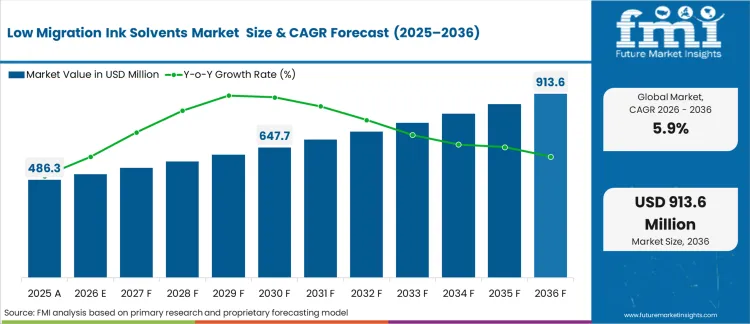
The low migration ink solvents market was valued at USD 485.4 million in 2025 and is expected to reach USD 515.0 million in 2026, reflecting a 5.9% CAGR. By 2036, the market is projected to grow to USD 913.6 million, driven by increasingly stringent regulatory requirements on organoleptic performance for primary packaging in direct food‑contact applications.
Brands that delay adopting these upgraded solvent formulations face immediate product recalls and exclusion from European and North American retail shelves under strict food contact material laws. Practitioners recognize that the most critical bottleneck is not ink adhesion, but rather the solvent's behavior during high-speed curing on non-porous flexible substrates, where trapped residues trigger taste and odor changes even without physical chemical transfer. This necessitates investment in advanced printing ink systems natively designed for absolute sensory neutrality.
This structural requirement establishes a mandatory compliance baseline before widespread market adoption becomes self-sustaining. Major global brand owners are now mandating full compositional disclosure and GMP certification from primary packaging converters ahead of the impending European printing audit cycle. Facilities completing this certification unlock access to premium pharmaceutical and food-grade printing contracts.
China advances at a 7.5% compound rate, driven by aggressive enforcement of its National Food Safety Standard (GB 4806.1-2016) across its massive domestic packaged food export sector. India tracks at 7.1% as domestic pharmaceutical manufacturing scales up its compliance with international packaging mandates. Germany expands at 6.2%, anchored by stringent European Food Safety Authority directives that continuously raise the bar for packaging safety. The United States follows at 5.8%, responding to strict FDA food contact substance notifications. The United Kingdom grows at 5.4%, driven by retail-led packaging safety initiatives. Japan records a 5.0% expansion, prioritizing high-purity packaging for its premium cosmetics sector. Brazil posts a 4.6% growth rate as regional food exporters align with global compliance requirements. This growth spread exists because distinct regional regulatory enforcement timelines dictate how quickly converters must abandon legacy ink systems.
The low migration ink solvents market encompasses high-purity carrier fluids, monomers, and specifically formulated solvent blends designed to ensure that printed packaging materials do not transfer chemical components into the packaged product. These solvents undergo specialized refining to eliminate volatile organic residues that could compromise the sensory properties or safety of food, pharmaceuticals, and cosmetics.
The market scope includes high-molecular-weight solvent carriers, specialized photoinitiator solvents, and polymeric carrier fluids utilized exclusively in low migration printing applications. Certified low-migration modifiers, specific aliphatic hydrocarbons, and high-purity specialized UV curable inks designed for absolute regulatory compliance fall within the defined boundaries. Formulations requiring GMP-certified production lines are fully incorporated.
