The low-dose hormone alternatives market is set to rise from USD 3.2 billion in 2026 to USD 6.8 billion by 2036, firmly establishing a CAGR of 7.8%. Clinical use is shaped by the balance between symptom control and long term risk management rather than by treatment intensity alone. Prescribers consider cardiovascular, metabolic, and oncology related safety profiles when selecting therapies, which shifts attention toward regimens that aim to reduce cumulative exposure. Demand is strongest in health systems where guidelines emphasize step wise escalation and regular reassessment. Regional uptake varies with physician training, patient monitoring capacity, and the structure of follow up care.
Treatment pathways and reimbursement rules play a central role in determining adoption patterns. In many markets, these products enter formularies as second line or maintenance options rather than as initial therapy. Payers and hospital committees evaluate outcomes data, adherence rates, and discontinuation behavior alongside acquisition cost. Once protocols are set, switching tends to be slow because patient stability and monitoring schedules are part of care planning. The market grows through gradual shifts in prescribing philosophy and risk tolerance, not through rapid replacement of conventional hormone therapies across all patient groups.
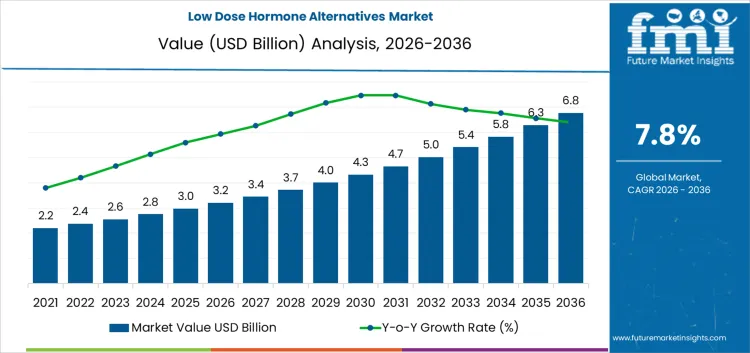
The low dose hormone alternatives market expands as both clinicians and patients seek symptom management options with lower systemic exposure and better long-term tolerability profiles. Around 2020, the market is still close to USD 1.9 billion and is mainly concentrated in early-stage menopause management, thyroid support adjacencies, and selected endocrine therapy support uses. The move to roughly USD 3.2 billion by 2026 reflects broader clinical comfort with lower-dose regimens, growing caution around long-term hormone exposure, and stronger patient demand for therapies positioned as gentler or risk-mitigated. At this point, the category is no longer limited to niche prescribing and begins to enter more routine treatment pathways, especially in primary care and outpatient settings.
After this inflection, growth is driven less by awareness and more by treatment volume and duration. The market reaches around USD 4.8 billion by 2032 as use expands across aging populations, longer treatment cycles, and wider acceptance in chronic symptom management rather than short-term interventions. By approximately USD 6.8 billion in 2036, low dose alternatives are embedded in a broad range of endocrine and women’s health protocols. Value expansion is supported by patient adherence, recurring therapy use, and incremental broadening of indications rather than by premium pricing. The trajectory is shaped mainly by clinical guidelines, risk-benefit perception, and long-term safety positioning rather than by breakthrough product launches.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 3.2 billion |
| Forecast Value (2036) | USD 6.8 billion |
| Forecast CAGR 2026 to 2036 | 7.8% |
Low-dose hormone alternatives are increasingly adopted to provide targeted hormonal therapy with minimized side effects for patients in menopause management, reproductive health, and endocrine-related conditions. Historically, conventional hormone therapies relied on higher dosages, which increased risks of adverse effects, including cardiovascular issues and hormone-sensitive tissue complications. Modern low-dose formulations incorporate precision dosing, bioidentical hormones, and novel delivery systems such as transdermal patches, gels, and sublingual tablets to ensure therapeutic efficacy while reducing systemic exposure.
Healthcare providers, clinics, and pharmaceutical manufacturers prioritize safety profiles, regulatory compliance, and patient adherence. Early adoption focused on specialized endocrine and gynecological care, while current demand spans primary care, wellness clinics, and women’s health programs, driven by patient preference for safer, personalized treatments. Dosage accuracy, delivery method, and hormone stability influence supplier selection.
Patient safety, treatment adherence, and therapeutic efficacy are shaping market growth. Compared with conventional hormone therapies, low-dose alternatives emphasize reduced side effects, precise hormonal control, and flexible administration routes. Cost structures depend on hormone sourcing, formulation complexity, and delivery system technology, concentrating margins among suppliers capable of delivering consistent, high-quality alternatives. Healthcare providers adopt these treatments to enhance patient comfort, mitigate risks, and support long-term health outcomes.
