified sourcing, and makes it easier for procurement teams to keep critical agents on formulary. It also helps innovators plan launches and evidence generation without fearing rapid market collapse, while stewardship policies can restrict use appropriately without undermining supplier viability. For clinicians, the benefit is practical: availability is protected, so prescribing decisions can follow guidelines and diagnostics rather than stock constraints. For manufacturers, predictable revenue supports quality investment and capacity retention across the full supply chain.
Germany Antimicrobials Market Shaped by Diagnostics-Led Stewardship Under DART 2030
Germany’s antimicrobials market is projected to grow at a 5.5% CAGR as national strategy ties prescribing more tightly to diagnostics, stewardship, and measurable appropriateness. German Federal Ministry of Health (BMG) focuses on the "Appropriate Use of Antibiotics including Laboratory Diagnostics." This forces brands into "visible formulation discipline" and ensures that antimicrobials are sold as science-backed solutions within the national drugstore ecosystem. In practice, this increases demand for products with clear spectra, strong evidence packages, and dosing forms that support guideline-concordant use in both outpatient and step-down care. It also elevates rapid testing and lab capacity as part of the purchasing logic, so therapies that pair well with diagnostic pathways gain preference. Parenteral agents remain vital for severe infections, but Germany’s growth is increasingly linked to precision use and consistent, system-wide implementation rather than sheer volume. As a result, manufacturers compete on clinical differentiation and supply reliability, while pharmacies and clinicians reinforce adherence through education and repeat refills at scale.
Which Companies Are Gaining Strategic Ground in the Antimicrobials Market?
The global antimicrobials market is increasingly shaped by companies that align innovation with policy-backed access, not just sales volume. Share leadership concentrates around firms that combine advanced infectious-disease R&D, regulatory readiness for resistant-pathogen indications, and secure supply for hospital and national procurement systems.
Pfizer has emerged as a structural leader due to its portfolio depth in hospital-grade anti-infectives and its success converting clinical evidence into formulary access. Its role in NHS England’s antimicrobial subscription model, where payment is tied to availability, not usage, marks a shift from volume dependency to resilience contracting. Shionogi, through its pathogen-targeted product cefiderocol (Fetcroja), also secured a UK subscription contract, highlighting how value is now monetised through access assurance.
GSK and Merck & Co. operate as scaled contenders. GSK’s regulatory success with Blujepa (gepotidacin) and Merck’s expansion of combination therapies signal strong readiness for resistance-focused commercial models. Emerging firms such as Venatorx Pharmaceuticals are gaining attention through BARDA and CARB-X partnerships, reinforcing the importance of early-stage stewardship-aligned innovation.
The market is now divided between access-contract leaders and pipeline-driven contenders, where success depends less on traditional volume dynamics and more on readiness for delinked, stewardship-compatible commercial pathways.
Recent Developments:
- In November 2025, GSK partnered with the Fleming Initiative to apply advanced AI and supercomputing against Gram-negative bacteria. Beginning operations in early 2026, the collaboration targets AMR grand challenges by discovering novel antibiotic classes and predicting drug-resistance emergence pathways. GSK and Fleming Initiative scientists unite to target AMR with advanced AI | GSK
- In December 2025, Sanofi completed acquisition of Vicebio, gaining an early-stage RSV and HMPV combination vaccine candidate. Vicebio’s Molecular Clamp technology strengthens Sanofi’s vaccine design capabilities and adds a non-mRNA respiratory option, expanding physician and patient choice significantly. Press Release: Sanofi completes acquisition of Vicebio
- In January 2026, Roche and Genentech launched the expanded pediatric label for Xofluza (baloxavir marboxil), extending eligibility to infants as young as three months, significantly broadening the antiviral’s clinical reach across early childhood influenza treatment settings. [Ad hoc announcement pursuant to Art. 53 LR] Roche reports strong 2025 results with 7% sales growth
Key Players in the Antimicrobials Market
- Pfizer Inc
- Merck & Co Inc
- Johnson & Johnson
- Novartis AG
- Bayer AG
- GlaxoSmithKline plc
- AstraZeneca plc
- AbbVie Inc
- Sanofi S.A.
