Hernia Mesh Devices Market
This report provides a thorough overview of the hernia mesh devices market, addressing market size, revenue forecast, competitive landscape, demand patterns, growth drivers, restraints, notable industry trends, supply chain evolution, and emerging growth opportunities.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Hernia Mesh Devices Market Size, Market Forecast and Outlook By FMI
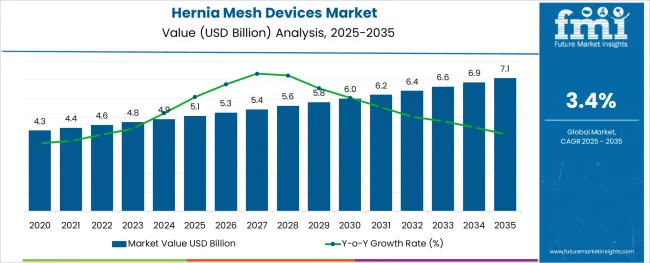
In 2025, the hernia mesh devices market was valued at USD 5.10 billion. Based on Future Market Insights' analysis, demand is estimated to grow to USD 5.27 billion in 2026 and USD 7.37 billion by 2036. FMI projects a CAGR of 3.40% during the forecast period.
Summary of Hernia Mesh Devices Market
- Hernia Mesh Devices Market Definition
- Hernia mesh devices are implantable surgical prostheses constructed from synthetic (polypropylene, polyester, ePTFE) or biological (acellular dermal matrix, porcine submucosa) materials used to reinforce weakened abdominal wall tissue during inguinal, incisional, femoral, and ventral hernia repair procedures.
- Demand Drivers in the Market
- Global hernia repair procedure volumes exceeding 20 million annually, per International Hernia Collaboration estimates, sustain baseline mesh device procurement, with inguinal hernia repair accounting for approximately 75% of total procedure volume.
- Lightweight large-pore polypropylene mesh adoption driven by clinical evidence demonstrating reduced chronic post-operative pain and lower foreign body sensation rates compared to heavyweight mesh predecessors is creating product replacement cycles within established surgical practices.
- Biological mesh demand for contaminated-field hernia repair (incisional hernia with concurrent bowel surgery) is growing as surgeons recognize that synthetic mesh placement in contaminated surgical fields carries unacceptable infection and explantation risk.
- Key Segments Analyzed in the FMI Report
- Synthetic Mesh mesh type: 61.0% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- Inguinal Hernia hernia type: 48.7% share in 2026, reflecting its structural position as the primary category across established procurement channels.
- China: 4.60% compound growth through 2036, indicating concentrated demand acceleration.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare observe that The hernia mesh devices market is a mature surgical implant category where product liability litigation has fundamentally reshaped manufacturer material development priorities. FMI analysts observe that synthetic mesh commands 61.0% type share because polypropylene and ePTFE materials deliver the tensile strength and tissue integration properties required for durable hernia repair at per-unit costs that biological mesh alternatives, priced 5x to 15x higher, cannot match for clean-field inguinal repair. Inguinal hernia at 48.7% hernia type share dominates because it is the most common surgical hernia presentation globally. The moderate 3.4% CAGR reflects the mature surgical volume base, with growth coming from developing-market access expansion and biological mesh adoption in contaminated-field applications rather than increased procedure rates in developed markets.
- Strategic Implications / Executive Takeaways
- Mesh manufacturers must invest in long-term post-market clinical registries documenting 5-year and 10-year recurrence and chronic pain rates to defend against litigation claims and satisfy surgeon procurement committee evidence requirements.
- Surgeons performing contaminated-field incisional hernia repair should evaluate biological mesh options where synthetic mesh infection risk is clinically unacceptable, despite the higher per-unit device cost.
- Medical device companies should develop composite mesh designs with absorbable anti-adhesion barriers for intraperitoneal placement, addressing the growing laparoscopic and robotic ventral hernia repair segment.
