Arthropod-borne Viral Infections Testing Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Arthropod-borne Viral Infections Testing Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Arthropod-borne Viral Infections Testing Market Size and Share Forecast Outlook 2025 to 2035
The Arthropod-borne Viral Infections Testing Market is estimated to be valued at USD 1.5 billion in 2025 and is projected to reach USD 2.6 billion by 2035, registering a compound annual growth rate (CAGR) of 5.6% over the forecast period.
Quick Stats for Arthropod-borne Viral Infections Testing Market
- Arthropod-borne Viral Infections Testing Market Industry Value (2025): USD 1.5 billion
- Arthropod-borne Viral Infections Testing Market Forecast Value (2035): USD 2.6 billion
- Arthropod-borne Viral Infections Testing Market Forecast CAGR: 5.6%
- Leading Segment in Arthropod-borne Viral Infections Testing Market in 2025: RT-PCR based Tests (49.2%)
- Key Growth Region in Arthropod-borne Viral Infections Testing Market: North America, Asia-Pacific, Europe
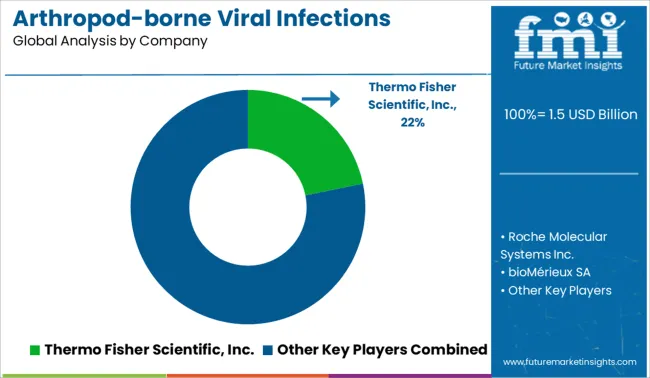
- Top Key Players in Arthropod-borne Viral Infections Testing Market: Thermo Fisher Scientific, Inc., Roche Molecular Systems Inc., bioMérieux SA, Abbott Laboratories, InBios International, Inc., NovaTec Immundiagnostica GmbH, Euroimmun AG (Sub. PerkinElmer), Quest Diagnostics, Certest Biotec S.L., Response Biomedical Corp., Chembio Diagnostic Systems, Inc.
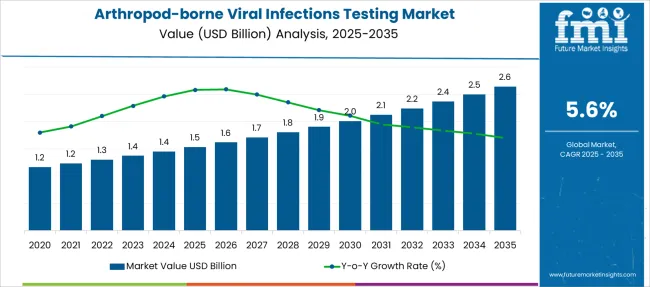
| Metric | Value |
|---|---|
| Arthropod-borne Viral Infections Testing Market Estimated Value in (2025 E) | USD 1.5 billion |
| Arthropod-borne Viral Infections Testing Market Forecast Value in (2035 F) | USD 2.6 billion |
| Forecast CAGR (2025 to 2035) | 5.6% |
Rationale for Segmental Growth in the Arthropod-borne Viral Infections Testing Market
The arthropod-borne viral infections testing market is experiencing steady growth due to increased incidence of vector-borne diseases and heightened focus on early diagnosis and disease control. The rising prevalence of infections transmitted by mosquitoes and ticks has driven demand for reliable and rapid diagnostic solutions.
Healthcare providers are placing emphasis on timely and accurate detection to contain outbreaks and improve patient outcomes. Advances in molecular diagnostics have made RT-PCR based tests the preferred method due to their high sensitivity and specificity.
Additionally, public health initiatives and surveillance programs have expanded testing capacity in hospitals and clinical centers. Growing awareness among healthcare practitioners about the importance of early testing has further fueled market expansion. The integration of testing with disease monitoring systems supports prompt intervention and resource allocation. Segment growth is expected to be led by RT-PCR based tests for their diagnostic accuracy and hospitals as the primary end users, reflecting the critical role of institutional healthcare settings in managing infectious diseases.
