Chronic Venous Occlusions Treatment Market
The Chronic Venous Occlusions Treatment Market is segmented by Treatment Type (Endovenous Laser Ablation, Radiofrequency Ablation, Sclerotherapy, Compression Therapy, Others), Product (Devices, Drugs), End User (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Others), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Chronic Venous Occlusions Treatment Market Size, Market Forecast and Outlook
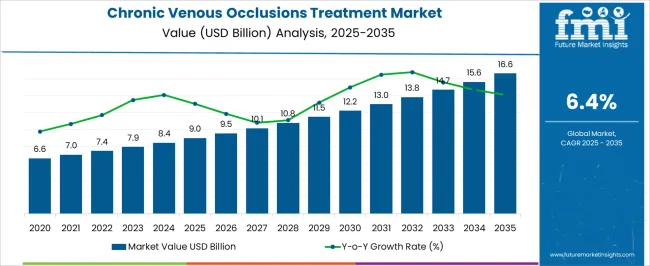
In 2025, the chronic venous occlusions treatment market was valued at USD 9.0 billion. Based on Future Market Insights' analysis, demand for chronic venous occlusions treatments is estimated to grow to USD 9.58 billion in 2026 and USD 17.84 billion by 2036. FMI projects a CAGR of 6.4% during the forecast period.
An absolute dollar growth of USD 8.26 billion between 2026 and 2036 reflects a structural shift from traditional surgical interventions toward minimally invasive endovenous techniques and combination therapies that address the underlying pathophysiology of venous occlusion while reducing procedure-related morbidity. Growth depends on the conversion of hospital-based procedures to ambulatory surgical center settings and the penetration of advanced devices such as drug-eluting stents and bioresorbable scaffolds into venous intervention practice. According to FMI, medical device manufacturers are capturing value not from hardware alone but from integrated treatment platforms that combine interventional devices with pharmacologic support and long-term patient monitoring.
Summary of the Chronic Venous Occlusions Treatment Market
- Market Definition:
- The market comprises endovenous ablation devices, venous stents, pharmacologic agents, and compression therapies designed to manage chronic venous obstructions resulting from deep vein thrombosis, post-thrombotic syndrome, and venous insufficiency across hospital, ambulatory surgical center, and specialty clinic settings.
- Demand Drivers:
- Aging global population with increased venous disease prevalence, with individuals over 65 years representing the fastest-growing segment of venous intervention recipients.
- Rising adoption of minimally invasive endovenous techniques offering faster recovery, reduced complication rates, and suitability for outpatient and ambulatory surgical center delivery.
- Increasing awareness of the systemic consequences of untreated venous disease including venous ulcers, pulmonary embolism risk, and quality of life impairment.
- Key Segments Analyzed
- Treatment Type: Endovenous laser ablation leads, accounting for 32.6% share in 2025 due to established clinical evidence, favorable reimbursement, and suitability for office-based procedure settings.
- Product: Devices dominate at 52.9% share, reflecting the technology-intensive nature of modern venous intervention and recurring revenue from single-use devices.
- End User: Hospitals lead at 45.2% share, though ambulatory surgical centers represent the fastest-growing segment as procedures migrate from inpatient to outpatient settings.
- Geography: North America leads revenue through high procedure volumes and favorable reimbursement; Asia Pacific leads growth through healthcare infrastructure expansion and rising treatment awareness.
- Analyst Opinion at FMI:
- Sabyasachi Ghosh, Principal Consultant for Healthcare at Future Market Insights, observes: “In this updated analysis, we see that the chronic venous occlusions treatment landscape has fundamentally transformed over the past decade. The 32.6% share held by endovenous laser ablation reflects not just technological preference but a systematic shift in care delivery from hospital-based surgery to office-based intervention. The next phase will be defined by combination products that integrate mechanical intervention with pharmacologic elution, addressing not just the occlusion but the underlying thrombotic tendency that caused it. Companies that master this integrated approach will define the standard of care for the next decade.”
- Strategic Implications/Executive Takeaways:
- Develop dedicated venous stent platforms optimized for the larger diameter, lower pressure, and higher flexibility requirements of the venous system rather than adapting arterial devices.
- Invest in physician training programs that support the migration of venous interventions to office-based settings, building loyalty among the next generation of interventionalists.
- Expand combination product pipelines integrating mechanical thrombectomy with localized drug delivery to address post-thrombotic syndrome risk reduction.