- Abbott
- BASF SE
- Biocote
- Mylan N.V
- F. Hoffmann-La Roche Ltd
- Zydus Cadila
- Gilead Sciences Inc
- Cipla Limited
- Teva Pharmaceutical Industries Ltd
- Bristol Myers Squibb Company
- Boehringer Ingelheim International GmbH
Market Definition
The antimicrobials market comprises revenues generated from pharmaceutical products formulated to kill or inhibit the growth of pathogenic microorganisms, including bacteria, fungi, viruses, and parasites, responsible for infectious diseases in humans. These products are intended for therapeutic use in the treatment, control, or clinical management of confirmed or suspected infections across inpatient and outpatient care settings. Market sizing is expressed in USD billion and evaluated over the 2026 to 2036 period.
The market covers systemic and topical antimicrobial agents prescribed or dispensed as part of medical treatment protocols. Products are classified by target organism and therapeutic mechanism, including antibacterials, antifungals, antivirals, and antiparasitic drugs with validated clinical efficacy. The analysis treats antimicrobials as a medical therapeutics category, distinct from infection prevention tools or hygiene products. Only finished pharmaceutical products with approved indications for infectious disease treatment are included in revenue calculations, ensuring alignment with regulated healthcare use rather than preventive or environmental applications.
Market Inclusion
Included within the antimicrobials market are finished pharmaceutical formulations approved for human use and sold through hospital pharmacies, retail pharmacies, online pharmacy platforms, and licensed healthcare provider channels. The scope covers prescription antimicrobials used under medical supervision, over-the-counter antimicrobial products indicated for minor infections, and hospital-administered therapies used in acute and critical care.
The market includes single-agent antimicrobials, fixed-dose combination products, and structured therapeutic regimens where antimicrobial activity is the primary mode of clinical action. Eligible products contain synthetic, natural, or semi-synthetic active ingredients with proven pathogen-targeting efficacy. Revenues derived from antimicrobials supplied as part of standardized treatment protocols, national formularies, or hospital stewardship programs are included where drug sales are directly attributable.
Geographic coverage spans developed and developing regions where regulatory authorization, healthcare infrastructure, and prescribing frameworks support antimicrobial access and use.
Market Exclusion
Excluded from the antimicrobials market are antiseptics, disinfectants, surface cleaners, and sanitizing agents used for environmental hygiene or infection prevention without therapeutic intent. Preventive medical products such as vaccines, immunoglobulins, and immune-modulating biologics are not included, regardless of infectious disease relevance.
The market also excludes diagnostic tools, laboratory testing kits, antimicrobial susceptibility tests, and monitoring devices that support infection management but do not deliver antimicrobial treatment. Medical devices, wound dressings, catheters, and infection-control materials without direct pharmacological antimicrobial action fall outside the scope.
Non-regulated products including nutritional supplements, herbal remedies, and alternative medicines lacking validated antimicrobial efficacy are excluded. Additionally, revenues from healthcare services, clinical procedures, professional consultations, and research or manufacturing infrastructure are not counted unless directly embedded in the pricing of finished antimicrobial pharmaceutical products.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Type | Antibacterial; Antifungal; Antiviral; Antiparasitic; Others |
| Origin | Synthetic; Natural; Semi-Synthetic; Others |
| Route of Administration | Oral; Topical; Parenteral; Others |
| Distribution Channel | Hospital Pharmacy; Retail Pharmacy; Online Pharmacy; Others |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, China, Germany, Japan, United Kingdom, France, and 40+ countries |
| Key Companies Profiled | Pfizer Inc; Merck & Co Inc; Johnson & Johnson; Novartis AG; Bayer AG; Others |
| Additional Attributes | Dollar sales by type, origin, route of administration, and distribution channel; clinical efficacy benchmarking across pathogen eradication, resistance prevention, safety profiles, and treatment outcomes; market drivers linked to resistance emergence, regulatory incentives, and global health security initiatives; impact on healthcare costs, clinical outcomes, and antimicrobial stewardship; development pipeline under precision medicine approaches; compatibility with diagnostic technologies, treatment protocols, and resistance monitoring across global healthcare systems |
Antimicrobials Market by Segment
Type:
- Antibacterial
- Antifungal
- Antiviral
- Antiparasitic
- Others
Origin:
- Synthetic
- Natural
- Semi-Synthetic
- Others
Route of Administration:
- Oral
- Topical
- Parenteral
- Others
Distribution Channel:
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- United States Food and Drug Administration (FDA). (2024). Antibacterial and antimicrobial drug products: Clinical development, approval pathways, and QIDP guidance. USA Department of Health and Human Services.
