The menopause induced insomnia treatment market starts 2026 at USD 3.4 billion and is expected to reach USD 7 billion by 2036 at a 6.2% CAGR. Adoption is influenced by physician preference, patient symptom severity, and formulation convenience rather than brand recognition. Clinicians choose therapies based on onset of action, safety profile, and hormone compatibility. Once a treatment is incorporated into standard care protocols, switching requires clinical justification and guideline review. Pharmacies and hospitals monitor stock levels, expiry dates, and batch traceability. Patient adherence programs and counseling services shape long-term uptake. Reimbursement policies affect formulary placement and availability.
Market expansion follows patient population growth, guideline adoption, and prescriber experience rather than promotional campaigns. Manufacturers focus on formulation stability, packaging integrity, and dose accuracy. Release requires potency checks, microbial testing, and documentation verification. Distribution networks stage stock near high-volume clinics and pharmacies to maintain uninterrupted access. Medical affairs teams support training, monitoring, and patient education. Profitability depends on predictable reorder cycles, low spoilage, and consistent clinical outcomes rather than aggressive pricing or frequent product reformulation. Program expansion aligns with awareness campaigns and new therapy
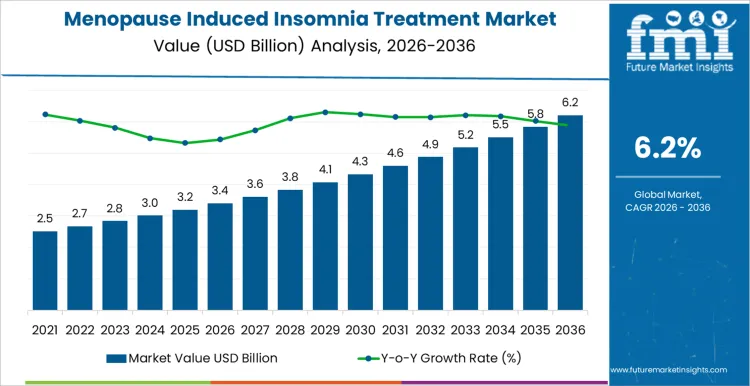
Treatment guideline adoption and patient awareness campaigns drive growth in the menopause induced insomnia treatment market more than overall sleep disorder prevalence. In 2026, at approximately USD 3.4 billion, demand is concentrated in women’s health clinics, primary care networks, and specialized sleep centers where therapy selection depends on hormonal profile, comorbidities, and patient preference. Uptake is influenced by physician familiarity with non-hormonal and cognitive behavioral therapies, insurance coverage, and integration into routine menopause management plans. Growth is supported by expanding patient education, earlier diagnosis of sleep disturbances, and inclusion of therapies in multi-modality care pathways rather than rapid replacement of existing medications.
Healthcare network planning and product differentiation shape the later phase of the menopause induced insomnia treatment market. As value approaches roughly USD 7 billion by 2036, stakeholders focus on therapy efficacy, safety, and adherence monitoring. Pharmaceutical and digital therapy providers that integrate clinical guidance, patient support tools, and follow-up mechanisms gain preference among prescribing physicians. The climb from mid three billion into the low seven billion reflects gradual adoption across clinics, expansion into telemedicine platforms, and increased awareness of targeted treatment options, rather than a single policy or clinical breakthrough. Companies offering reliable therapy access, educational support, and evidence-backed protocols are positioned to maintain long-term market presence.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 3.4 billion |
| Forecast Value (2036) | USD 6.2 billion |
| Forecast CAGR 2026 to 2036 | 6.2% |
Treatments for menopause-related insomnia target sleep disruption caused by hormonal changes, including declining estrogen and progesterone levels. Sleep-specific protocols are increasingly being written into broader menopause treatment plans managed in gynecology and primary care settings. Earlier approaches relied primarily on general sedative-hypnotics or over-the-counter sleep aids, which often produced inconsistent relief and undesirable side effects. This has increased trial use of sleep supplements among patients seeking non-prescription options before stepping up to long-term pharmacotherapy. Modern therapies include selective melatonin receptor agonists, non-benzodiazepine hypnotics, and hormone-based interventions tailored to midlife physiology. For milder symptoms, some care pathways still include melatonin supplements as an OTC option when next-day impairment risk needs to stay low. Clinicians assess options based on efficacy, safety profile, interaction with comorbid conditions, and suitability for long-term use. Adoption is concentrated in sleep clinics, gynecology practices, and primary care settings where individualized treatment planning is critical to improve sleep quality, daytime functioning, and overall quality of life. Therapy effectiveness also influences adherence and patient satisfaction.
