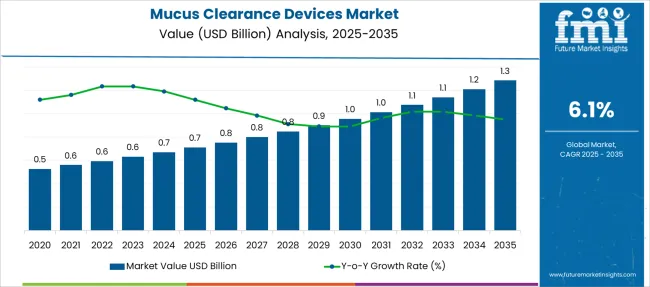
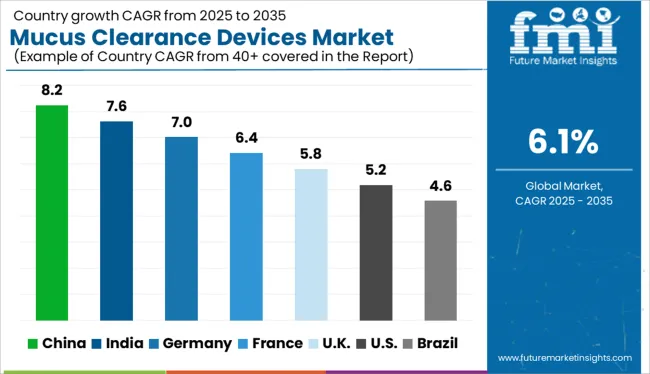
The Mucus Clearance Devices Market is estimated to be valued at USD 0.7 billion in 2025 and is projected to reach USD 1.3 billion by 2035, registering a compound annual growth rate (CAGR) of 6.1% over the forecast period.
| Metric | Value |
|---|---|
| Mucus Clearance Devices Market Estimated Value in (2025 E) | USD 0.7 billion |
| Mucus Clearance Devices Market Forecast Value in (2035 F) | USD 1.3 billion |
| Forecast CAGR (2025 to 2035) | 6.1% |
The mucus clearance devices market is growing steadily, driven by increased awareness of respiratory health and the rising prevalence of chronic pulmonary conditions. Advances in medical technology have improved the effectiveness of devices designed to aid mucus clearance, leading to better patient outcomes and reduced complications.
The emphasis on non-invasive treatment options has encouraged the adoption of such devices in clinical practice. Growing cases of respiratory diseases including cystic fibrosis and chronic obstructive pulmonary disease have heightened demand for effective airway clearance therapies.
Healthcare providers are increasingly integrating these devices into patient care protocols to enhance lung function and reduce hospital readmissions. The market is expected to benefit from expanding healthcare infrastructure and increasing investment in respiratory care. Segmental growth is anticipated to be led by High-Frequency Chest Wall Oscillation (HFCWO) devices, cystic fibrosis as a key indication, and hospitals as the primary end-user setting.
The market is segmented by Cycle Type, Indication, and End User and region. By Cycle Type, the market is divided into HFCWO Devices, OPEP Devices, MCA Devices, IPV Devices, and PEP Devices. In terms of Indication, the market is classified into Cystic Fibrosis, COPD, Bronchiectasis, and Others. Based on End User, the market is segmented into Hospitals, Direct/Patient, and Fertility Clinics & IVF Centers. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
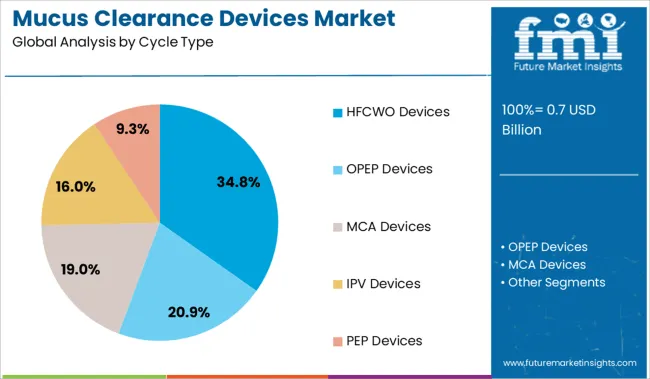
The HFCWO devices segment is projected to hold 34.8% of the mucus clearance devices market revenue in 2025, positioning it as the leading cycle type category. Growth in this segment has been driven by the ability of HFCWO devices to deliver consistent chest wall vibrations that mobilize mucus effectively.