- Methodology:
- Built on primary interviews with vascular surgeons and interventional radiologists.
- Benchmarked against venous disease prevalence studies and procedure volume databases.
- Validated using shipment audit data and internal peer review as per FMI modelling standards.
The global demographic shift toward an aging population, with the World Bank projecting a 13% increase in the population aged 65 and over by 2035, directly correlates with higher venous disease incidence. Venous disorders affect an estimated 30 million people in the United States alone, with chronic venous disease prevalence estimated to be ten times higher than peripheral artery disease, creating sustained demand for effective treatment solutions across all regions.
China leads global expansion with a projected CAGR of 7.2%, supported by rapid healthcare infrastructure development, increasing adoption of Western treatment protocols, and government initiatives to expand access to minimally invasive procedures. India follows at 6.8% CAGR, driven by rising medical tourism, expansion of specialty vascular centers in metropolitan areas, and growing awareness of venous disease complications. Germany grows at 6.2% CAGR on the back of robust clinical guideline implementation, strong reimbursement frameworks, and early adoption of novel endovenous technologies.
Mature markets such as the United States and the United Kingdom generate comparatively slower expansion where demand is shaped primarily by technology upgrades and procedure volume growth rather than first-time treatment access. The U.S. market shows a 5.8% CAGR supported by high procedure volumes, favorable reimbursement for minimally invasive treatments, and concentrated expertise in specialized vascular centers, while the UK at 5.3% CAGR demonstrates steady adoption through the National Health Service's commitment to expanding day-case vascular procedures.
Chronic Venous Occlusions Treatment Market Definition
The chronic venous occlusions treatment market covers the global supply of therapeutic interventions and products designed to manage long-term venous blockages resulting from deep vein thrombosis, post-thrombotic syndrome, venous stenosis, and congenital abnormalities. These treatments encompass endovenous thermal ablation techniques including laser and radiofrequency ablation, mechanochemical ablation, sclerotherapy, compression therapy, pharmacologic agents including anticoagulants and thrombolytics, and venous stenting procedures.
Market Inclusions
The report includes global and regional market size estimates in value terms, with a forecast period from 2026 to 2036. It provides segmentation by Treatment Type, Product, End User, and Region. The analysis incorporates technology trends including endovenous device innovation, pharmacologic advancement, and digital health integration for post-treatment monitoring. Regional demand dynamics, country level growth rates, and competitive positioning are also evaluated as per FMI methodology.
Market Exclusions
The scope excludes treatments for acute deep vein thrombosis where intervention occurs within 14 days of symptom onset, as these represent a distinct clinical pathway with different treatment algorithms. It also excludes compression garments sold through retail channels without medical prescription. Surgical bypass procedures for venous occlusion are not considered due to their declining utilization and replacement by endovenous techniques. Wound care products for venous ulcer management when sold separately from comprehensive treatment protocols are excluded from market size calculations.
Research Methodology
- Primary Research: Interviews were conducted with vascular surgeons, interventional radiologists, phlebologists, hospital procurement managers, and medical device distributors.
- Desk Research: Public company filings, clinical practice guideline documents, vascular surgery society publications, and healthcare utilization databases were reviewed.
- Market Sizing and Forecasting: A hybrid top down and bottom up model was constructed, reconstructing demand from venous disease prevalence estimates, procedure volume data, and per procedure device utilization patterns.
- Data Validation and Update Cycle: Outputs were subjected to variance testing against production shipment data, trade flow benchmarking under relevant HS codes, and structured internal review prior to publication.
Segment Analaysis
Chronic Venous Occlusions Treatment Market Analysis by Treatment Type
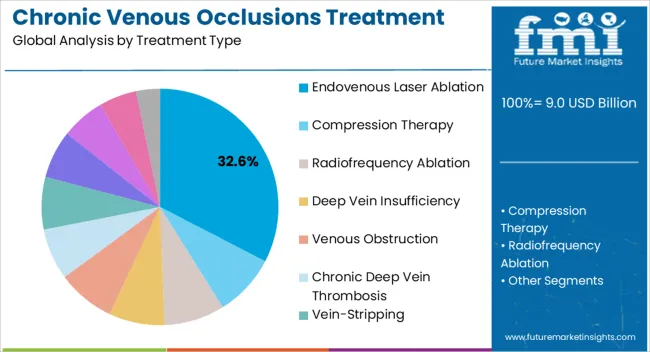
Based on FMI's chronic venous occlusions treatment market report, demand for endovenous laser ablation remains the leading treatment category, capturing 32.6% of the market share. This dominance is sustained by the extensive clinical evidence base supporting its efficacy, favorable reimbursement coverage across major markets, and suitability for delivery in office-based settings without general anesthesia. EVLA provides consistent occlusion rates exceeding 95% with minimal complication rates, establishing it as the reference standard against which newer technologies are compared.
