Pneumococcal Testing Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Pneumococcal Testing Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Pneumococcal Testing Market Size and Share Forecast Outlook 2025 to 2035
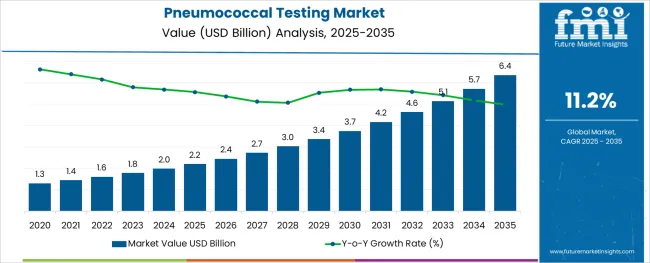
The Pneumococcal Testing Market is estimated to be valued at USD 2.2 billion in 2025 and is projected to reach USD 6.4 billion by 2035, registering a compound annual growth rate (CAGR) of 11.2% over the forecast period.
Quick Stats for Pneumococcal Testing Market
- Pneumococcal Testing Market Industry Value (2025): USD 2.2 billion
- Pneumococcal Testing Market Forecast Value (2035): USD 6.4 billion
- Pneumococcal Testing Market Forecast CAGR: 11.2%
- Leading Segment in Pneumococcal Testing Market in 2025: Immunodiagnostics (43.6%)
- Key Growth Region in Pneumococcal Testing Market: North America, Asia-Pacific, Europe
- Top Key Players in Pneumococcal Testing Market: Thermo Fisher Scientific Inc., Abbott Laboratories, F-Hoffmann la Roche Ltd., Hologic Inc., bioMerieux, Becton, Dickenson and Company, Bio-Rad Laboratories Inc., Meridian Biosciences, Quest Diagnostics, Quidel
Rationale for Segmental Growth in the Pneumococcal Testing Market
Market Overview
The pneumococcal testing market is witnessing steady expansion as healthcare providers focus on early detection of pneumococcal infections that contribute significantly to respiratory illnesses worldwide. Clinical awareness campaigns and immunization programs have emphasized the importance of rapid and accurate diagnosis to prevent disease progression.
Laboratories and diagnostic centers are increasingly adopting modern testing solutions that offer improved sensitivity and faster turnaround times. This market has further gained momentum due to the growing elderly population and the rising incidence of pneumococcal pneumonia among immunocompromised patients.
Developments in assay technologies and laboratory automation have enhanced test efficiency, supporting the broader use of pneumococcal diagnostics in both hospital and outpatient settings. Future growth will be fueled by continuous innovations in detection methods and the increasing need for scalable diagnostic solutions. Segmental growth is expected to be driven by Immunodiagnostics in method, Consumables in product, and Enzyme Linked Immunosorbent Assay (ELISA) in technology, reflecting evolving clinical demand and laboratory practices.
Segmental Analysis
The market is segmented by Method, Product, Technology, and End User and region. By Method, the market is divided into Immunodiagnostics, Molecular Diagnostic, and Point of Care Testing. In terms of Product, the market is classified into Consumables and Analyzers.
Based on Technology, the market is segmented into Enzyme Linked Immunosorbent Assay (ELISA), Immunofluorescence, Western Blot Test, Nucleic Acid Sequence based Amplification, Immunohistochemistry, Polymerase Chain Reaction, and Others. By End User, the market is divided into Hospitals, Ambulatory Surgical Centers, and Clinics. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Method Segment: Immunodiagnostics
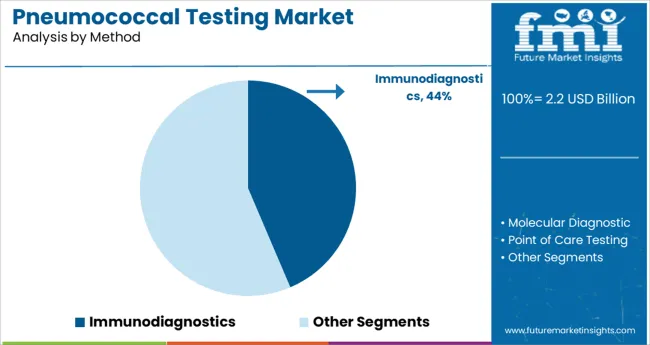
The Immunodiagnostics segment is projected to contribute 43.6% of the pneumococcal testing market revenue in 2025, establishing itself as the dominant method. Growth of this segment is attributed to the ability of immunodiagnostic tests to provide reliable pathogen detection through antigen-antibody interactions. Laboratories have increasingly relied on these tests for their accuracy, scalability, and compatibility with automated systems.