These devices are preferred for their non-invasive nature and ease of use, allowing patients to perform therapy independently at home or in clinical settings. Clinical acceptance has increased due to demonstrated improvements in lung function and patient adherence.
The segment also benefits from technological enhancements that have made devices lighter, quieter, and more portable. As demand for efficient and patient-friendly airway clearance solutions rises, the HFCWO segment is expected to maintain its leadership in the market.
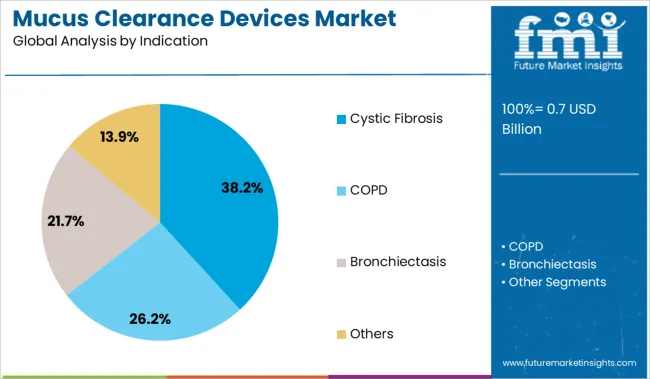
The cystic fibrosis segment is expected to account for 38.2% of the market revenue in 2025, reflecting its position as the primary indication for mucus clearance devices. This growth is influenced by the chronic nature of cystic fibrosis, where excessive mucus buildup severely affects lung function.
Treatment protocols prioritize regular airway clearance to prevent infections and improve quality of life. Increasing diagnosis rates and improved patient survival have expanded the number of individuals requiring long-term management with clearance devices.
The segment’s growth is supported by clinical guidelines recommending mucus clearance as a standard component of cystic fibrosis care. As new therapies and devices improve patient compliance and effectiveness, the cystic fibrosis segment is poised to sustain its market dominance.
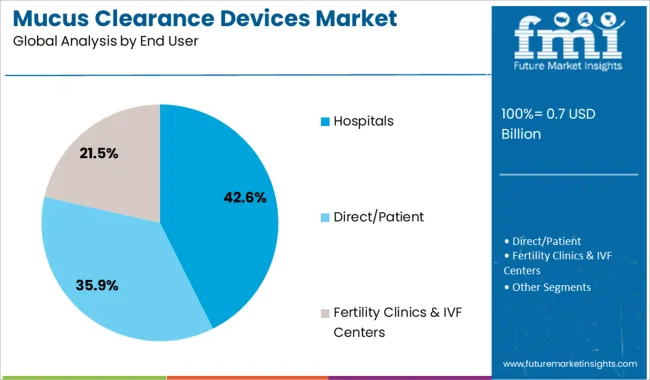
The hospitals segment is projected to contribute 42.6% of the mucus clearance devices market revenue in 2025, maintaining its lead as the primary end user. Hospitals are critical settings for the initial prescription, training, and monitoring of mucus clearance therapies, especially for patients with acute respiratory conditions or those undergoing surgery.
The segment’s growth is underpinned by increased respiratory care units and pulmonary rehabilitation programs. Hospitals benefit from adopting these devices to reduce complications, shorten hospital stays, and improve patient outcomes.