Pharmaceutical and biotech developers differentiate products through receptor specificity, pharmacokinetic stability, and tolerability rather than cost alone. Earlier interventions offered limited duration or broad sedative effects, which restricted applicability in patients with chronic sleep disruption. Current programs emphasize formulations that maintain circadian rhythm regulation, minimize next-day residual effects, and integrate patient education for behavioral support. Providers evaluate total treatment value through improvements in sleep duration, reduction in nighttime awakenings, and minimal adverse events rather than per-dose cost. Supply partners add value via formulation reliability, patient guidance materials, and adherence monitoring solutions. Market growth is expected to track increasing awareness of menopause-related sleep disorders, expanded diagnostic screening, and demand for therapies that balance efficacy with safety in long-term midlife care. Expanded access to sleep studies supports differential diagnosis when insomnia overlaps with apnea, restless legs, or medication-related sleep disruption.
Treatment demand in the Menopause Induced Insomnia Market is influenced by symptom severity, patient safety considerations, and guideline adherence rather than short-term purchasing fluctuations. Healthcare providers prioritize therapies that improve sleep quality, minimize adverse effects, and integrate with menopause management programs. Segment adoption varies according to mechanism of action, regulatory status, and patient acceptability. Suppliers offering training on therapy selection, adherence support, and reliable product supply can secure long-term adoption. Market growth is concentrated in prescription therapies and OTC sleep aids, providing predictable demand for manufacturers that combine therapeutic efficacy, patient education, and consistent service delivery in menopausal care.
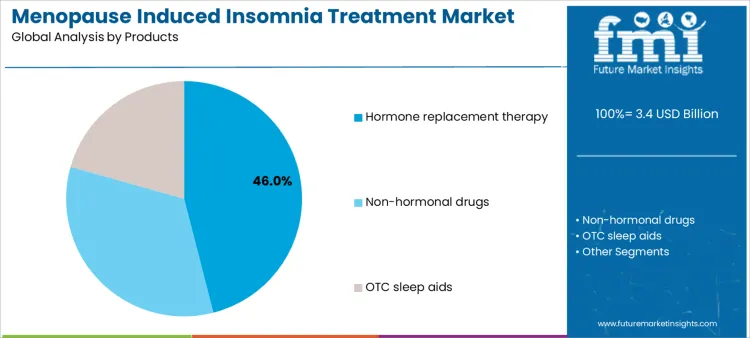
Hormone replacement therapies account for approximately 46% of demand due to their efficacy in managing sleep disturbances linked to hormonal imbalance. Formulary decisions for hormone replacement therapy increasingly weigh sleep outcomes alongside vasomotor symptom control in midlife care pathways. Suppliers supporting these therapies must ensure dosing guidance, adherence programs, and regulatory compliance support. Non-hormonal drugs provide alternative pharmacologic options for patients with contraindications, requiring education on therapy selection, monitoring, and adverse effect management. Some providers also review low dose hormone alternatives when risk profiles limit standard regimens but symptom relief still needs pharmacologic support. OTC sleep aids are widely used for self-management, creating supplier opportunities in packaging, labeling, and consumer guidance. In retail channels, menopause supplements are often positioned as non-prescription adjuncts for women who want symptom support alongside sleep hygiene changes. Product type determines supplier involvement in training, patient adherence support, and inventory management, influencing adoption across clinical and self-care settings.
Clinical workflow and patient management strategies vary by product type. Hormone replacement therapies require monitoring, personalized dosing, and integration with broader menopause care plans, creating recurring supplier touchpoints. Non-hormonal drugs emphasize safety monitoring, adherence tracking, and patient guidance, increasing the need for technical support. OTC sleep aids require consistent supply, clear labeling, and optional patient education, particularly in retail or pharmacy settings. Facilities and providers adopting specific therapies depend on suppliers for education, device or medication reliability, and consistent inventory. Companies providing comprehensive training, adherence guidance, and supply management gain a competitive advantage in the menopausal insomnia market.