Hernia Mesh Devices Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.27 Billion |
| Industry Value (2036) | USD 7.37 Billion |
| CAGR (2026 to 2036) | 3.40% |
Source: Future Market Insights, 2026
The market is set to add approximately USD 2.09 billion in absolute terms between 2026 and 2036. Growing hernia repair surgical volumes driven by aging populations and rising obesity rates sustain mesh device demand, while litigation-driven product liability concerns (transvaginal mesh recall precedent) are compelling manufacturers to invest in biocompatible material development and post-market surveillance programmes. Lightweight large-pore polypropylene mesh designs are displacing heavyweight small-pore predecessors based on clinical evidence of reduced chronic pain and foreign body sensation. As per FMI, country-level growth rates through 2036 are projected as follows: China at 4.60%, India at 4.30%, Germany at 3.90%, France at 3.60%, UK at 3.20%, USA at 2.90%, Brazil at 2.60%. China records the fastest expansion driven by concentrated institutional investment and infrastructure development.
Hernia Mesh Devices Market Definition
The hernia mesh devices market encompasses implantable surgical prostheses engineered for abdominal wall reinforcement during hernia repair procedures. Synthetic mesh products utilize polypropylene, polyester, and ePTFE materials in lightweight large-pore and heavyweight small-pore configurations, while biological mesh products employ decellularized extracellular matrix from human or animal tissue sources. FMI is of the opinion that the market is in a material specification evolution phase where lightweight synthetic designs and biological alternatives are displacing first-generation heavyweight mesh products, driven by clinical outcome data and product liability risk management considerations.
Hernia Mesh Devices Market Inclusions
Market scope covers synthetic polypropylene, polyester, and ePTFE mesh implants, biological mesh derived from acellular dermal matrix and porcine submucosa, and composite mesh devices combining synthetic and absorbable components for all hernia repair indications.
Hernia Mesh Devices Market Exclusions
Suture-only hernia repair without mesh reinforcement, hernia trusses and external support garments, and general surgical suture materials not classified as mesh implants are excluded.
Hernia Mesh Devices Market Research Methodology
- Primary Research: Analysts conducted structured interviews with procurement managers, product engineers, and distribution channel operators to map purchase decision triggers and specification requirements across key verticals.
- Desk Research: Data collection aggregated regulatory filings, industry standards documentation, patent registries, trade body publications, and company annual reports across all target geographies.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of product shipment volumes and average selling prices, validated against publicly disclosed revenue figures from leading suppliers.
- Data Validation and Update Cycle: Projections are cross-referenced against quarterly earnings data, import-export trade statistics, and industry association production reports to maintain forecast integrity.
Key Factors Driving Global Hernia Mesh Devices Market
According to a research published in the United Kingdom, the prevalence of hernias rises with age. The prevalence of hernia was found to be 5%, 10%, 18%, 24%, 31%, and 45% among the age groups of 25 - 34 years, 35 - 44 years, 45 - 54 years, 55 - 64 years, 65 - 74 years and 75 years and above, respectively.
The advancements such as self-fixating meshes and articulating fixation are achieved to overcome the issues, which makes it a key factor for the growth in the adoption of hernia mesh devices. The cost of raw material for hernia mesh though subjected to market volatility has never crossed USD 1,500 in any year providing a necessary profit margin to the manufacturer of hernia mesh devices.
Potential of 3D-Printed Biological Mesh in Hernia Repair
Over 4.9 Million hernia operations take place worldwide annually. Non-degradable hernia surgical mesh implants are characterized by poor healing response, and in vivo erosion which leads to over 10% failure rate and an estimated 42% of recurrent hernias. In 4.921, the average global price of polypropylene was USD 1,285 per ton as compared to 4.94.9, while prices have increased roughly by 36% between these years.
This increase in the price of raw materials for making synthetic mesh adversely affected the market revenue of polypropylene hernia mesh devices.
The 3-D printed biological mesh minimized post-surgical complications of hernia repair and its raw material such as human or porcine dermis is widely available and hence not subjected to market volatility effectively making their price constant. The manufacturing of biological mesh is highly reproducible and scalable and it can be modified depending on the patient’s background.
The cost-utility of a biological mesh device with a 3-D printing technique for the treatment of hernia is not cost-effective for the hospital or surgeons who pay the retail price of the product but it is cost-effective from a third-party payer perspective like medical reimbursement by the government interventions. The cost of 3D printed biological mesh will be USD 1,813 which is cost-effective regardless of size, furthermore making it significantly lower than the current retail cost of synthetic mesh.
Regional Performance and Growth Trends in the Hernia Mesh Devices Market
Why does North America have a growing Hernia Mesh Market?