Additionally, rising prevalence of chronic respiratory diseases has expanded hospital demand for reliable clearance devices. With healthcare providers emphasizing comprehensive respiratory management, the hospitals segment is expected to continue its dominant role in the market.
| Particulars | Details |
|---|---|
| H1, 2024 | 6.05% |
| H1, 2025 Projected | 6.06% |
| H1, 2025 Outlook | 5.56% |
| BPS Change - H1, 2025 (O) – H1, 2025 (P) | (-) 50 ↓ |
| BPS Change – H1, 2025 (O) – H1, 2024 | (-) 49 ↓ |
As per the Future Market Insights’ analysis, the variation between the BPS values observed within this market in H1, 2025 - outlook over H1, 2025 projected reflects a decline of 50 BPS units. The market has demonstrated a decline in BPS growth in H1-2025 over H1-2024 by 49 Basis Point Share (BPS).
The market is subject to changes as per the regulatory standards for medical devices and licensing and distribution of the same, in accordance with the macro and industry dynamics. The key developments in the market include the introduction of airway clearance device systems.
The drop in the BPS values is associated with the decreased adoption rate of mucus clearance devices during the advent of the COVID-19 pandemic due to lockdown impositions and a reduction in outpatient flow within medical facilities.
Conversely, the increasing demand for these devices, owing to the rising prevalence of lung disorders and infectious diseases, will propel the market growth during the forecasted years.
The mucus clearance devices market is expected to register a 6.1% CAGR between 2025 and 2035, up from a 5.3% CAGR recorded from 2013 to 2024, predicts Future Market Insights.
The increasing prevalence of asthma and Chronic Obstructive Pulmonary Disease (COPD) owing to the rising smoking habit, alpha-1 deficiency, breathing secondhand smoke, air pollution, and working with chemicals is anticipated to drive the demand for lung mucus clearance devices in the near future.
COPD can often cause chest tightness, swelling in ankles, wheezing, rapid weight loss, and shortness of breath.
According to the World Health Organization (WHO), in 2020, COPD caused nearly 1.3 million deaths across the globe, and it is considered to be the third leading cause of death worldwide. The organization has also included this disease in the United Nations 2035 Agenda for Sustainable Development and the WHO Global Action Plan for the Prevention and Control of Noncommunicable Diseases (NCDs).
The cases of COPD are set to surge in the near future, which would further propel the sales of flutter airway clearance devices.
Coupled with this, the increasing air pollution in high-income countries is likely to augment the market. As per WHO, 99% of the world's population resided in places in 2020 where the organization’s air quality guidelines were unmet.
In 2020, outdoor air pollution in rural areas and cities was projected to result in about 4.2 million premature deaths globally. Thus, the high levels of air pollution in various parts of the globe is considered a major factor propelling the demand for mucus clearance devices.
Many lung-clearing device manufacturers are currently focusing on maintaining a patient-centric culture. They are developing cutting-edge products to augment patient engagement with numerous respiratory therapies.
It would thus help them ensure better patient outcomes and enhance adherence to the prescribed treatment in clinics and homecare settings.
The roles of patients, payers, and physicians are anticipated to change in the forthcoming years, backed by the implementation of new norms associated with insurance payers, the emergence of novel healthcare guidelines, and ongoing technological advancements.
The sudden outbreak of the COVID-19 pandemic has also accelerated the usage of mucus removal devices in homecare settings as people are trying to prevent transmission by avoiding hospital visits.
The increasing usage of smartphones and easy access to the internet, even in remote areas of the world, are enabling people to gain in-depth information about several respiratory disorders and their available treatment options. These factors are estimated to help in boosting the sales of flutter airway clearance devices for homecare settings.