- Technology evolution: Laser ablation systems have evolved from fixed wavelength devices to systems offering wavelength optimization based on vein diameter and depth. Modern generators incorporate automated pullback mechanisms that standardize energy delivery, reducing operator dependence and improving consistency across procedures.
- Radiofrequency ablation competition: Radiofrequency ablation maintains a strong position as the second most utilized thermal ablation technique, offering comparable efficacy with potentially reduced post-procedural discomfort. RFA systems have benefited from technological refinements including segmental ablation catheters that treat longer vein segments with each application.
- Non-thermal alternatives: Emerging non-thermal, non-tumescent techniques including mechanochemical ablation and cyanoacrylate closure are gaining traction among physicians seeking to eliminate the need for tumescent anesthesia entirely. These approaches, while representing smaller current market share, are growing rapidly as clinical evidence accumulates.
- Combination approaches: Sophisticated practitioners increasingly combine thermal ablation of incompetent saphenous trunks with concurrent sclerotherapy of tributary varicosities, creating integrated treatment plans that address both the underlying cause and visible manifestations of venous disease in a single session.
Chronic Venous Occlusions Treatment Market Analysis by Product
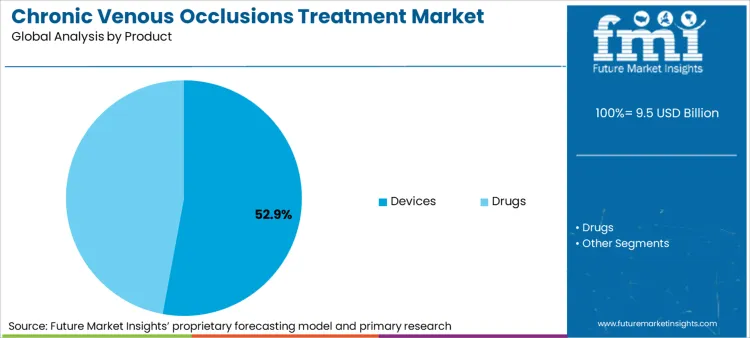
The devices segment accounts for 52.9% of the chronic venous occlusions treatment market, reflecting the technology-intensive nature of modern venous intervention and the recurring revenue model associated with single-use devices. This dominance is reinforced by continuous innovation cycles that introduce improved catheters, ablation fibers, and venous stents, creating replacement demand even as procedure volumes stabilize.
- Venous stent development: Dedicated venous stents designed specifically for the unique hemodynamic environment of the venous system have replaced adapted arterial stents in contemporary practice. These devices feature larger diameters, higher radial strength, and greater flexibility to accommodate venous anatomy while resisting compression from surrounding structures.
- Drug-eluting technologies: The integration of pharmacologic agents with mechanical devices represents the frontier of venous intervention innovation. Drug-eluting balloons and stents designed to reduce restenosis rates are under active investigation, with early data suggesting potential for improved long-term patency in challenging venous lesions.
- Thrombectomy device advancement: Mechanical thrombectomy devices optimized for venous applications have improved the ability to remove acute and subacute thrombus, potentially reducing the incidence of post-thrombotic syndrome. These devices combine rheolytic, rotational, or aspiration mechanisms to debulk thrombus before residual stenosis is addressed with angioplasty or stenting.
- Imaging integration: Intravascular ultrasound has become integral to venous intervention planning and execution, providing detailed assessment of venous anatomy, lesion morphology, and stent deployment that is not available with conventional venography. IVUS utilization is associated with improved procedural outcomes and is increasingly considered standard of care for complex venous interventions.
