The Gynecology Gloves market is projected at USD 1.3 billion in 2026 and is expected to reach USD 2.2 billion by 2036, growing at a CAGR of 5.5%. Latex gloves account for 45% of the product mix, primarily used in examinations (65%), while hospitals represent the largest end-user segment at 55%. Clinics and retail channels constitute the remainder. Leading players include Top Glove, Hartalega, Ansell, Kossan, and Supermax. Demand is rising with increasing gynecological procedures.
Market growth is supported by rising awareness of hygiene, infection prevention, and safety in healthcare facilities. Nitrile gloves are gaining adoption for surgical applications due to enhanced chemical resistance and durability. Expansion of healthcare infrastructure and clinics worldwide further drives demand. Retail distribution and disposable usage for minor procedures also contribute to steady adoption. These factors collectively sustain growth across the 2026–2036 forecast period.
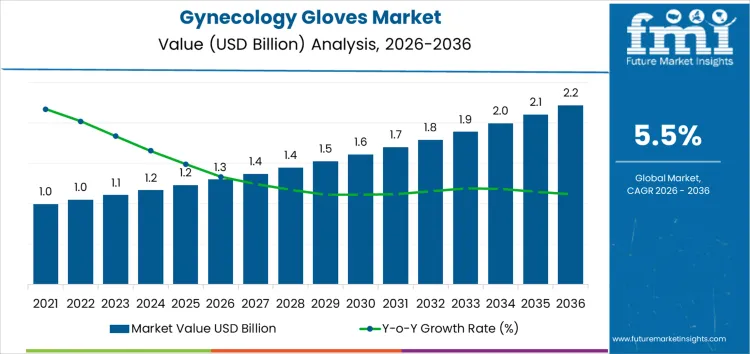
From 2026 to 2031, the gynecology gloves market grows from USD 1.3 billion to approximately USD 1.6 billion, representing the early adoption phase. Annual value additions increase from USD 0.08 billion in 2026 to USD 0.12 billion in 2031. Growth is driven by rising demand in hospitals, clinics, and maternity centers, where high-quality, sterile gloves are critical for surgical and examination procedures. North America and Europe account for roughly 60% of early adoption, supported by regulatory compliance, hygiene protocols, and rising procedural volumes. Uptake is concentrated in urban hospitals and specialized obstetrics-gynecology centers.
Between 2031 and 2036, the market expands from roughly USD 1.6 billion to USD 2.2 billion, illustrating the late adoption phase characterized by broader commercialization and multi-site penetration. Annual increments rise steadily from USD 0.12 billion to USD 0.14 billion, driven by repeat procurement, portfolio-wide adoption across hospitals and clinics, and expansion in emerging regions in Asia Pacific and Latin America. Growth is reinforced by increased surgical and diagnostic procedures, regulatory emphasis on infection control, and normalized adoption across gynecology and obstetrics care settings. Early growth relied on selective high-volume centers, while later expansion reflects structural deployment across global healthcare facilities.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.3 billion |
| Forecast Value (2036) | USD 2.2 billion |
| Forecast CAGR (2026–2036) | 5.5% |
Demand for gynecology gloves developed from procedural realities rather than general growth in medical consumables. Gynecologic examinations and interventions require sustained tactile precision, controlled grip, and reliable barrier protection during intimate and often prolonged contact. General examination gloves frequently compromised sensitivity or fit, affecting clinician performance during cervical screening, intrauterine device placement, and obstetric assessments. Clinicians began favouring gloves designed for anatomical fit, extended cuff length, and consistent thickness to maintain control and reduce hand fatigue. Historical demand reflected clinician driven selection within hospitals and clinics, shaped by procedure complexity, patient comfort considerations, and infection control experience rather than centralised procurement mandates.
Future demand for gynecology gloves is expected to reflect formalisation of procedure specific standards and workforce protection priorities. Healthcare systems are placing greater emphasis on exposure reduction, allergy management, and ergonomic performance for clinicians performing repetitive examinations. Procurement policies increasingly differentiate gloves by use case rather than treating them as interchangeable supplies. Material choices such as non latex polymers and low extractable formulations are being specified to address sensitivity risks for both patients and staff. Demand growth will align with guideline updates, occupational safety requirements, and standardisation of obstetric and gynecologic care protocols rather than simple volume expansion in healthcare services.