- European Medicines Agency (EMA). (2024). Guideline on the evaluation of medicinal products indicated for treatment of bacterial infections. European Union.
- Centers for Disease Control and Prevention (CDC). (2024). Antimicrobial resistance threats report and stewardship program framework. USA CDC.
Frequently Asked Questions
How big is the global antimicrobials market?
The global antimicrobials market is valued at USD 117.6 billion in 2026 and is forecast to reach USD 197.0 billion by 2036.
What is the growth outlook for the antimicrobials market over the next 10 years?
The category is projected to expand at a 5.3% CAGR from 2026 to 2036, reflecting increasing infectious disease burden and development of next-generation therapeutic approaches.
Which antimicrobial types or therapeutic categories drive demand in this market?
Antibacterial agents lead demand with a 45.3% share, supported by their central role in treating bacterial infections across all healthcare settings and patient populations.
How does market development differ by region?
Growth is strongest where research investment and regulatory support align with clinical needs, with United States (5.9% CAGR), China (5.7%), Germany (5.5%), Japan (5.3%), and United Kingdom (5.1%) leading global development during 2026-2036.
What are the main challenges and constraints affecting this market?
Growth is constrained where economic viability conflicts with public health needs, regulatory complexity delays approval, or clinical trial challenges limit evidence generation for infectious disease applications.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Type, 2026 to 2036
- Antibacterial
- Antifungal
- Antiviral
- Antiparasitic
- Others
- Y-o-Y Growth Trend Analysis By Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Origin
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Origin, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Origin, 2026 to 2036
- Synthetic
- Natural
- Semi-Synthetic
- Others
- Y-o-Y Growth Trend Analysis By Origin, 2021 to 2025
- Absolute $ Opportunity Analysis By Origin, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Route of Administration, 2026 to 2036
- Oral
- Topical
- Parenteral
- Others
- Y-o-Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
- Y-o-Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Billion) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Origin
- By Route of Administration
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Pfizer Inc
- Merck & Co Inc
- Johnson & Johnson
- Novartis AG
- Bayer AG
- GlaxoSmithKline plc
- AstraZeneca plc
- AbbVie Inc
- Sanofi S.A.
- Abbott
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Antimicrobials Market Key Takeaways
- Global Antimicrobials Market Size (USD Billion), 2021-2036
- Global Antimicrobials Market Y-o-Y Growth (%), 2021-2036
- Global Antimicrobials Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Antimicrobials Market Pricing Analysis (USD/Unit), 2021-2036
- Global Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- Global Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- Global Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- Global Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- Global Antimicrobials Market Size (USD Billion) By Region, 2021-2036
- North America Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- North America Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- North America Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- North America Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- North America Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- North America Antimicrobials Market Attractiveness Analysis By Country
- North America Antimicrobials Market Attractiveness Analysis By Type
- North America Antimicrobials Market Attractiveness Analysis By Origin
- North America Antimicrobials Market Attractiveness Analysis By Route of Administration
- North America Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Latin America Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- Latin America Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- Latin America Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- Latin America Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- Latin America Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- Latin America Antimicrobials Market Attractiveness Analysis By Country
- Latin America Antimicrobials Market Attractiveness Analysis By Type
- Latin America Antimicrobials Market Attractiveness Analysis By Origin
- Latin America Antimicrobials Market Attractiveness Analysis By Route of Administration
- Latin America Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Western Europe Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- Western Europe Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- Western Europe Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- Western Europe Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- Western Europe Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- Western Europe Antimicrobials Market Attractiveness Analysis By Country
- Western Europe Antimicrobials Market Attractiveness Analysis By Type
- Western Europe Antimicrobials Market Attractiveness Analysis By Origin
- Western Europe Antimicrobials Market Attractiveness Analysis By Route of Administration
- Western Europe Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- Eastern Europe Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- Eastern Europe Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- Eastern Europe Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- Eastern Europe Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Country