Standard industrial printing solvents lacking specific food-contact compliance certifications are explicitly excluded. General-purpose volatile organic compounds used in commercial publication printing, standard textile inks, and architectural coatings fall outside the defined parameters. Base raw materials not specifically refined and marketed for low migration applications are omitted from the valuation.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 515.0 million |
| Industry Value (2036) | USD 913.6 million |
| CAGR (2026-2036) | 5.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
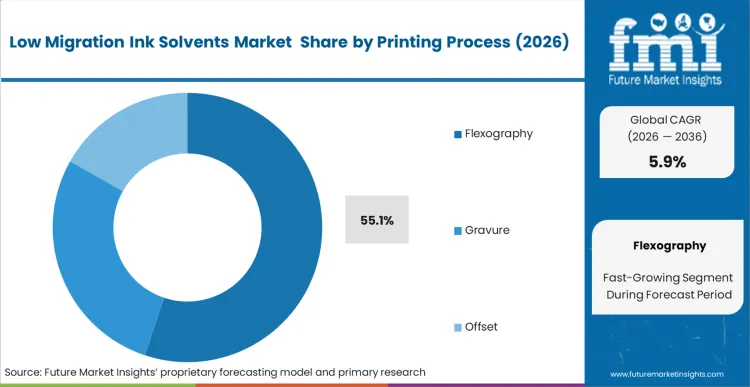
The widespread utilization of continuous web formats for flexible packaging forces primary converters to demand highly stable ink formulations. Flexography commands a dominant 55.1% share in 2026, reflecting the absolute requirement for rapid solvent evaporation during high-volume label and wrapper production. Ink formulators supplying these processes must precisely balance the carrier fluid's volatility to prevent premature drying on the printing plates while ensuring complete evacuation before the final rewind. This delicate equilibrium dictates the exact chemical makeup of the formulation. According to FMI's estimates, facilities transitioning to these optimized carriers significantly reduce their scrap rates related to set-off and blocking. Plant managers failing to validate their solvent evaporation curves risk significant organoleptic failures in the final packaged product, jeopardizing multi-million dollar contracts. Proper integration requires rigorous high speed flexographic integration protocols.
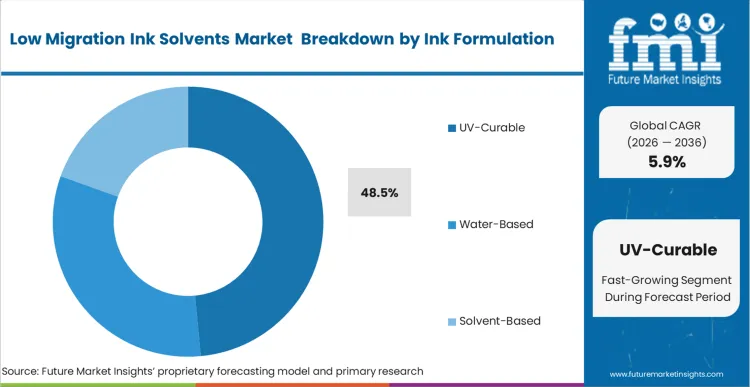
The fundamental transition from evaporative drying to localized photopolymerization drives the adoption of advanced ink architectures. UV-Curable formulations hold a 48.5% share in 2026, as the instantaneous transformation from liquid to solid effectively locks all chemical components into a cross-linked matrix. This structural mechanism physically prevents residual monomers from migrating through the packaging substrate. Based on FMI's assessment, the deployment of large-molecule photoinitiators that cannot easily penetrate flexible films forms the foundation of this segment's success. Formulation scientists must utilize specialized, high-purity monomers that react completely upon exposure to specific light wavelengths. Converters relying on outdated, low-molecular-weight photoinitiators face the immediate threat of unbound chemicals migrating into the packaged product, triggering regulatory intervention. Forward-looking facilities are also exploring water based ink transitions to further minimize chemical hazards.
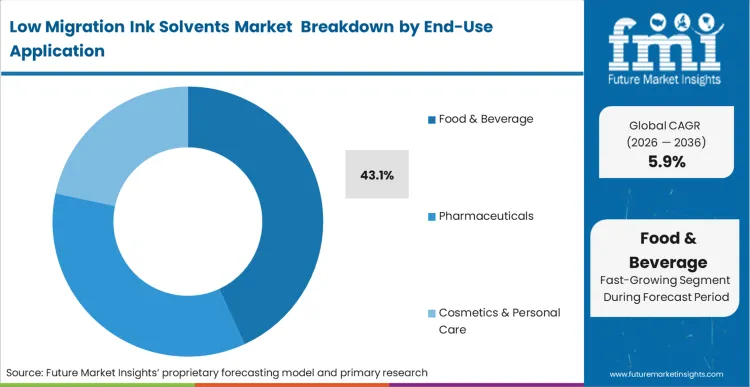
Global FMCG brands responding to intense consumer scrutiny mandate absolute chemical neutrality across their entire packaging portfolio. The Food & Beverage segment emerges as the dominant application area, representing 43.1% of the total market share in 2026. Rigid safety directives compel brand owners to specify high-purity solvent systems that guarantee zero organoleptic interference with the packaged product. As per FMI's projection, the sheer volume of continuously produced consumable goods requires massive quantities of certified, low-migration ink formulations to sustain production lines. Any deviation from these strict chemical profiles introduces unacceptable liability risks. Packaging converters failing to provide verifiable migration testing data for their food-grade outputs immediately lose their Tier-1 supplier status. Parallel requirements are reshaping sterile pharmaceutical packaging layers to ensure medicine efficacy.
