The demand for structured product labels in the USA is experiencing significant growth, expected to rise from USD 26.9 billion in 2026 to USD 92.7 billion in 2036, reflecting an 11.90% CAGR. The 10-year growth comparison highlights consistent increases in demand year over year. Starting at USD 15.4 billion in 2026, the demand steadily grows, reaching USD 17.2 billion in 2027 and USD 19.2 billion in 2028, indicating strong growth across various industries. As businesses continue to prioritize transparency, traceability, and compliance in product labeling, structured product labels are increasingly adopted in sectors such as healthcare, food and beverages, and consumer goods.
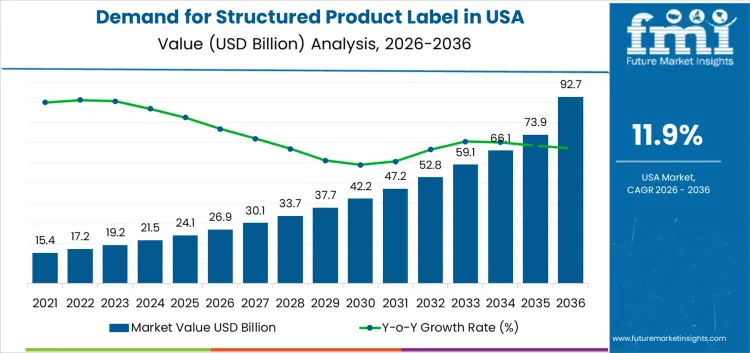
The demand continues to expand, reaching USD 21.5 billion in 2029, USD 24.1 billion in 2030, and USD 26.9 billion in 2031, maintaining a strong upward trajectory. From 2032 onwards, the demand for structured product labels accelerates further, reaching USD 30.1 billion in 2032, USD 33.7 billion in 2033, and continuing the rise through USD 47.2 billion in 2035. By 2036, the demand is expected to reach USD 92.7 billion, reflecting a more than threefold increase over the forecast period. This sustained growth is driven by the rising importance of regulatory compliance, consumer demand for more detailed product information, and technological advancements in label printing and tracking solutions.
| Metric | Value |
|---|---|
| Demand for Structured Product Label in USA Value (2026) | USD 26.9 billion |
| Demand for Structured Product Label in USA Forecast Value (2036) | USD 92.7 billion |
| Demand for Structured Product Label in USA Forecast CAGR (2026-2036) | 11.9% |
The demand for structured product labels in the USA is growing due to the increasing need for more informative, transparent, and standardized labeling in various industries, including pharmaceuticals, food and beverages, chemicals, and cosmetics. Structured product labels, which provide clear and organized information in a standardized format, are essential for meeting regulatory requirements, improving consumer understanding, and ensuring the safety and traceability of products. As consumers become more informed and concerned about the ingredients, sourcing, and environmental impact of products, the demand for structured labels that provide this information in an accessible and standardized format is increasing.
Key drivers for this growth include the growing emphasis on regulatory compliance, particularly in industries like pharmaceuticals and food and beverage, where detailed product information is required for safety and quality assurance. Structured product labels help companies meet regulatory standards, such as those from the FDA, and ensure that information such as dosage, warnings, expiration dates, and nutritional facts is clearly presented. The rise in demand for clean-label products, where consumers expect transparency about the ingredients and sourcing, is also driving the need for more detailed and structured labeling.
Technological advancements in label design and printing, such as the integration of digital and QR code-enabled labels, are expanding the functionality of structured labels. These technologies allow for more dynamic, interactive, and accessible information, enhancing the consumer experience and offering manufacturers a way to provide real-time updates on product data. Despite challenges such as the cost of compliance and the complexity of label design for different regions and products, the growing focus on transparency, traceability, and consumer engagement is expected to drive steady growth in the demand for structured product labels in the USA through 2036.
Demand for structured product labels (SPL) in the USA is segmented by solution type, end-user, and region. By solution type, software leads with 85% of the demand, followed by services. In terms of end-users, pharmaceutical companies account for 39% of the demand, with biotechnology companies, medical device companies, regulatory authorities, and contract research organizations (CROs) contributing to the remainder. Regionally, demand is distributed across West USA, South USA, Northeast USA, and Midwest USA.
