Venous Thromboembolism Treatment Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
The venous thromboembolism treatment market is segmented by Product Type (Retrievable Inferior Vena Cava Filters, Permanent Inferior Vena Cava Filters, Thrombectomy Devices, Catheter-directed Thrombolysis Devices, Compression Systems), Disease Indication (Deep Venous Thrombosis, Pulmonary Embolism), End User (Hospitals, Catheterization Laboratories, Ambulatory Surgical Centers), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Venous Thromboembolism Treatment Market Size, Market Forecast and Outlook By FMI
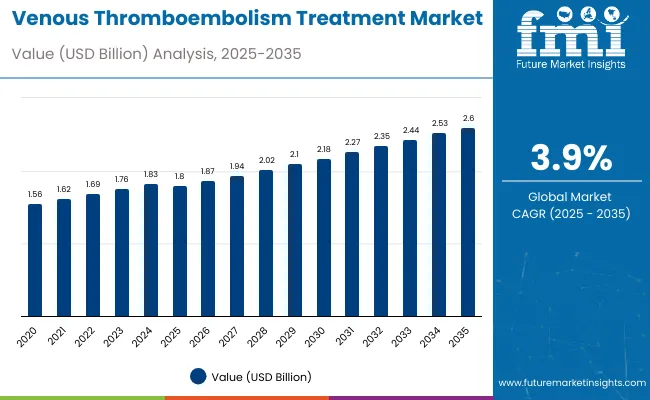
The venous thromboembolism treatment market stood at USD 1.9 billion in 2026 and is projected to reach USD 2.7 billion by 2036. The market is projected to register a 3.9% during the forecast period. Retrievable inferior vena cava filters are expected to hold about 52.0% share in 2026. Hospitals are projected to account for around 43.0% share.
Summary of the Venous Thromboembolism Treatment Market
- Demand and Expansion Drivers
- Patient access and diagnosis growth support use across care settings.
- Clinical workflow needs raise demand for reliable products and services.
- Specialist care expansion improves treatment and testing availability.
- Provider focus on safety and outcomes supports product evaluation.
- Product and Segment View
- Leading product categories support routine clinical use and repeat demand.
- Specialist applications improve value across higher-acuity care settings.
- Hospital and clinic use remains central to product adoption.
- Service support improves provider confidence across care pathways.
- Geography and Competitive Outlook
- North America remains important through specialist access and reimbursement.
- Europe supports demand through mature healthcare infrastructure.
- Asia Pacific expands through hospital capacity and diagnosis growth.
- Companies with clinical evidence and provider training hold stronger positions.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant at FMI states: ""Venous Thromboembolism Treatment demand is shaped by clinical need and provider access. Leading categories remain central because care pathways need reliable products and consistent outcomes. Hospitals and specialist settings influence adoption across daily practice. Product quality and training support affect provider confidence. Companies with evidence and service strength can improve market position.""
- Venous Thromboembolism Treatment Market Value Analysis
- Clinical use creates value through recurring treatment and testing needs.
- Hospital adoption supports premium demand and service contracts.
- Specialist settings improve product utilization across targeted care pathways.
- Training and evidence strengthen provider preference.
The industry faces challenges related to bleeding risks, long term patient adherence, and pricing pressure from generic alternatives. Reimbursement constraints and safety monitoring requirements in certain regions also influence prescribing patterns. To address these issues, pharmaceutical companies are investing in safer drug profiles, simplified treatment regimens, and stronger clinical evidence to support extended use. These efforts are expected to support sustained, moderate growth in the venous thromboembolism treatment market over the long term.
Venous Thromboembolism Treatment Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.9 billion |
| Market Forecast Value (2036) | USD 2.7 billion |
| Forecast CAGR (2026 to 2036) | 3.9% |
How Are the Segments Classified in the Venous Thromboembolism Treatment Market?
