Transcatheter Mitral Valve Market
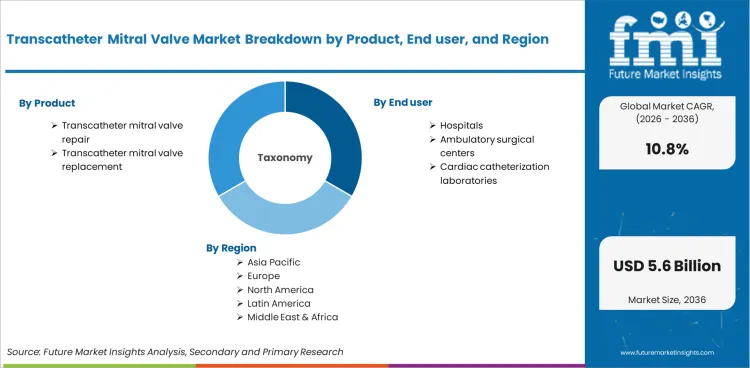
Transcatheter Mitral Valve Market is segmented by Product (Transcatheter mitral valve repair, Transcatheter mitral valve replacement), End user (Hospitals, Ambulatory surgical centers, Cardiac catheterization laboratories), Indication (Mitral stenosis, Mitral regurgitation), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Transcatheter Mitral Valve Market Size, Market Forecast and Outlook By FMI
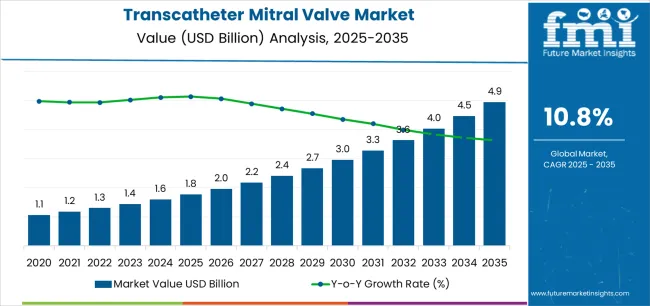
The transcatheter mitral valve market stood at USD 1.8 billion in 2025 and is projected to reach USD 4.9 billion by 2035. The market is anticipated to register a 10.8% CAGR during the forecast period. Repair was likely to lead product with a 57.4% share in 2025, while mitral regurgitation was expected to account for 68.1% share of indication revenue in 2025.
Summary of the Transcatheter Mitral Valve Market
- Demand and Growth Drivers
- Minimally invasive cardiac procedures are expected to support wider use of transcatheter mitral valve systems.
- Mitral valve disease treatment is likely to expand as older patients need safer alternatives to open-heart surgery.
- Imaging-guided procedures are anticipated to improve physician confidence in mitral valve repair and replacement.
- Product and Segment View
- Repair was expected to lead the product segment due to lower procedural risk and valve preservation benefits.
- Mitral regurgitation was likely to remain the main indication because of higher diagnosis and intervention volumes.
- Hospitals are anticipated to remain the leading end user due to advanced cardiac infrastructure and surgical backup.
- Geography and Competitive Outlook
- The United States is expected to lead country-level growth due to coverage support and strong cardiac procedure capacity.
- China is likely to record strong expansion as mitral valve treatment access improves across major hospitals.
- Companies with stronger repair platforms and clinical evidence are anticipated to improve their competitive position.
- Analyst Opinion
- Sabyasachi Ghosh, Principal consultant for Healthcare at FMI, suggests, “The transcatheter mitral valve market is moving toward safer intervention for high-risk cardiac patients. Device makers with durable repair systems and strong physician training support are likely to gain wider hospital adoption.”
- Transcatheter Mitral Valve Market Value Analysis
- The transcatheter mitral valve market is shifting from limited high-risk use toward broader structural heart intervention.
- Demand is likely to rise as hospitals treat more mitral regurgitation patients through catheter-based procedures.
- Adoption is expected to improve as repair devices reduce recovery burden for patients unsuitable for surgery.
- Spending is anticipated to be supported by clinical trials and next-generation valve system approvals.
Transcatheter Mitral Valve Market Definition
The transcatheter mitral valve market encompasses all commercially traded products, solutions, and services categorized by product, end user, indication. The market covers revenue generated from sales across North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa during the forecast period from 2026 to 2036.