Chronic Venous Occlusions Treatment Market Analysis by End User
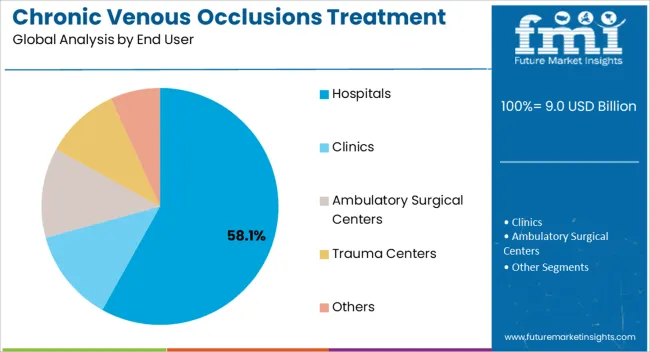
Hospitals currently account for the largest share of venous intervention procedures at 45.2%, reflecting the traditional concentration of complex vascular care in inpatient settings. However, ambulatory surgical centers represent the fastest-growing segment as procedure migration accelerates.
- Procedure migration economics: The shift from hospital-based to ambulatory surgical center delivery reduces costs by 30 to 50% while improving patient convenience and satisfaction. This economic advantage, combined with favorable reimbursement policies, has driven rapid growth in outpatient venous intervention.
- Office-based laboratory expansion: Interventionalists have increasingly invested in office-based laboratories offering the full range of venous procedures without hospital facility fees. These settings enable same-day treatment with minimal disruption to patient schedules and have become particularly prevalent in geographic regions with concentrated populations.
- Specialty clinic emergence: Dedicated vein centers staffed by phlebologists and vascular specialists have proliferated, offering comprehensive venous care from initial consultation through treatment and follow-up. These facilities optimize workflow for venous intervention and have driven standardization of treatment protocols.
- Integrated care models: Over 58% of institutions now implement shared care models combining surgical, radiologic, and post-operative support across multiple care settings. These integrated approaches improve efficiency, reduce duplication, and ensure consistent follow-up after intervention.
Chronic Venous Occlusions Treatment Market Drivers, Restraints, and Opportunities
The market has historically functioned as a surgical specialty where open venous bypass and stripping procedures represented the standard of care for symptomatic venous occlusion. Its baseline scale was constrained by procedure invasiveness, lengthy recovery periods, and patient reluctance to undergo surgical intervention.
FMI analysts observe a complete transformation of the treatment paradigm, with endovenous techniques now representing the majority of venous interventions. This shift has expanded the addressable patient population by offering treatment options acceptable to patients who would have declined surgical approaches.
- Venous disease prevalence and recognition: According to the National Center for Biotechnology Information, more than 30 million people in the United States have some form of chronic venous disease, with prevalence estimated to be ten times higher than peripheral artery disease. Despite this high prevalence, treatment rates remain low, indicating substantial room for market expansion as awareness increases and treatment options improve.
- Minimally invasive procedure adoption: Over 59% of venous procedures now rely on digital imaging and precision targeting, reflecting the integration of advanced imaging technologies into routine interventional practice. This evolution supports significant growth as healthcare providers integrate novel devices for safer interventions with faster patient recovery.
- Ambulatory surgical center expansion: The shift of venous interventions from hospital operating rooms to ambulatory surgical centers and office-based laboratories has fundamentally altered the economics of venous treatment. Over 58% of institutions now implement shared care models that combine surgical, radiologic, and post-operative support, improving efficiency and expanding treatment capacity.
- Personalized medicine approaches: More than 60% of treatment decisions are now guided by detailed imaging analytics and patient-specific factors, reflecting the growing sophistication of venous care. These advances are improving therapeutic precision and setting the foundation for tailored treatment protocols based on individual patient characteristics.
Regional Analysis
The chronic venous occlusions treatment market is segmented geographically across North America, Europe, Asia Pacific, Latin America, and Middle East and Africa. Growth dynamics differ based on venous disease awareness, healthcare infrastructure maturity, reimbursement frameworks, and adoption of minimally invasive technologies.
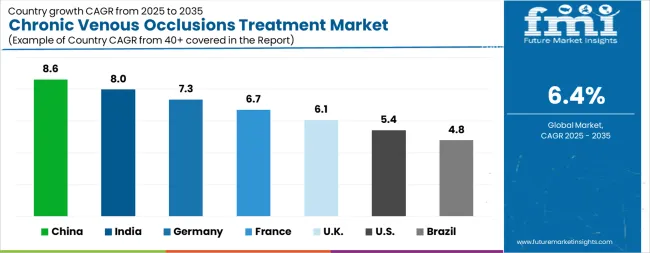
| Country | CAGR 2026 to 2036 |
| China | 7.2% |
| India | 6.8% |
| Germany | 6.2% |
| France | 5.9% |
| United States | 5.8% |
| United Kingdom | 5.3% |
| Brazil | 4.9% |
Source: Future Market Insights analysis, based on proprietary forecasting model and primary research
North America Market Analysis
North America represents the largest regional market for chronic venous occlusions treatment, accounting for approximately 40% of global revenue. This leadership position is supported by well-established healthcare infrastructure, high awareness levels among both physicians and patients, and early adoption of novel therapeutic technologies. The region benefits from favorable reimbursement policies that support office-based procedure delivery and innovation adoption.