The venous thromboembolism treatment market is structured around product type, disease indication, end user, and region, reflecting the full care pathway for VTE management. By product, the industry includes retrievable inferior vena cava filters, permanent IVC filters, thrombectomy devices, catheter-directed thrombolysis devices, and compression systems. Based on disease indication, demand is divided between deep venous thrombosis and pulmonary embolism. From an end-user perspective, treatment delivery spans hospitals, catheterization laboratories, and ambulatory surgical centers. Regionally, the industry covers North America, Latin America, Europe, East Asia, South Asia & Pacific, and the Middle East & Africa, capturing both mature and emerging healthcare systems.
Which Product Segment Accounts for the Largest Share of Global Demand?
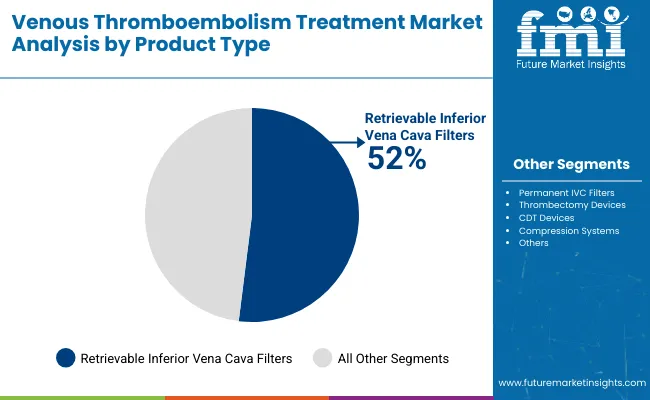
Retrievable inferior vena cava filters account for about 52% of total market share, making them the leading product segment globally. Their dominance is driven by the ability to provide temporary protection against pulmonary embolism while allowing removal once the risk subsides. Compared with permanent filters, retrievable devices offer greater clinical flexibility and a lower risk of long-term complications. Increasing guideline support, wider adoption in both emergency and elective settings, and continued innovation in filter design and retrieval mechanisms reinforce the segment’s leadership.
Which End User Segment Represents the Largest Share of Global Demand?
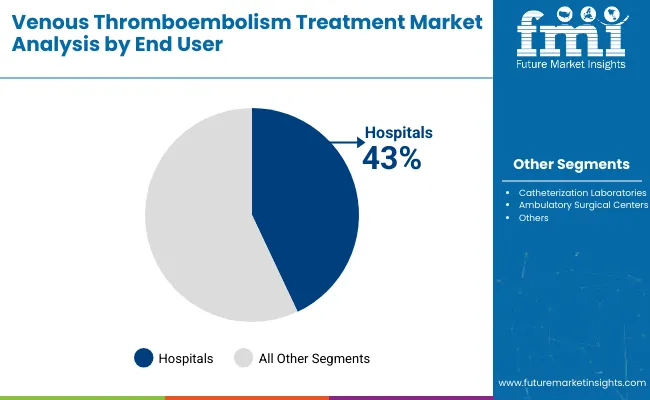
Hospitals represent the largest end-user segment, accounting for around 43% of overall demand. This leadership reflects their central role in diagnosing, treating, and managing venous thromboembolism, particularly in acute and high-risk cases. Hospitals offer advanced imaging, specialist expertise, and integrated care pathways that are essential for complex VTE interventions. High procedural volumes and expanding interventional capabilities continue to position hospitals as the primary setting for VTE treatment worldwide.
What Are the Key Dynamics in the Venous Thromboembolism Treatment Market?
Global demand for venous thromboembolism treatment is rising steadily as the incidence of deep venous thrombosis and pulmonary embolism increases, particularly among aging populations, hospitalized patients, and individuals with cancer, obesity, or prolonged immobility. Healthcare systems are placing stronger emphasis on early diagnosis, rapid intervention, and prevention of fatal complications, which is driving adoption of advanced VTE treatment devices alongside conventional anticoagulation therapy. The industry is increasingly shaped by the need for minimally invasive solutions that deliver reliable clot management while improving patient safety and recovery outcomes.