Transcatheter Mitral Valve Market Inclusions
Market scope includes all transcatheter mitral valve products and services sold through commercial channels, covering Transcatheter mitral valve repair, Transcatheter mitral valve replacement; Hospitals, Ambulatory surgical centers, Cardiac catheterization laboratories; Mitral stenosis, Mitral regurgitation. Revenue is tracked from 2026 to 2036 across all identified regions and countries.
Transcatheter Mitral Valve Market Exclusions
The scope excludes non-commercial transactions, internal transfers, prototype or research-stage products, and investigational devices used in clinical trials without commercial approval. Government-funded research programs without commercial procurement are also excluded.
Transcatheter Mitral Valve Market Research Methodology
- Primary Research: Interviews with device manufacturers, hospital procurement teams, interventional physicians, and regulatory consultants across key markets.
- Desk Research: Analysis of clinical trial databases, regulatory approval records, and reimbursement policy documents.
- Market Sizing and Forecasting: Bottom-up aggregation of procedural volumes, reimbursement data, and clinical adoption rates, with regional adoption curves and top-down validation.
- Data Validation: Cross-checked quarterly against procedural volume databases and manufacturer revenue disclosures.
Why is the Transcatheter Mitral Valve Market Growing?
- Aging demographics and expanding clinical adoption are increasing procedural volumes, supporting a CAGR of 10.8% through 2036.
- USA leads growth at 12.8% owing to large patient populations and advanced healthcare infrastructure.
- Transcatheter mitral valve repair accounts for 82.4% of the product segment, reflecting clinical adoption patterns and procedural volume concentration.
Market growth is directly tied to procedural volumes across clinical settings. Aging populations, expanding disease prevalence, and improving diagnostic capabilities are increasing the number of procedures performed annually. Growth is concentrated in regions with established healthcare infrastructure and favorable reimbursement frameworks. The relationship between demographic trends and procedural demand provides long-term growth visibility for device manufacturers.
Clinical adoption of new devices and procedures is heavily influenced by reimbursement decisions. Coverage determinations by government payers and private insurers define the economic viability of new treatment approaches. Favorable reimbursement accelerates adoption by reducing the financial risk for hospitals and physicians. The reimbursement approval timeline varies by region and product category, creating geographic variation in adoption rates.
Regulatory approval and clinical evidence requirements create significant barriers to market entry. New devices must demonstrate safety and efficacy through clinical trials, which require substantial investment and multi-year timelines. The evidence burden varies by regulatory jurisdiction, with the FDA and EU MDR representing the most demanding approval pathways. Established companies with clinical trial infrastructure and regulatory expertise maintain competitive advantages.
Market Segmentation Analysis
- The transcatheter mitral valve market is segmented by product, end user, indication, covering all commercially relevant product and service categories.
- Transcatheter mitral valve repair maintains the largest share at 82.4% of the product segment in 2026.
- Hospitals accounts for 36.7% of the end user category.
The transcatheter mitral valve market segmentation reflects the distribution of demand across product categories, application types, and end-user groups. Market structure is concentrated in the top segments, with Transcatheter mitral valve repair and Hospitals accounting for the majority of revenue.
Insights into the Transcatheter mitral valve repair Segment
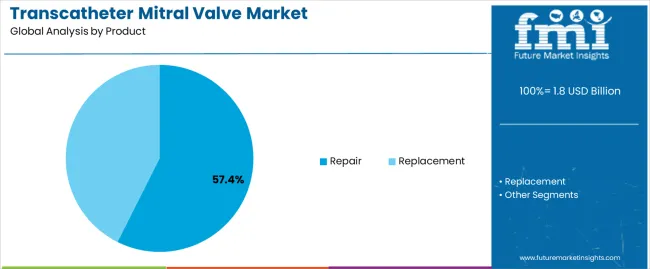
Transcatheter mitral valve repair accounts for 82.4% of the product segment in 2026. Demand for this category reflects clinical adoption patterns and procedural volume concentration. Adoption is concentrated in regions with established healthcare systems and favorable reimbursement. Growth in this segment is expected to remain consistent through the forecast period.