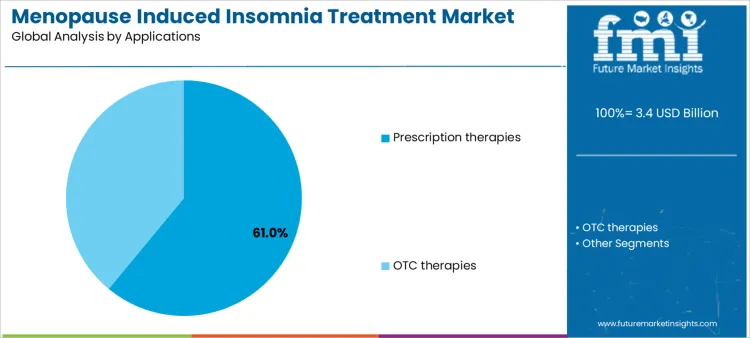
Prescription therapies account for approximately 61% of demand because patients with moderate-to-severe insomnia require clinician-guided interventions, generating high-volume, predictable adoption. OTC therapies serve patients seeking self-managed solutions or mild symptom relief, creating smaller but stable markets. Application-specific demand informs supplier inventory allocation, educational initiatives, and service offerings. Aligning product availability and support services with both prescription and OTC applications enables suppliers to maximize adoption, procedural efficiency, and revenue stability, while supporting diverse patient needs across healthcare and retail channels.
Consumer behavior and clinical workflow further shape supplier strategy. High-volume prescription use emphasizes consistent supply, staff training, and adherence monitoring programs to ensure therapeutic effectiveness. OTC applications require clear labeling, packaging reliability, and optional patient education to encourage proper use and repeat purchase. Segment distribution depends on symptom prevalence, treatment complexity, and patient preference rather than short-term procurement cycles. Suppliers offering training, product reliability, and adherence support can secure broader adoption and long-term engagement across prescription and self-care channels.
The Menopause Induced Insomnia Treatment Market is shaped by hospitals, clinics, and specialty healthcare providers managing sleep disturbances related to hormonal changes. Treatments are evaluated based on effectiveness, safety profile, ease of administration, and patient adherence. Selection decisions involve gynecologists, sleep specialists, and pharmacy procurement teams. Suppliers that provide therapies with consistent efficacy, clear dosing instructions, and minimal side effects are preferred. This positions insomnia treatments as essential components of patient care rather than optional interventions, emphasizing reliable outcomes, quality of life, and predictable therapeutic benefits.
Healthcare providers increasingly follow evidence-based care guidelines to ensure consistent patient outcomes and minimize adverse events. This drives demand in the Menopause Induced Insomnia Treatment Market toward therapies that integrate with standard treatment algorithms, provide predictable improvement, and reduce complications. Buyers prefer products supported by clinical data, clear administration protocols, and patient education materials. Suppliers are evaluated on product consistency, regulatory compliance, and technical support rather than minor formulation differences. Treatments aligned with guidelines are more likely to be adopted across multiple clinics and hospital systems.
Recurring demand comes from hospitals, outpatient clinics, and specialty women’s health centers managing ongoing therapy for patients experiencing menopause-related sleep disturbances. Each treatment cycle generates predictable repeat orders. The Menopause Induced Insomnia Treatment Market also benefits from multi-site hospital networks standardizing on approved therapies. Suppliers that provide patient adherence programs, dosing support, and consistent supply chains secure long-term engagement. Embedding treatments into standard care pathways ensures ongoing demand tied to patient volume and treatment duration.
Insomnia treatments for menopause carry cost and safety considerations, which can limit adoption in smaller clinics or low-volume healthcare centers. The market also faces constraints from regulatory approvals, formulary inclusion, and clinician familiarity. Physicians are cautious about switching therapies mid-treatment due to patient comfort and safety concerns. Suppliers entering the market must provide clinical guidance, patient education, and reliable product availability. Adoption is concentrated in high-volume women’s health centers and specialized clinics where efficacy, adherence, and patient outcomes are critical.