United States
The United States chronic venous occlusions treatment market is forecasted to advance at 5.8% CAGR through 2036. Growth is driven by several converging factors including the high prevalence of venous disease affecting an estimated 30 million Americans, the continued migration of procedures to office-based settings, and favorable reimbursement through Medicare and commercial payers. The Centers for Disease Control and Prevention estimates that 900,000 people are affected by deep vein thrombosis annually, with 60,000 to 100,000 deaths from venous thromboembolism, creating substantial demand for both acute and chronic venous care.
The U.S. market is distinctive for its rapid adoption of new technologies, with innovative devices gaining traction through early adopter networks and clinical evidence generation. Physician entrepreneurship has driven the proliferation of office-based laboratories and dedicated vein centers, creating concentrated expertise that attracts patient referrals.
Canada
The Canadian market demonstrates steady growth supported by public healthcare coverage of medically necessary venous interventions and increasing awareness of venous disease impact. Provincial health authorities have recognized the cost-effectiveness of ambulatory venous procedures, supporting expanded access through outpatient funding models.
Europe Market Analysis
Europe represents a mature but innovative regional market characterized by strong clinical guideline development, robust health technology assessment processes, and increasing adoption of day-case procedure models. The European market benefits from collaboration across national vascular societies and harmonization of training standards.
Germany
Germany leads the European market with projected 6.2% CAGR through 2036, supported by the country's strong medical technology sector, comprehensive health insurance coverage, and early adoption of novel venous interventions. German vascular surgeons and interventional radiologists have contributed substantially to the clinical evidence base for endovenous techniques through rigorous clinical studies and registry participation.
The German healthcare system's emphasis on evidence-based medicine ensures that new technologies achieve broad adoption only after demonstrated clinical and cost-effectiveness. This approach has favored technologies with strong clinical trial support while limiting penetration of unproven alternatives.
France
France demonstrates 5.9% CAGR, reflecting the country's robust healthcare system and increasing utilization of minimally invasive venous procedures. French vascular specialists have been early adopters of non-thermal ablation techniques, contributing to the evidence base through multicenter studies.
United Kingdom
The United Kingdom market grows at 5.3% CAGR, shaped by the National Health Service's commitment to expanding day-case procedure capacity and reducing surgical waiting lists. NICE clinical guidelines provide clear pathways for venous intervention, supporting consistent adoption across the country.
Asia Pacific Market Analysis
Asia Pacific represents the fastest-growing regional market for chronic venous occlusions treatment, driven by rapid healthcare infrastructure development, increasing adoption of Western treatment protocols, and rising awareness of venous disease among both physicians and patients. The region benefits from medical tourism flows and government initiatives to expand access to advanced medical technologies.
China
China leads Asia Pacific growth with projected 7.2% CAGR through 2036, supported by massive healthcare infrastructure investment, government initiatives to expand access to minimally invasive procedures, and increasing adoption of Western treatment protocols. China's aging population and rising chronic disease burden have focused attention on venous disease management as a healthcare priority.
Chinese physicians have rapidly adopted endovenous techniques, with training programs and international collaborations accelerating technology transfer. Domestic manufacturing capabilities have developed to serve the local market with cost-competitive devices, expanding access beyond major urban centers.
India
India demonstrates 6.8% CAGR through 2036, driven by rising medical tourism, expansion of specialty vascular centers in metropolitan areas, and growing awareness of venous disease complications. India's large and growing middle class has increased demand for private healthcare services, including elective venous procedures.
Indian interventionalists have developed expertise in managing advanced venous disease, often treating patients with more severe presentations due to delayed diagnosis. This experience has contributed to technical refinements applicable to challenging cases globally.
Japan
Japan maintains a mature but innovative market with steady growth supported by the country's aging population and strong medical technology sector. Japanese investigators have contributed importantly to venous stent development, with dedicated venous devices receiving early approval in the Japanese market.