Immunodiagnostics have become integral in screening patients across emergency departments, outpatient clinics, and routine health checkups. Moreover, the simplicity of these tests has supported their use in resource-limited settings where rapid detection is essential for timely clinical intervention.
As healthcare systems continue to prioritize fast and reliable testing for respiratory infections, the Immunodiagnostics segment is expected to sustain its market leadership.
Insights into the Product Segment: Consumables
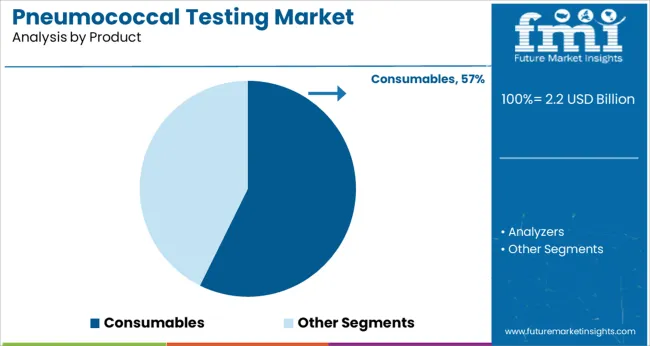
The Consumables segment is expected to account for 57.3% of the pneumococcal testing market revenue in 2025, retaining its position as the leading product segment. Consumables such as reagents, assay kits, and test cartridges are critical components of pneumococcal testing workflows. Diagnostic laboratories have demonstrated a consistent need for these products due to the repeat nature of testing procedures and the demand for maintaining high-throughput operations.
Frequent replenishment of consumables ensures consistent test accuracy and system performance. Product development has also focused on enhancing the stability and ease of handling of these consumables to support better workflow management.
With growing diagnostic volumes and expanding test menus in healthcare laboratories, the Consumables segment is expected to remain the primary revenue contributor in the market.
Insights into the Technology Segment: Enzyme Linked Immunosorbent Assay (ELISA)
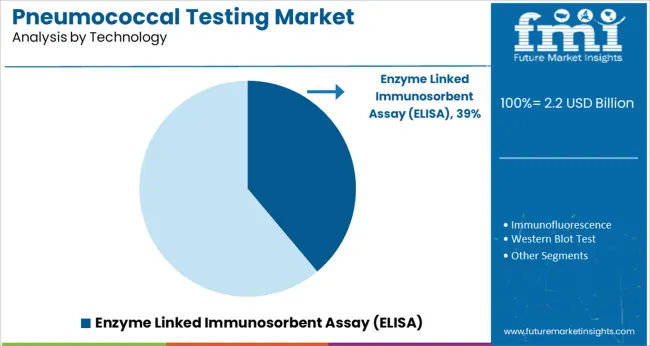
The Enzyme Linked Immunosorbent Assay (ELISA) segment is projected to hold 38.9% of the pneumococcal testing market revenue in 2025, maintaining its leadership among technologies. ELISA has been widely adopted for its sensitivity and specificity in detecting pneumococcal antigens and antibodies. Clinical laboratories have favored ELISA for its ability to process multiple samples simultaneously, making it efficient for routine testing.
The technology’s compatibility with automation platforms has further improved laboratory throughput and minimized manual errors. In addition, ELISA kits have been continuously optimized for better reproducibility and reduced assay time.