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Type
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Origin
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Route of Administration
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Distribution Channel
- East Asia Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- East Asia Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- East Asia Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- East Asia Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- East Asia Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- East Asia Antimicrobials Market Attractiveness Analysis By Country
- East Asia Antimicrobials Market Attractiveness Analysis By Type
- East Asia Antimicrobials Market Attractiveness Analysis By Origin
- East Asia Antimicrobials Market Attractiveness Analysis By Route of Administration
- East Asia Antimicrobials Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- South Asia and Pacific Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- South Asia and Pacific Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- South Asia and Pacific Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- South Asia and Pacific Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Country
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Type
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Origin
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Route of Administration
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Antimicrobials Market Size (USD Billion) By Country, 2021-2036
- Middle East & Africa Antimicrobials Market Size (USD Billion) By Type, 2021-2036
- Middle East & Africa Antimicrobials Market Size (USD Billion) By Origin, 2021-2036
- Middle East & Africa Antimicrobials Market Size (USD Billion) By Route of Administration, 2021-2036
- Middle East & Africa Antimicrobials Market Size (USD Billion) By Distribution Channel, 2021-2036
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Country
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Type
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Origin
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Route of Administration
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Distribution Channel
- USA Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- USA Antimicrobials Market Share Analysis (%) By Type, 2025
- USA Antimicrobials Market Share Analysis (%) By Origin, 2025
- USA Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- USA Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Canada Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Canada Antimicrobials Market Share Analysis (%) By Type, 2025
- Canada Antimicrobials Market Share Analysis (%) By Origin, 2025
- Canada Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Canada Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Mexico Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Mexico Antimicrobials Market Share Analysis (%) By Type, 2025
- Mexico Antimicrobials Market Share Analysis (%) By Origin, 2025
- Mexico Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Mexico Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Brazil Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Brazil Antimicrobials Market Share Analysis (%) By Type, 2025
- Brazil Antimicrobials Market Share Analysis (%) By Origin, 2025
- Brazil Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Brazil Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Chile Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Chile Antimicrobials Market Share Analysis (%) By Type, 2025
- Chile Antimicrobials Market Share Analysis (%) By Origin, 2025
- Chile Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Chile Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Germany Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Germany Antimicrobials Market Share Analysis (%) By Type, 2025
- Germany Antimicrobials Market Share Analysis (%) By Origin, 2025
- Germany Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Germany Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- UK Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- UK Antimicrobials Market Share Analysis (%) By Type, 2025
- UK Antimicrobials Market Share Analysis (%) By Origin, 2025
- UK Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- UK Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Italy Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Italy Antimicrobials Market Share Analysis (%) By Type, 2025
- Italy Antimicrobials Market Share Analysis (%) By Origin, 2025
- Italy Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Italy Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Spain Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Spain Antimicrobials Market Share Analysis (%) By Type, 2025
- Spain Antimicrobials Market Share Analysis (%) By Origin, 2025
- Spain Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Spain Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- France Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- France Antimicrobials Market Share Analysis (%) By Type, 2025
- France Antimicrobials Market Share Analysis (%) By Origin, 2025
- France Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- France Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- India Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- India Antimicrobials Market Share Analysis (%) By Type, 2025