Segmental Analysis
The market is segmented by Test Type and End User and region. By Test Type, the market is divided into RT-PCR based Tests, ELISA-based Tests, and Immunochromatography Rapid Diagnosis. In terms of End User, the market is classified into Hospitals, Public Health Laboratory, Diagnostic Centers, and Others. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Test Type Segment: RT-PCR Based Tests
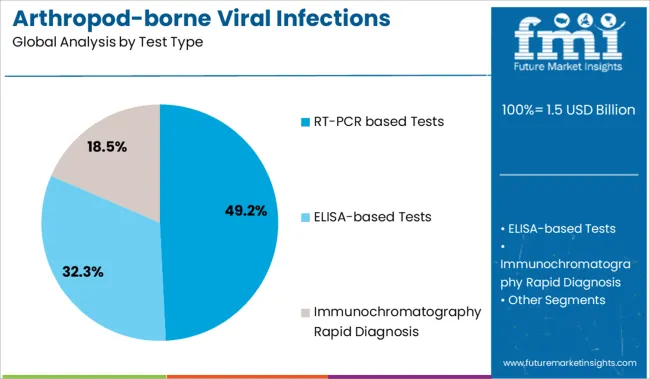
The RT-PCR based tests segment is projected to hold 49.2% of the market revenue in 2025, positioning it as the leading test type. Growth has been driven by the superior diagnostic performance of RT-PCR technology in detecting viral RNA with high precision. These tests allow for early detection during the acute phase of infection and can differentiate between closely related viral strains.
The ability to process multiple samples with automated platforms has increased throughput and testing efficiency. Furthermore, RT-PCR tests have become standard practice in hospital laboratories for confirmation of arthropod-borne viral infections, supporting rapid clinical decision-making.
As emerging viral strains continue to pose diagnostic challenges, the reliance on RT-PCR technology is expected to remain strong.
Insights into the End User Segment: Hospitals
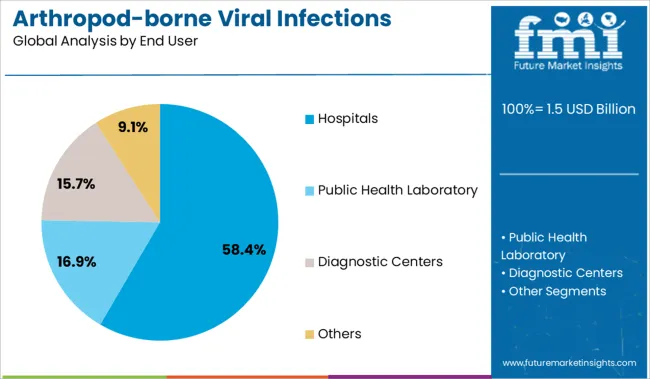
Hospitals are projected to represent 58.4% of the market revenue in 2025, maintaining their position as the primary end users of arthropod-borne viral infections testing. The growth in this segment is attributed to the critical need for institutional testing facilities equipped with advanced molecular diagnostic capabilities.
Hospitals serve as frontline centers for patient care during viral outbreaks and require reliable diagnostic tools to manage and isolate infected individuals promptly. The availability of specialized laboratory infrastructure and trained personnel in hospital settings supports the widespread use of RT-PCR and other advanced tests.
Additionally, hospitals play a key role in public health surveillance and reporting, further driving demand for efficient diagnostic testing. With continuous investment in healthcare infrastructure and growing disease burden, hospitals are expected to sustain their lead as the main consumers of arthropod-borne viral infections tests.
How The Market Progressed Till June 2025?
| Particulars | Details |
|---|---|
| H1, 2024 | 5.64% |
| H1, 2025 Projected | 5.63% |
| H1, 2025 Outlook | 3.83% |
| BPS Change - H1, 2025 (O) - H1, 2025 (P) | (-) 180 ↓ |
| BPS Change - H1, 2025 (O) - H1, 2024 | (-) 181 ↓ |
The variation between the BPS values observed within the arthropod-borne viral infections testing market in H1, 2025 - outlook over H1, 2025 projected reflects a decline by 180 BPS units. Additionally, a value BPS decline in H1-2025 over H1-2024 by 181 Basis Point Share (BPS) is demonstrated by the market.