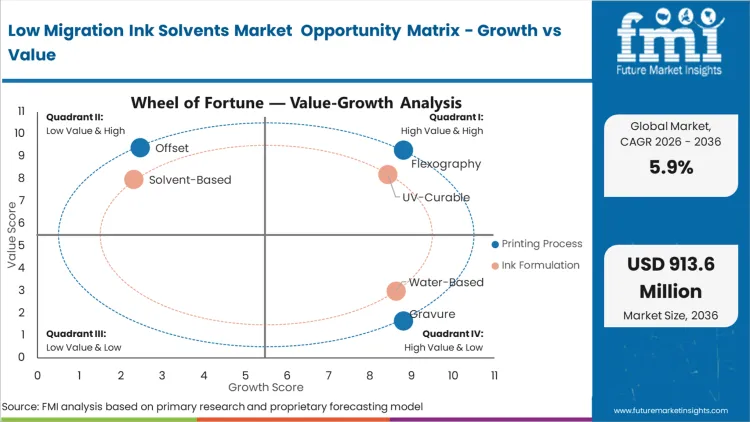
Strict enforcement of the European Food Safety Authority (EFSA) directives and the Swiss Ordinance (RS 817.023.21) forces primary packaging converters to eliminate standard volatile organic compounds from all direct and indirect food-contact printing operations. This regulatory mechanism requires converters to adopt high-molecular-weight, highly refined solvent systems that are physically incapable of migrating through flexible films. Asset owners upgrading their press operations face a strict mandate: deploy certified low-migration formulations or forfeit access to the lucrative FMCG supply chain. Facilities that fail to modernize their ink architectures face catastrophic product recalls and immediate exclusion from major retail markets.
The intricate chemical engineering required to balance rapid curing speeds with low migration properties creates significant operational friction for traditional ink formulators. Developing highly refined, large-molecule solvent carriers that perform efficiently on high-speed flexographic presses demands massive R&D expenditure and dedicated GMP-certified production lines to prevent cross-contamination. While novel polymeric carriers are emerging as a workaround, their inherently higher viscosity limits application speeds and requires costly press modifications, constraining rapid adoption among mid-tier converters.
Based on the regional analysis, the Low Migration Ink Solvents Market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.5% |
| India | 7.1% |
| Germany | 6.2% |
| United States | 5.8% |
| United Kingdom | 5.4% |
| Japan | 5.0% |
| Brazil | 4.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
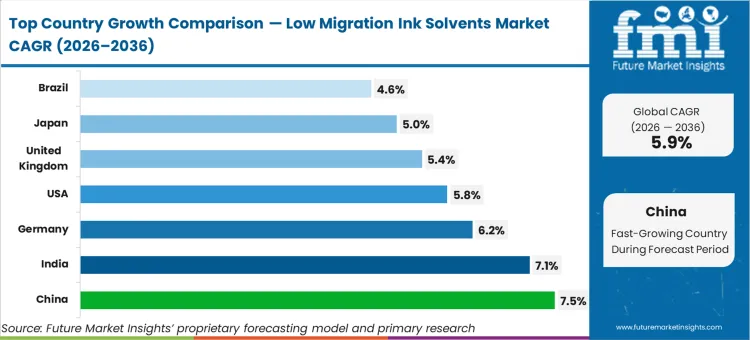
Aggressive enforcement of national food safety protocols radically shifts the procurement landscape across East Asia. Government regulatory bodies updating domestic standards to align with rigorous international export requirements are forcing local packaging converters to abandon cheap, legacy solvent systems. This policy-led transformation necessitates substantial investments in advanced chemical architectures capable of meeting zero-tolerance contamination thresholds. In FMI's view, this top-down regulatory pressure rapidly scales the adoption of high-purity solvent formulations across the region's massive manufacturing base. Regional converters must adapt quickly to secure their positions within global supply chains, heavily driving the implementation of digital packaging presses.