North America is anticipated to grow at 3.4% CAGR during the forecast period 2026 to 2036. In North America, the reimbursement policy for hernia mesh is favorable to the patients, which encourages them to choose laparoscopic surgery.
Patients have simple access to detailed payment codes and ratios that governments and industry players make public. In addition, technical improvements and the introduction and acceptance of new products, such as TELA Bio, are boosting the market forward.
Analysis of Top Countries Using Hernia Mesh Devices
.webp)
How does the United States continue to be the Nucleus of the Global Hernia Mesh Devices Market?
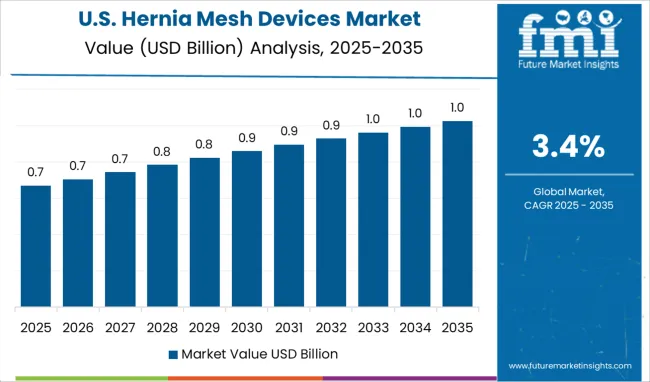
The United States will account for over 34% of the global Hernia Mesh Devices Market by the year 2036, representing it as a fertile ground for hernia mesh device manufacturers, especially synthetic mesh devices. Hernia Mesh Devices Market will grow a USD 2.9% CAGR between 2026 and 2036 for the United States.
The global annual death rate of hernia per 4.300 thousand individuals is 0.5, according to Health Grove. According to statistics by FDA, more than 4.3 Million hernia procedures happen annually in the United States out of which, about 800 thousand procedures are of the inguinal type, making it one of the largest market for Hernia Mesh Devices.
How is China Gearing Up to Become the Next Hotspot for Hernia Mesh Devices?
Hernia mesh devices revenue in China is expected to witness an absolute dollar opportunity of USD 5.4.3 Million during 2026 - 2036. With a projected CAGR of 2.6%, China will become a USD 7.4.3 Million Hernia Mesh Devices Market by the end of 2036.
It was observed that the number of tension-free hernia repair operations in China reached 4.3 Million in 204.35 with an increase of 20%-30% in such operations each year. In particular, an inguinal hernia is more prominent with a male-to-female ratio of 8.2 to 4.3.
According to China’s Hernia Registry, adult inguinal hernia also accounts for the largest proportion of 83.3%. A report by medical service information, 204.38, the cost of a single case of inguinal hernia was USD 922.6, indicating that cost control in county-wide hospitals was good, further creating a robust demand for hernia mesh devices in China.
Hernia Mesh Devices Industry Analysis by Top Investment Segments
Why does Revenue through Inguinal Hernia Mesh Stands Highest?
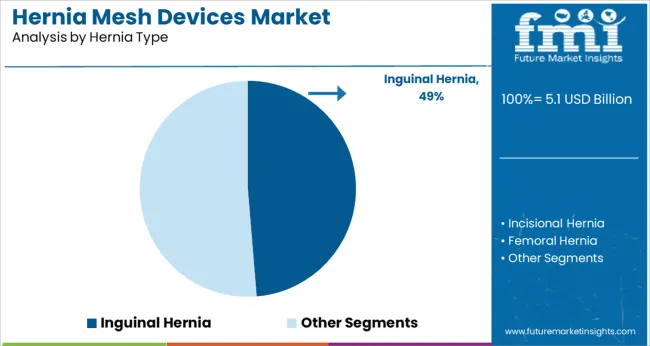
Inguinal hernia constitutes the most revenue from sales of hernia mesh devices. Inguinal hernias account for over 80% of all hernias. It is also 10X more common in men than in women, thus necessitating the need for correction.
Hernia repair for primary and incisional hernias is the most common abdominal surgery performed all around the globe, according to the International Journal of Abdominal Wall and Hernia Surgery. Men have a 27-43% and women have a 3-6% lifetime risk of developing an inguinal hernia. It is expected to trigger the growth of hernia repair market.