The market observes a decline in the BPS values owed to the poor accessibility of detection methods in low-income economies with a large disease burden. As a result, high costs of healthcare facilities along with a lack of awareness about infectious diseases are expected to restrain the market's growth.
The market is subjected to several macro economical and industry variables, which include disease epidemiology, biologics licensing, product patenting and distribution licensing.
Factors such as climate change, urbanization, and global trade can expand the geographic distribution of vectors and associated arboviruses, thus increasing the potential disease burden in susceptible hosts. This factor can present an opportunity for a positive growth outlook for the market
Demand Analysis of Arthropod-borne Viral Infections Testing 2020 to 2025 Vs Market Metrics 2025 to 2035
Healthcare has become an area of prime concern across the world and this is due to the rising diseases and infections across the world. There has been a substantial rise in arboviral diseases over the past few years and this has influenced demand for arthropod-borne viral infections testing.
Arthropod-borne viral infections testing demand rose at a CAGR of 4.9% from 2014 to 2024 and ended up with a market worth of USD 1.4 Billion in 2024.
Most common arthropod-borne viral infection in the world according to WHO (World Health Organization) is dengue, with the potential risk to infect more than 30% of the world’s population.
Rising focus on public healthcare, increasing travel and trade activities on a global scale, changing lifestyle trends, environmental and climatic changes, rapid urbanization across the world, etc. are some major factors that influence arthropod-borne viral infections testing market potential on a global scale.
The market for arthropod-borne viral infections testing is anticipated to rise at a CAGR of 5.6% over the forecast period of 2025 to 2035.
Which Factors Favor Arthropod-borne Viral Infections Testing Market Potential?
Re-emergence of Arthropod-borne Viral Infections to Drive Demand
Arthropod-borne infections have proven to be responsible for widespread epidemics across the world. Some of the notable arboviral epidemics are of dengue and Zika virus which led to further complications in the population infected.
These epidemics have been dealt with to a certain extent but have proven their re-emergence in recent times which is a major factor that drives demand for arthropod-borne viral infections testing on a global level. The unpredictable nature of these infections promotes arthropod-borne viral infections testing providers to develop better diagnostic solutions to detect and avoid a community spread of these infections.
Increasing Government Initiatives to Limit Risk of Arboviral Infections
Governments across the world are working in sync with health organizations to mitigate the risk of arthropod-borne viral infections. As a matter of public safety, the focus on these diseases is increasing in the past few years with severe outbreaks being recorded across the world. The emergence of coronavirus in 2024 has further bolstered the importance of government inclusion in matters of public health.
For instance, a pilot program funded by Global Emerging Infectious Surveillance (GEIS) a division of Armed Forces Health Surveillance Branch of the USA is set to launch in spring of 2025 to identify emerging infectious diseases and mitigate risk. This is expected to be attained by a global laboratory network that is designed to enhance global health security.
Which Regions Should Arthropod-borne Viral Infections Testing Providers Focus On?
MEA and Latin America to Provide Major Opportunities for Market Players
This FMI industry analysis provides metrics for the arthropod-borne viral infections testing market across regions such as North America, Latin America, Europe, East Asia, South Asia & Pacific, and the Middle East & Africa (MEA).
The market for arthropod-borne viral infections testing in Latin America is anticipated to see high demand over the forecast period as it accounts for a notable number of infections across the world.
Developing healthcare infrastructure and rising awareness about arthropod-borne infections is anticipated to influence arthropod-borne viral infections testing demand in this region.
Demand for arthropod-borne viral infections testing in North America is expected to be low as the incidence of arboviral infections and diseases in this region is low. Advanced healthcare infrastructure and proper preventive care reduce the chances of infection in this region.
East Asia and South Asia are expected to see high demand for arthropod-borne viral infections testing over the forecast period. Implementation of stringent regulations to curb the spread of infectious diseases in this region is anticipated to drive market potential in these regions.
Densely populated countries like India and China are expected to emerge as highly lucrative markets for arthropod-borne viral infections testing vendors.