Competitive Aligners for Market Players
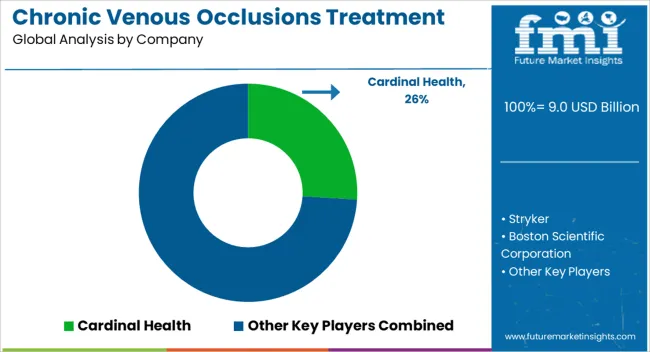
The chronic venous occlusions treatment market is moderately consolidated with Boston Scientific Corporation, Medtronic, and Cardinal Health representing leading positions across multiple product categories. Stryker has established strength in thrombectomy devices, while Cook Medical maintains a strong venous stent portfolio. Edwards Lifesciences has focused on surgical venous products while expanding into endovascular categories.
The competitive landscape has been shaped by strategic acquisitions as larger companies seek to build comprehensive venous product portfolios. In May 2023, Royal Philips completed the acquisition of Vesper Medical Inc., a USA-based medical device company specializing in minimally invasive peripheral vascular solutions. The deal expanded Philips' diagnostic and therapeutic portfolio with an advanced venous stent system for deep venous disease treatment, strengthening its leadership in the peripheral vascular space.
In December 2021, Integer Holdings Corporation acquired Oscor, a private medical device company specializing in the design, development, and manufacturing of active implantable leads for cardiac rhythm management and neurostimulation, along with vascular access and specialty catheter technologies relevant to venous intervention.
Buyer behavior in this market reflects the influence of physician preference, clinical evidence, and economic factors. Interventionalists develop preferences for specific devices based on training, experience, and perceived performance characteristics. Group purchasing organizations negotiate contracts that influence hospital purchasing, while ambulatory surgical centers and office-based laboratories make independent purchasing decisions based on clinical and economic factors.
Recent Developments:
- Royal Philips Acquisition May 2023: Philips completed acquisition of Vesper Medical Inc., adding advanced venous stent system for deep venous disease treatment to its peripheral vascular portfolio.
- Integer Holdings Acquisition December 2021: Integer acquired Oscor, integrating vascular access and specialty catheter technologies relevant to venous intervention.
- Boston Scientific Corporation Pipeline Development: Boston Scientific has advanced development of drug-eluting venous stent technology through clinical trials evaluating restenosis reduction in challenging venous lesions.
- Medtronic Product Expansion: Medtronic expanded its venous product portfolio with next-generation ablation catheters and venous stent systems designed specifically for venous anatomy.
Key Players in the Chronic Venous Occlusions Treatment Market
- Cardinal Health
- Stryker
- Boston Scientific Corporation
- Edward Lifesciences
- Cook Medical
- Medtronic
- AngioDynamics
- Gore
- Bayer AG
- Sanofi
Scope of Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 9.58 billion 2026 to USD 17.84 billion 2036 at a CAGR of 6.4% |
| Market Definition | The chronic venous occlusions treatment market comprises the global supply of therapeutic interventions and products designed to manage long-term venous blockages through endovenous ablation, venous stenting, pharmacologic agents, and compression therapies delivered across hospital, ambulatory surgical center, and specialty clinic settings. |
| Treatment Type Segmentation | Endovenous Laser Ablation, Radiofrequency Ablation, Sclerotherapy, Compression Therapy, Others |
| Product Segmentation | Devices, Drugs |
| End User Segmentation | Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Others |
| Region Segmentation | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, China, India, Japan, South Korea, Brazil and 40 plus countries |
| Key Companies Profiled | Cardinal Health, Stryker, Boston Scientific Corporation, Edward Lifesciences, Cook Medical, Medtronic, AngioDynamics, Gore, Bayer AG, Sanofi |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with vascular surgeons, interventional radiologists, and hospital procurement managers supported by trade benchmarking and procedure volume analysis |
Chronic Venous Occlusions Treatment Market Analysis by Segments
Treatment Type:
- Endovenous Laser Ablation
- Radiofrequency Ablation
- Sclerotherapy
- Compression Therapy
- Others
Product:
- Devices
- Drugs
End User:
- Hospitals
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
Region:
- North America (USA, Canada, Mexico)
- Latin America (Brazil, Chile, Rest of Latin America)
- Western Europe (Germany, UK, Italy, Spain, France, Nordic, BENELUX, Rest of Western Europe)
- Eastern Europe (Russia, Poland, Hungary, Balkan and Baltic, Rest of Eastern Europe)
- East Asia (China, Japan, South Korea)
- South Asia and Pacific (India, ASEAN, Australia and New Zealand, Rest of South Asia and Pacific)
- Middle East and Africa (Kingdom of Saudi Arabia, Other GCC Countries, Turkiye, South Africa, Other African Union, Rest of Middle East and Africa)
Bibliography
- [1] World Bank. (2024). Global Population Projections and Demographic Trends 2024-2040. Washington, D.C.: World Bank Group.