Patient feasibility due to tension-free procedures and post-operative complications involved in classic surgical techniques are also contributing to the demand for Inguinal Hernia Mesh. In addition, the recurrence rate for inguinal hernia is 1-3%, which further imparts strain on the hernia mesh devices revenue.
Which Hernia Mesh Type accounts for Highest Revenue in the Hernia Mesh Devices Market?
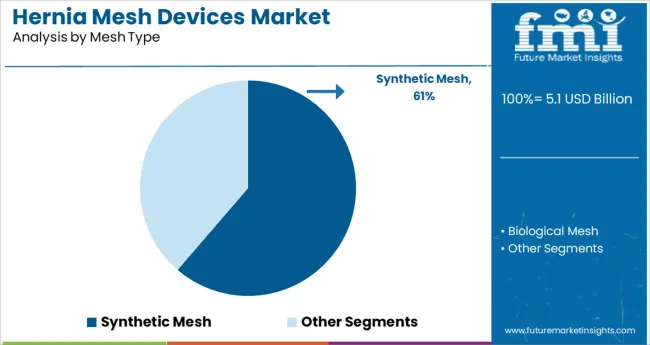
A procedure of synthetic mesh for hernia repair surgery which ranges from USD 4,200 to USD 12,500 has hurt the capitalization of the Hernia Mesh Devices Market. Synthetic Mesh is used in around 90% of hernia surgeries according to FDA. In the Asia Pacific where 99% of Hernia surgery is performed with Synthetic Mesh, there is a significant growth opportunity for biological mesh for based hernia repair devices.
On the other hand, the biological mesh has yet to reach the majority of the consumers as it is still in the research process hence it captures only around 1% of the global Hernia Mesh Devices market.
Competitive Landscape of the Hernia Mesh Devices Market
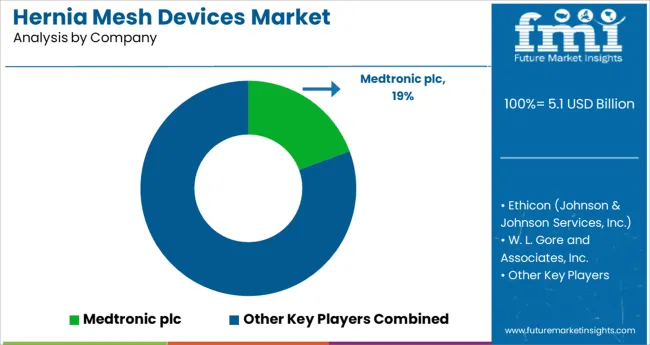
Most of the leading players in hernia mesh devices market have been focusing on obtaining approvals from regulatory authorities.
The key companies operating in the Hernia Mesh Devices market include Medtronic plc, Ethicon, and W. L. Gore and Associates, Inc., Atrium Medical Technologies, B. Braun Melsungen AG, PRIMEQUAL SA, Becton, Dickinson, and Company, Deep Blue Medical Inc., Dipromed Srl, BioCer Entwicklungs-GmbH, Betatech Medical, Sinolinks Medical Innovation, Inc, Aspide Medical S.A.S., Meril Life Sciences Pvt. Ltd., Herniamesh Srl., Changzhou Medical Equipment General Factory Co., Ltd, Gem Srl, SAMYANG HOLDING CORPORATION, Katsan Katgut Sanayi Ve Ticaret Anonim Sirketi, Novus Scientific AB.
Similarly, recent developments related to companies manufacturing hernia Mesh Devices have been tracked by the team at Future Market Insights, which is available in the full report.