FMI's report includes extensive coverage of South Korea and Taiwan. The prevailing dynamic across these nations is the rapid localization of highly certified chemical production, allowing domestic ink manufacturers to directly compete with established European compliance leaders.
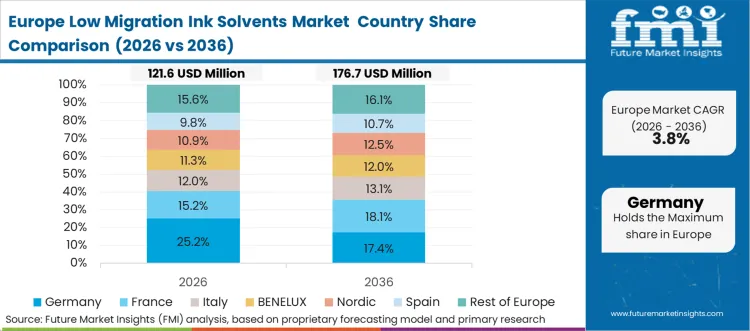
Stringent, continuously evolving directives from the European Food Safety Authority (EFSA) define the operational boundaries for the entire European packaging sector. Regulatory agencies explicitly mandate the use of positively listed chemical substances for any material in indirect or direct contact with food, establishing an exceptionally high barrier to entry for ink suppliers. According to FMI's estimates, this uncompromising policy framework forces packaging converters to utilize only GMP-certified low migration formulations, regardless of the underlying printing technology. Asset operators attempting to circumvent these standards face severe legal repercussions and immediate brand blacklisting.
FMI's report includes thorough investigation of France, Italy, and the Benelux region. A recurring structural pattern across these nations is the proactive phase-out of traditional free-radical photoinitiators, pushing the market heavily toward advanced polymeric solvent structures.
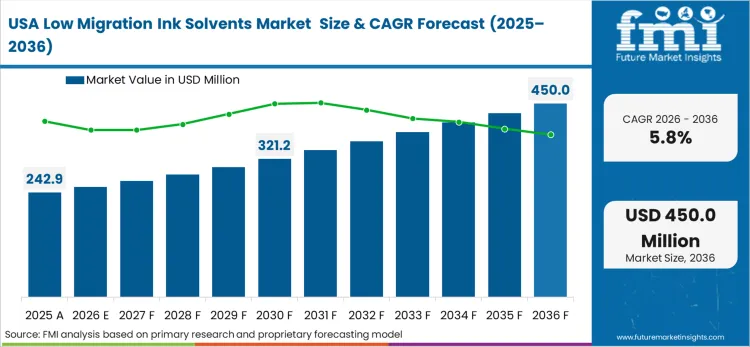
Strict adherence to FDA Food Contact Substance (FCS) notifications dictates the chemical architectures permitted within the North American packaging supply chain. Pharmaceutical and food manufacturing conglomerates enforce rigorous testing protocols to ensure zero chemical transfer into their products, effectively banning legacy evaporative solvents from sensitive applications. Based on FMI's assessment, this buyer behavior-led environment requires ink formulators to invest heavily in comprehensive toxicological testing before launching new solvent carrier platforms. Companies competing in this space must prove the absolute stability of their formulations under extreme supply chain conditions.
FMI's report includes specific analysis of the Canadian and Mexican industrial markets. The defining dynamic in these countries involves the integration of cross-border FMCG supply chains, which requires standardized, highly certified ink protocols to coordinate continuous packaging operations across multiple regulatory jurisdictions.