- India Antimicrobials Market Share Analysis (%) By Origin, 2025
- India Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- India Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- ASEAN Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- ASEAN Antimicrobials Market Share Analysis (%) By Type, 2025
- ASEAN Antimicrobials Market Share Analysis (%) By Origin, 2025
- ASEAN Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- ASEAN Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Australia & New Zealand Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Antimicrobials Market Share Analysis (%) By Type, 2025
- Australia & New Zealand Antimicrobials Market Share Analysis (%) By Origin, 2025
- Australia & New Zealand Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Australia & New Zealand Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- China Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- China Antimicrobials Market Share Analysis (%) By Type, 2025
- China Antimicrobials Market Share Analysis (%) By Origin, 2025
- China Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- China Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Japan Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Japan Antimicrobials Market Share Analysis (%) By Type, 2025
- Japan Antimicrobials Market Share Analysis (%) By Origin, 2025
- Japan Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Japan Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- South Korea Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- South Korea Antimicrobials Market Share Analysis (%) By Type, 2025
- South Korea Antimicrobials Market Share Analysis (%) By Origin, 2025
- South Korea Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- South Korea Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Russia Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Russia Antimicrobials Market Share Analysis (%) By Type, 2025
- Russia Antimicrobials Market Share Analysis (%) By Origin, 2025
- Russia Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Russia Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Poland Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Poland Antimicrobials Market Share Analysis (%) By Type, 2025
- Poland Antimicrobials Market Share Analysis (%) By Origin, 2025
- Poland Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Poland Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Hungary Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Hungary Antimicrobials Market Share Analysis (%) By Type, 2025
- Hungary Antimicrobials Market Share Analysis (%) By Origin, 2025
- Hungary Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Hungary Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Kingdom of Saudi Arabia Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Antimicrobials Market Share Analysis (%) By Type, 2025
- Kingdom of Saudi Arabia Antimicrobials Market Share Analysis (%) By Origin, 2025
- Kingdom of Saudi Arabia Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Kingdom of Saudi Arabia Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Turkiye Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- Turkiye Antimicrobials Market Share Analysis (%) By Type, 2025
- Turkiye Antimicrobials Market Share Analysis (%) By Origin, 2025
- Turkiye Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- Turkiye Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- South Africa Antimicrobials Pricing Analysis (USD/Unit), 2021-2036
- South Africa Antimicrobials Market Share Analysis (%) By Type, 2025
- South Africa Antimicrobials Market Share Analysis (%) By Origin, 2025
- South Africa Antimicrobials Market Share Analysis (%) By Route of Administration, 2025
- South Africa Antimicrobials Market Share Analysis (%) By Distribution Channel, 2025
- Antimicrobials Market Competition Dashboard
- Antimicrobials Market Competition Benchmarking Analysis
- Antimicrobials Market Share Analysis of Top Players By Regional, 2025
- Antimicrobials Market Share Analysis of Top Players By Type, 2025
- Antimicrobials Market Share Analysis of Top Players By Origin, 2025
- Antimicrobials Market Share Analysis of Top Players By Route of Administration, 2025
- Antimicrobials Market Share Analysis of Top Players By Distribution Channel, 2025
- Pfizer Inc - Company Profile
- Merck & Co Inc - Company Profile
- Johnson & Johnson - Company Profile
- Novartis AG - Company Profile
- Bayer AG - Company Profile
- GlaxoSmithKline plc - Company Profile
- AstraZeneca plc - Company Profile
- AbbVie Inc - Company Profile
- Sanofi S.A. - Company Profile
- Abbott - Company Profile
- Assumptions & Acronyms Used in Antimicrobials Market Report
List of Figures
- Antimicrobials Market Size (USD Billion), 2021-2036
- Antimicrobials Market Y-o-Y Growth (%), 2021-2036
- Antimicrobials Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Antimicrobials Market Value Chain Analysis
- Antimicrobials Market Supply Chain Analysis
- Antimicrobials Market Investment Feasibility Matrix
- Antimicrobials Market PESTLE Analysis
- Antimicrobials Market Porter's Five Forces Analysis
- Antimicrobials Market Product Life Cycle Analysis
- Antimicrobials Market Opportunity Map Analysis
- Antimicrobials Market Scenario Forecast Analysis
- Antimicrobials Market Production and Consumption Statistics
- Antimicrobials Market Import and Export Statistics
- Global Antimicrobials Market Share (%) By Type, 2025
- Global Antimicrobials Market Share (%) By Type, 2036
