Demand for chemotherapy induced myelosuppression treatment in Japan is valued at USD 429.1 million in 2025 and is projected to reach USD 624.5 million by 2035 at a CAGR of 3.8%. Neutropenia represents the largest treated indication, followed by anemia and thrombocytopenia across solid tumor and hematological cancer regimens. Growth factors dominate drug class utilization, while erythropoietin stimulating agents, thrombopoietic agents, and iron supplements support adjunct management. Oral therapies account for the larger treatment share, supported by injectable protocols in hospital oncology units. Demand is concentrated across Kanto, Kansai, and Chubu where high-volume cancer centers, university hospitals, and regional oncology networks operate at scale.
From 2030, demand expansion reflects steady chemotherapy procedure volumes, longer treatment durations, and broader protocol adoption in aging patient groups. Hospital pharmacies remain the primary distribution channel, while online pharmacies and drug stores record rising prescription fulfillment for maintenance therapies. Pricing stability is shaped by national reimbursement frameworks and biosimilar penetration in supportive oncology care. Key companies active in the competitive landscape include Novartis, Amgen, Pfizer, Teva Pharmaceutical Industries, and Myelo Therapeutics. Competitive strategies focus on biosimilar portfolio expansion, dosing optimization, and clinical evidence generation supporting reduced infection risk and hospitalization burden during multi-cycle chemotherapy programs.
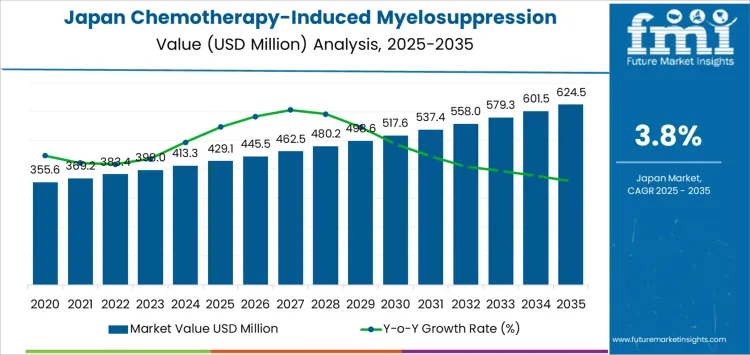
Demand for chemotherapy induced myelosuppression treatment in Japan reaches USD 429.1 million in 2025 and climbs to USD 498.6 million by 2030, generating USD 69.5 million in incremental value during this phase. The rise from USD 355.6 million in 2020 reflects the steady expansion of cancer treatment volumes linked to Japan aging population and the increasing use of intensive multi-cycle chemotherapy protocols. Growth is closely tied to higher utilization of granulocyte colony-stimulating factors and supportive biologics across solid tumors and hematological malignancies. Oncology centers are extending prophylactic usage earlier in treatment cycles to stabilize dose intensity and reduce hospitalization risk. The resulting demand pattern is driven by protocol standardization and improved adherence to febrile neutropenia prevention guidelines.
From 2030 to 2035, demand advances from USD 498.6 million to USD 624.5 million, adding a larger USD 125.9 million in new value. Annual growth gains strength as patient volumes continue to rise and treatment intensification expands into later-line cancer care. Longer survival in breast, lung, and colorectal cancers leads to repeated chemotherapy exposure, sustaining consistent need for myelosuppression control. Biosimilar penetration improves access while maintaining high treatment frequency across regional oncology networks. Demand is further supported by outpatient chemotherapy expansion, which shifts more responsibility to proactive blood count stabilization. By 2035, chemotherapy induced myelosuppression treatment demand in Japan reflects structurally higher cancer therapy intensity rather than episodic oncology cycle fluctuations.
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 429.1 million |
| Forecast Value (2035) | USD 624.5 million |
| Forecast CAGR (2025–2035) | 3.8% |
The demand for chemotherapy induced myelosuppression treatment in Japan is shaped by the structure of oncology care, an aging cancer population, and the clinical priority placed on maintaining chemotherapy intensity. Historically, treatment interruptions due to neutropenia, anemia, and thrombocytopenia were managed conservatively through dose reductions and inpatient monitoring. As cancer care shifted toward outpatient chemotherapy and protocol driven dosing, supportive treatments to stabilize blood counts became central to keeping patients on schedule. Japan strong adoption of guideline based oncology care reinforced early use of colony stimulating factors and related supportive therapies. The rise in combination chemotherapy regimens and longer treatment durations further increased reliance on myelosuppression management to prevent infection risk, hospital admissions, and unplanned treatment delays.