As pneumococcal testing becomes a routine component of respiratory disease diagnosis, the Enzyme Linked Immunosorbent Assay (ELISA) segment is expected to remain a preferred choice for laboratories seeking scalable and accurate diagnostic solutions.
How The Market Progressed Till June 2025?
| Particulars | Details |
|---|---|
| H1, 2024 | 11.08% |
| H1, 2025 Projected | 11.21% |
| H1, 2025 Outlook | 11.11% |
| BPS Change - H1, 2025 (O) - H1, 2025 (P) | (-) 10 ↓ |
| BPS Change - H1, 2025 (O) - H1, 2024 | (+) 3 ↑ |
The comparative analysis and market growth rate of global pneumococcal testing market as studied by Future Market Insights, will show a negative BPS growth in H1-2025 outlook as compared to H1-2025 projected period by 10 BPS.
As per the FMI Analysis, the global pneumococcal testing market will observe an increased change in the Basis Point Share (BPS) by nearly 03 units in H1- 2025 (O) as compared to H1-2024.
This was majorly due to the prevalence of high pneumococcal carriage rate among adults and children, adoption of multi-locus sequence analysis (MLSA) for pathogen identification, and high diagnostic yield of pneumococcal testing, promoting this rise in the BPS value.
The key factors boosting the demand for pneumococcal testing are the development of novel biomarkers for rapid disease testing, and the structuring of detection methods for pneumococcal antigens.
Additionally, introduction of pneumococcal conjugate vaccines for invasive pneumococcal disease, and devising of immunization strategies that induce optimum immunogenicity and protection against invasive pneumococcal disease in the elderly have further promoted this surge in the BPS value.
Conversely, challenges linked with the accurate diagnosis of pneumococcal pneumonia will restrain the growth of pneumococcal testing market.
2015-2025 Pneumococcal Testing Market Demand Outlook Compared to 2025-2035 Forecast
Sales of pneumococcal testing grew at a CAGR of 9.8% between 2020 and 2024.
The pneumonia testing market is developing at a decent rate as pneumonia is a serious disease in which testing aids to detect the disease sooner and further, they can be treated rapidly. Furthermore, the awareness level is rising gradually which leads to early diagnosis, thus promoting the development of pneumonia testing market.
The main clinical syndromes of pneumococcal disease are meningitis, bacteremia, and pneumonia. According to the Centers for Diseases Control and Prevention in 2020, reports that 150,000 hospitalizations occurred from pneumococcal pneumonia yearly in the USA
Pneumococci hold for about 30% of community-acquired pneumonia for adults. Bacteremia happens about 25-30% of pneumococcal pneumonia patients. The case-mortality rate is 5-7% and might be much higher amongst geriatric persons. This is expected to boost the demand for the market.
Considering this, FMI expects the global pneumococcal testing market to grow at a CAGR of 11.2% through 2035.
Which Factors Are Restraining Demand for Pneumococcal Testing?
The high cost of testing and limited reimbursement for new or expensive diagnostic tests are the primary factors hampering the pneumococcal testing market growth.
Diagnostic tools for Legionella comprise antigen detection in urine, culture, immunohistochemistry or direct fluorescent antibody staining, serological testing, and polymerase chain reaction (PCR) testing.
Various commercially accessible urinary antigen tests for Legionella provide a quick diagnosis with high specificity. However, a 2020 study to evaluate the Legionella incidence and to understand the cost effectiveness of the test in Central Texas revealed that the projected cost of screening of population with Legionella urinary antigen testing was around USD 214,871.76 above 13 years, with a price per positive patient of USD 12,640.
Concerns and difficulties about reimbursement for expensive or new diagnostic tests may hinder innovation and are an important challenge to the market growth. Coverage for detection tests differs regionally, and given the absence of standards in determining coverage, this can present challenges for dissemination and development of a new diagnostic method.