Insights into the Hospitals Segment
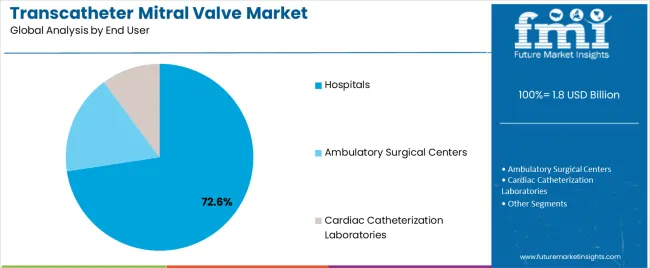
Hospitals accounts for 36.7% of the end user segment in 2026. Demand for this category reflects evolving clinical protocols and treatment pathway preferences. Adoption is concentrated in regions with established healthcare systems and favorable reimbursement. Growth in this segment is expected to remain consistent through the forecast period.
Transcatheter Mitral Valve Market Drivers, Restraints, and Opportunities
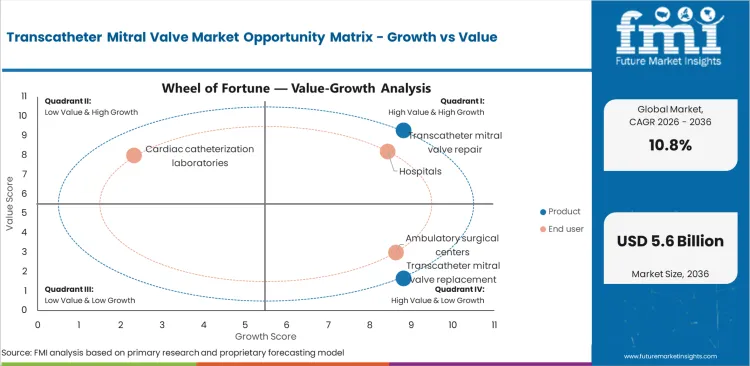
- Expanding procedural volumes and favorable reimbursement are creating structured demand.
- Regulatory approval timelines and reimbursement uncertainty remain the primary restraints limiting faster adoption.
- Next-generation device development and emerging market expansion represent the most significant growth opportunities through 2036.
The transcatheter mitral valve market continues to expand as aging populations, increasing procedural volumes, and expanding clinical adoption of minimally invasive techniques create sustained procurement demand. Growth is tempered by regulatory complexity and reimbursement variability in certain segments and regions.
Procedural Volume Growth Defines Market Trajectory
Market growth is directly tied to procedural volumes across clinical settings. Aging populations, expanding disease prevalence, and improving diagnostic capabilities are increasing the number of procedures performed annually. Growth is concentrated in regions with established healthcare infrastructure and favorable reimbursement frameworks. The relationship between demographic trends and procedural demand provides long-term growth visibility for device manufacturers.
Reimbursement Frameworks Control Adoption Speed
Clinical adoption of new devices and procedures is heavily influenced by reimbursement decisions. Coverage determinations by government payers and private insurers define the economic viability of new treatment approaches. Favorable reimbursement accelerates adoption by reducing the financial risk for hospitals and physicians. The reimbursement approval timeline varies by region and product category, creating geographic variation in adoption rates.
Clinical Evidence Requirements Raise Entry Barriers
Regulatory approval and clinical evidence requirements create significant barriers to market entry. New devices must demonstrate safety and efficacy through clinical trials, which require substantial investment and multi-year timelines. The evidence burden varies by regulatory jurisdiction, with the FDA and EU MDR representing the most demanding approval pathways. Established companies with clinical trial infrastructure and regulatory expertise maintain competitive advantages.
Hospital Capital Budgets Constrain Equipment Adoption
Hospital capital expenditure budgets influence the pace of equipment adoption and technology upgrades. Budget cycles, competing capital priorities, and financial performance affect purchasing decisions. Vendors that offer flexible financing, leasing arrangements, and value-based pricing models reduce the capital barrier for adoption. The shift toward outpatient and ambulatory settings is creating new procurement channels outside traditional hospital budgets.
Analysis of Transcatheter Mitral Valve Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| USA | 12.8% |
| China | 11.7% |
| Germany | 10% |
| France | 9.6% |
| Italy | 9.2% |
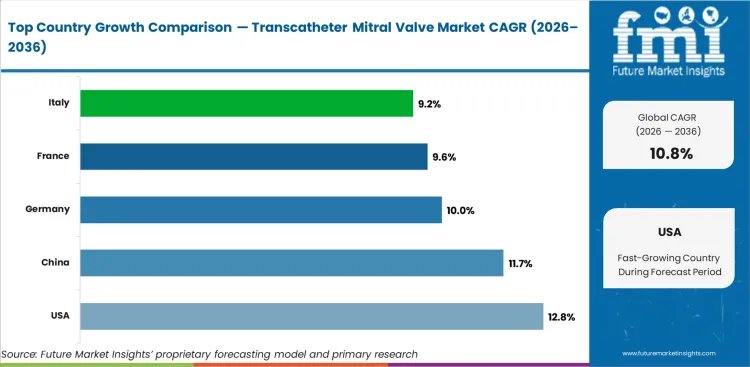
- USA leads at 12.8% CAGR, reflecting procedural volume growth.