How Are Clinical Protocols and Hospital-Based Care Models Influencing VTE Treatment Adoption?
Evolving clinical guidelines and hospital workflows are strongly influencing VTE treatment patterns. Hospitals and catheterization laboratories are prioritizing retrievable inferior vena cava filters, thrombectomy devices, and catheter-directed thrombolysis systems that allow physicians to tailor treatment duration and intensity based on patient risk. These technologies support immediate protection against pulmonary embolism while offering flexibility to remove or adjust devices as clinical conditions improve. Growing awareness of VTE as a leading cause of preventable hospital deaths has reinforced protocol-driven adoption, especially in emergency care, interventional cardiology, and vascular surgery settings. Expanding interventional capabilities in Asia Pacific and Latin America further support broader uptake of device-based therapies.
How Do Regulatory Requirements, Cost Pressures, and Operational Constraints Affect Market Scalability?
Regulatory oversight plays a critical role in shaping the scalability of the venous thromboembolism treatment market. Stringent approval requirements, clinical validation standards, and post-market surveillance obligations extend development timelines and increase compliance costs for device manufacturers. High device prices and the need for specialized physician training can limit adoption in cost-sensitive healthcare systems and emerging markets. Operational challenges, including uneven access to trained interventional specialists and reimbursement variability across regions, also affect market expansion. Long-term growth will depend on balancing innovation with affordability, expanding clinical training, and aligning device performance with evolving regulatory and patient safety expectations.
How Is Demand for the Venous Thromboembolism Treatment Market Evolving Across Key Countries?
Demand for venous thromboembolism treatment is rising steadily as healthcare systems place greater emphasis on early diagnosis, preventive care, and timely intervention for deep venous thrombosis and pulmonary embolism. Aging populations, increasing hospitalization rates, and growth in surgical procedures are key contributors to market expansion. Adoption of advanced treatment options such as thrombectomy devices, catheter-directed thrombolysis systems, and retrievable inferior vena cava filters continues to increase, particularly in tertiary and specialty care settings. While developed economies show stable, guideline-driven growth, emerging markets are witnessing faster expansion due to improving healthcare access and infrastructure development.
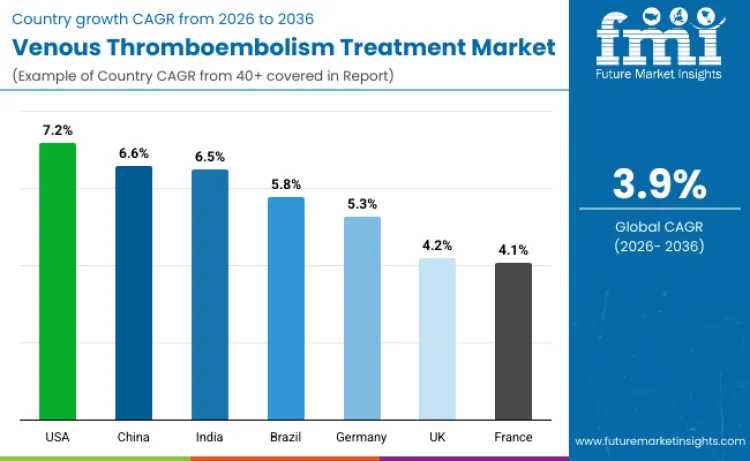
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 7.2% |
| China | 6.6% |
| India | 6.5% |
| Brazil | 5.8% |
| Germany | 5.3% |
| UK | 4.2% |
| France | 4.1% |
Why Does the United States Lead the Venous Thromboembolism Treatment Market?
