Cytotoxic Chemotherapy Market
Cytotoxic Chemotherapy Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Cytotoxic Chemotherapy Market Forecast and Outlook 2026 to 2036
The cytotoxic chemotherapy market is likely to be valued at USD 24,628.0 million in 2026 and reach USD 39,358.0 million by 2036, at a CAGR of 4.8%. Value formation remains closely tied to the role of cytotoxic agents as foundational therapies across a wide range of solid tumors and hematological malignancies. Despite advances in targeted and immune-based oncology treatments, cytotoxic drugs continue to anchor first-line and combination regimens due to their broad mechanism of action and established clinical evidence. Utilization is concentrated in hospital-led oncology care, where infusion capacity, pharmacy compounding infrastructure, and supportive care protocols enable safe administration.
Generic availability across multiple drug classes supports affordability and inclusion in national treatment guidelines, particularly in public health systems. Pricing dynamics are influenced more by volume throughput and tender-based procurement than by innovation-driven premium positioning, reinforcing stable demand patterns across mature and emerging healthcare systems.
Quick Stats for Cytotoxic Chemotherapy Market
- Cytotoxic Chemotherapy Market Value (2026): USD 24,628.0 million
- Cytotoxic Chemotherapy Market Forecast Value (2036): USD 39,358.0 million
- Cytotoxic Chemotherapy Market Forecast CAGR 2026 to 2036: 4.8%
- Leading Product Category by Demand Share: Alkylating Agents
- Fastest-Growing Countries: India, China, Brazil, USA, UK
- Top Players in Global Demand: Roche, Pfizer, Novartis, Bristol Myers Squibb, Teva
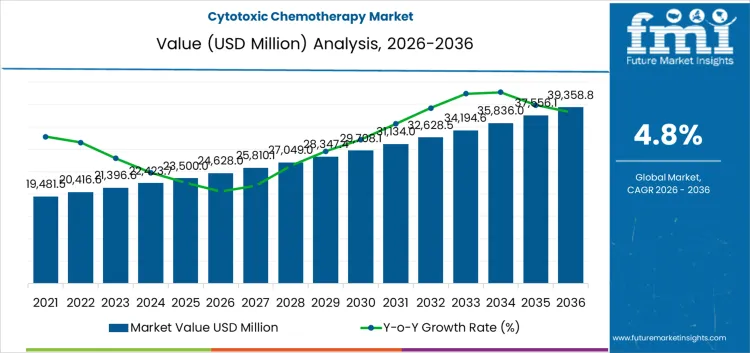
Across the forecast period, expansion reflects patient volume growth rather than structural change in treatment paradigms. Rising cancer incidence, driven by aging populations and improved diagnostic reach, sustains consistent chemotherapy cycles across treatment stages. Clinical practice continues to integrate cytotoxic agents with targeted therapies and immunotherapies, preserving their relevance within multimodal regimens. Constraints persist around toxicity management, resistance development, and infrastructure intensity, which limit unrestricted scaling in resource-constrained settings. Reimbursement policies and formulary decisions strongly shape usage patterns, with public systems prioritizing cost-effective cytotoxic options where access to newer therapies remains selective. Manufacturing scale, supply reliability, and hazardous drug handling compliance remain critical operational factors. Overall growth remains anchored in protocol continuity, healthcare access expansion, and sustained reliance on established chemotherapy classes rather than rapid displacement by novel oncology treatments.
Cytotoxic Chemotherapy Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 24,628.0 million |
| Market Forecast Value (2036) | USD 39,358.0 million |
| Forecast CAGR 2026 to 2036 | 4.8% |
Why is the Demand for Cytotoxic Chemotherapy Growing?
Demand for cytotoxic chemotherapy is increasing because oncology treatment pathways continue to rely on cytotoxic agents to manage a broad range of malignant conditions where tumor cell proliferation must be controlled with systemic therapy. Clinicians specify cytotoxic regimens to reduce tumor burden, prevent metastatic progression, and support curative or palliative intent when surgical or targeted options are limited by tumor biology or stage at diagnosis. Healthcare systems invest in infusion infrastructure, pharmacy preparation capabilities, and supportive care protocols to administer these agents with attention to safety and dosing accuracy. Procurement teams evaluate product stability, shelf life, and compatibility with standard infusion platforms to ensure supply continuity across hospitals, cancer centers, and outpatient oncology clinics.