Future demand for chemotherapy induced myelosuppression treatment in Japan will be shaped by cancer care decentralization, increased use of aggressive multi drug protocols, and pressure to reduce inpatient burden. As more elderly patients receive active chemotherapy, vulnerability to bone marrow suppression will rise due to frailty and comorbidities. Expansion of home based supportive care and ambulatory oncology centers will also increase structured use of blood count stabilizing therapies outside hospital wards. Barriers include cautious prescribing due to thrombotic risk concerns, cost containment pressure within the national insurance system, and protocol driven restrictions on prophylactic use. Long term demand will depend on how effectively Japan balances chemotherapy intensity, infection prevention, and cost control within its evolving outpatient focused cancer treatment framework.
The demand for chemotherapy induced myelosuppression treatment in Japan is shaped by national cancer incidence, aging population structure, and standardized inpatient oncology care pathways. Neutropenia leads by indication due to its high frequency during cytotoxic chemotherapy and the need for infection risk control. Growth factors dominate by drug class because they directly stimulate white blood cell recovery and shorten hospitalization duration. Procurement is driven by hospital oncology departments operating under national insurance reimbursement schedules. Import reliance remains significant for biologic drug inputs. Substitution pressure between supportive care options remains limited by protocol driven prescribing. Demand stability is reinforced by consistent chemotherapy utilization and centralized cancer treatment infrastructure.
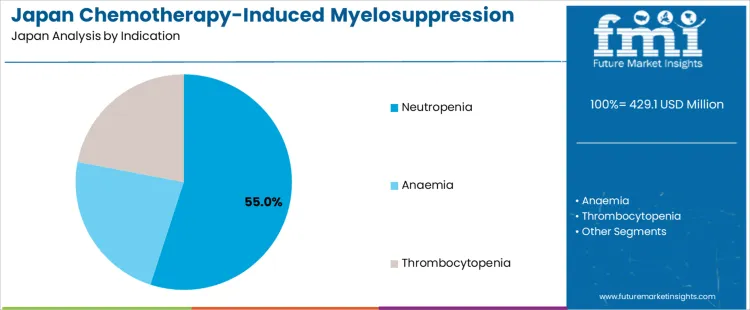
Neutropenia accounts for 55% of the demand for chemotherapy induced myelosuppression treatment in Japan by indication, reflecting its high incidence across solid tumor and hematologic cancer regimens. Consumption intensity is driven by infection risk management during dose dense chemotherapy schedules. Usage remains stable because neutrophil suppression occurs predictably across multiple drug combinations. Procurement is dominated by acute care hospitals and cancer specialty centers operating inpatient infusion units. Price sensitivity remains moderate because infection prevention directly influences hospitalization cost and patient survival. Specification control emphasizes timing accuracy, blood count monitoring integration, and predictable neutrophil recovery profiles to minimize febrile complications during treatment cycles.
Neutropenia management also generates consistent repeat demand through cyclical chemotherapy protocols that require supportive therapy with each treatment round. Repeat utilization remains predictable because patients often complete multiple cycles under identical regimens. Providers favor standardized dosing algorithms to reduce treatment variability and infection rates. Margin structure remains controlled under national reimbursement frameworks. Regulatory exposure remains elevated due to adverse event monitoring and blood disorder reporting obligations. Import reliance persists for biologic active substances. Substitution pressure from dose reduction remains limited because maintaining chemotherapy intensity remains central to treatment outcomes across major tumor types nationwide.
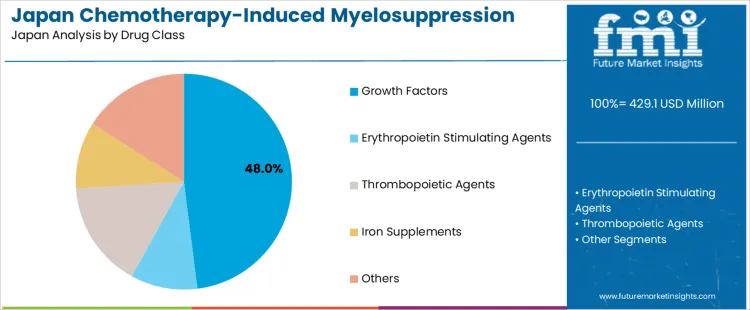
Growth factors represent 48.0% of the demand for chemotherapy induced myelosuppression treatment in Japan by drug class, reflecting their frontline role in stimulating white blood cell regeneration. Consumption intensity is driven by prophylactic and therapeutic use during moderate to high risk chemotherapy regimens. Usage remains stable because national oncology guidelines favor growth factor administration to reduce febrile neutropenia rates. Procurement is led by hospital pharmacies operating under diagnosis linked treatment groups. Price sensitivity remains moderate because growth factor use reduces downstream infection management cost. Specification control emphasizes bioactivity consistency, dose response reliability, and predictable bone marrow stimulation timing across patient populations.