The rapid expansion of domestic pharmaceutical manufacturing and packaged food exports dictates a massive shift in procurement standards across South Asia. Industry leaders constructing new production facilities are bypassing legacy ink systems entirely, specifying internationally certified low-migration formulations from day one. FMI analysts opine that this infrastructure-led modernization strategy is fundamentally required to pass stringent audits from foreign regulatory bodies like the US FDA and the European Medicines Agency. Asset operators recognize that utilizing uncertified solvent systems critically endangers their ability to export high-margin consumable products to Western markets.
FMI's report includes detailed analysis of the broader ASEAN region and Oceania. A primary trend shaping these nations is the aggressive capital investment by global printing press manufacturers to establish localized support hubs for advanced low-migration curing technologies.
The consolidation of regional food and beverage production into massive, export-oriented conglomerates is transforming the Latin American packaging sector. Procurement teams within these unified entities demand standardized, highly certified material inputs to ensure their products clear strict border inspections in North America and Europe. Based on FMI's assessment, this economics-led consolidation empowers large buyers to dictate compliance terms to local ink suppliers, forcing rapid technological upgrades across the continent. Smaller packaging converters must either adopt these advanced low-migration solvent systems or face exclusion from the most lucrative export supply chains.
FMI's report includes comprehensive evaluation of Argentina, Chile, and Mexico. A consistent structural condition across these markets is the increasing dominance of multinational FMCG brands, which enforce their own global packaging safety standards regardless of looser local regulations.
The low migration ink solvents sector exhibits a highly concentrated market structure. This consolidation exists because developing and validating high-purity, low-migration solvent systems requires immense capital investment in dedicated, GMP-certified production facilities to eliminate trace cross-contamination. Major industry leaders such as Sun Chemical Corporation, hubergroup Deutschland, and Siegwerk Druckfarben dominate the landscape by leveraging their massive global scale to amortize the high costs of continuous toxicological testing and regulatory compliance. Buyers utilize comprehensive compositional transparency and third-party migration certifications as the primary competitive variable to distinguish qualified from unqualified vendors during the tender process.
Companies holding proprietary green solvent extraction methods possess a distinct structural advantage in formulation purity. This capability allows them to reliably produce high-molecular-weight carriers that execute rapid curing without leaving volatile residues behind. For a challenger to replicate this advantage, they must construct entirely segregated manufacturing lines and navigate years of complex EFSA and FDA approval cycles for new chemical architectures. Consequently, smaller ink formulators are increasingly forced into niche, non-food applications, leaving the lucrative Tier-1 FMCG contracts to the dominant, highly certified multinationals.
To prevent vendor lock-in, massive FMCG brand owners actively promote open compliance frameworks and standardized testing protocols, allowing them to qualify multiple global ink suppliers simultaneously. This structural tension between the buyers' desire for supply chain redundancy and the dominant vendors' push for integrated, single-source ecosystems heavily limits pricing power even in this highly concentrated market. Moving toward 2036, the competitive trajectory points toward further consolidation, as the escalating costs of maintaining global regulatory compliance outpace the financial capabilities of regional independent ink manufacturers.

| Metric | Value |
|---|---|
| Quantitative Units | USD 515.0 million to USD 913.6 million, at a CAGR of 5.9% |
| Market Definition | The Low Migration Ink Solvents Market provides specialized, highly refined carrier fluids and monomers that prevent the chemical transfer of printing components into packaged consumables, ensuring absolute regulatory compliance and sensory neutrality. |
| Printing Process Segmentation | Flexography, Gravure, Offset, Digital |
| Ink Formulation Segmentation | UV-Curable, Water-Based, Solvent-Based |
| End-Use Application Segmentation | Food & Beverage, Pharmaceuticals, Cosmetics & Personal Care, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, United States, United Kingdom, Japan, Brazil, and 40 plus countries |
| Key Companies Profiled | Sun Chemical Corporation, hubergroup Deutschland, Siegwerk Druckfarben, INX International Ink Co., Artience Co., Ltd., Agfa-Gevaert, Flint Group, ALTANA AG |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of certified low-migration solvent shipments, applying region-specific regulatory enforcement curves to project the future adoption velocity. Segment forecasts undergo cross-validation against quarterly specialty chemical shipment volumes. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the low migration ink solvents market in 2026?