| Country | CAGR |
|---|---|
| USA | 8.3% |
| Brazil | 10.2% |
| China | 11.1% |
| India | 11.3% |
| Germany | 8.1% |
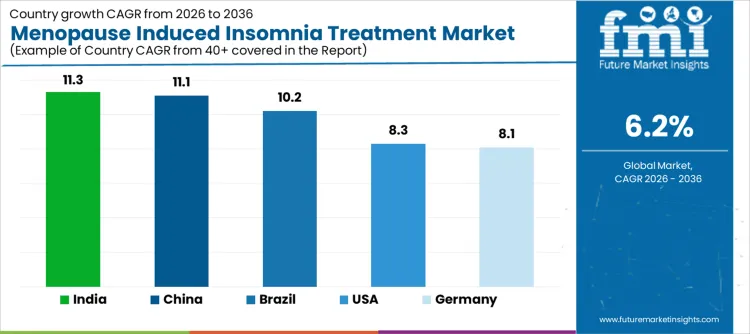
Demand for menopause induced insomnia treatment is rising as healthcare providers and clinics address sleep disturbances in aging female populations through pharmacological therapies and behavioral interventions. India leads with an 11.3% CAGR, supported by growing awareness of menopausal health issues, expansion of gynecology and sleep disorder clinics, and increased access to treatment options. China follows at 11.1%, driven by higher prevalence, improved diagnosis rates, and adoption of targeted therapies. Brazil records 10.2%, reflecting growing healthcare access and patient acceptance of treatment programs. The USA grows at 8.3%, shaped by established treatment protocols and routine management of menopausal insomnia. Germany, at 8.1%, reflects steady demand in a mature healthcare system with structured patient care pathways.
United States demand for menopause induced insomnia treatments is increasing along an 8.3% CAGR due to rising patient volumes, expanded gynecology services, and outpatient sleep wellness programs. Procurement authority rests with hospital pharmacy committees, gynecology department heads, and clinical protocol boards. Adoption emphasizes non-hormonal therapies, cognitive behavioral interventions, and pharmacologic options for post-menopausal sleep disturbances. Once selected, treatment protocols are integrated across multiple hospitals and wellness clinics. Clinical guideline adherence, insurance coverage, and patient monitoring influence adoption. Import products dominate high specification therapy options, while domestic suppliers provide standard treatments. Capital allocation follows program expansion and patient volume growth. Replacement cycles track therapy adherence and dosage schedules. Commercial success depends on inclusion in hospital formularies, wellness program protocols, and supplier support agreements.
Brazil is experiencing a 10.2% CAGR in menopause induced insomnia treatments due to expanding private hospital gynecology units, outpatient sleep clinics, and wellness programs. Procurement authority rests with hospital pharmacy boards, gynecology directors, and clinical committees. Adoption focuses on non-hormonal sleep therapies, cognitive behavioral programs, and pharmacologic interventions tailored for menopausal patients. Import products dominate high specification therapies, while domestic suppliers provide standard regimens. Once approved, treatment protocols are implemented across multiple affiliated hospitals and clinics. Physician and patient education programs, workflow integration, and adherence monitoring influence adoption. Capital allocation follows hospital expansion and outpatient program launches. Replacement cycles track therapy duration and patient adherence. Commercial success depends on inclusion in network formularies, reference site adoption, and multi-year supply agreements rather than isolated clinic purchases.
China is experiencing an 11.1% CAGR in menopause induced insomnia treatments due to expansion of hospital gynecology programs, outpatient wellness centers, and chronic care management initiatives. Major hospitals and clinical networks control procurement. Adoption emphasizes non-hormonal and pharmacologic therapies, patient monitoring, and integration with sleep wellness programs. Domestic suppliers cover standard therapies, while international brands retain positions in advanced treatment protocols. Procurement follows multi-year hospital planning and centralized tenders. Once validated, treatments are implemented across multiple hospitals and outpatient centers. Physician training, patient adherence programs, and protocol alignment influence utilization. Replacement cycles follow therapy schedules rather than product expiration. Capital allocation aligns with hospital program growth and new clinic openings. Commercial success depends on inclusion in provincial and national hospital formularies and network treatment protocols.
India is experiencing an 11.3% CAGR in menopause induced insomnia treatments due to expansion of hospital gynecology programs, specialty women’s health centers, and outpatient wellness clinics. Procurement authority resides with hospital pharmacy boards, gynecology committees, and program directors. Therapy selection emphasizes non-hormonal and pharmacologic sleep treatments tailored for menopausal patients. Import products dominate high specification regimens while domestic suppliers cover standard treatments. Once approved, protocols are implemented across multiple affiliated hospitals and clinics. Training programs, protocol adherence, and patient monitoring influence utilization. Replacement cycles track therapy schedules and patient adherence. Capital allocation aligns with hospital expansion and program launches. Commercial success depends on inclusion in network approved formularies, reference program adoption, and multi-year supply contracts rather than individual clinic sales.