Regulatory frameworks for chemotherapy preparation and administration influence facility readiness and reinforce demand for cytotoxic formulations that align with documented safety standards. Growth in cancer incidence, linked to aging populations and ongoing screening efforts, reinforces use of cytotoxic chemotherapy because many tumor types respond to agents that disrupt cellular replication mechanisms. Oncologists integrate cytotoxic drugs within multimodal treatment schedules that balance efficacy and tolerability, and adherence to evidence-based protocols supports consistent prescribing. Research activity focused on optimizing dosing schedules and reducing adverse event profiles encourages continued clinical reliance on cytotoxic classes alongside emerging therapies. These clinical and operational requirements are contributing to sustained demand growth in the cytotoxic chemotherapy market.
How Are the Segments Classified in the Cytotoxic Chemotherapy Market?
Demand for cytotoxic chemotherapy is shaped by cancer incidence patterns, established treatment protocols, and inclusion within standard oncology regimens. Utilization remains anchored in first-line and combination therapies across solid tumors and hematological indications. Adoption reflects clinical guideline adherence, availability of generics, and hospital-based administration practices. Treatment selection prioritizes proven efficacy, dosing flexibility, and compatibility with multimodal oncology care. Segment classification reflects differentiation by drug class, therapeutic application, and end-user setting, each influencing prescribing behavior, procurement structures, and treatment delivery pathways.
Which Product Category Holds the Largest Share in Cytotoxic Chemotherapy?
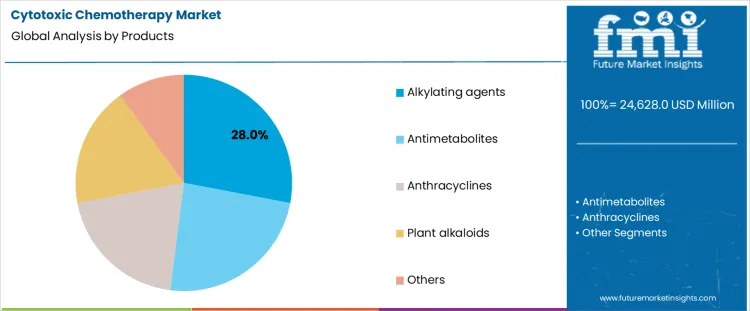
Alkylating agents account for 28.0%, representing the largest share among cytotoxic product categories. These agents demonstrate broad-spectrum activity across multiple cancer types and remain foundational in combination regimens. Mechanisms involving DNA cross-linking support effectiveness against rapidly dividing cells. Long-standing clinical familiarity sustains continued prescribing across treatment lines. Antimetabolites follow closely due to role in solid tumors and hematological malignancies. Anthracyclines maintain importance in breast and blood cancers with cardiotoxicity monitoring. Plant alkaloids support mitotic inhibition across selected indications. Product segmentation reflects reliance on established drug classes with wide therapeutic applicability and protocol integration.
Key Points
- Alkylating agents support broad-spectrum oncology regimens.
- Clinical familiarity sustains long-term utilization.
- Other classes address specific mechanisms and cancer profiles.
How Does Application Segmentation Influence Cytotoxic Chemotherapy Demand?
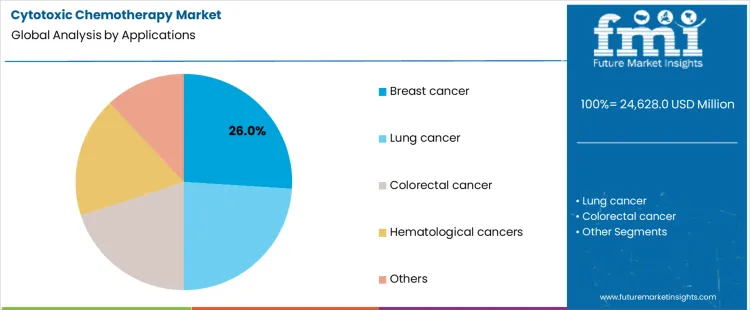
Breast cancer applications account for 26.0%, representing the largest share among therapeutic uses. High global prevalence and structured treatment pathways drive consistent chemotherapy utilization. Cytotoxic agents remain integral within adjuvant and neoadjuvant protocols. Lung cancer follows due to late-stage diagnosis patterns requiring systemic therapy. Colorectal cancer applications rely on combination regimens with defined chemotherapy backbones. Hematological cancers require intensive cytotoxic protocols for induction and consolidation phases. Application segmentation reflects alignment between disease burden, standardized regimens, and sustained reliance on cytotoxic therapies within oncology practice.