The market is estimated to reach USD 515.0 million in 2026 as global brand owners enforce strict GMP certification requirements on primary packaging converters.
What will it be valued at by 2036?
The market is projected to reach USD 913.6 million by 2036 as the transition to high-molecular-weight carriers eliminates legacy evaporative solvents entirely.
What CAGR is projected?
A CAGR of 5.90% is projected, structurally supported by the mandatory nature of food contact material laws across major global economies.
Which printing process segment leads?
Flexography commands a 55.1% share due to its unparalleled efficiency in continuous web printing for flexible food and beverage packaging lines.
Which ink formulation segment leads?
The UV-Curable segment leads because instantaneous cross-linking effectively locks chemical components into a solid matrix, preventing sub-surface migration.
Which end-use application segment leads?
Food & Beverage leads due to the sheer volume of continuously produced consumable goods requiring absolute chemical safety and sensory neutrality.
What drives rapid growth?
Strict enforcement of the Swiss Ordinance and heightened organoleptic testing requirements drive the rapid phase-out of traditional industrial solvents.
What is the primary restraint?
The intricate chemical engineering required to balance rapid curing speeds with low migration properties creates significant operational friction for formulators.
Which country grows fastest?
China advances at a 7.5% compound rate, driven by aggressive enforcement of its National Food Safety Standard across its massive food export sector.
What specific regulatory framework dictates compliance in Europe?
The European Food Safety Authority (EFSA) directives and the Swiss Ordinance explicitly mandate the use of positively listed chemical substances.
What specific technology shift is underway in ink architectures?
The industry is transitioning away from standard volatile organic compounds toward large-molecule photo initiators and natively stable polymeric solvent systems.
What specific competitive variable separates market leaders from challengers?
Possessing dedicated, GMP-certified production lines physically isolated from standard industrial ink manufacturing is the critical differentiator.
Why is India expanding so rapidly?
India is tracking 7.1% growth as its domestic pharmaceutical export sector scales up compliance to pass stringent US FDA and European Medicines Agency audits.
What forces United Kingdom packaging converters to upgrade?
Retail-led packaging safety initiatives enforced by major UK supermarket chains compel suppliers to surpass baseline government chemical regulations.
How does Germany maintain its strong market position?
Stringent national printing ordinances impose strict liability on packaging converters utilizing non-compliant systems, accelerating hardware and formulation replacement.
What compliance standard drives the United States market?
Strict adherence to FDA Food Contact Substance notifications forces pharmaceutical and food manufacturing conglomerates to enforce zero chemical transfer protocols.
Why does Japan require specialized solvent systems?
Japan's premium cosmetics sector mandates ultra-refined carrier fluids to guarantee absolute sensory neutrality in luxury personal care packaging.
How is Brazil integrated into the global compliance landscape?
Brazil's massive agricultural and processed food export sector must deploy advanced low-migration formulations to ensure products pass European and North American border inspections.
What is the role of GMP certification in this market?
GMP certification ensures that low-migration formulations are produced on dedicated lines, eliminating the risk of cross-contamination from legacy industrial inks.
What are organoleptic failures?
Organoleptic failures occur when trapped solvent residues trigger taste and odor changes in the packaged consumable, severely damaging brand reputation.
Why are UV-curable inks highly preferred?
UV-curable inks cure instantly upon light exposure, turning liquid monomers into a stable solid matrix that cannot migrate into the packaging substrate.
What is included in the scope of this market analysis?
The scope encompasses high-purity carrier fluids, specialized photoinitiator solvents, and polymeric carrier fluids utilized exclusively in certified low migration applications.
What products are explicitly excluded from this report?
General-purpose volatile organic compounds used in commercial publication printing and standard industrial solvent systems without food-contact certifications are excluded.
How does FMI cross-validate its market projections?
Projections undergo cross-validation against quarterly specialty chemical shipment volumes and publicly reported capital expenditure guidance from major global ink manufacturing conglomerates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.