- [2] National Center for Biotechnology Information. (2024). Chronic Venous Disease Prevalence and Impact: Updated Epidemiology Review. Bethesda, MD: NCBI.
- [3] Royal Philips. (2023, May 15). Philips completes acquisition of Vesper Medical to expand venous intervention portfolio. Philips Press Release.
- [4] Centers for Disease Control and Prevention. (2024). Venous Thromboembolism Data and Statistics. Atlanta: CDC.
- [5] European Society for Vascular Surgery. (2024). Clinical Practice Guidelines on Venous Intervention Management. ESVS Guidelines Committee.
- [6] American College of Phlebology. (2025). Venous Procedure Volume Survey 2024-2025. San Diego: ACP.
- [7] National Institute for Health and Care Excellence. (2024). Varicose Veins: Diagnosis and Management. NICE Guideline NG235. London: NICE.
- [8] Journal of Vascular Surgery: Venous and Lymphatic Disorders. (2025). Global Registry of Endovenous Ablation Procedures: 5-Year Outcomes. JVS-VL, 13(2), 245-258.
- [9] USA Food and Drug Administration. (2024). 510(k) Clearances for Venous Stent Systems 2023-2024. Silver Spring, MD: FDA.
- [10] World Health Organization. (2024). Global Health Estimates: Burden of Venous Disease. Geneva: WHO.
This Report Addresses
- Market intelligence to enable structured strategic decision making across medical device manufacturers, pharmaceutical companies, and healthcare provider organizations.
- Market size estimation and 10 year revenue forecasts from 2026 to 2036 including validated CAGR and absolute dollar opportunity sizing.
- Growth opportunity mapping across Treatment Type, Product, End User, and regional demand pockets with emphasis on minimally invasive techniques and combination product development.
- Segment and regional forecasting covering endovenous ablation, venous stenting, and pharmacologic approaches alongside demand trajectories across hospital and ambulatory surgical center settings.
- Competition strategy assessment including technology development, clinical evidence generation, and strategic acquisition activity shaping market positioning.
- Product and compliance tracking focused on venous-specific device design, drug-eluting technology validation, and regulatory pathway navigation.
- Regulatory impact analysis covering FDA breakthrough device designation, CE marking under Medical Device Regulation, and national reimbursement policies influencing market access.
- Report delivery in PDF, Excel, and presentation ready formats to support executive planning, product development alignment, and market entry strategy development.
Frequently Asked Questions
How large is the demand for chronic venous occlusions treatments in the global market in 2026?
Demand for chronic venous occlusions treatments is estimated to be valued at USD 9.58 billion in 2026.
What will be the market size of chronic venous occlusions treatments in the global market by 2036?
Market size for chronic venous occlusions treatments is projected to reach USD 17.84 billion by 2036.
What is the expected demand growth for chronic venous occlusions treatments between 2026 and 2036?
Demand for chronic venous occlusions treatments is expected to grow at a 6.4% CAGR between 2026 and 2036.
Which treatment type segment is poised to lead global sales?
Endovenous laser ablation is expected to remain the leading treatment type, anchored by established clinical evidence, favorable reimbursement, and suitability for office-based delivery, holding 32.6% share in 2025.
Which product segment dominates the chronic venous occlusions treatment market?
The devices segment dominates at 52.9% share in 2025, reflecting the technology-intensive nature of modern venous intervention and recurring revenue from single-use devices.
Which end user segment leads chronic venous occlusions treatment adoption?