Key Points
- Breast cancer drives consistent chemotherapy utilization.
- Structured protocols sustain cytotoxic use across stages.
- Other cancers rely on combination-based systemic therapy.
What Role Does End User Distribution Play in Cytotoxic Chemotherapy Delivery?
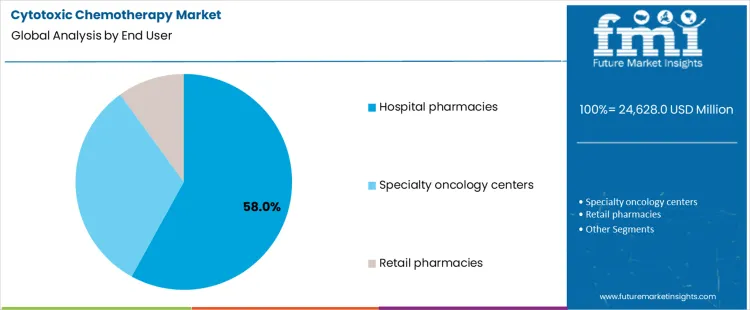
Hospital pharmacies account for 58.0%, representing the largest share among end-user settings. Inpatient and outpatient infusion services centralize chemotherapy preparation and dispensing. Handling requirements and safety controls favor hospital-based compounding. Oncology pharmacists support dose adjustment and protocol compliance. Specialty oncology centers follow with focused cancer treatment delivery and ambulatory infusion capacity. Retail pharmacies supply limited oral cytotoxic agents under controlled dispensing frameworks. End-user segmentation reflects concentration where infrastructure, safety requirements, and clinical oversight support complex chemotherapy administration and monitoring.
Key Points
-
Hospitals centralize chemotherapy preparation and delivery.
- Safety and compounding requirements favor controlled settings.
- Other channels support specialized or limited dispensing needs.
What are the Key Dynamics in the Cytotoxic Chemotherapy Market?
Demand for cytotoxic chemotherapy reflects foundational treatment requirements for solid tumors and hematologic malignancies across care settings. Adoption concentrates in hospital oncology departments, cancer centers, and infusion clinics. Global scope aligns with established treatment protocols and broad tumor applicability. Usage centers on alkylating agents, antimetabolites, anthracyclines, and taxanes administered as monotherapy or in combination regimens within curative and palliative intent pathways.
How do treatment breadth and protocol integration shape adoption?
Cytotoxic agents retain relevance due to activity across multiple cancer types and disease stages. Demand increases where rapid tumor burden reduction remains clinically necessary. Combination regimens integrate cytotoxics with targeted therapies, immunotherapies, and radiotherapy to enhance response rates. Neoadjuvant and adjuvant settings rely on cytotoxic backbones supported by long-term survival evidence. Dose intensity and scheduling flexibility allow tailoring to patient performance status and comorbidities. Generic availability supports broad access within public health systems. Pediatric oncology continues dependence on cytotoxic protocols with established cure rates. Adoption reflects reliance on predictable cytoreductive efficacy and integration within standardized oncology treatment algorithms.
How do toxicity burden, resistance, and cost pressures influence market scalability?
Cytotoxic chemotherapy causes dose-limiting toxicities affecting bone marrow, gastrointestinal tract, and cardiac function. Demand sensitivity rises where supportive care capacity limits safe administration. Acquired resistance reduces long-term effectiveness in recurrent disease. Narrow therapeutic windows require intensive monitoring and infrastructure. Patient quality-of-life concerns influence regimen selection and duration. Emergence of targeted and immune-based alternatives shifts use toward combination or later-line settings. Manufacturing complexity and sterile supply requirements affect availability. Price erosion from generics constrains innovation incentives. Scalability remains limited by toxicity management demands, resistance patterns, and evolving oncology treatment paradigms.