Rising instances of arboviral infections and epidemics in the MEA region are expected to majorly drive demand for arthropod-borne viral infections testing in this region. This region is expected to be one of the most important markets on a global level owing to the re-emergence of multiple infections like Zika virus and others.
Underdeveloped healthcare infrastructure is a major restraining factor for arthropod-borne viral infections testing market in the MEA region.
Country-wise Analysis
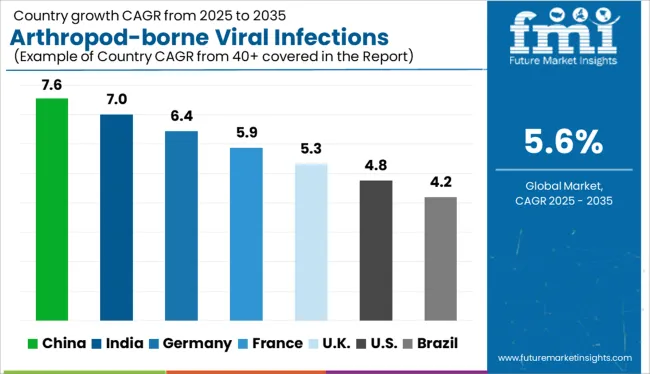
Why is the UK a Lucrative Market for Arthropod-borne Viral Infections Testing Providers?
Increasing Threat of Arboviral Infections Augmenting Market Expansion
Instances of arthropod-borne viral infections have been increasing in the UK in recent times and this is anticipated to drive arthropod-borne viral infections testing demand in the nation. Lyme disease has become a prominent issue in the UK over recent years and other arthropod-borne diseases have also seen an increase in incidence.
Increasing global travel and trade are expected to fuel the adoption of arthropod-borne viral infections testing in the UK
What is the Outlook for Arthropod-borne Viral Infections Testing in Brazil?
Developing Healthcare Infrastructure to Provide Lucrative Market Opportunities
Brazil is anticipated to be a highly rewarding market for arthropod-borne viral infections testing vendors due to high incidence of multiple arthropod-borne viral infections like Zika virus and dengue. Brazil is a developing economy and has an emerging healthcare infrastructure that has a lot of untapped potential that arthropod-borne viral infections testing providers can unlock if they enter this market.
Category-wise Analysis
Which Test Type is Expected to Perform the Best?
High Preference for ELISA-based Test Kits to Drive Sales through 2035
Enzyme-linked immunosorbent assay (ELISA)-based tests have been immensely popular in the arthropod-borne viral infections testing marketplace over the past few years and currently account for a dominant market share of 52% in the global landscape.
Increasing preference for use of ELISA-based test kits by patients, healthcare professionals, researchers, etc. is expected to propel its popularity further.
In 2025, reverse-transcription polymerase chain reaction (RT-PCR) kits hold a market share of 33.5% in the global arthropod-borne viral infections testing industry. The COVID-19 pandemic boosted demand for RT-PCR test kits and this trend is expected to be prevalent over the forecast period as well.
Competitive Landscape
Arthropod-borne viral infections testing providers are investing in the research and development of rapid test kits that are effective in diagnosing infections in a short period of time with minimal error.
- In November 2024, Achico, a healthcare technology firm headquartered in Switzerland, announced the development of dengue fever diagnostic kits. This new testing kit would be based on the company’s DNA aptamer technology platform that has proven its efficiency in COVID-19 diagnosis.