Country-wise Insights

What Makes the USA a Large Market for Pneumococcal Testing?
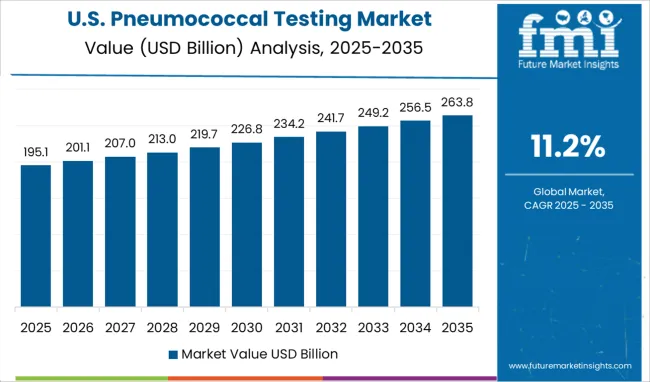
The USA is estimated to account for around 82.9% of the North America pneumococcal testing market in 2025.
USA is anticipated to dominate the pneumonia testing market owing to increasing popularity among the healthcare industry for POC tests, increasing geriatric population, and increasing growth of advanced PCR-based technologies.
For instance, as per the Centers for Disease Control and Prevention (CDC), pneumonia is an eighth primary cause for fatality in U.S. which further result into growth in awareness regarding the disease and further boosts the market.
Also, there are growing product approval and rising product launches that are boosting the overall market in the country. Moreover, with the growing healthcare expenditure and the availability of well-developed healthcare infrastructure is driving the growth of the market in USA.
Why is Germany Considered a Lucrative Pneumococcal Testing Market?
Germany is estimated to account for 31.3% of the Europe pneumococcal testing market in 2025.
The development of the market in Germany is owing to the initiatives undertaken by the government regarding the growth of healthcare infrastructure, and availability of favorable regulations coupled with the high disease incidence levels in the country. Point-of-care testing is widely used in developed countries owing to high awareness as compared to the developing countries and improved reimbursement policies.
What are the Factors Fueling the Demand for Pneumococcal Testing in China?
The pneumococcal testing market in China is estimated to be worth USD 2.2 Million in 2025 in the global market.
The key factors fueling the development of the market include growing technological advancement in point-of-care diagnostics devices coupled with rising adoption of inorganic development strategies like agreements and collaborations to produce novel devices. This is projected to drive the market development in China. In addition, rising prevalence of pneumonia and the growing demand for its diagnosis via testing is another key factor adding to the development of the pneumococcal testing market in China.
How will the Pneumococcal Testing Market Expand across India?
Demand for pneumococcal testing in India is expected to rise at around 13.7% CAGR over the forecast period.
The rising elderly population and the rising awareness concerning pneumococcal testing are some of the key factors that will drive the market development in the country. The rising geriatric population is more susceptible to pneumonia which is growing the demand for pneumococcal testing.
Category-wise Insights
Demand for Point of Care Testing to continue gaining Traction
By method, point of care testing is anticipated to hold the maximum share of 43.0% in 2025, expanding at rate of nearly 11.7% during the forecast period.
POC testing is an innovative technology that is presently an ideal technique for diagnosing pneumonia. The rising adoption of POC testing is owing to growing cases of pneumonia, higher efficiency of POC testing and the rapid test results, growing its preference over other testing methods.
Thus, with the increasing prevalence of chronic diseases and huge customer base adopting the inexpensive and instant diagnostic tests, is boosting the point of care testing. The benefits of POC test to render accurate and rapid results is also aiding in encouraging the patient-centered healthcare at home care settings which leads to adoption of point of care testing and thus encouraging the development of pneumonia testing market.
Why Analyzers will hold the most of the market share?
By Product, analyzers will lead the market and is projected to account for 59.7% of the total market revenue share in 2025.
