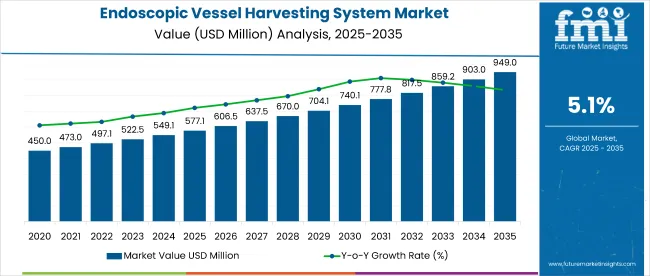
The endoscopic vessel harvesting system market is valued at USD 603.7 million in 2026 and is projected to reach USD 993.4 million by 2036, reflecting a CAGR of 5.1%. Demand rises as cardiovascular surgeons, hospital systems, and cardiac surgery centers adopt minimally invasive harvesting techniques that reduce surgical trauma and improve patient outcomes in coronary artery bypass grafting procedures. Adoption strengthens across developed healthcare markets where procedural efficiency and patient safety requirements drive investment in advanced surgical technologies.
Disposable systems lead product-type usage due to infection control benefits, sterility assurance, and elimination of reprocessing requirements that streamline surgical workflows. Saphenous vein harvesting dominates vessel type applications because anatomical characteristics and surgical familiarity support reliable graft outcomes in cardiovascular procedures. Hospital facilities control end-user segments through centralized procurement decisions and integrated surgical care protocols that prioritize patient safety and procedural standardization.
United States, Germany, Japan, India, and Brazil emerge as core growth regions driven by expanding cardiovascular disease prevalence, aging populations, and healthcare infrastructure investments supporting advanced surgical capabilities. Getinge AB, Terumo Corporation, LivaNova PLC, KARL STORZ SE & Co., and Saphena Medical Inc. shape competition through product innovation, surgical training programs, and distribution networks that enhance procedural adoption across cardiovascular surgery practices.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 603.7 million |
| Market Forecast Value (2036) | USD 993.4 million |
| Forecast CAGR (2026-2036) | 5.1% |
The global market is classified into product type, vessel type, application, end user, and region. By product type, the market is divided into disposable and reusable systems. In terms of vessel type, the market is classified into saphenous vein, radial artery, and others. By application, the market is segmented into coronary artery bypass grafting (CABG), peripheral artery disease surgery, and other cardiovascular surgeries. Based on end user, the market is divided into hospitals, ambulatory surgical centers, and specialty clinics. By region, the market is divided into North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa.
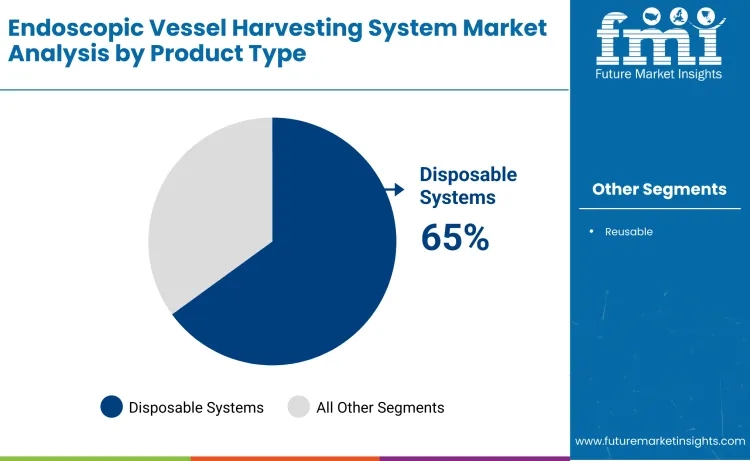
Disposable systems hold 65%, making them the leading product-type segment globally. Disposable system platforms support infection control protocols, sterility assurance, and streamlined surgical workflows across cardiovascular procedure applications. Reusable systems maintain demand in cost-conscious healthcare facilities where sterilization infrastructure and long-term equipment utilization are prioritized. Disposable system distribution reflects safety requirements, regulatory compliance goals, and integration with modern surgical safety standards that prioritize patient protection.