The United States dominates the VTE treatment market, growing at a CAGR of 7.2% from 2026 to 2036, supported by advanced healthcare infrastructure and high procedural volumes. Widespread insurance coverage and reimbursement for interventional therapies encourage early adoption of thrombectomy devices and retrievable vena cava filters. Large academic hospitals and specialty centers continue to drive innovation through clinical trials and technology partnerships. Increasing focus on patient safety, faster recovery, and reduced hospital readmissions further strengthens demand for advanced VTE treatment solutions across both emergency and elective care settings.
What Is Driving Strong Growth for VTE Treatment in China?
China is emerging as a high-growth VTE treatment market with a projected CAGR of 6.6%, driven by healthcare modernization and rising disease awareness. Urban hospitals in major cities are expanding interventional capabilities and adopting advanced devices to manage acute thrombotic events. Government-backed healthcare investment and physician training programs are improving diagnosis and treatment consistency. In parallel, domestic manufacturing of medical devices is enhancing affordability and supply stability. These factors collectively support wider adoption of VTE treatment technologies across both public and private healthcare facilities.
How Is India Expanding Access to Venous Thromboembolism Treatment?
India is expected to grow at a CAGR of 6.5% as VTE treatment shifts from niche specialty care to broader hospital adoption. Rising awareness, improved diagnostic availability, and growth in tertiary care centers are key contributors. Large metropolitan hospitals are increasingly equipped for catheter-based interventions and emergency thrombus management. Public healthcare programs and private sector investment are improving access, while collaborations with global device manufacturers support technology transfer. Together, these developments are strengthening India’s VTE treatment ecosystem and expanding patient reach.
What Factors Are Supporting VTE Treatment Growth in Brazil?
Brazil’s VTE treatment market is projected to expand at a CAGR of 5.8%, supported by gradual improvements in healthcare infrastructure and device availability. Demand is concentrated in major urban centers where private hospitals and advanced public facilities are increasing adoption of minimally invasive procedures. Expanded distribution networks and physician training are improving access beyond primary cities. However, economic limitations and reliance on imported devices continue to moderate overall penetration, resulting in steady but controlled market growth across the country.
Why Is Germany Seeing Stable Growth in VTE Treatment Demand?
Germany is forecast to grow at a CAGR of 5.3%, reflecting a mature healthcare system with strong adherence to evidence-based clinical protocols. Hospitals benefit from comprehensive insurance coverage and established interventional expertise, supporting consistent use of advanced VTE treatment devices. An aging population and emphasis on early diagnosis further sustain demand. German medical centers also play a role in refining procedural standards and integrating high-quality domestic devices, ensuring reliable adoption without rapid market volatility.
How Is the United Kingdom Shaping Its VTE Treatment Market?
The United Kingdom is expected to record a CAGR of 4.2%, driven largely by National Health Service initiatives and standardized clinical guidelines. Hospitals focus on prevention, early intervention, and cost-effective treatment pathways rather than rapid technology expansion. Procurement frameworks emphasize proven outcomes and long-term value, supporting measured adoption of VTE treatment devices. Growing awareness campaigns and improved post-discharge monitoring also contribute to steady demand across hospital and outpatient settings.
What Is Influencing VTE Treatment Adoption in France?
France is projected to grow at a CAGR of 4.1%, reflecting a well-established and clinically mature VTE treatment market. Demand is concentrated in large medical centers where cardiovascular expertise and interventional capabilities are well developed. Growth is driven by incremental innovation, including device refinement and improved procedural efficiency, rather than broad capacity expansion. Strong healthcare coverage and consistent clinical standards support stable adoption, while digital health integration enhances long-term patient management.
What Is the Competitive Landscape of the Venous Thromboembolism Treatment Market?