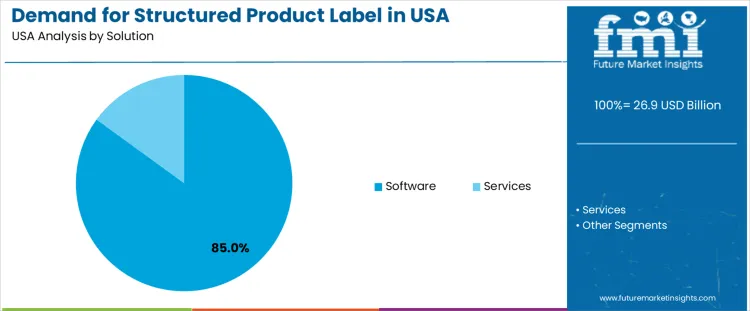
Software solutions account for 85% of the demand for structured product labels in the USA, driven by the increasing need for efficient and accurate labeling in regulated industries like pharmaceuticals and biotechnology. Software enables companies to create and manage product labels in compliance with regulatory standards, ensuring that labels are consistent, accurate, and up to date. The demand for SPL software is further fueled by the rising complexity of product labeling requirements, particularly in the pharmaceutical and medical device industries. With software solutions, companies can streamline the process of label creation, distribution, and compliance, reducing the risk of errors and regulatory issues. As regulatory bodies continue to enforce stricter standards for product labeling, the demand for specialized software solutions to manage SPLs is expected to grow, solidifying its dominant position in the industry.
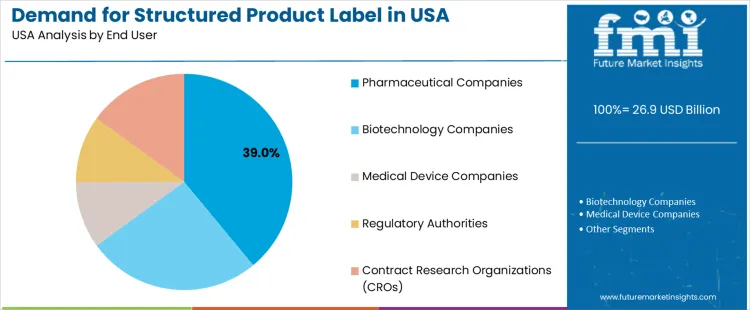
Pharmaceutical companies account for 39% of the demand for structured product labels in the USA, driven by the need to comply with stringent regulatory requirements for drug packaging and labeling. Accurate and standardized labeling is critical in the pharmaceutical industry to ensure patient safety, regulatory compliance, and clear communication of product information. Structured product labels provide a streamlined and efficient way for pharmaceutical companies to manage complex product information, including dosage instructions, warnings, and ingredients. With the growing number of drugs entering the industry and the increasing global demand for pharmaceutical products, pharmaceutical companies rely on SPL solutions to meet regulatory guidelines set by agencies like the FDA. The increasing emphasis on data integrity, safety, and efficient supply chain management continues to drive the demand for SPL solutions in the pharmaceutical sector, further establishing its importance in the industry.
Demand for Structured Product Label (SPL) in the USA is growing as regulatory and healthcare stakeholders emphasize standardized, machine‑readable product information. Key trends include integration of SPL with electronic health records and clinical decision support systems, and use of SPL for biologics and complex therapies. Drivers include stringent FDA requirements for submission and updates, increased use of digital therapeutics, and demand for accurate prescribing and safety information.
Demand for Structured Product Label (SPL) in the USA is increasing because healthcare providers, patients, and regulators require consistent access to accurate, standardized drug information. SPL provides a uniform format for product labeling data, enabling easier dissemination through electronic prescribing systems, mobile health applications, and patient information portals. FDA mandates for SPL submissions and frequent updates to labeling content drive adoption by pharmaceutical manufacturers. The shift toward digital health records and interoperability across health systems further supports demand, as SPL enables seamless integration of labeling into clinical workflows. As drug development becomes more complex and personalized, structured, machine‑readable labels help ensure safety and compliance throughout a product’s life cycle.
Technological advances are accelerating demand for Structured Product Label (SPL) in the USA by making creation, validation and distribution more efficient. Tools that automate SPL generation from regulatory, clinical and safety data reduce manual effort and errors. Integration with regulatory submission systems and electronic health records promotes real‑time access to up‑to‑date labeling. Enhanced data standards and validation tools help ensure SPLs meet FDA requirements and support advanced queries and analytics for safety monitoring and risk management. Cloud‑based solutions and APIs facilitate sharing SPL data with healthcare IT systems, improving interoperability. These innovations lower barriers to adoption and support broader use across pharmaceutical, biotech and healthcare software companies.