- China at 11.7% CAGR represents the second-fastest growth corridor.
- Germany (10%) and France (9.6%) show sustained growth supported by regional policy frameworks.
The global transcatheter mitral valve market is expected to expand at a CAGR of 10.8% from 2026 to 2036. The analysis covers more than 30 countries, with the primary markets detailed below.
Demand Outlook for Transcatheter Mitral Valve Market in USA
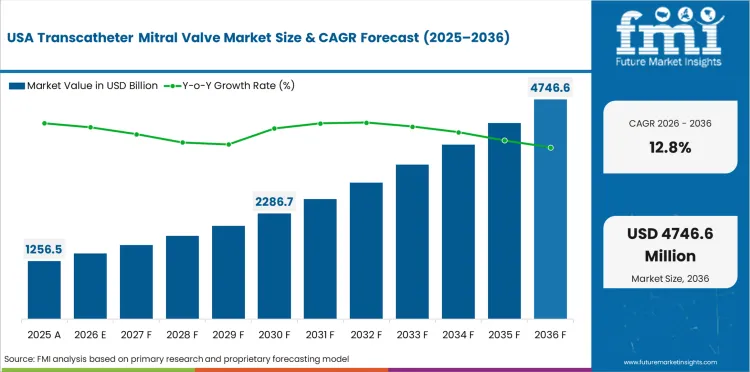
- USA is projected to register a CAGR of 12.8% through 2036. Growth reflects expanding procedural volumes, favorable reimbursement frameworks, and increasing clinical adoption of advanced treatment modalities.
- Aging demographics are increasing procedural volumes across cardiovascular and interventional specialties.
- Reimbursement frameworks are supporting adoption of newer treatment approaches.
- Hospital infrastructure investment is enabling access to advanced procedural technologies.
Future Outlook for Transcatheter Mitral Valve Market in China
- China is projected to register a CAGR of 11.7% through 2036. Growth reflects expanding procedural volumes, favorable reimbursement frameworks, and increasing clinical adoption of advanced treatment modalities.
- Aging demographics are increasing procedural volumes across cardiovascular and interventional specialties.
- Reimbursement frameworks are supporting adoption of newer treatment approaches.
- Hospital infrastructure investment is enabling access to advanced procedural technologies.
Opportunity Analysis of Transcatheter Mitral Valve Market in Germany
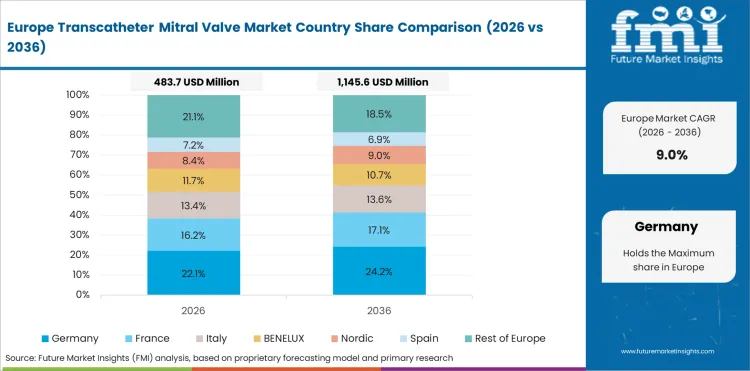
- Germany is projected to register a CAGR of 10% through 2036. Growth reflects expanding procedural volumes, favorable reimbursement frameworks, and increasing clinical adoption of advanced treatment modalities.
- Aging demographics are increasing procedural volumes across cardiovascular and interventional specialties.
- Reimbursement frameworks are supporting adoption of newer treatment approaches.
- Hospital infrastructure investment is enabling access to advanced procedural technologies.
In-depth Analysis of Transcatheter Mitral Valve Market in France
- France is projected to register a CAGR of 9.6% through 2036. Growth reflects expanding procedural volumes, favorable reimbursement frameworks, and increasing clinical adoption of advanced treatment modalities.