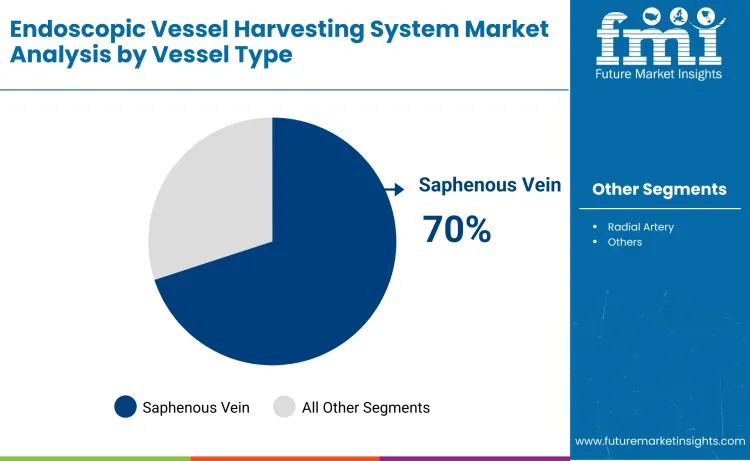
Saphenous vein holds 70%, making it the largest vessel-type segment globally. Saphenous vein harvesting supports coronary artery bypass procedures, anatomical accessibility, and proven clinical outcomes across diverse patient populations. Radial artery harvesting serves specialized applications requiring alternative graft options. Vessel type distribution reflects surgical familiarity, clinical evidence, and procedural success rates that influence surgeon preferences and patient outcomes.
Global demand grows as cardiovascular surgeons, hospital systems, and surgical teams explore advanced harvesting technologies to enhance procedural outcomes, reduce patient trauma, and improve surgical efficiency. Endoscopic vessel harvesting supports patient care improvements in cardiovascular procedures where surgical precision directly affects recovery outcomes. Manufacturers deploy disposable systems, reusable platforms, and AI-enhanced technologies for comprehensive surgical applications. Technology platforms manage device selection, quality control, and procedural coordination. Interest rises as surgical programs evaluate minimally invasive options suited to diverse cardiovascular procedure categories.
How are Cardiovascular Surgery Trends and Healthcare Provider Preferences Influencing Adoption of Endoscopic Vessel Harvesting Systems?
Cardiovascular surgery centers experience growing demand for minimally invasive procedures, which strengthens interest in advanced harvesting technologies that enhance patient outcomes across coronary and peripheral vascular surgery segments. Surgeons and healthcare teams use harvesting systems for comprehensive vessel procurement to improve graft quality during complex cardiovascular interventions. Harvesting systems deliver consistent performance across procedural variations that affect vessel integrity and long-term graft success. Hospital systems, cardiac surgery centers, and specialized facilities adopt harvesting solutions to support advanced cardiovascular care requirements. Technology suppliers oversee development using precision engineering for improved ergonomics and procedural efficiency. Digital integration platforms connect harvesting data with surgical record systems, enabling procedural tracking and reliable outcome measurement. Quality assurance systems and regulatory compliance modules improve device standards across varied cardiovascular surgery environments.
How do Regulatory Requirements, Cost Considerations, and Training Limitations Affect Scalability?
Medical device regulations shape technology adoption through approval processes, safety requirements, clinical testing protocols, and surgical training standards. Manufacturers assess capital investment for device development, clinical trials, regulatory submissions, and training program implementation. Market dynamics influence pricing strategies, affecting technology accessibility in cost-sensitive healthcare segments. Technology complexity and training requirements create adoption challenges across different surgical skill levels. Healthcare facilities balance harvesting system adoption with procedural volume requirements and surgical outcome expectations. Manufacturing and distribution facilities require coordination with healthcare supply networks, affecting technology availability and market penetration. Regulatory programs in several regions maintain device safety and efficacy standards, which influence long-term investment strategies for specialized cardiovascular technology operations.