- Global Antimicrobials Market Share (%) By Origin, 2025
- Global Antimicrobials Market Share (%) By Origin, 2036
- Global Antimicrobials Market Share (%) By Route of Administration, 2025
- Global Antimicrobials Market Share (%) By Route of Administration, 2036
- Global Antimicrobials Market Share (%) By Distribution Channel, 2025
- Global Antimicrobials Market Share (%) By Distribution Channel, 2036
- Global Antimicrobials Market Share (%) By Region, 2025
- Global Antimicrobials Market Share (%) By Region, 2036
- Global Antimicrobials Market Attractiveness Analysis By Region
- North America Antimicrobials Market Size (USD Billion), 2021-2036
- North America Antimicrobials Market Share (%) By Country, 2025
- North America Antimicrobials Market Share (%) By Country, 2036
- North America Antimicrobials Market Share (%) By Type, 2025
- North America Antimicrobials Market Share (%) By Type, 2036
- North America Antimicrobials Market Share (%) By Origin, 2025
- North America Antimicrobials Market Share (%) By Origin, 2036
- North America Antimicrobials Market Share (%) By Route of Administration, 2025
- North America Antimicrobials Market Share (%) By Route of Administration, 2036
- North America Antimicrobials Market Share (%) By Distribution Channel, 2025
- North America Antimicrobials Market Share (%) By Distribution Channel, 2036
- North America Antimicrobials Market Attractiveness Analysis By Country
- North America Antimicrobials Market Attractiveness Analysis By Type
- North America Antimicrobials Market Attractiveness Analysis By Origin
- North America Antimicrobials Market Attractiveness Analysis By Route of Administration
- North America Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Latin America Antimicrobials Market Size (USD Billion), 2021-2036
- Latin America Antimicrobials Market Share (%) By Country, 2025
- Latin America Antimicrobials Market Share (%) By Country, 2036
- Latin America Antimicrobials Market Share (%) By Type, 2025
- Latin America Antimicrobials Market Share (%) By Type, 2036
- Latin America Antimicrobials Market Share (%) By Origin, 2025
- Latin America Antimicrobials Market Share (%) By Origin, 2036
- Latin America Antimicrobials Market Share (%) By Route of Administration, 2025
- Latin America Antimicrobials Market Share (%) By Route of Administration, 2036
- Latin America Antimicrobials Market Share (%) By Distribution Channel, 2025
- Latin America Antimicrobials Market Share (%) By Distribution Channel, 2036
- Latin America Antimicrobials Market Attractiveness Analysis By Country
- Latin America Antimicrobials Market Attractiveness Analysis By Type
- Latin America Antimicrobials Market Attractiveness Analysis By Origin
- Latin America Antimicrobials Market Attractiveness Analysis By Route of Administration
- Latin America Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Western Europe Antimicrobials Market Size (USD Billion), 2021-2036
- Western Europe Antimicrobials Market Share (%) By Country, 2025
- Western Europe Antimicrobials Market Share (%) By Country, 2036
- Western Europe Antimicrobials Market Share (%) By Type, 2025
- Western Europe Antimicrobials Market Share (%) By Type, 2036
- Western Europe Antimicrobials Market Share (%) By Origin, 2025
- Western Europe Antimicrobials Market Share (%) By Origin, 2036
- Western Europe Antimicrobials Market Share (%) By Route of Administration, 2025
- Western Europe Antimicrobials Market Share (%) By Route of Administration, 2036
- Western Europe Antimicrobials Market Share (%) By Distribution Channel, 2025
- Western Europe Antimicrobials Market Share (%) By Distribution Channel, 2036
- Western Europe Antimicrobials Market Attractiveness Analysis By Country
- Western Europe Antimicrobials Market Attractiveness Analysis By Type
- Western Europe Antimicrobials Market Attractiveness Analysis By Origin
- Western Europe Antimicrobials Market Attractiveness Analysis By Route of Administration
- Western Europe Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Eastern Europe Antimicrobials Market Size (USD Billion), 2021-2036
- Eastern Europe Antimicrobials Market Share (%) By Country, 2025
- Eastern Europe Antimicrobials Market Share (%) By Country, 2036
- Eastern Europe Antimicrobials Market Share (%) By Type, 2025
- Eastern Europe Antimicrobials Market Share (%) By Type, 2036
- Eastern Europe Antimicrobials Market Share (%) By Origin, 2025
- Eastern Europe Antimicrobials Market Share (%) By Origin, 2036
- Eastern Europe Antimicrobials Market Share (%) By Route of Administration, 2025
- Eastern Europe Antimicrobials Market Share (%) By Route of Administration, 2036
- Eastern Europe Antimicrobials Market Share (%) By Distribution Channel, 2025
- Eastern Europe Antimicrobials Market Share (%) By Distribution Channel, 2036
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Country
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Type
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Origin
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Route of Administration
- Eastern Europe Antimicrobials Market Attractiveness Analysis By Distribution Channel
- East Asia Antimicrobials Market Size (USD Billion), 2021-2036
- East Asia Antimicrobials Market Share (%) By Country, 2025
- East Asia Antimicrobials Market Share (%) By Country, 2036
- East Asia Antimicrobials Market Share (%) By Type, 2025
- East Asia Antimicrobials Market Share (%) By Type, 2036
- East Asia Antimicrobials Market Share (%) By Origin, 2025
- East Asia Antimicrobials Market Share (%) By Origin, 2036