The Low Dose Hormone Alternatives Market in 2026 is segmented by therapy type and by application. By therapeutic approach, demand in the Low Dose Hormone Alternatives Market is divided into bioidentical hormone therapies, phytoestrogen and plant based alternatives, selective estrogen receptor modulators, and low dose hormone replacement formulations, each reflecting different risk profiles, regulatory positioning, and patient acceptance levels.
By application, demand in the Low Dose Hormone Alternatives Market is organized around menopausal symptom management, androgen deficiency therapies, osteoporosis and bone health, and sexual wellness or libido support, which differ in treatment duration, patient monitoring intensity, and sensitivity to side effect concerns. These segments show how treatment choices increasingly focus on dose minimization and long term tolerability.
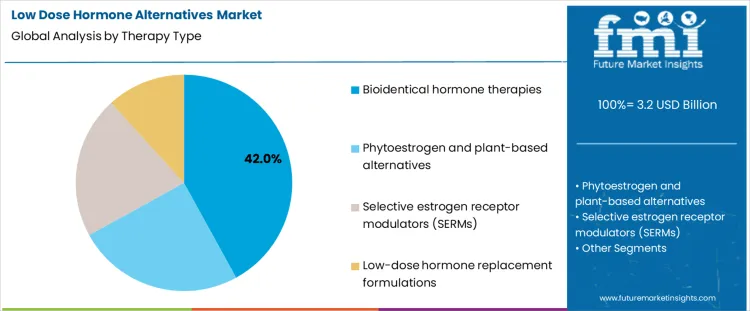
Bioidentical hormone therapies account for about 42% of demand in the Low Dose Hormone Alternatives Market in 2026 because they are widely perceived by clinicians and patients as closer to natural physiological hormones. This perception supports acceptance in long term treatment of menopausal symptoms and age related hormonal changes. In the Low Dose Hormone Alternatives Market, these therapies are often prescribed in customized or flexible dosing regimens, which fits the broader shift toward individualized treatment plans. Many patients who are cautious about conventional hormone replacement still accept bioidentical options as a compromise between efficacy and perceived safety. This combination of clinical familiarity, patient confidence, and adaptable dosing keeps bioidentical therapies as the leading therapy category.
Plant based alternatives, SERMs, and low dose conventional formulations in the Low Dose Hormone Alternatives Market serve more specific roles. Phytoestrogens appeal to patients seeking non pharmaceutical solutions but often deliver milder effects. SERMs are used in defined clinical indications such as bone health, yet they require closer monitoring. Low dose hormone replacement products follow established protocols but still face the same risk perception issues as standard HRT. These options are essential in targeted settings, but none matches the broad acceptance and prescribing flexibility of bioidentical therapies in the Low Dose Hormone Alternatives Market.
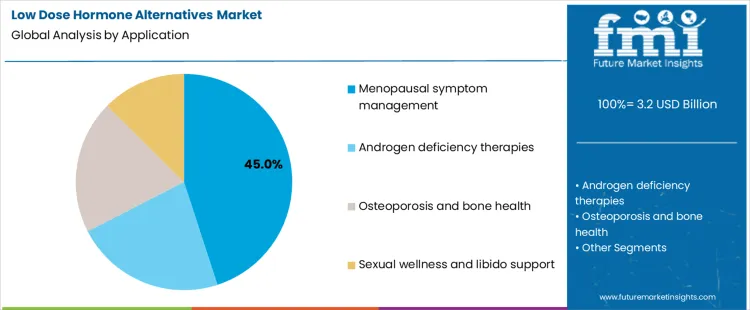
Menopausal symptom management represents about 45% of demand in the Low Dose Hormone Alternatives Market in 2026 because it involves a large and growing patient population seeking long term symptom relief. Hot flashes, sleep disturbance, mood changes, and bone density concerns often require multi year management rather than short courses of treatment. In the Low Dose Hormone Alternatives Market, many patients and clinicians aim to control these symptoms while minimizing cumulative hormone exposure, which supports demand for lower dose and alternative therapies. Regular follow up visits and therapy adjustments also sustain repeat prescribing.
Androgen deficiency, bone health, and sexual wellness applications in the Low Dose Hormone Alternatives Market follow different treatment patterns. Androgen deficiency therapies involve smaller patient groups and more tightly controlled protocols. Osteoporosis treatment is often combined with non-hormonal medications. Sexual wellness treatments are more episodic and discretionary. These segments contribute to overall growth, but none matches the scale, duration, and continuity of care associated with menopausal symptom management in the Low Dose Hormone Alternatives Market.