The several advantages offered by analyzers is increasing the demand for analyzers thus increasing the market share of this segment. The advantages offered by analyzers include optimized time-consuming analytical and testing processes, reliable diagnostic information, accurate and timely analysis of samples and improved patient and wellness care.
Why Enzyme Linked Immunosorbent Assay (ELISA) contribute the highest market share by application?
In terms of technology, Enzyme Linked Immunosorbent Assay (ELISA) segment in projected to account for 29.7% of the total market share in 2025.
Immunofluorescence, even though, has been the traditional laboratory method in various biological applications, this technology is high-priced and requires trained professionals. ELISA (Enzyme linked immunosorbent assay) has evolved as a substitute method, as it offers high specificity and sensitivity, and is easier to carry out in comparison to other methods for diagnosis of substances in the body.
Why Hospitals projected maximum growth in the Pneumococcal Testing Market?
By end user, hospitals segment is anticipated to hold the maximum share of 55.3% in 2025, expanding at rapid rate of 11.5% CAGR during the forecast period.
The hospital segment is anticipated to dominate due to the higher implementation of advanced diagnostic equipment in hospitals. Also, trends like growing adoption of robotic technology and automation in instruments that enhances the results of diagnostic tests is projected to drive the segment.
Moreover, favorable reimbursement policies for several diagnostic tests is also expected to support hospital segment retain its market share during the forecast period.
Competitive Landscape
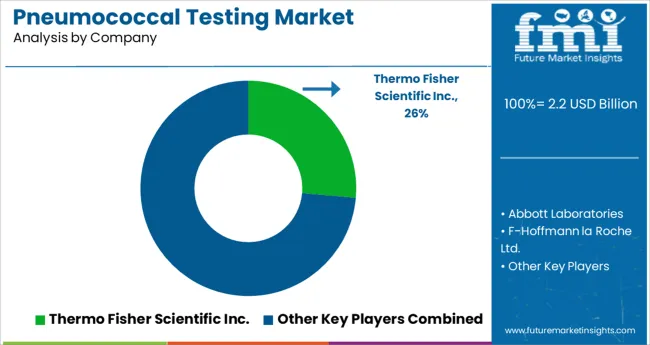
Companies operating in the pneumococcal testing market are competitive by nature, with a presence of few players. These players are involved in a number of strategic alliances.
The product launch and acquisition accelerates the manufacturer’s strategy to capitalize on the market share and capture the significant share of market. Some of the recent instances include:
- In January 2025, Quest Diagnostics announced that they will now offer consumer-initiated laboratory testing powered by QuestDirect™ in collaboration with Walmart. The new solution allows people to take control of their health care and purchase the same high quality laboratory tests ordered by healthcare providers through a consumer-friendly website. This collaboration builds upon a long-standing relationship between Walmart and Quest Diagnostics to broaden access to high quality health services.
- In December 2024, Quidel Corporation entered into a definitive agreement in which Quidel will acquire Ortho, one of the world’s largest in vitro diagnostics companies, for USD 24.68 per share of common stock using a combination of cash and newly issued shares in the combined company, representing a 25% premium over Ortho’s closing price and an equity value of approximately USD 6.0 billion.