Hospitals currently lead at 45.2% share, though ambulatory surgical centers represent the fastest-growing segment as procedures migrate from inpatient to outpatient settings.
What is driving demand in the United States?
High venous disease prevalence affecting 30 million Americans, procedure migration to office-based settings, and favorable Medicare reimbursement for minimally invasive procedures are driving demand in the United States.
What is the United States growth outlook in this report?
The United States is projected to expand at a 5.8% CAGR during 2026 to 2036.
What is driving demand in Germany?
Strong medical technology sector, comprehensive health insurance coverage, early adoption of novel venous interventions, and rigorous clinical evidence development are driving demand in Germany.
What is Germany's growth outlook in this report?
Germany is projected to expand at a 6.2% CAGR during 2026 to 2036.
Does the report cover the United Kingdom in its regional analysis?
Yes, the United Kingdom is included within Europe under the regional coverage framework.
What is the United Kingdom's growth outlook in this report?
The United Kingdom is projected to expand at a 5.3% CAGR during 2026 to 2036.
What is driving demand in China?
Healthcare infrastructure investment, government initiatives expanding access to minimally invasive procedures, and adoption of Western treatment protocols are driving demand in China.
What is China's growth outlook in this report?
China is projected to expand at a 7.2% CAGR during 2026 to 2036.
What is driving demand in India?
Medical tourism growth, expansion of specialty vascular centers in metropolitan areas, and rising awareness of venous disease complications are driving demand in India.
What is India's growth outlook in this report?
India is projected to expand at a 6.8% CAGR during 2026 to 2036.
Does the report cover France in its regional analysis?
Yes, France is included within Europe under the regional coverage framework.
What is France's growth outlook in this report?
France is projected to expand at a 5.9% CAGR during 2026 to 2036.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America and is covered in the country wise assessment.
What is Brazil's growth outlook in this report?
Brazil is projected to expand at a 4.9% CAGR during 2026 to 2036.
What are chronic venous occlusions treatments and what are they mainly used for?
Chronic venous occlusions treatments encompass endovenous ablation techniques, venous stenting, pharmacologic agents, and compression therapies designed to manage long-term venous blockages from deep vein thrombosis, post-thrombotic syndrome, and venous insufficiency, restoring blood flow and preventing complications.
What does the chronic venous occlusions treatment market mean in this report?
It refers to the global supply of therapeutic interventions and products for chronic venous occlusions by treatment type, product, end user, and region, measured in value terms at the manufacturer shipment layer.
What is included in the scope of this chronic venous occlusions treatment market report?
Scope includes market sizing and forecasting for 2026 to 2036, segmentation by treatment type, product, end user, and region, along with competitive assessment, technology trend analysis, and country wise outlook.
What is excluded from the scope of this report?
The scope excludes acute deep vein thrombosis interventions within 14 days of symptom onset, retail compression garments without prescription, surgical bypass procedures, and wound care products sold separately.
How are regulatory developments shaping the chronic venous occlusions treatment market?
Regulatory developments including FDA breakthrough device designation for venous stents, CE marking requirements under Medical Device Regulation, and national reimbursement policy updates are accelerating innovation while increasing market entry barriers for companies without robust clinical evidence and regulatory expertise.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Treatment Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment Type , 2026 to 2036
- Endovenous Laser
- Radiofrequency Ablation
- Sclerotherapy
- Compression Therapy
- Endovenous Laser
- Y to o to Y Growth Trend Analysis By Treatment Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Treatment Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Devices
- Drugs
- Devices
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospital
- Ambulatory Surgical Centers
- Specialty Clinics
- Others
- Hospital
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Treatment Type
- By Product
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Treatment Type
- By Product
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Treatment Type
- By Product
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Treatment Type
- By Product
- By End User
- Competition Analysis
- Competition Deep Dive
- Cardinal Health
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Stryker
- Boston Scientific Corporation
- Edward Lifesciences
- Cook Medical
- Medtronic
- AngioDynamics
- Cardinal Health
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Treatment Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Treatment Type
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Treatment Type
- Figure 26: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product
- Figure 29: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End User
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Treatment Type
- Figure 36: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Product
- Figure 39: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End User
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Treatment Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Product
- Figure 49: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End User
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Treatment Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Product
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End User
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Treatment Type
- Figure 66: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Product
- Figure 69: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End User
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Treatment Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Treatment Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Treatment Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Treatment Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