How Is Demand for Cytotoxic Chemotherapy Evolving Globally?
Demand for cytotoxic chemotherapy is expanding globally due to rising cancer incidence, broader treatment access, and continued reliance on established drug classes across solid and hematological malignancies. Public oncology programs and hospital formularies sustain usage where targeted and immunotherapies remain constrained by cost or indication scope. Treatment protocols continue to combine cytotoxic agents with newer modalities. Growth rates in India at 6.5%, China at 6.3%, Brazil at 6.0%, USA at 5.4%, and the UK at 4.3% indicate sustained expansion driven by patient volume growth, reimbursement coverage, and protocol continuity rather than innovation-led therapy substitution.
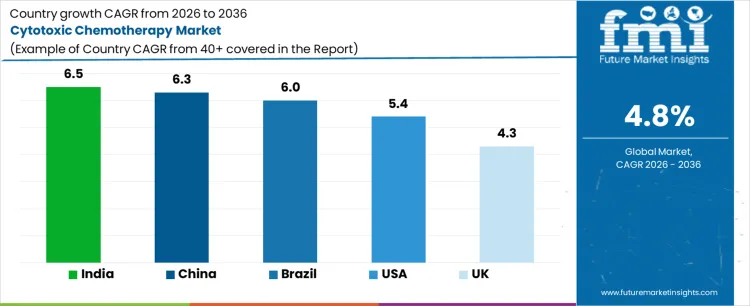
| Country | CAGR (%) |
|---|---|
| India | 6.5% |
| China | 6.3% |
| Brazil | 6.0% |
| USA | 5.4% |
| UK | 4.3% |
What Is Driving Expansion of Cytotoxic Chemotherapy Demand in India?
Demand for cytotoxic chemotherapy in India is growing at a CAGR of 6.5%, supported by rising cancer diagnosis rates and expanding access to oncology treatment. Public hospitals and regional cancer centers rely on cytotoxic drugs as first-line therapy due to affordability and inclusion in national treatment guidelines. Government-funded insurance schemes improve patient coverage for essential chemotherapy regimens. High prevalence of late-stage diagnosis sustains multi-cycle cytotoxic usage. Domestic generic manufacturing ensures stable supply and price control. Growth reflects access expansion and treatment volume increase rather than rapid transition toward premium oncology therapies.
- Rising cancer diagnosis and treatment volumes
- Public oncology center reliance on cytotoxic regimens
- Government insurance coverage expansion
- Strong domestic generic drug supply
Why Is China Sustaining Growth in Cytotoxic Chemotherapy Utilization?
Cytotoxic chemotherapy demand in China is expanding at a CAGR of 6.3%, driven by increasing cancer burden and standardized treatment pathways. Public hospital networks continue to prescribe cytotoxic agents across multiple tumor types. Inclusion of essential chemotherapy drugs in reimbursement lists supports sustained utilization. Combination regimens pairing cytotoxic drugs with targeted therapies remain common. Expansion of oncology capacity in secondary cities increases treatment access. Growth reflects system-wide treatment scale-up and protocol adherence rather than displacement by novel therapies.
- Expanding national cancer treatment capacity
- Reimbursement support for essential chemotherapy drugs
- Continued use within combination treatment protocols
- Oncology service expansion beyond tier-one cities
How Is Brazil Supporting Growth in Cytotoxic Chemotherapy Demand?
Demand for cytotoxic chemotherapy in Brazil is growing at a CAGR of 6.0%, supported by public healthcare oncology services and increasing diagnosis rates. Public hospitals depend on cytotoxic agents as cost-effective backbone therapies. National treatment guidelines prioritize established chemotherapy regimens for common cancers. Limited access to advanced biologics sustains cytotoxic usage. Regional oncology center expansion improves patient reach. Growth reflects public healthcare delivery scale and affordability considerations rather than rapid adoption of high-cost oncology innovations.