Scope of the Report
| Attribute | Details |
|---|---|
| Forecast Period | 2020 to 2024 |
| Historical Data Available for | 2025 to 2035 |
| Market Analysis | million for Value |
| Key Countries Covered | USA, Canada, Brazil, Mexico, Germany, UK, France, Italy, Spain, BENELUX, Russia, China, Japan, South Korea, India, Thailand, Malaysia, Indonesia, Australia, New Zealand, GCC Countries, Turkey, South Africa, Northern Africa |
| Key Segments Covered | Method, Product, Technology, End User and Region |
| Key Companies Profiled | Thermo Fisher Scientific Inc; Becton, Dickinson and Company; Abbott Laboratories; F-Hoffmann la Roche Ltd; Hologic, Inc.; bioMerieux; Bio-Rad Laboratories, Inc.; Meridian Biosciences; Quest Diagnostics; Quidel |
| Report Coverage | Market Forecast, Competition Intelligence, DROT Analysis, Market Dynamics and Challenges, Strategic Growth Initiatives |
| Customization & Pricing | Available upon Request |
Pneumococcal Testing Market by Category
By Method:
- Immunodiagnostics
- Molecular Diagnostic
- Point of Care Testing
By Product:
- Consumables
- Analyzers
By Technology:
- Immunofluorescence
- Enzyme Linked Immunosorbent Assay (ELISA)
- Western Blot Test
- Nucleic Acid Sequence based Amplification
- Immunohistochemistry
- Polymerase Chain Reaction
- Others
By End User:
- Hospitals
- Ambulatory Surgical Centers
- Clinics
By Region:
- North America
- Latin America
- Europe
- South Asia
- East Asia
- Oceania
- Middle East and Africa (MEA)
Frequently Asked Questions
How big is the pneumococcal testing market in 2025?
The global pneumococcal testing market is estimated to be valued at USD 2.2 billion in 2025.
What will be the market size for the pneumococcal testing market in 2035?
It is projected to reach USD 6.4 billion by 2035.
How much will be the CAGR of the pneumococcal testing market between 2025 and 2035?
The market is expected to grow at a 11.2% CAGR between 2025 and 2035.
What are the key product types in the pneumococcal testing market?
The key product types are immunodiagnostics, molecular diagnostic and point of care testing.
Which consumables segment is expected to dominate the pneumococcal testing market in 2025?
consumables segment is expected to dominate with a 57.3% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020-2024 and Forecast, 2025-2035
- Historical Market Size Value (USD Million) Analysis, 2020-2024
- Current and Future Market Size Value (USD Million) Projections, 2025-2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020-2024 and Forecast 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Method
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Method , 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Method , 2025-2035
- Immunodiagnostics
- Molecular Diagnostic
- Point of Care Testing
- Immunodiagnostics
- Y-o-Y Growth Trend Analysis By Method , 2020-2024
- Absolute $ Opportunity Analysis By Method , 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2025-2035
- Consumables
- Analyzers
- Consumables
- Y-o-Y Growth Trend Analysis By Product, 2020-2024
- Absolute $ Opportunity Analysis By Product, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2025-2035
- Enzyme Linked Immunosorbent Assay (ELISA)
- Immunofluorescence
- Western Blot Test
- Nucleic Acid Sequence based Amplification
- Immunohistochemistry
- Polymerase Chain Reaction
- Others
- Enzyme Linked Immunosorbent Assay (ELISA)
- Y-o-Y Growth Trend Analysis By Technology, 2020-2024
- Absolute $ Opportunity Analysis By Technology, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020-2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025-2035
- Hospitals
- Ambulatory Surgical Centers
- Clinics
- Hospitals
- Y-o-Y Growth Trend Analysis By End User, 2020-2024
- Absolute $ Opportunity Analysis By End User, 2025-2035
- Global Market Analysis 2020-2024 and Forecast 2025-2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020-2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025-2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- USA
- Canada
- Mexico
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- Latin America Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- East Asia Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- China
- Japan
- South Korea
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020-2024 and Forecast 2025-2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020-2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025-2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Method
- By Product
- By Technology
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Method
- By Product
- By Technology
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Method
- By Product
- By Technology
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Method
- By Product
- By Technology
- By End User
- Competition Analysis
- Competition Deep Dive
- Thermo Fisher Scientific Inc.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Abbott Laboratories
- F-Hoffmann la Roche Ltd.
- Hologic Inc.
- bioMerieux
- Becton, Dickenson and Company
- Bio-Rad Laboratories Inc.
- Meridian Biosciences
- Quest Diagnostics
- Quidel
- Thermo Fisher Scientific Inc.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020–2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025–2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025–2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025–2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025–2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025–2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025–2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025–2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025–2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market – Tier Structure Analysis
- Figure 21: Global Market – Company Share Analysis