- Aging demographics are increasing procedural volumes across cardiovascular and interventional specialties.
- Reimbursement frameworks are supporting adoption of newer treatment approaches.
- Hospital infrastructure investment is enabling access to advanced procedural technologies.
Sales Analysis of Transcatheter Mitral Valve Market in Italy
- Italy is projected to register a CAGR of 9.2% through 2036. Growth reflects expanding procedural volumes, favorable reimbursement frameworks, and increasing clinical adoption of advanced treatment modalities.
- Aging demographics are increasing procedural volumes across cardiovascular and interventional specialties.
- Reimbursement frameworks are supporting adoption of newer treatment approaches.
- Hospital infrastructure investment is enabling access to advanced procedural technologies.
Competitive Landscape and Strategic Positioning
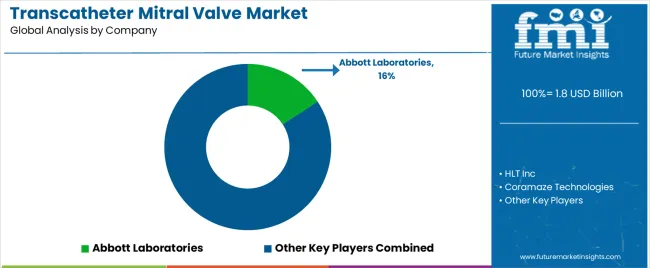
- HLT Inc maintains a strong position through clinical evidence depth and physician training networks.
- Coramaze Technologies and Medtronic compete through specialized capabilities and targeted market focus.
- New entrants are targeting minimally invasive solutions and emerging clinical applications.
- The competitive landscape reflects a mix of global players with broad portfolios and regional specialists with deep application expertise. Market concentration is moderate, with the top 5 companies accounting for a significant share of revenue.
- HLT Inc leads through comprehensive device portfolios and strong clinical data. Coramaze Technologies competes through focused product innovation and clinical partnerships.
- Barriers to entry include clinical trial investment, regulatory approval timelines, and physician training requirements.
Key Companies in the Transcatheter Mitral Valve Market
Key global companies in the transcatheter mitral valve market include:
- HLT Inc, Coramaze Technologies, Medtronic, Biotronik maintain strong market positions through established infrastructure and multi-regional capabilities.
- NeoChord, Inc., Colibri Heart Valve LLC, Neovasc have established regional positions through specialized capabilities.
- Edwards Lifesciences Corporation, Abbott Laboratories, LIVANOVA PLC represent emerging participants with growth potential through innovation and niche positioning.
Competitive Benchmarking: Transcatheter Mitral Valve Market
| Company | Product Portfolio | Clinical Evidence | Physician Access | Geographic Reach |
|---|---|---|---|---|
| HLT Inc | High | High | Strong | Global |
| Coramaze Technologies | High | High | Strong | Global |
| Medtronic | High | Medium | Strong | Global |
| Biotronik | Medium | Medium | Moderate | Global |
| NeoChord, Inc. | Medium | Medium | Moderate | Regional |
| Colibri Heart Valve LLC | Medium | Low | Moderate | Regional |
| Neovasc | Low | Low | Moderate | Regional |
| Edwards Lifesciences Corporation | Low | Low | Limited | Niche |
| Abbott Laboratories | Low | Low | Limited | Niche |
| LIVANOVA PLC | Low | Low | Limited | Niche |
Source: Future Market Insights competitive analysis, 2026
Key Players in the Transcatheter Mitral Valve Market
Major Global Players
- HLT Inc
- Coramaze Technologies
- Medtronic
- Biotronik
- NeoChord, Inc.
- Colibri Heart Valve LLC
- Neovasc
Emerging Players/Startups
- Edwards Lifesciences Corporation
- Abbott Laboratories
- LIVANOVA PLC
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.99 Billion to USD 5.56 Billion, at a CAGR of 10.8% |
| Market Definition | The transcatheter mitral valve market encompasses transcatheter mitral valve products and services across product, end user, indication. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, China, Germany, France, Italy, 30 plus countries |
| Key Companies Profiled | HLT Inc, Coramaze Technologies, Medtronic, Biotronik, NeoChord, Inc., Colibri Heart Valve LLC, Neovasc, Edwards Lifesciences Corporation, Abbott Laboratories, LIVANOVA PLC |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
Segmentation
Transcatheter Mitral Valve Market Segmented by Product:
- Transcatheter mitral valve repair
- Transcatheter mitral valve replacement
Transcatheter Mitral Valve Market Segmented by End user:
- Hospitals
- Ambulatory surgical centers
- Cardiac catheterization laboratories
Transcatheter Mitral Valve Market Segmented by Indication:
- Mitral stenosis
- Mitral regurgitation
Transcatheter Mitral Valve Market by Region:
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Research Sources and Bibliography
- World Health Organization. (2025). Global Health Expenditure Database. WHO.