Arthropod-borne Viral Infections Testing Industry Report Scope
| Attribute | Details |
|---|---|
| Forecast Period | 2025 to 2035 |
| Historical Data Available for | 2014 to 2024 |
| Market Analysis | USD Million for Value |
| Key Regions Covered | North America; Latin America; Europe; East Asia; South Asia & Pacific; Middle East & Africa (MEA) |
| Key Countries Covered | United States, Canada, Brazil, Mexico, Germany, UK, France, Spain, Italy, China, Japan, South Korea, India, Indonesia, Malaysia, Singapore, Australia, New Zealand, Turkey, South Africa, and GCC Countries |
| Key Market Segments Covered | Test Type, End User, Region |
| Key Companies Profiled | Thermo Fisher Scientific Inc.; Roche Molecular Systems Inc.; bioMérieux SA; InBios International Inc.; NovaTec Immundiagnostica GmbH; Euroimmun AG (Sub. PerkinElmer); Quest Diagnostics; Certest Biotec S.L.; Response Biomedical Corp.; Chembio Diagnostic Systems Inc.; Abbott Laboratories |
| Pricing | Available upon Request |
Key Segments of Arthropod-borne Viral Infections Testing Industry Survey
ByTest Type:
- ELISA-based Tests for Arthropod-borne Viral Infections Testing
- RT-PCR-based Tests for Arthropod-borne Viral Infections Testing
- Immunochromatography Rapid Diagnosis for Arthropod-borne Viral Infections Testing
ByEnd User:
- Arthropod-borne Viral Infections in Testing Hospitals
- Arthropod-borne Viral Infections in Public Health Laboratories
- Arthropod-borne Viral Infections in Diagnostic Centres
- Others
ByRegion:
- North America Arthropod-borne Viral Infections Testing Market
- Latin America Arthropod-borne Viral Infections Testing Market
- Europe Arthropod-borne Viral Infections Testing Market
- East Asia Arthropod-borne Viral Infections Testing Market
- South Asia & Pacific Arthropod-borne Viral Infections Testing Market
- Middle East & Africa (MEA) Arthropod-borne Viral Infections Testing Market
Frequently Asked Questions
How big is the arthropod-borne viral infections testing market in 2025?
The global arthropod-borne viral infections testing market is estimated to be valued at USD 1.5 billion in 2025.
What will be the size of arthropod-borne viral infections testing market in 2035?
The market size for the arthropod-borne viral infections testing market is projected to reach USD 2.6 billion by 2035.
How much will be the arthropod-borne viral infections testing market growth between 2025 and 2035?
The arthropod-borne viral infections testing market is expected to grow at a 5.6% CAGR between 2025 and 2035.
What are the key product types in the arthropod-borne viral infections testing market?
The key product types in arthropod-borne viral infections testing market are rt-pcr based tests, elisa-based tests and immunochromatography rapid diagnosis.
Which end user segment to contribute significant share in the arthropod-borne viral infections testing market in 2025?
In terms of end user, hospitals segment to command 58.4% share in the arthropod-borne viral infections testing market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Mn) Analysis, 2020-2024
- Current and Future Market Size Value (USD Mn) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Arthropod-borne Viral Infections Testing Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Test Type
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By Test Type , 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By Test Type , 2025-2035
- RT-PCR based Tests
- ELISA-based Tests
- Immunochromatography Rapid Diagnosis
- Y-o-Y Growth Trend Analysis By Test Type , 2020-2024
- Absolute $ Opportunity Analysis By Test Type , 2025-2035
- Global Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Mn) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Mn) Analysis and Forecast By End User, 2025-2035
- Hospitals
- Public Health Laboratory
- Diagnostic Centers
- Others
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Mn) Analysis By Region, 2020-2024
- Current Market Size Value (USD Mn) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- U.S.
- Canada
- Mexico
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- Latin America Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- Western Europe Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- U.K.
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- Eastern Europe Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- East Asia Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- South Asia and Pacific Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- Middle East & Africa Arthropod-borne Viral Infections Testing Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Mn) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Mn) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Test Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Test Type
- By End User
- Key Takeaways
- Key Countries Arthropod-borne Viral Infections Testing Market Analysis
- U.S.
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- U.K.
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Test Type
- By End User
- U.S.
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Test Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific, Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Roche Molecular Systems Inc.
- bioMérieux SA
- Abbott Laboratories
- InBios International, Inc.
- NovaTec Immundiagnostica GmbH
- Euroimmun AG (Sub. PerkinElmer)
- Quest Diagnostics
- Certest Biotec S.L.
- Response Biomedical Corp.
- Chembio Diagnostic Systems, Inc.
- Thermo Fisher Scientific, Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
x- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: Global Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 3: Global Market Value (USD Million) Forecast by End User, 2020-2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: North America Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 6: North America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Latin America Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2020-2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2020-2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 17: East Asia Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2020-2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2020-2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Test Type , 2020-2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