Germany is experiencing an 8.1% CAGR in menopause induced insomnia treatments due to structured clinical guidelines, reimbursement frameworks, and standardized hospital gynecology protocols. University hospitals and regional wellness centers control most procurement. Adoption focuses on non-hormonal therapies, pharmacologic interventions, and patient adherence programs. Device and therapy evaluation emphasizes safety, efficacy, and integration into hospital protocols. Procurement follows health technology assessments and capital approval processes. Import suppliers dominate high specification treatments while domestic suppliers provide standard regimens. Once approved, therapies are deployed across multiple departments and hospitals. Training, patient education, and workflow alignment influence utilization. Replacement cycles track therapy duration. Capital allocation aligns with hospital program expansion. Commercial success depends on framework inclusion, reference site adoption, and integrated supplier support rather than isolated clinic purchases.
Treatment selection for menopause induced insomnia follows clinical guideline alignment, patient symptom profiling, and formulary review rather than simple product listing. Clinicians define severity, comorbidity considerations, dosing schedules, and safety monitoring before approving therapies. Pfizer, Bayer, Eisai, Merck, AbbVie compete during evaluation, clinical trial evidence review, and guideline integration stages. Once a therapy enters treatment pathways, prescribing protocols, patient education, and pharmacy stocking anchor adoption. Pfizer leverages sleep and CNS experience alongside safety data in menopausal populations. Bayer focuses on hormonal and non hormonal therapy integration. Eisai emphasizes receptor selective and novel mechanism approaches. Merck positions products with well characterized pharmacokinetics and tolerability. AbbVie integrates therapies with broader women’s health programs and support services.
Competitive outcomes reflect efficacy, safety, patient adherence, and support programs rather than pricing. Physicians assess sleep latency improvement, duration, adverse events, and quality of life outcomes during evaluation. Pharmacists review dosing complexity, storage requirements, and dispensing compatibility. Pfizer secures adoption through clinical data, physician familiarity, and patient support programs. Bayer gains share where hormonal integration and guideline recommendations align. Eisai competes in centers valuing novel mechanism therapy options. Merck holds positions in institutions emphasizing predictable tolerability and adherence. AbbVie benefits from programs linking insomnia treatment to wider menopausal symptom management. Market positions vary by clinical practice patterns, guideline adoption, patient demographics, and insurance coverage frameworks.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Hormone replacement therapy; Non-hormonal drugs; OTC sleep aids |
| Application | Prescription therapies; OTC therapies |
| End User / Sales Channel | Retail pharmacies; E-commerce; Hospitals |
| Region | Asia Pacific; Europe; North America; Latin America; Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Brazil, Chile, Saudi Arabia, Turkey, South Africa, Rest of regions |
| Key Companies Profiled | Pfizer; Bayer; Eisai; Merck; AbbVie |
| Additional Attributes | Dollar by sales by product and application, prescription versus OTC mix, HRT versus non-hormonal usage split, retail versus e-commerce channel trends, adherence and persistence patterns, regulatory and reimbursement impact, shelf life and inventory rotation, patient education influence, guideline adoption effects |
How big is the menopause induced insomnia treatment market in 2026?
The global menopause induced insomnia treatment market is estimated to be valued at USD 3.4 billion in 2026.
What will be the size of menopause induced insomnia treatment market in 2036?
The market size for the menopause induced insomnia treatment market is projected to reach USD 6.2 billion by 2036.
How much will be the menopause induced insomnia treatment market growth between 2026 and 2036?
The menopause induced insomnia treatment market is expected to grow at a 6.2% CAGR between 2026 and 2036.
What are the key product types in the menopause induced insomnia treatment market?
The key product types in menopause induced insomnia treatment market are hormone replacement therapy, non-hormonal drugs and otc sleep aids.
Which applications segment to contribute significant share in the menopause induced insomnia treatment market in 2026?
In terms of applications, prescription therapies segment to command 61.0% share in the menopause induced insomnia treatment market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.