- USA Food and Drug Administration. (2025). FDA Medical Device Approvals Database. FDA.
- European Commission. (2025). EU Medical Device Regulation Implementation Report. EC.
- Organisation for Economic Co-operation and Development. (2025). OECD Health Statistics. OECD.
- World Bank. (2025). World Development Indicators: Health Expenditure. World Bank Group.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
This Report Answers
- Estimating the size of the transcatheter mitral valve market and revenue potential from 2026 to 2036.
- Segmentation by product, end user, indication.
- Regional insights across more than 30 markets.
- Analysis of procedural volumes and clinical adoption patterns.
- Competitive landscape assessment and company benchmarking.
- Identification of investment opportunities across clinical application areas and geographic growth corridors.
- Supply chain and distribution channel tracking.
- Delivery of data in PDF and Excel formats.
Frequently Asked Questions
What is the global market demand for Transcatheter Mitral Valve Market in 2026?
In 2026, the global transcatheter mitral valve market is expected to be valued at USD 1.99 billion.
How big is the Transcatheter Mitral Valve Market expected to be in 2036?
By 2036, the transcatheter mitral valve market is projected to reach USD 5.56 billion.
What is the expected growth rate for the Transcatheter Mitral Valve Market between 2026 and 2036?
The transcatheter mitral valve market is projected to grow at a CAGR of 10.8% from 2026 to 2036.
Which product segment is expected to lead in 2026?
Transcatheter mitral valve repair is expected to account for 82.4% of the product segment in 2026, supported by aging populations, increasing procedural volumes, and expanding clinical adoption of minimally invasive techniques.
What is driving demand in USA?
USA is projected to grow at 12.8% CAGR through 2036, supported by expanding procedural volumes and favorable reimbursement.
What is driving demand in China?
China is projected to grow at 11.7% CAGR through 2036, driven by growing clinical adoption and healthcare infrastructure expansion.
What does FMI mean by Transcatheter Mitral Valve Market definition?
The transcatheter mitral valve market includes all commercially traded products and solutions categorized by product, end user, indication, across the forecast period from 2026 to 2036.
How does FMI build and validate the Transcatheter Mitral Valve Market forecast?
Forecasting uses a hybrid bottom-up and top-down methodology, starting with procedural volumes, reimbursement data, and clinical adoption rates and cross-checking against industry production statistics and manufacturer disclosures.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Transcatheter mitral valve repair
- Transcatheter mitral valve replacement
- Transcatheter mitral valve repair
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End user
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End user, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End user, 2026 to 2036
- Hospitals
- Ambulatory surgical centers
- Cardiac catheterization laboratories
- Hospitals
- Y to o to Y Growth Trend Analysis By End user, 2021 to 2025
- Absolute $ Opportunity Analysis By End user, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication, 2026 to 2036
- Mitral stenosis
- Mitral regurgitation
- Mitral stenosis
- Y to o to Y Growth Trend Analysis By Indication, 2021 to 2025
- Absolute $ Opportunity Analysis By Indication, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By End user
- By Indication
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By End user
- By Indication
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By End user
- By Indication
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By End user
- By Indication
- Competition Analysis
- Competition Deep Dive
- HLT Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Coramaze Technologies
- Medtronic
- Biotronik
- NeoChord, Inc.
- Colibri Heart Valve LLC
- Neovasc
- Edwards Lifesciences Corporation
- Abbott Laboratories
- LIVANOVA PLC
- HLT Inc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Indication, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by End user, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Indication, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End user
- Figure 9: Global Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Indication
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: North America Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End user
- Figure 29: North America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Indication
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Product
- Figure 36: Latin America Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by End user
- Figure 39: Latin America Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Indication
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Product
- Figure 46: Western Europe Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by End user
- Figure 49: Western Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Indication
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Product
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by End user
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Product
- Figure 66: East Asia Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by End user
- Figure 69: East Asia Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Indication
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End user
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End user, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by End user, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End user
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Indication, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Indication, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