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 5.27 Bn to USD 7.37 Bn, at a CAGR of 3.40% |
| Market Definition | Hernia mesh devices are implantable surgical prostheses constructed from synthetic (polypropylene, polyester, ePTFE) or biological (acellular dermal matrix, porcine submucosa) materials used to reinforce weakened abdominal wall tissue during inguinal, incisional, femoral, and ventral hernia repair procedures. |
| Mesh Type Segmentation | Synthetic Mesh, Biological Mesh |
| Hernia Type Segmentation | Inguinal Hernia, Incisional Hernia, Femoral Hernia, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | Medtronic plc, Ethicon (Johnson & Johnson), W. L. Gore and Associates, Inc., Atrium Medical Technologies, B. Braun Melsungen AG, Cook Medical, Integra Lifesciences, Dipro Medical Devices S.R.L., Via Surgical, PRIMEQUAL SA, Becton Dickinson, Meril Life Sciences Pvt. Ltd. |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down modeling validated against primary research inputs and publicly available industry data. |
Top Segments Studied in the Hernia Mesh Devices Market Research Report
By Mesh Type:
- Biological Mesh
- Synthetic Mesh
By Hernia Type:
- Inguinal Hernia
- Incisional Hernia
- Femoral Hernia
- Others
By Region:
- North America
- Latin America
- Europe
- South Asia
- East Asia
- Oceania
- Middle East & Africa
Bibliography
- [1] International Hernia Collaboration. (2024). International Guidelines for Groin Hernia Management Update. Hernia Journal.
- [2] USA Food and Drug Administration. (2024). Surgical Mesh for Hernia Repair: Device Classification and Guidance. FDA.
- [3] European Commission. (2024). Medical Device Regulation (EU) 2017/745: Implantable Mesh Classification. European Commission.
- [4] Ethicon (Johnson & Johnson). (2024). Hernia Mesh Product Portfolio Technical Specifications. Ethicon.
- [5] Medtronic plc. (2024). Annual Report 2024: Surgical Innovations Division. Medtronic.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Hernia Mesh Devices in the global market in 2026?
Demand for Hernia Mesh Devices in the global market is estimated to be valued at USD 5.27 billion in 2026.
What will be the market size of Hernia Mesh Devices by 2036?
Market size for Hernia Mesh Devices is projected to reach USD 7.37 billion by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 3.40% between 2026 and 2036.
Which Mesh Type is poised to lead by 2026?
Synthetic Mesh accounts for 61.0% in 2026.
How significant is Inguinal Hernia in driving adoption?
Inguinal Hernia represents 48.7% of segment share in 2026.
What country records the fastest growth?
China is projected to grow at a CAGR of 4.60% during 2026 to 2036.
What is the projected growth for India?
India is projected to expand at a CAGR of 4.30% during 2026 to 2036.
What is included in the scope of this report?
Market scope covers synthetic polypropylene, polyester, and ePTFE mesh implants, biological mesh derived from acellular dermal matrix and porcine submucosa, and composite mesh devices combining synthetic and absorbable components for all hernia repair indications.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Mesh Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Mesh Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Mesh Type , 2026 to 2036
- Synthetic Mesh
- Biological Mesh
- Synthetic Mesh
- Y to o to Y Growth Trend Analysis By Mesh Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Mesh Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Hernia Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Hernia Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Hernia Type, 2026 to 2036
- Inguinal Hernia
- Incisional Hernia
- Femoral Hernia
- Others
- Inguinal Hernia
- Y to o to Y Growth Trend Analysis By Hernia Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Hernia Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Mesh Type
- By Hernia Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Mesh Type
- By Hernia Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Mesh Type
- By Hernia Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Mesh Type
- By Hernia Type
- Competition Analysis
- Competition Deep Dive
- Medtronic plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Ethicon (Johnson & Johnson Services, Inc.)
- W. L. Gore and Associates, Inc.
- Atrium Medical Technologies
- B. Braun Melsungen AG
- Cook Medical
- Integra Lifesciences
- Dipro Medical Devices S.R.L.
- Via Surgical
- PRIMEQUAL SA
- Becton
- Dickinson and Company
- Deep Blue Medical Inc.
- BioCer Entwicklungs-GmbH
- Betatech Medica
- Sinolinks Medical Innovation Inc.
- Aspide Medical S.A.S.
- Meril Life Sciences Pvt. Ltd.
- Herniamesh Srl.
- Changzhou Medical Equipment General Factory Co. Ltd.
- Medtronic plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Mesh Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Hernia Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Mesh Type
- Figure 6: Global Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Hernia Type
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Mesh Type
- Figure 23: North America Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Hernia Type
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Mesh Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Hernia Type
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Mesh Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Hernia Type
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Mesh Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Hernia Type
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Mesh Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Hernia Type
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Mesh Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Hernia Type
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Mesh Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Mesh Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Mesh Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Hernia Type, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Hernia Type, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Hernia Type
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