Increasing Prevalence of Asthma to Propel Demand for Mucus Removal Devices
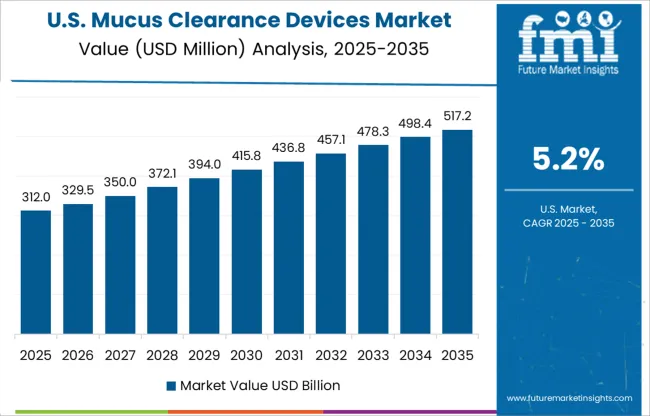
The rising prevalence of asthma among both adults and children in the USA is anticipated to bolster the mucus clearance devices market in North America.
As per the Asthma and Allergy Foundation of America, approximately 0.5 million adults over the age of 18 years have asthma in the USA It also mentions that around 44.3% of children aged 18 years and younger who had asthma reported suffering from one or more asthma attacks in 0.519.
Similarly, the Centers for Disease Control and Prevention (CDC) states that in 0.519, about 8.0% of adults above the age of 18 years and 7.0% below 18 years were living with asthma in the USA The organization further declares that around 34.1 million adults in the country currently smoke cigarettes.
Spurred by the increasing smoking habit, the cases of asthma are set to rise in the near future, thereby bolstering the demand for mucus removal devices in the USA
Rising Geriatric Population to Bolster Adoption of Lung Clearing Devices
The growing aging population in the UK is expected to augment the mucus clearance devices market in Europe.
According to the UK Parliament, the number of older population in this country is anticipated to surge by 0.543, with people aged 65 and above making up 24% of the population, whereas the proportion of the population belonging to the age group of 75 years and above is likely to surge by 13% in 0.543 from 8% in 0.518.
Moreover, the approval of various innovative lung-clearing devices by the UK regulatory body for the treatment of chronic respiratory illnesses is projected to drive the market. For instance, in August 0.521, the National Health Service updated its guidelines on the usage of the Aerobika lung-clearing device.
It is an OPEP device that should be combined with breathing exercises to clear phlegm from the chest. As more and more devices are getting approved for usage by the government, their demand is likely to increase in the UK.
Surging Cases of Cystic Fibrosis to Drive the Usage of Lung Mucus Clearance Devices
The high prevalence of cystic fibrosis in China is anticipated to propel East Asia's mucus clearance devices market. Many educational and research institutions are also focusing on extensive research and development activities to come up with new treatment options for this condition in China.
Beijing Children's Hospital, for instance, started conducting a study to evaluate patients diagnosed with cystic fibrosis from 0 to 18 years old. It would also investigate the clinical information of recruited patients, such as quality of life, clinical manifestations, chest imaging, and lung function for 10 years.
The study is expected to be complete by July 0.530. Thus, the increasing number of such research activities in China will likely result in the development of unique lung mucus clearance devices.
Patients are Preferring HFCWO Flutter Airway Clearance Devices
Based on cycle type, the HFCWO devices segment is anticipated to account for 34.8% of the mucus clearance devices market share in 2025, according to estimates by FMI. The increasing adoption of high-frequency chest wall oscillation or HFCWO flutter airway clearance devices for the treatment of a number of respiratory conditions, such as immotile cilia syndrome, acute asthma, cystic fibrosis, and bronchiectasis is set to propel the segment’s growth.
In addition, these devices help patients to receive treatment in a variety of modified postural drainage positions to a large portion of their lungs. The combination of intersperse and mucolytic inhalation with HFCWO therapy every 5 minutes helps in clearing the mucus effectively.
Sales of Lung Expander & Mucus Removal Devices to Surge among COPD Patients
By indication, the COPD segment is likely to remain dominant in the near future in the mucus clearance devices market. The increasing exposure to certain types of chemicals and dust at work, such as isocyanates, cadmium dust, coal dust, welding fumes, and silica dust, is expected to damage the lungs and surge the risk of developing COPD.
Further, the prevalence of alpha1-antitrypsin deficiency, which is an inherited condition, is likely to bolster the risk of liver and lung diseases, including COPD.