Clinical decision making around hormone therapy is becoming more conservative and more structured at the same time. Concern about cumulative exposure, long treatment horizons, and patient specific risk profiles is pushing prescribers to reconsider dose intensity as a design variable, not just a tuning parameter. At the same time, health systems are tightening formulary governance and standardizing treatment sequences. This combination creates space for lower dose and alternative regimens, but only where evidence and operational clarity exist. The category therefore grows through institutional acceptance and protocol inclusion, not through spontaneous patient demand, and remains paced by how quickly proof, guidance, and reimbursement frameworks evolve.
Why Is Risk Management Changing Prescribing Behavior?
In long term hormone management, avoidance of adverse effects often matters as much as symptom control. Many patients remain on therapy for years, which makes cumulative exposure a central concern in clinical planning. Physicians increasingly adjust starting doses downward, escalate more slowly, or prefer alternative delivery approaches when safety margins are narrow. This is reinforced by patient caution and by medico legal defensiveness in borderline cases. When a lower intensity regimen proves sufficient, it tends to become the preferred baseline. As a result, demand follows changes in prescribing philosophy and training rather than marketing pressure or short term shifts in patient preferences.
What Is Holding Back Faster Adoption?
Despite interest, proof remains uneven. Long horizon outcome data is limited for many alternative or lower dose strategies, and comparative studies against established regimens are often small or inconsistent. Payers and formulary committees therefore hesitate to endorse broad substitution. In routine practice, switching stable patients introduces clinical uncertainty without obvious operational benefit. Monitoring requirements and coding limitations also complicate use. These frictions slow translation from specialist experimentation to general practice. The category expands cautiously, case by case, instead of through rapid guideline driven shifts.
How Is Institutional Governance Reshaping This Market?
Treatment choices are increasingly being made at system level rather than by individual clinicians alone. Hospitals and payer networks now rely on defined step therapy sequences, preferred options, and escalation rules. For low dose alternatives, inclusion in these structures is more important than brand recognition. Education, documentation, and auditability matter as much as clinical nuance. Once a regimen is embedded in these pathways, usage becomes routine and repeatable. This turns growth into a governance problem rather than a promotional one, with success depending on evidence quality, committee acceptance, and fit within standardized care flows.
| Country | CAGR (%) |
|---|---|
| USA | 7.2 |
| UK | 6.8 |
| China | 8.5 |
| India | 9.0 |
| Brazil | 7.5 |
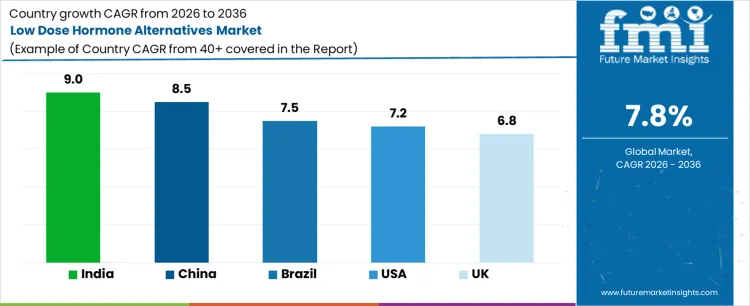
Demand for low dose hormone alternatives is rising as healthcare providers and patients adopt safer, targeted therapies for hormone-related conditions, aging, and metabolic health. India leads with a 9.0% CAGR, driven by rising awareness of hormone therapies, growing healthcare access, and adoption of alternative dosing strategies. China follows at 8.5%, supported by expanding healthcare infrastructure and use of low dose hormone formulations. Brazil records 7.5% growth, shaped by adoption in clinical and outpatient settings. The USA grows at 7.2%, influenced by replacement and therapeutic optimization. The UK shows 6.8% CAGR, reflecting steady adoption of low dose hormone alternatives in healthcare and wellness programs.
United States is seeing a CAGR of 7.2% as hospitals and outpatient clinics in states like California, Texas, and Florida increasingly integrate low dose hormone therapies into women’s health programs. Patients prefer therapies with reduced systemic impact, encouraging adoption in gynecology and endocrinology departments. Investments focus on advanced delivery systems, patient monitoring tools, and compliance with FDA safety standards. Urban medical centers and specialized clinics provide concentrated demand for modern hormone alternatives. Growth is driven by the aging population, heightened awareness of hormone therapy side effects, and the push for personalized treatment plans within well-established healthcare networks.
United Kingdom is experiencing a CAGR of 6.8%, as private clinics and urban hospitals in London, Manchester, and Edinburgh adopt low dose hormonal solutions to complement conventional treatments. Rising patient interest in preventive care and minimally invasive therapies has led to stronger uptake. Investments emphasize formulation stability, European regulatory compliance, and staff training to ensure proper administration. Concentrated demand exists in metropolitan healthcare hubs where outpatient care is high. Growth reflects the combination of lifestyle-related hormonal conditions, patient education initiatives, and increasing availability of specialized treatment packages within private and public healthcare networks.