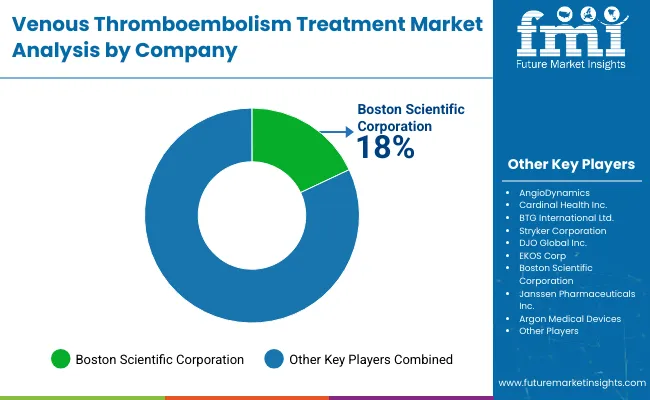
The venous thromboembolism treatment market is shaped by a small group of well established medical device companies that bring together clinical experience, regulatory expertise, and strong hospital relationships. Competition is closely tied to how reliably companies can support physicians in high risk, time sensitive treatment settings. Product safety, ease of use, and proven clinical outcomes play a central role in purchasing decisions, while the ability to meet regulatory standards across different regions continues to define market leadership. Companies that stay aligned with evolving treatment guidelines tend to retain long term demand.
Boston Scientific Corporation holds a leading position due to its wide range of VTE treatment devices and deep presence in interventional cardiology and vascular care. Its strength comes from consistent product performance, strong clinical data, and long standing relationships with hospitals and clinicians. Other players such as Stryker Corporation, AngioDynamics, Cardinal Health Inc., and Argon Medical Devices compete by focusing on specific therapeutic needs, procedural efficiency, and targeted innovation. These companies invest in physician training, product refinement, and clinical research to strengthen adoption across diverse care settings.
Competitive pressure remains high as new technologies must clear strict regulatory and clinical validation requirements before reaching the industry. Success increasingly depends on delivering dependable devices, supporting clinicians beyond the procedure itself, and building trust through long term service and clinical collaboration rather than scale alone.
Key Players in the Venous Thromboembolism Treatment Market
- AngioDynamics
- Cardinal Health Inc.
- Stryker Corporation
- DJO Global Inc.
- EKOS Corp
- Boston Scientific Corporation
- Janssen Pharmaceuticals Inc.
- Argon Medical Devices
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Products Analyzed | Retrievable inferior vena cava filters, permanent inferior vena cava filters, thrombectomy devices, catheter directed thrombolysis devices, compression systems |
| Disease Indications | Deep venous thrombosis, pulmonary embolism |
| End Users | Hospitals, catheterization laboratories, ambulatory surgical centers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, China, Japan, South Korea, India, Australia, Brazil, and 40 plus countries |
| Key Companies Profiled | AngioDynamics, Cardinal Health Inc., Stryker Corporation, DJO Global Inc., EKOS Corp, Boston Scientific Corporation, Janssen Pharmaceuticals Inc., Argon Medical Devices |
| Additional Attributes | Revenue analysis by product and disease indication, country wise treatment adoption trends, regulatory and reimbursement landscape, competitive landscape and company market share analysis, technology innovation trends, integration of VTE therapies within hospital care pathways |
Venous Thromboembolism Treatment Market by Segment
By Product:
- Non segmented pneumatic compression pumps
- Segmented pneumatic compression pumps without gradient
- Segmented pneumatic compression pumps with calibrated gradient
- Upper pneumatic compression sleeves
- Lower pneumatic compression sleeves
- Permanent inferior vena cava filters
- Retrievable inferior vena cava filters
- Embolectomy balloon catheters
- Percutaneous thrombectomy devices
- Catheter directed thrombolysis devices
By Disease Indication:
- Deep venous thrombosis
- Pulmonary embolism
By End User:
- Hospitals
- Catheterisation laboratories
- Ambulatory surgical centres
By Region:
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Europe
- United Kingdom
- Germany
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- Japan
- India
- South Korea
- Australia
- Rest of Asia Pacific
- Middle East and Africa
- GCC
- South Africa
- Rest of Middle East and Africa
Frequently Asked Questions
How big is the venous thromboembolism treatment market in 2026?