Growth factors also generate strong recurring demand because many treatment protocols require repeat administration with each chemotherapy cycle. Repeat utilization remains high across both solid tumor and blood cancer care pathways. Providers favor long acting formulations to reduce injection burden and inpatient nursing time. Margin structure remains controlled under reimbursement ceilings and biosimilar competition. Regulatory exposure remains elevated because of biologic safety surveillance and immunogenicity monitoring. Import reliance persists for recombinant protein manufacturing inputs. Substitution pressure from dose adjusted chemotherapy exists but remains limited because growth factor support allows maintenance of planned treatment intensity.
Demand for chemotherapy-induced myelosuppression treatment in Japan is driven by high chemotherapy utilization across gastric, lung, colorectal, and breast cancers, combined with strict infection prevention protocols. Hospitals prioritize early neutrophil recovery to avoid treatment delays and inpatient complications. The aging cancer population increases vulnerability to bone marrow suppression and febrile neutropenia. Dose-dense chemotherapy regimens further amplify risk exposure. Demand is therefore anchored in treatment continuity, complication avoidance, and inpatient resource protection rather than rescue-driven critical care alone.
Japan oncology practice increasingly adopts dose-dense and combination chemotherapy regimens to improve survival outcomes. These protocols elevate the risk of severe neutropenia and thrombocytopenia, making preventive myelosuppression control a routine requirement. Prophylactic intervention reduces unscheduled hospitalizations and antibiotic dependency. Oncology departments integrate preventive treatment planning into cycle scheduling rather than waiting for cytopenia to develop. This proactive clinical strategy structurally strengthens demand through planned, protocol-led administration rather than reactive case management.
Japan cancer demographic is heavily skewed toward elderly patients, who face higher mortality from neutropenic infections. Even transient white cell suppression can lead to prolonged hospitalization and treatment discontinuation. Physicians therefore place strong emphasis on maintaining immune resilience throughout chemotherapy. Elderly patients are closely monitored with rapid intervention thresholds. This risk-avoidance framework elevates myelosuppression control as a core safety requirement in routine oncology practice rather than a secondary supportive therapy.
Demand for myelosuppression treatment in Japan is restrained by strict reimbursement controls and pressure to shorten hospital stays. Repeated injections create outpatient compliance challenges, especially for frail patients. Some mid-risk regimens still rely on close monitoring instead of universal prophylaxis to control cost. Prescription thresholds remain tightly protocol-bound. These structural and administrative limits prevent blanket use despite strong clinical emphasis on complication prevention.
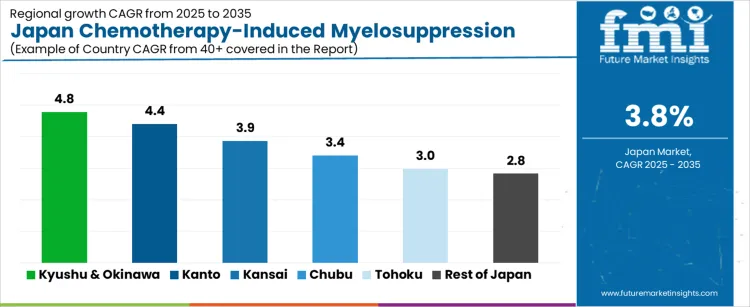
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 4.8% |
| Kanto | 4.4% |
| Kansai | 3.9% |
| Chubu | 3.4% |
| Tohoku | 3.0% |
| Rest of Japan | 2.8% |
The demand for chemotherapy induced myelosuppression treatment in Japan is rising steadily across oncology care networks, led by Kyushu and Okinawa at a 4.8% CAGR. Growth in this region is supported by expanding cancer treatment capacity, higher chemotherapy utilization in regional hospitals, and wider access to colony stimulating factors and supportive injectables. Kanto follows at 4.4%, driven by dense concentrations of tertiary oncology centers, advanced treatment protocols, and higher patient throughput. Kansai records 3.9% growth, reflecting stable use across university hospitals and cancer specialty institutes. Chubu at 3.4% shows moderate uptake linked to regional cancer care programs. Tohoku and the Rest of Japan, at 3.0% and 2.8%, reflect slower growth shaped by lower treatment volumes and longer therapy adoption cycles.