Demand for endoscopic vessel harvesting systems is rising due to increased cardiovascular disease prevalence, advancing surgical techniques, and growing preference for minimally invasive procedures that improve patient outcomes. United States records a CAGR of 2.1% supported by established cardiac surgery infrastructure and advanced healthcare technology adoption. Germany shows a 3.8% CAGR driven by precision engineering capabilities and high healthcare quality standards. India posts a 3.9% CAGR supported by healthcare infrastructure development and growing cardiovascular disease burden. Japan holds a 4.6% CAGR linked with technological innovation and aging population healthcare needs. Brazil records a 3.5% CAGR aligned with healthcare service improvements and cardiovascular awareness programs.
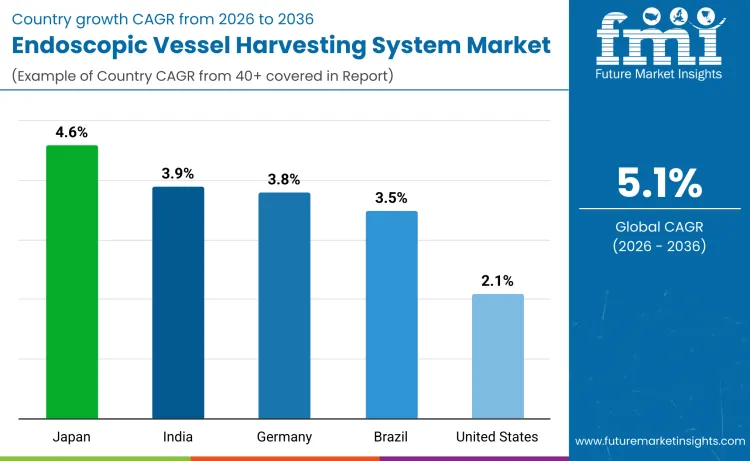
| Country | CAGR (2026-2036) |
|---|---|
| United States | 2.1% |
| Germany | 3.8% |
| India | 3.9% |
| Japan | 4.6% |
| Brazil | 3.5% |
United States drives demand through high cardiovascular disease prevalence, advanced healthcare infrastructure, and strong adoption of minimally invasive surgical techniques across established cardiac surgery programs. The USA endoscopic vessel harvesting system market is slated to grow at CAGR of 2.1% during the study period. This reflects steady deployment of advanced harvesting technologies across hospitals, cardiac centers, and specialty surgical facilities. Healthcare providers use harvesting systems to enhance procedural efficiency and improve patient outcomes effectively. Insurance coverage supports access to advanced surgical technologies enabling widespread procedural adoption. Technology suppliers introduce sophisticated platforms suited for high-volume surgical environments. Strong clinical acceptance of minimally invasive approaches reinforces operational expansion across major cardiac surgery centers.
Germany supports rising demand due to precision engineering capabilities, high healthcare quality standards, and systematic approach to surgical technology integration across cardiac care facilities. German endoscopic vessel harvesting system market is poised to grow at a CAGR of 3.8% during the study period, showing consistent adoption of harvesting technologies across medical centers and specialized cardiovascular facilities. Growing surgical precision requirements benefit from technologies that improve procedural outcomes at optimal quality standards. Healthcare platforms integrate advanced and traditional surgical approaches to address diverse procedural requirements. Regulatory frameworks and quality assurance programs support technology adoption across expanding cardiovascular surgery networks.
India drives demand through healthcare infrastructure development, increasing cardiovascular disease awareness, and expanding access to advanced cardiac surgery procedures across urban and metropolitan areas. The demand for endoscopic vessel harvesting systems is anticipated to grow at a CAGR of 3.9% during the study period, reflecting growing adoption of harvesting technologies across public and private healthcare facilities, specialty hospitals, and cardiac surgery centers. Healthcare expansion patterns favor procedures that improve outcomes while managing cost considerations for diverse patient populations. Healthcare providers integrate harvesting systems to address growing cardiovascular disease burden and procedural demand. Medical tourism development and international healthcare standards support technology adoption across major medical centers.