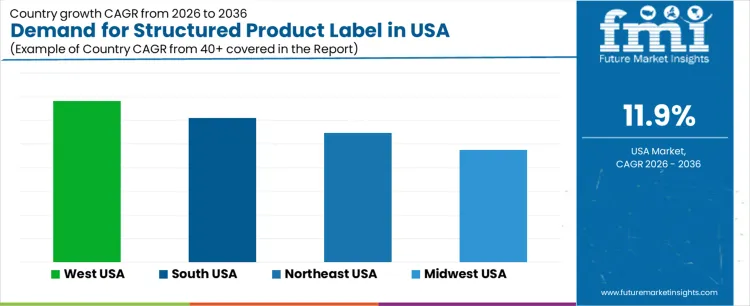
| Region | CAGR (%) |
|---|---|
| West USA | 13.7% |
| South USA | 12.2% |
| Northeast USA | 10.9% |
| Midwest USA | 9.5% |
Demand for structured product labels in the USA is growing steadily, with West USA leading at a 13.7% CAGR, driven by its strong retail, e-commerce, and tech sectors. South USA follows with a 12.2% CAGR, fueled by the region’s expanding manufacturing base and focus on consumer engagement. Northeast USA shows a 10.9% CAGR, supported by its pharmaceutical, food, and retail industries, all requiring detailed and compliant labeling. Midwest USA experiences a 9.5% CAGR, with steady growth driven by the region’s manufacturing, agriculture, and retail sectors.
West USA leads the demand for structured product labels, growing at a 13.7% CAGR. The region’s strong focus on consumer products, e-commerce, and high-tech industries is driving the adoption of structured labeling solutions. States like California, Oregon, and Washington, home to major tech hubs and large retail industrys, are seeing widespread use of structured product labels in industries such as electronics, food, and pharmaceuticals. Structured labels help businesses streamline inventory management, ensure regulatory compliance, and improve the consumer experience with more detailed and easily accessible product information. The region’s emphasis on sustainability and environmental concerns is also encouraging businesses to adopt eco-friendly labeling solutions. As e-commerce continues to grow and product transparency becomes increasingly important, the demand for structured product labels in West USA will continue to expand, particularly in sectors that require precise and scalable product information.
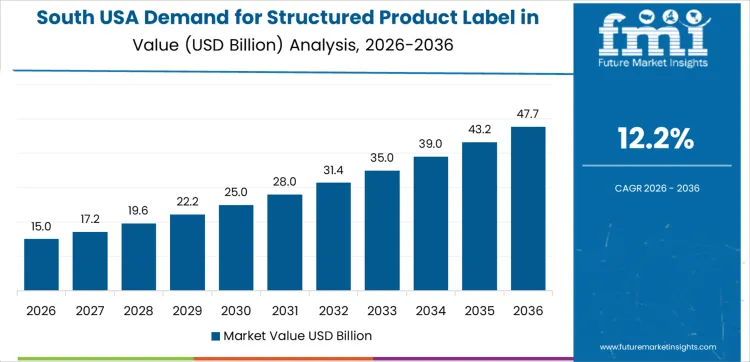
South USA is experiencing strong demand for structured product labels, with a 12.2% CAGR. The region’s large manufacturing and retail sectors are key drivers behind this growth, particularly in states like Texas, Florida, and Georgia. Structured product labels are essential for industries such as food and beverage, healthcare, and automotive, as they ensure accurate product tracking, compliance with regulations, and improved consumer communication. The growing trend toward e-commerce and omnichannel retailing is further fueling the need for structured labels to streamline product identification and improve the customer experience. As the region continues to expand its manufacturing capabilities and retail networks, the demand for structured product labels will rise, driven by the need for detailed and reliable product information across various industries. The growing emphasis on sustainability and cost-effective solutions is also pushing the adoption of these labels in South USA.
Northeast USA is seeing steady demand for structured product labels, with a 10.9% CAGR. The region’s strong presence in pharmaceuticals, food and beverage, and retail is contributing to this growth. States like New York, Massachusetts, and Pennsylvania are home to many industries that require structured product labels for regulatory compliance, accurate tracking, and consumer engagement. The increasing focus on product transparency and consumer safety, especially in the pharmaceutical and food sectors, is driving the need for detailed, structured labeling solutions. As businesses focus on improving supply chain efficiency and ensuring product traceability, structured product labels play a critical role. The region’s commitment to digital transformation and technological adoption, including the use of QR codes and smart labels, further supports the adoption of structured product labels. As these industries continue to expand and innovate, demand for these labels will continue to grow.