Demand for chemotherapy induced myelosuppression treatment in Kyushu and Okinawa is advancing at a CAGR of 4.8% through 2035, supported by rising regional oncology caseloads, expanding hospital chemotherapy capacity, and improving access to supportive care medicines. Cancer centers in Fukuoka and Okinawa are increasing use of colony stimulating factors and transfusion support for solid tumor and hematologic cancer patients. This region contrasts with Kanto through stronger dependence on public hospital delivery networks rather than private cancer clinics. Demand reflects steady chemotherapy volume growth and broader adoption of prophylactic supportive therapies.
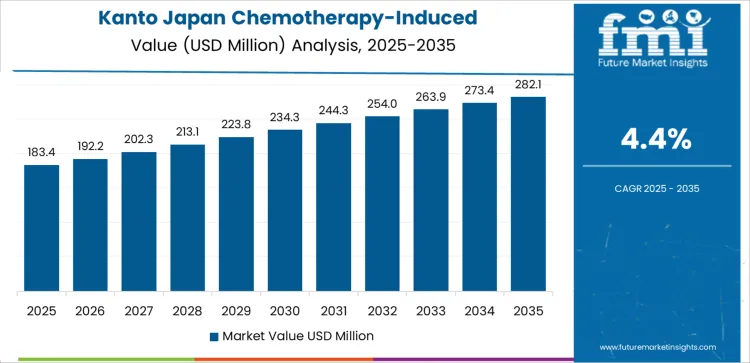
Demand for chemotherapy induced myelosuppression treatment in Kanto is advancing at a CAGR of 4.4% through 2035, supported by dense concentration of cancer hospitals, strong private oncology clinics, and high annual chemotherapy cycles in Tokyo and nearby prefectures. Kanto leads in early adoption of long acting supportive care biologics and hospital protocol driven prophylaxis. This region differs from Kyushu and Okinawa through stronger participation in clinical guideline driven care and specialist managed oncology pathways. Demand reflects rising outpatient chemotherapy use and improved survival driving longer supportive treatment duration.
Demand for chemotherapy induced myelosuppression treatment in Kansai is advancing at a CAGR of 3.9% through 2035, supported by stable cancer incidence, strong university hospital networks, and integrated regional oncology care in Osaka and Kyoto. Kansai shows balanced use of growth factors, blood products, and infection prevention therapies. This region contrasts with Kanto through steadier growth without strong private cancer center expansion. Demand is sustained by multidisciplinary cancer care models and consistent inpatient chemotherapy utilization across teaching hospitals.
Demand for chemotherapy induced myelosuppression treatment in Chubu is progressing at a CAGR of 3.4% through 2035, supported by industrial workforce driven healthcare access, steady oncology referrals, and gradual expansion of cancer treatment facilities in Aichi. Chubu differs from Kansai through stronger linkage to general hospitals rather than specialized cancer institutes. Demand remains focused on essential supportive therapies for solid tumor chemotherapy regimens, with moderate uptake of higher cost long acting biologics.
Demand for chemotherapy induced myelosuppression treatment in Tohoku is advancing at a CAGR of 3.0% through 2035, supported by aging population cancer incidence, limited oncology infrastructure expansion, and stable hospital based chemotherapy services. Miyagi serves as the main regional treatment hub. Tohoku contrasts with Kanto through lower patient turnover and fewer private oncology facilities. Supportive care use remains centered on short acting growth factors and hospital based blood support for inpatient chemotherapy patients.
Demand for chemotherapy induced myelosuppression treatment in Rest of Japan is advancing at a CAGR of 2.8% through 2035, supported by small city hospital networks, stable regional cancer treatment access, and gradual adoption of standardized chemotherapy supportive care protocols. These regions differ from Kanto through limited access to advanced oncology biologics and lower chemotherapy cycle density. Demand remains focused on essential supportive medicines supplied through public hospitals and regional cancer treatment centers. Growth is stable and guided by demographic driven oncology care needs rather than technology led expansion.