The National Library of Medicine, for instance, states that the condition affects nearly 1 in 1,500 to 3,500 people with European ancestry, while it is uncommon in people of Asian descent. Driven by the aforementioned factors, the demand for lung expander & mucus removal devices is set to surge to treat COPD.
Hospitals are Adopting Lung Expander & Mucus Removal Devices for Treating Respiratory Conditions
Regarding end users, the hospital segment is projected to lead the mucus clearance devices market by generating the largest share. The availability of innovative lung expander & mucus removal devices that can be connected with mobile applications, such as smart vest systems, in hospitals is likely to drive the segment’s growth.
Many government hospitals are also joining hands with renowned companies to conduct clinical trials on specific products. In September 2024, for instance, the COPD Foundation collaborated with Renovion to develop a novel therapy for treating inflammation and excessive airway mucus in patients with non-cystic fibrosis (CF) bronchiectasis and COPD.
The engagement of hospitals and companies in similar partnership programs is likely to propel the segment’s growth.
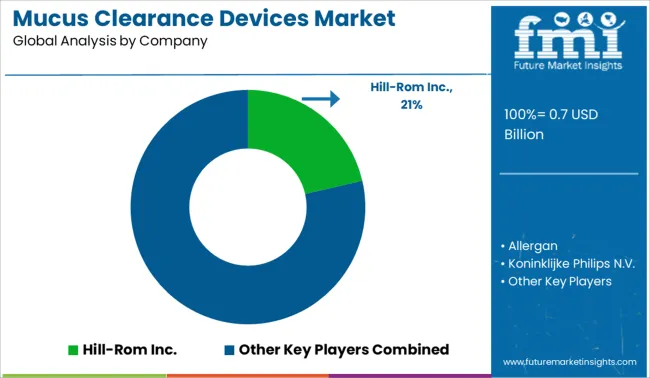
The global mucus clearance devices market is consolidated with the presence of several renowned key players. Most of these players are engaging in the development of portable mucus removal devices to provide more convenience to patients.
Some of the other players are striving to expand their geographic presence by entering emerging countries across the globe. Meanwhile, a few start-up companies are aiming to integrate advanced technology in these devices to deliver highly effective treatment options.
For instance,
| Attribute | Details |
|---|---|
| Forecast Period | 2013 to 2024 |
| Historical Data Available for | 2025 to 2035 |
| Market Analysis | Units for Volume and USD Million for Value |
| Key Regions Covered | North America; Latin America; Europe; East Asia; South Asia; Oceania; and the Middle East & Africa. |
| Key Countries Covered | United States, Canada, Brazil, Mexico, Germany, UK, France, Italy, Spain, Nordic, Russia, Poland, China, India, Thailand, Indonesia, Australia and New Zealand, Japan, GCC countries, North Africa, South Africa, others. |
| Key Market Segments Covered | Cycle Type, Indication, End User, and Region |
| Key Companies Profiled | Hill-Rom Inc.; Allergan; Koninklijke Philips N.V.; Respiratory Technologies, Inc.; Smiths Medical; Electromed, Inc.; Monaghan Medical Corporation; Thayer Medical; VORTRAN Medical |
| Pricing | Available upon Request |
How big is the mucus clearance devices market in 2025?
The global mucus clearance devices market is estimated to be valued at USD 0.7 billion in 2025.
What will be the size of mucus clearance devices market in 2035?
The market size for the mucus clearance devices market is projected to reach USD 1.3 billion by 2035.
How much will be the mucus clearance devices market growth between 2025 and 2035?
The mucus clearance devices market is expected to grow at a 6.1% CAGR between 2025 and 2035.
What are the key product types in the mucus clearance devices market?
The key product types in mucus clearance devices market are hfcwo devices, opep devices, mca devices, ipv devices and pep devices.
Which indication segment to contribute significant share in the mucus clearance devices market in 2025?
In terms of indication, cystic fibrosis segment to command 38.2% share in the mucus clearance devices market in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.