The gynecology gloves market is driven by high procedure volumes in women’s healthcare, infection control protocols, and routine examination requirements across clinical settings. Demand concentrates on glove types that balance tactile sensitivity, barrier protection, and cost efficiency. Application patterns are shaped by frequency of use, procedural risk level, and regulatory hygiene standards. Market leadership reflects daily clinical practice needs rather than episodic surgical demand, with purchasing decisions influenced by safety compliance, clinician preference, and supply reliability.
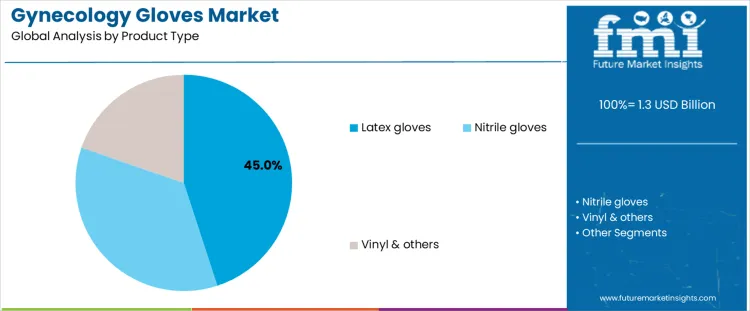
Latex gloves account for about 45% of demand because they offer superior elasticity, tactile sensitivity, and fit, which are critical during gynecological examinations. Clinicians rely on latex gloves for precision during pelvic exams, cervical screenings, and routine diagnostic procedures where manual dexterity is essential. Their widespread availability and cost effectiveness further reinforce adoption in high volume clinical environments. Despite concerns around latex sensitivity, controlled use and powder free variants have sustained acceptance in many regions. Nitrile gloves are increasingly adopted where latex allergy risk management is prioritized, particularly in hospitals with strict occupational safety policies. They provide strong puncture resistance and chemical protection but are generally less elastic. Vinyl and other materials serve low risk, short duration tasks but lack durability. Product type leadership therefore reflects balance between tactile performance, clinician familiarity, and unit cost rather than material innovation alone.
Latex dominance is also supported by entrenched procurement contracts and predictable supply chains. Nitrile growth remains steady but supplementary. Vinyl and alternative materials remain marginal due to performance limitations.
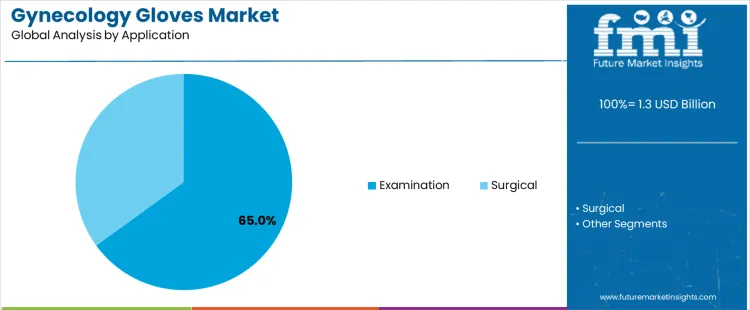
Examination applications represent about 65% of demand because gynecological care involves frequent routine assessments rather than surgical intervention. Pelvic exams, prenatal checkups, fertility consultations, and outpatient diagnostic procedures require gloves for every patient interaction, driving consistent consumption. These settings prioritize comfort, sensitivity, and rapid glove changes, favoring products optimized for examinations. Surgical applications account for a smaller share because gynecologic surgeries occur less frequently and use specialized sterile glove protocols with lower turnover volumes. Examination driven demand is reinforced by preventive care programs, screening initiatives, and rising outpatient visit volumes in women’s health clinics.
Hospitals and clinics stock examination gloves in significantly higher quantities than surgical gloves due to daily patient flow. Surgical use remains critical but episodic, often involving separate procurement channels. Application leadership therefore reflects frequency of clinical contact and routine care intensity rather than procedural complexity. As preventive gynecology services expand, examination driven glove demand continues to anchor overall market consumption patterns.
Use appears in hospitals, clinics, and specialized gynecology centers where sterile barrier protection is essential during examinations, procedures, and surgeries. Healthcare professionals adopt gloves to prevent cross-contamination between patients and providers while maintaining tactile sensitivity for diagnostic and operative tasks. Obstetrics and reproductive health services use gloves during deliveries, intrauterine procedures, and assisted reproductive technologies. These applications reflect operational and infection-control priorities rather than aesthetic purposes, with adoption driven by sterility, material performance, and patient safety.