- Public healthcare reliance on cost-effective chemotherapy
- National guideline support for cytotoxic regimens
- Limited biologic therapy penetration
- Expansion of regional oncology centers
What Factors Are Influencing Cytotoxic Chemotherapy Demand in United States?
Cytotoxic chemotherapy demand in the United States is expanding at a CAGR of 5.4%, supported by large treated patient populations and protocol integration. Despite growth of targeted therapies, cytotoxic agents remain central within combination regimens. Broad insurance coverage sustains access across care settings. High incidence of solid tumors maintains consistent utilization volumes. Hospital and outpatient oncology centers ensure steady administration capacity. Growth reflects treatment continuity and patient volume stability rather than increasing reliance on cytotoxic monotherapy.
- Continued role within combination oncology regimens
- Broad insurance coverage across care settings
- High incidence of solid tumor cancers
- Extensive hospital and outpatient oncology infrastructure
Why Is the UK Experiencing Moderate Growth in Cytotoxic Chemotherapy Use?
Demand for cytotoxic chemotherapy in United Kingdom is growing at a CAGR of 4.3%, driven by NHS treatment protocols and rising cancer prevalence. National clinical guidelines continue to include cytotoxic agents for multiple indications. Cost-effectiveness assessments favor established chemotherapy drugs within public procurement. Access to newer therapies remains selectively controlled. Aging population demographics increase treatment volumes. Growth remains moderate, reflecting structured healthcare delivery and controlled adoption of alternative oncology treatments.
- NHS guideline inclusion of cytotoxic therapies
- Cost-effectiveness prioritization in public procurement
- Controlled access to novel oncology drugs
- Aging population increasing treatment demand
What is the competitive landscape of demand for the cytotoxic chemotherapy market globally?
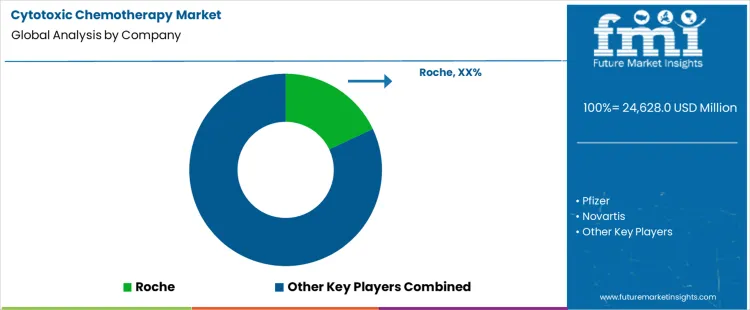
Demand for cytotoxic chemotherapy is driven by sustained cancer incidence, standardized treatment protocols, and broad applicability across tumor types. Buyers evaluate clinical efficacy across indications, safety profiles, dosing flexibility, formulation stability, and compatibility with combination regimens. Hospital procurement teams assess supplier reliability, regulatory approvals, manufacturing scale, and continuity of essential drug supply. Purchasing behavior emphasizes cost containment, tender participation strength, generic availability, and inclusion within national reimbursement frameworks. Trend in the cytotoxic chemotherapy market reflects stable utilization in solid tumors and hematologic cancers, alongside gradual pressure from targeted therapies and biosimilars influencing treatment sequencing and procurement economics.
Roche leads competitive positioning through a strong oncology portfolio integrating cytotoxic agents within combination treatment strategies. Pfizer maintains relevance via established chemotherapeutic products supported by global manufacturing and hospital access. Novartis competes through oncology specialization and integration of cytotoxic therapies within broader cancer care offerings. Bristol Myers Squibb participates by leveraging oncology expertise and long-standing presence in hospital-administered cancer treatments. Teva supports demand through large-scale generic cytotoxic drug production, enabling cost-sensitive access across global healthcare systems. Competitive differentiation centers on supply reliability, regulatory breadth, portfolio depth, and alignment with institutional oncology treatment pathways.