Japan drives demand due to technological innovation leadership, aging population healthcare requirements, and commitment to advanced medical device development and surgical excellence. The demand for endoscopic vessel harvesting systems in Japan is slated to expand at a CAGR of 4.6% during the forecast period, showing strong adoption of harvesting technologies supporting precision surgery requirements, advanced cardiovascular care, and demographic healthcare needs. Technology development firms produce high-quality harvesting systems suited for demanding surgical applications. Healthcare authorities collaborate with device manufacturers to evaluate technologies meeting strict performance and safety standards. Aging population demographics support increased reliance on cardiovascular interventions and specialized surgical solutions. Research and development networks adopt harvesting technologies to support predictable outcomes and long-term procedural success.
Brazil drives demand through healthcare service improvements, cardiovascular disease awareness campaigns, and expanding access to advanced surgical procedures across public and private healthcare sectors. The demand for endoscopic vessel harvesting systems is poised to grow at a CAGR of 3.5% during the study period, showing steady adoption of harvesting technologies supporting domestic healthcare development and procedural capability expansion. Healthcare improvement strategies encourage advanced technology adoption to achieve clinical excellence in competitive medical markets. Healthcare providers and surgical teams employ harvesting solutions to support procedural quality commitments and patient care requirements. Government healthcare programs support controlled expansion evaluating technology performance, clinical outcomes, and healthcare delivery efficiency. Growth in medical infrastructure reinforces the role of harvesting systems in comprehensive cardiovascular surgery operations.
The endoscopic vessel harvesting system market is highly competitive, with several key players driving innovation and technological advancement. Getinge AB is a leading player, known for its Vasoview Hemopro series featuring simultaneous cut-and-seal technology that enhances vessel quality while minimizing thermal injury. The company operates globally, focusing on providing advanced harvesting solutions for cardiovascular applications across diverse surgical environments. Getinge AB's strong engineering capabilities and continuous improvements in surgical technology give it a competitive advantage.
Terumo Corporation competes in the cardiovascular surgery market through its Virtuo Saph Plus system, emphasizing ergonomic design and intuitive operation for enhanced surgical efficiency. Liva Nova PLC has established a strong position in cardiovascular technology, particularly focusing on solutions that improve surgeon ergonomics and procedural outcomes. KARL STORZ SE & Co. concentrates on precision engineering and endoscopic technologies that provide superior visualization and surgical performance.
Saphena Medical Inc. targets surgical efficiency and procedural simplification through unified harvesting systems designed to reduce operative time and improve patient recovery outcomes. These companies leverage advanced manufacturing capabilities to deliver high-quality materials for demanding cardiovascular surgery requirements.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product Type | Disposable, Reusable |
| Vessel Type | Saphenous Vein, Radial Artery, Others |
| Application | Coronary Artery Bypass Grafting (CABG), Peripheral Artery Disease Surgery, Other Cardiovascular Surgeries |
| End User | Hospitals, Ambulatory Surgical Centers, Specialty Clinics |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, Germany, Japan, India, Brazil, United Kingdom, France, Italy, and 40+ countries |
| Key Companies Profiled | Getinge AB, Terumo Corporation, LivaNova PLC, KARL STORZ SE & Co., Saphena Medical Inc. |
| Additional Attributes | Dollar sales by product type and application; deployment trends across cardiovascular surgery and minimally invasive procedures; adoption driven by growth in cardiac surgery volumes, patient safety improvements, and procedural efficiency; integration with advanced surgical technologies . |
How big is the endoscopic vessel harvesting system market in 2026?
The global endoscopic vessel harvesting system market is valued at USD 603.7 million in 2026.
What will be the size of the endoscopic vessel harvesting system market in 2036?
The market is projected to reach USD 993.4 million by 2036.
How fast will the endoscopic vessel harvesting system market grow between 2026 and 2036?
The market will grow at a CAGR of 5.1% from 2026 to 2036.
Which product type leads the endoscopic vessel harvesting system market in 2026?
Disposable systems lead the market due to infection control benefits and streamlined surgical workflows.
Who are the major players in the endoscopic vessel harvesting system market?
Key players include Getinge AB, Terumo Corporation, LivaNova PLC, KARL STORZ SE & Co., and Saphena Medical Inc.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.