Selection aligns with gloves that provide barrier protection against pathogens while ensuring comfort, flexibility, and reduced allergic potential. Materials such as nitrile, latex, or synthetic blends are optimized for sensitivity, tensile strength, and chemical resistance. Healthcare teams monitor proper sizing, donning procedures, and single-use protocols to maintain hygiene. Regulatory compliance with standards such as ASTM, EN, or ISO ensures consistent sterility and performance. These conditions emerge from operational efficiency, clinical safety, and regulatory adherence priorities in structured gynecology workflows.
Cost per pair may influence selection in resource-constrained facilities. Allergies to latex or certain additives require alternative materials. Supply chain reliability is critical for maintaining stock during high-volume procedures. Improper usage, storage, or handling can compromise sterility. These factors lead to selective deployment where infection control, tactile performance, and patient safety justify incremental cost and operational oversight.
| Country | CAGR (%) |
|---|---|
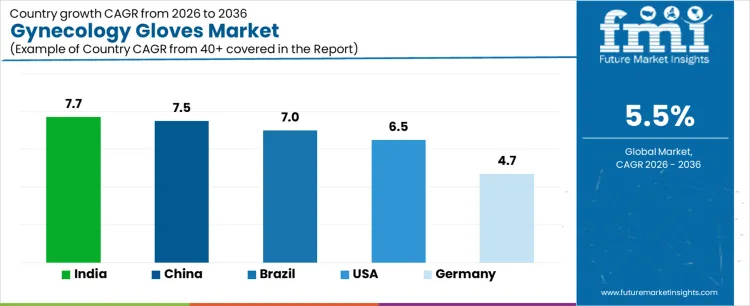
| USA | 6.5% |
| Brazil | 7.0% |
| China | 7.5% |
| India | 7.7% |
| Germany | 4.7% |
The demand for gynecology gloves varies across countries, driven by healthcare infrastructure, surgical procedures, and rising awareness of infection control. India leads with a 7.7% CAGR, supported by increasing gynecological procedures, expanding hospital networks, and growing adoption of disposable medical gloves. China follows at 7.5%, driven by large scale healthcare facilities and increasing surgical interventions. Brazil records 7.0%, shaped by rising healthcare access and hospital infrastructure expansion. The USA grows at 6.5%, reflecting steady adoption in hospitals and specialized clinics. Germany posts 4.7%, supported by mature healthcare systems and regulated glove usage in gynecology and surgical applications.
In the United States, revenue from the Gynecology Gloves Market is growing at a CAGR of 6.5% through 2036, driven by adoption of single-use, high-quality gloves in hospitals, clinics, and outpatient gynecology centers. Manufacturers are supplying sterile, hypoallergenic gloves to improve procedural hygiene, reduce infection risks, and enhance clinician safety. Demand is concentrated in maternity wards, gynecological surgical units, and diagnostic centers. Domestic suppliers provide medical-grade gloves compatible with existing clinical protocols and sterilization standards. Rising patient volumes, healthcare regulations, and recurring glove usage are sustaining predictable procurement nationwide.
In Brazil, the market is growing at a CAGR of 7% through 2036, supported by increasing investment in maternal care, gynecology services, and outpatient clinics. Manufacturers are providing sterile, high-performance gloves to maintain procedural hygiene and reduce contamination during gynecological exams and surgeries. Demand is concentrated in maternity wards, specialized gynecology units, and hospitals. Local suppliers deliver gloves that meet medical standards and regulatory requirements. Rising patient care programs, increased procedural volumes, and recurring glove usage are sustaining adoption nationwide.
China is witnessing growth at a CAGR of 7.5% through 2036, driven by rapid expansion of hospitals, women’s health clinics, and outpatient gynecology services. Manufacturers are integrating sterile, single-use gloves into clinical workflows to enhance infection control and clinician safety. Demand is concentrated in urban hospitals, specialty clinics, and maternity care centers. Domestic suppliers are scaling production of high-performance, medical-grade gloves compatible with sterilization and hygiene protocols. Expanding healthcare facilities, patient awareness, and recurring clinical usage are sustaining rapid adoption nationwide.