Midwest USA is experiencing moderate demand for structured product labels, with a 9.5% CAGR. The region’s established manufacturing, agriculture, and retail sectors are driving the demand for structured labels. States like Michigan, Illinois, and Ohio, with their large production bases, are increasingly adopting structured product labels to enhance inventory management, ensure regulatory compliance, and streamline operations. The growing trend toward automation and digital solutions in manufacturing and logistics is also contributing to the increased use of structured labels, which help improve product traceability and reduce errors in supply chains. The rise in e-commerce and the need for consistent, detailed product information is pushing demand for structured product labels in the retail and consumer goods sectors. As businesses in the Midwest continue to focus on efficiency, sustainability, and compliance, the demand for structured product labels will continue to rise steadily.
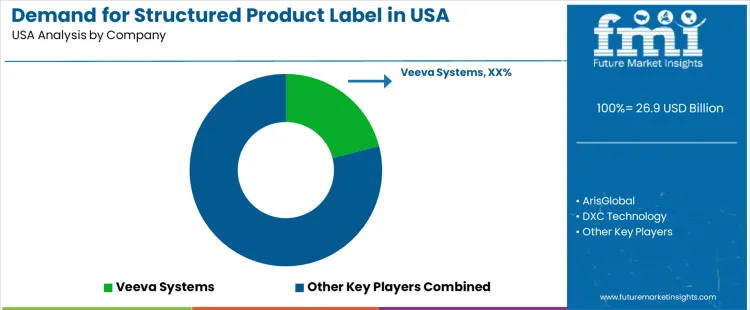
Demand for structured product labels (SPL) in the USA is increasing as regulatory compliance and accurate, standardized labeling become more critical in industries such as pharmaceuticals, life sciences, and medical devices. SPL is an essential part of ensuring that product information is accurate, consistent, and compliant with global regulatory requirements. As companies seek to streamline labeling processes and improve efficiency in global industrys, the demand for SPL software solutions is expected to grow, particularly as industries face stricter regulations and the need for faster time-to-industry.
Veeva Systems is a leading player in the SPL industry, offering cloud-based solutions that help life sciences companies create, manage, and distribute structured product labels. The company’s strategy focuses on providing scalable and flexible solutions that integrate with regulatory affairs and compliance processes. Veeva’s strong industry presence is supported by its deep expertise in the life sciences industry, offering solutions that ensure compliance with regulatory standards and streamline label lifecycle management.
ArisGlobal, DXC Technology, and MasterControl also play significant roles in the SPL industry. ArisGlobal offers software solutions that focus on regulatory compliance and content management, providing a comprehensive platform for SPL management. DXC Technology delivers end-to-end solutions for life sciences companies, helping them digitize labeling and regulatory processes for better efficiency and accuracy. MasterControl specializes in quality and regulatory compliance solutions, helping businesses automate labeling processes and ensure that they meet industry standards. Amplexor (Acolad) provides specialized solutions for structured product labeling, focusing on enhancing translation, compliance, and content management.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Key Segments | Software, Services |
| Deployment | Cloud-based, On-Premise |
| End User Industry | Automotive, Aerospace & Defense, Electronics & Semiconductors, Healthcare, Food & Beverage, Others |
| Key Country | USA |
| Region | West USA, South USA, Northeast USA, Midwest USA |
| Key Players Profiled | Veeva Systems, ArisGlobal, DXC Technology, MasterControl, Amplexor (Acolad) |
| Additional Attributes | Dollar sales by solution, deployment, and end-user industry; regional CAGR and growth trends in Structured Product Label demand in the USA |
How big is the demand for structured product label in USA in 2026?
The demand for structured product label in USA is estimated to be valued at USD 26.9 billion in 2026.
What will be the size of structured product label in USA in 2036?
The market size for the structured product label in USA is projected to reach USD 92.7 billion by 2036.
How much will be the demand for structured product label in USA growth between 2026 and 2036?
The demand for structured product label in USA is expected to grow at a 11.9% CAGR between 2026 and 2036.
What are the key product types in the structured product label in USA?
The key product types in structured product label in USA are software and services.
Which deployment segment is expected to contribute significant share in the structured product label in USA in 2026?
In terms of deployment, cloud-based segment is expected to command 55.0% share in the structured product label in USA in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.