The global venous thromboembolism treatment market is valued at around USD 1.9 billion in 2026.
What will be the size of the venous thromboembolism treatment market in 2036?
The industry is projected to reach approximately USD 2.7 billion by 2036.
How much will the venous thromboembolism treatment market grow between 2026 and 2036?
The industry is expected to grow at a CAGR of about 3.9% during the forecast period.
Which product segment leads the venous thromboembolism treatment market?
Retrievable inferior vena cava filters hold the largest share, accounting for about 52% of the industry.
Which end user dominates demand in the venous thromboembolism treatment market?
Hospitals lead demand, contributing roughly 43% of total market share.
Table of Content
- Venous Thromboembolism Treatment Market Analysis Size and Share Forecast Outlook 2026 to 2036
- Venous Thromboembolism Treatment Market Forecast and Outlook (2026 to 2036)
- Quick Stats for Venous Thromboembolism Treatment Market
- Venous Thromboembolism Treatment Market Key Takeaways
- Venous Thromboembolism Treatment Market Overview
- Market Introduction
- Market Size and Growth Trends
- Value Chain Overview
- How Are the Segments Classified in the Venous Thromboembolism Treatment Market?
- Product Analysis
- Retrievable Inferior Vena Cava Filters
- Permanent Inferior Vena Cava Filters
- Thrombectomy Devices
- Catheter Directed Thrombolysis Devices
- Compression Systems
- Disease Indication Analysis
- Deep Venous Thrombosis
- Pulmonary Embolism
- End User Analysis
- Hospitals
- Catheterization Laboratories
- Ambulatory Surgical Centers
- Which Product Segment Accounts for the Largest Share of Global Demand?
- Which End User Segment Represents the Largest Share of Global Demand?
- What Are the Key Dynamics in the Venous Thromboembolism Treatment Market?
- Market Drivers
- Market Restraints
- Market Opportunities
- Market Challenges
- How Are Clinical Protocols and Hospital-Based Care Models Influencing VTE Treatment Adoption?
- How Do Regulatory Requirements, Cost Pressures, and Operational Constraints Affect Market Scalability?
- Regional Market Analysis
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East and Africa
- Country Level Analysis
- United States
- China
- India
- Brazil
- Germany
- United Kingdom
- France
- Competitive Landscape of the Venous Thromboembolism Treatment Market
- Market Structure Analysis
- Market Share Analysis
- Strategic Developments
- Key Players in the Venous Thromboembolism Treatment Market
- Scope of the Report
- Venous Thromboembolism Treatment Market by Segment
- Frequently Asked Questions
List of Tables
- Table 1: Venous Thromboembolism Treatment Market Size and Forecast 2026 to 2036
- Table 2: Key Takeaways of Venous Thromboembolism Treatment Market
- Table 3: Venous Thromboembolism Treatment Market Segmentation by Product
- Table 4: Venous Thromboembolism Treatment Market Segmentation by Disease Indication
- Table 5: Venous Thromboembolism Treatment Market Segmentation by End User
- Table 6: Regional Venous Thromboembolism Treatment Market Analysis
- Table 7: Country Wise CAGR Analysis 2026 to 2036
- Table 8: Competitive Landscape and Key Player Positioning
- Table 9: Scope of the Report
List of Figures
- Figure 1: Global Venous Thromboembolism Treatment Market Value 2026 to 2036
- Figure 2: Venous Thromboembolism Treatment Market Value Based CAGR 2026 to 2036
- Figure 3: Venous Thromboembolism Treatment Market Share by Product
- Figure 4: Venous Thromboembolism Treatment Market Share by Disease Indication
- Figure 5: Venous Thromboembolism Treatment Market Share by End User
- Figure 6: Regional Share of Venous Thromboembolism Treatment Market
- Figure 7: Country Level CAGR Comparison 2026 to 2036
- Figure 8: Competitive Landscape and Market Concentration Analysis