Key Players in the Cytotoxic Chemotherapy Market
- Roche
- Pfizer
- Novartis
- Bristol Myers Squibb
- Teva
- Others
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Products | Alkylating agents; Antimetabolites; Anthracyclines; Plant alkaloids; Others |
| Applications | Breast cancer; Lung cancer; Colorectal cancer; Hematological cancers; Others |
| End User | Hospital pharmacies; Specialty oncology centers; Retail pharmacies |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, UK, and 40+ countries |
| Key Companies Profiled | Roche; Pfizer; Novartis; Bristol Myers Squibb; Teva; Others |
| Additional Attributes | Dollar sales by product class, application, end user, and sales channel; regimen intensity, dosing cycles, and combination use across solid and hematological cancers; patent status and generic penetration shaping pricing; cold-chain and hazardous drug handling requirements; formulary access via hospital pharmacies and oncology centers; procurement patterns aligned with treatment protocols and reimbursement frameworks. |
Cytotoxic Chemotherapy Market by Segment
Products:
- Alkylating agents
- Antimetabolites
- Anthracyclines
- Plant alkaloids
- Others
Applications:
- Breast cancer
- Lung cancer
- Colorectal cancer
- Hematological cancers
- Others
End User:
- Hospital pharmacies
- Specialty oncology centers
- Retail pharmacies
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2023). WHO model list of essential medicines: 23rd list (includes cytotoxic anticancer medicines). World Health Organization.
- International Agency for Research on Cancer. (2024). Global cancer burden 2024: Cancer incidence, mortality, and prevalence worldwide. World Health Organization.
- American Society of Clinical Oncology. (2023). ASCO guideline update on systemic anticancer therapy safety, dosing, and supportive care. ASCO.
- National Cancer Institute. (2024). Chemotherapy and other systemic therapies for cancer treatment (NCI Cancer Treatment Fact Sheets). USA National Institutes of Health.
- European Medicines Agency. (2023). Guideline on the clinical evaluation of anticancer medicinal products (EMA/CHMP updated guidance). EMA.
Frequently Asked Questions
How big is the cytotoxic chemotherapy market in 2026?
The global cytotoxic chemotherapy market is estimated to be valued at USD 24,628.0 million in 2026.
What will be the size of cytotoxic chemotherapy market in 2036?
The market size for the cytotoxic chemotherapy market is projected to reach USD 39,358.8 million by 2036.
How much will be the cytotoxic chemotherapy market growth between 2026 and 2036?
The cytotoxic chemotherapy market is expected to grow at a 4.8% CAGR between 2026 and 2036.
What are the key product types in the cytotoxic chemotherapy market?
The key product types in cytotoxic chemotherapy market are alkylating agents, antimetabolites, anthracyclines, plant alkaloids and others.
Which applications segment to contribute significant share in the cytotoxic chemotherapy market in 2026?
In terms of applications, breast cancer segment to command 26.0% share in the cytotoxic chemotherapy market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Products
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Products , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Products , 2026 to 2036
- Alkylating agents
- Antimetabolites
- Anthracyclines
- Plant alkaloids
- Others
- Alkylating agents
- Y to o to Y Growth Trend Analysis By Products , 2021 to 2025
- Absolute $ Opportunity Analysis By Products , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Applications, 2026 to 2036
- Breast cancer
- Lung cancer
- Colorectal cancer
- Hematological cancers
- Others
- Breast cancer
- Y to o to Y Growth Trend Analysis By Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital pharmacies
- Specialty oncology centers
- Retail pharmacies
- Hospital pharmacies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Products
- By Applications
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Products
- By Applications
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Products
- By Applications
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Products
- By Applications
- By End User
- Competition Analysis
- Competition Deep Dive
- Roche
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Pfizer
- Novartis
- Bristol Myers Squibb
- Teva
- Others
- Roche
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Products , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Products
- Figure 6: Global Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by Applications
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by Products
- Figure 26: North America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 28: North America Market Attractiveness Analysis by Applications
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 35: Latin America Market Attractiveness Analysis by Products
- Figure 36: Latin America Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 38: Latin America Market Attractiveness Analysis by Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 45: Western Europe Market Attractiveness Analysis by Products
- Figure 46: Western Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 48: Western Europe Market Attractiveness Analysis by Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Products
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 65: East Asia Market Attractiveness Analysis by Products
- Figure 66: East Asia Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 68: East Asia Market Attractiveness Analysis by Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Products
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Products , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Products , 2026 to 2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Products
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Applications, 2026 to 2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