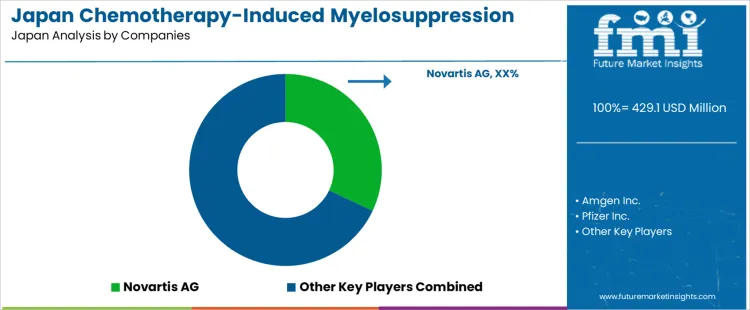
The demand for chemotherapy induced myelosuppression treatment in Japan is shaped by an aging cancer population, high chemotherapy utilization, and strict inpatient safety standards for infection and anemia management. Chugai Pharmaceutical holds a central domestic role through oncology supportive care products supplied across major cancer centers and regional hospitals. Kyowa Kirin supports demand through hematology focused therapies used in neutropenia and bone marrow recovery protocols. Takeda maintains a strong presence through oncology and supportive care integration across hospital formularies. Novartis participates through growth factor related therapies aligned with chemotherapy support protocols. Amgen supports demand through long established biologics used in neutropenia management under standardized dosing schedules.
Pfizer participates through injectable supportive care products supplied to hospital oncology pharmacies. Teva Pharmaceutical Industries supports access through generic oncology adjuncts used in cost controlled hospital settings. Myelo Therapeutics GmbH remains limited to research and early clinical exposure rather than routine hospital use. Prescribing in Japan is governed by Japanese Society of Medical Oncology guidelines, inpatient reimbursement rules, and PMDA labeling. Buyer preference favors suppliers with uninterrupted cold chain supply, stable lot quality, and domestic medical affairs coverage. Demand visibility tracks cancer incidence trends, chemotherapy protocol intensity, expansion of outpatient infusion centers, and continued government support for advanced oncology treatment infrastructure across prefectural hospital networks.
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Indication | Neutropenia, Anaemia, Thrombocytopenia |
| Drug Class | Growth Factors, Erythropoietin Stimulating Agents, Thrombopoietic Agents, Iron Supplements, Others |
| Route of Administration | Oral Chemotherapy-Induced Myelosuppression Treatment, Injectable Chemotherapy-Induced Myelosuppression Treatment |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Drug Stores |
| Region | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Novartis AG, Amgen Inc., Pfizer Inc., Teva Pharmaceutical Industries Ltd., Myelo Therapeutics GmbH |
| Additional Attributes | Dollar by sales by indication, drug class, and route; Regional CAGR and growth drivers; Hospital and outpatient oncology distribution; Biosimilar penetration and reimbursement impact; Prophylactic vs therapeutic usage; Recurrent demand across chemotherapy cycles; Specialty hospital vs general hospital adoption; Dose density and protocol standardization impact; Aging population influence; Regulatory compliance under PMDA; Cold chain and supply reliability; Margin and price sensitivity under national insurance coverage |
How big is the demand for chemotherapy induced myelosuppression treatment in Japan in 2025?
The demand for chemotherapy induced myelosuppression treatment in Japan is estimated to be valued at USD 429.1 million in 2025.
What will be the size of chemotherapy induced myelosuppression treatment in Japan in 2035?
The market size for the chemotherapy induced myelosuppression treatment in Japan is projected to reach USD 624.5 million by 2035.
How much will be the demand for chemotherapy induced myelosuppression treatment in Japan growth between 2025 and 2035?
The demand for chemotherapy induced myelosuppression treatment in Japan is expected to grow at a 3.8% CAGR between 2025 and 2035.
What are the key product types in the chemotherapy induced myelosuppression treatment in Japan?
The key product types in chemotherapy induced myelosuppression treatment in Japan are neutropenia, anaemia and thrombocytopenia.
Which drug class segment is expected to contribute significant share in the chemotherapy induced myelosuppression treatment in Japan in 2025?
In terms of drug class, growth factors segment is expected to command 48.0% share in the chemotherapy induced myelosuppression treatment in Japan in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.