China is growing at a CAGR of 8.5% with increasing usage in tier-1 city hospitals such as Beijing, Shanghai, and Shenzhen, where specialized endocrinology departments are expanding services. Patients are shifting toward low dose hormone therapies to reduce side effects from traditional treatments. Hospitals and private clinics are investing in modern dosing equipment, quality monitoring systems, and workflow integration. Demand concentrates in urban centers with high patient volumes, and regional manufacturing hubs ensure reliable product supply. Growth is linked to rising awareness of endocrine health, urban lifestyle pressures affecting hormonal balance, and modernization of outpatient treatment protocols.
India is recording a CAGR of 9%, with urban hospital networks in Mumbai, Delhi, and Bengaluru actively introducing low dose hormone therapies in women’s health programs. Increased patient focus on safer and more cost-effective alternatives has strengthened demand in fertility clinics, endocrinology centers, and private hospitals. Investments prioritize durable formulations, scalable distribution, and clinical staff training. Urban centers serve as adoption hubs while expanding infrastructure reaches semi-urban areas. Growth is shaped by rising healthcare access, greater middle-class awareness of hormone therapy options, and government initiatives supporting modern endocrine treatment availability.
Brazil is seeing a CAGR of 7.5%, with metropolitan healthcare facilities in São Paulo, Rio de Janeiro, and Curitiba introducing low dose hormone therapies for preventive and wellness-focused care. Clinics are incorporating safer, patient-friendly alternatives to conventional hormone treatments. Investments center on formulation consistency, compliance with ANVISA regulations, and operator training. Urban healthcare hubs show concentrated adoption, while smaller cities are gradually expanding use. Growth reflects cultural emphasis on female health, increased disposable income, and the expansion of private clinics providing advanced endocrine care and wellness services across Brazil.
Theramex supplies low dose hormone alternatives formulated for symptom management in hormone related conditions, with an emphasis on tolerability and consistent dosing profiles. AbbVie offers products engineered to deliver controlled hormone activity while supporting established safety frameworks for long term use. Pfizer develops low dose options that integrate with its broader women’s health portfolio, focusing on predictable pharmacokinetics and patient adherence. Bayer provides hormone alternatives backed by clinical evidence and regulatory approvals across multiple regions. Mylan, now part of Viatris, delivers cost effective low dose hormone formulations designed for broad accessibility and generic substitution. Amgen contributes biologically derived products that address hormone modulation needs in niche patient segments. Each supplier aligns product attributes with safety standards and clinical utility in chronic therapy contexts.
Differentiation in this market arises from dosing precision, side effect profiles, and post marketing support. Theramex and Bayer emphasize quality assurance and long term safety tracking. AbbVie and Pfizer focus on established brand trust and clinician familiarity with their formulations. Viatris leverages scale to provide affordable alternatives, while Amgen advances targeted biologic hormone modulators for select indications. Other regional manufacturers may offer specialized low dose hormone alternatives tailored to local regulatory environments and prescribing practices. Adoption depends on evidence of efficacy, tolerability in sensitive populations, and alignment with treatment guidelines for hormone replacement or alternative therapy pathways. Suppliers with robust clinical data and accessible delivery formats maintain relevance across patient care settings.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Therapy Type | Bioidentical hormone therapies, Phytoestrogen and plant-based alternatives, Selective estrogen receptor modulators (SERMs), Low-dose hormone replacement formulations |
| Application | Menopausal symptom management, Androgen deficiency therapies, Osteoporosis and bone health, Sexual wellness and libido support |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Theramex, AbbVie, Pfizer, Bayer, Mylan (Viatris), Amgen |
| Additional Attributes | Dollar by sales by therapy type and application; demand led by menopausal symptom management and bioidentical therapies; adoption shaped by guideline positioning, step-therapy protocols, and risk management; growth driven by long-term use, patient adherence, and gradual inclusion in formularies rather than rapid substitution of conventional hormone therapies. |
How big is the low dose hormone alternatives market in 2026?
The global low dose hormone alternatives market is estimated to be valued at USD 3.2 billion in 2026.
What will be the size of low dose hormone alternatives market in 2036?
The market size for the low dose hormone alternatives market is projected to reach USD 6.8 billion by 2036.
How much will be the low dose hormone alternatives market growth between 2026 and 2036?
The low dose hormone alternatives market is expected to grow at a 7.8% CAGR between 2026 and 2036.
What are the key product types in the low dose hormone alternatives market?
The key product types in low dose hormone alternatives market are bioidentical hormone therapies , phytoestrogen and plant‑based alternatives, selective estrogen receptor modulators (serms) and low‑dose hormone replacement formulations.
Which application segment to contribute significant share in the low dose hormone alternatives market in 2026?
In terms of application, menopausal symptom management segment to command 45.0% share in the low dose hormone alternatives market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.