- East Asia Antimicrobials Market Share (%) By Route of Administration, 2025
- East Asia Antimicrobials Market Share (%) By Route of Administration, 2036
- East Asia Antimicrobials Market Share (%) By Distribution Channel, 2025
- East Asia Antimicrobials Market Share (%) By Distribution Channel, 2036
- East Asia Antimicrobials Market Attractiveness Analysis By Country
- East Asia Antimicrobials Market Attractiveness Analysis By Type
- East Asia Antimicrobials Market Attractiveness Analysis By Origin
- East Asia Antimicrobials Market Attractiveness Analysis By Route of Administration
- East Asia Antimicrobials Market Attractiveness Analysis By Distribution Channel
- South Asia and Pacific Antimicrobials Market Size (USD Billion), 2021-2036
- South Asia and Pacific Antimicrobials Market Share (%) By Country, 2025
- South Asia and Pacific Antimicrobials Market Share (%) By Country, 2036
- South Asia and Pacific Antimicrobials Market Share (%) By Type, 2025
- South Asia and Pacific Antimicrobials Market Share (%) By Type, 2036
- South Asia and Pacific Antimicrobials Market Share (%) By Origin, 2025
- South Asia and Pacific Antimicrobials Market Share (%) By Origin, 2036
- South Asia and Pacific Antimicrobials Market Share (%) By Route of Administration, 2025
- South Asia and Pacific Antimicrobials Market Share (%) By Route of Administration, 2036
- South Asia and Pacific Antimicrobials Market Share (%) By Distribution Channel, 2025
- South Asia and Pacific Antimicrobials Market Share (%) By Distribution Channel, 2036
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Country
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Type
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Origin
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Route of Administration
- South Asia and Pacific Antimicrobials Market Attractiveness Analysis By Distribution Channel
- Middle East & Africa Antimicrobials Market Size (USD Billion), 2021-2036
- Middle East & Africa Antimicrobials Market Share (%) By Country, 2025
- Middle East & Africa Antimicrobials Market Share (%) By Country, 2036
- Middle East & Africa Antimicrobials Market Share (%) By Type, 2025
- Middle East & Africa Antimicrobials Market Share (%) By Type, 2036
- Middle East & Africa Antimicrobials Market Share (%) By Origin, 2025
- Middle East & Africa Antimicrobials Market Share (%) By Origin, 2036
- Middle East & Africa Antimicrobials Market Share (%) By Route of Administration, 2025
- Middle East & Africa Antimicrobials Market Share (%) By Route of Administration, 2036
- Middle East & Africa Antimicrobials Market Share (%) By Distribution Channel, 2025
- Middle East & Africa Antimicrobials Market Share (%) By Distribution Channel, 2036
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Country
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Type
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Origin
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Route of Administration
- Middle East & Africa Antimicrobials Market Attractiveness Analysis By Distribution Channel
- USA Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Canada Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Mexico Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Brazil Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Chile Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Germany Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- UK Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Italy Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Spain Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- France Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- India Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- ASEAN Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Australia & New Zealand Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- China Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Japan Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- South Korea Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Russia Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Poland Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Hungary Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Kingdom of Saudi Arabia Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Turkiye Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- South Africa Antimicrobials Pricing Trend Analysis (USD/Unit), 2021-2036
- Antimicrobials Market Competitive Landscape
- Antimicrobials Market Structure Analysis
- Antimicrobials Market Competition Dashboard
- Antimicrobials Market Competition Benchmarking Analysis
- Antimicrobials Market Share Analysis of Top Players By Regional
- Antimicrobials Market Share Analysis of Top Players By Type
- Antimicrobials Market Share Analysis of Top Players By Origin
- Antimicrobials Market Share Analysis of Top Players By Route of Administration
- Antimicrobials Market Share Analysis of Top Players By Distribution Channel
- Pfizer Inc - Revenue Analysis (USD Billion), 2019-2025
- Merck & Co Inc - Revenue Analysis (USD Billion), 2019-2025
- Johnson & Johnson - Revenue Analysis (USD Billion), 2019-2025
- Novartis AG - Revenue Analysis (USD Billion), 2019-2025
- Bayer AG - Revenue Analysis (USD Billion), 2019-2025
- Pfizer Inc - Market Position Analysis
- Merck & Co Inc - Market Position Analysis
- Johnson & Johnson - Market Position Analysis
- Novartis AG - Market Position Analysis
- Bayer AG - Market Position Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