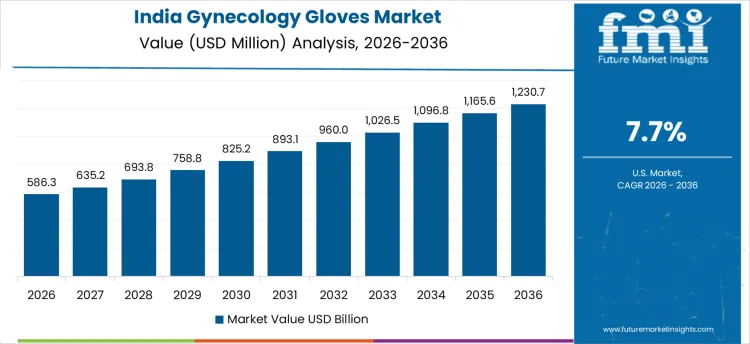
In India, revenue from the Gynecology Gloves Market is expanding at a CAGR of 7.7% through 2036, supported by rising maternal care services, gynecological procedures, and outpatient clinics. Hospitals and clinics are adopting sterile, high-quality gloves to improve hygiene, prevent infections, and support safe clinical practice. Demand is concentrated in maternity wards, gynecology surgery units, and diagnostic centers. Domestic suppliers provide medical-grade gloves compatible with sterilization standards and clinical protocols. Expansion of women’s health services, procedural volumes, and recurring glove usage are sustaining robust adoption nationwide.
Germany is witnessing growth at a CAGR of 4.7% through 2036, driven by adoption of sterile gynecology gloves in hospitals, clinics, and specialized outpatient centers. Manufacturers are providing high-quality gloves to maintain hygiene, reduce contamination, and support safe procedures in gynecological and obstetric care. Demand is concentrated in maternity wards, hospital gynecology units, and diagnostic centers. Domestic suppliers provide medical-grade, sterilization-compatible gloves that meet stringent regulatory standards. Healthcare compliance, recurring procedural requirements, and clinician safety protocols are sustaining consistent procurement nationwide.
Competition in the Gynecology Gloves Market is shaped by procurement discipline, regulatory compliance, and consistency of supply rather than by product differentiation alone. Top Glove, Hartalega, Kossan, and Supermax compete primarily on manufacturing scale, quality control, and the ability to meet large volume hospital tenders with stable specifications. These suppliers emphasize automation, yield control, and standardized nitrile and latex formulations that satisfy medical examination and gynecological procedure requirements. Ansell follows a different strategy, focusing on clinical assurance, brand trust, and portfolio depth across surgical and examination gloves, which supports premium positioning in regulated healthcare settings. Purchasing decisions are driven by defect rates, tactile sensitivity, barrier reliability, and audit readiness rather than incremental material innovation.
Strategic positioning also reflects geographic reach and risk management. Large Asian manufacturers prioritize diversified customer bases across public hospitals, private clinics, and distributors to reduce exposure to regional demand cycles. Capacity expansion is paced conservatively to avoid price volatility, while investments in compliance documentation and traceability support access to regulated markets. Quality incidents and supply disruptions carry long term consequences, making reliability a core competitive asset. Smaller and regional producers participate through niche contracts or local distribution advantages rather than scale. Across the market, competition favors operational discipline, cost control, and regulatory credibility. Success depends on meeting institutional procurement standards consistently over time rather than on marketing intensity or rapid product line expansion.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Product Type | Latex gloves, Nitrile gloves, Vinyl & others |
| Application | Examination, Surgical |
| End-Use Industry | Hospitals, Clinics, Retail/others |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | Top Glove, Hartalega, Ansell, Kossan, Supermax, Others |
| Additional Attributes | Dollar by sales by product type, Dollar by sales by application, Dollar by sales by end-user, Dollar by sales by region, Annual procurement cycles, Glove sterility and compliance, Allergy-free / low-protein formulations, Tactile sensitivity and dexterity ratings, Material durability and puncture resistance, Hospital and clinic stocking levels, Regulatory adherence (ASTM, ISO, EN standards), Procedural volume coverage, Disposable usage rates, Training and handling protocols, Repeat procurement scheduling, Packaging and distribution formats, Integration with infection control policies, Domestic vs. imported product distribution, Supply chain reliability and batch traceability |
How big is the gynecology gloves market in 2026?
The global gynecology gloves market is estimated to be valued at USD 1.3 billion in 2026.
What will be the size of gynecology gloves market in 2036?
The market size for the gynecology gloves market is projected to reach USD 2.2 billion by 2036.
How much will be the gynecology gloves market growth between 2026 and 2036?
The gynecology gloves market is expected to grow at a 5.5% CAGR between 2026 and 2036.
What are the key product types in the gynecology gloves market?
The key product types in gynecology gloves market are latex gloves, nitrile gloves and vinyl & others.
Which application segment to contribute significant share in the gynecology gloves market in 2026?
In terms of application, examination segment to command 65.0% share in the gynecology gloves market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.