Anti-Aging Creams and Serums Market
Anti-Aging Creams and Serums Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Anti-Aging Creams and Serums Market Forecast and Outlook 2026 to 2036
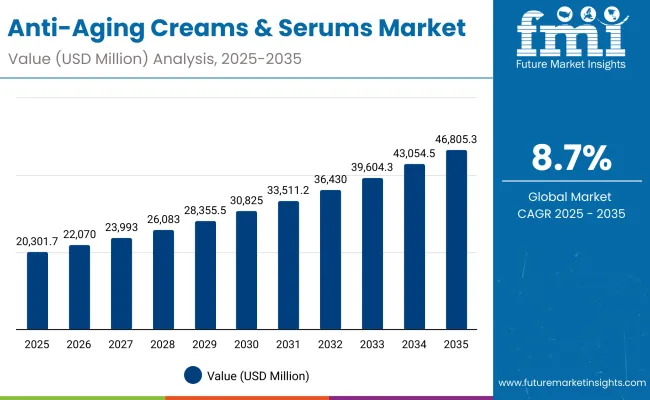
The Anti-Aging Creams and Serums Market is projected to expand from USD 78.6 Billion in 2026 to USD 151.7 Billion by 2036, at a 6.8% CAGR. The growth pathway is being reinforced by large operators scaling science-backed skincare platforms and moving innovation from episodic hero launches to repeatable pipelines. L’Oréal positions this shift explicitly through its Dermatological Beauty strategy and scale with healthcare professional engagement, stating: ‘We lead skincare innovation, partnering with 310,000 healthcare professionals, to provide life-changing products and services for all.’
Ingredient governance is also tightening in a way that changes formulation roadmaps for anti-aging. The EU amended the cosmetics framework to restrict Vitamin A compounds, setting maximum concentrations for Retinol Equivalent in body lotions and other leave-on and rinse-off products, and introducing labelling expectations to manage exposure risk. This is directly relevant to retinoid-led anti-aging portfolios.
Innovation is simultaneously being commercialised through disclosed corporate R&D and launches. Beiersdorf’s Annual Report 2024 documents the launch of NIVEA Q10 Dual Action Serum incorporating a new anti-glycation ingredient platform and positions the work as the output of multi-year screening and research. These signals indicate that scaled incumbents are treating anti-aging as an investable science platform under rising regulatory scrutiny, not a marketing-only segment.
Summary of the Anti-Aging Creams and Serums Market
What is the Anti-Aging Creams and Serums Market size in 2026 and 2036 as per FMI projection?
It is projected to be USD 78.6 Billion in 2026 and USD 151.7 Billion by 2036, expanding at a 6.8% CAGR.
FMI Research Approach: Forecast anchored in category consumption, governance-driven ingredient limits, and scaled brand investment patterns.
Which product type leads demand in 2026 and what structurally supports it?
Anti-aging creams lead with a 61.3% share in 2026, supported by high-repeat routines and mass reach in multi-format retail where day-cream regimens remain the primary habit loop.
FMI Research Approach: Share mapped to routine frequency, retail shelf throughput, and brand portfolio weighting.
Which ingredient type has the largest share in 2026 and why does it persist?
Retinoids lead with a 28.7% share in 2026 because they remain a core actives platform for anti-aging portfolios, while governance is shifting products toward compliant concentrations and clearer exposure labelling.
FMI Research Approach: Ingredient share modelled using regulatory thresholds, product reformulation cycles, and claims discipline.
Which distribution channel is largest in 2026 and what is its role in conversion?
Specialty stores lead with a 34.9% share in 2026 because conversion for anti-aging remains driven by assisted selling, regimen building, and credibility signalling at shelf.
FMI Research Approach: Channel split sized by conversion mechanics and premium mix.
Which end user dominates in 2026 and what explains the concentration?
Women lead with a 73.5% share in 2026 due to higher category penetration, broader regimen adoption, and stronger engagement with specialty retail and derm-inspired skincare narratives.
FMI Research Approach: End-user split estimated through penetration, regimen intensity, and channel preference.
Anti-Aging Creams and Serums Market Key Takeaways
| Items | Values |
|---|---|
| Market Size (2026) | USD 78.6 Billion |
| Forecast Size (2036) | USD 151.7 Billion |
| CAGR (2026 to 2036) | 6.8% |
What is Driving Growth in Anti-Aging Creams and Serums?
Growth is being driven by a tightening governance perimeter around high-impact actives, which is forcing product redevelopment and resetting competitive advantage toward companies that can reformulate without losing efficacy signalling. The EU’s 2024 amendment to cosmetics rules restricts Vitamin A compounds, including Retinol and related esters, and requires warnings to manage overexposure risk, pushing brands toward compliant retinoid architectures and clearer labelling. In parallel, scaled incumbents are investing in new anti-aging pathways beyond classic actives. Beiersdorf documents that it launched an anti-aging product containing an anti-glycation ingredient platform (NAHP) and links it to a multi-year research and screening effort, signalling that anti-aging is being treated as an R&D-led platform category. In the United States, MoCRA-linked FDA guidance on facility registration and product listing raises compliance expectations for cosmetics manufacturers and brand owners, increasing the value of disciplined operational systems for large-scale anti-aging portfolios.
How is the Anti-Aging Creams and Serums Market Segmented?
The market for anti-aging creams and serums is segmented into Product Type (anti-aging creams and anti-aging serums), Ingredient Type (retinoids, hyaluronic acid, peptides, antioxidants, and botanical extracts), Distribution Channel (specialty stores, supermarkets and hypermarkets, pharmacies and drug stores, online retail, and direct-to-consumer channels), and End User (women and men). This segmentation reflects how anti-aging consumption is structured around daily-use routines, governed by ingredient safety and claims compliance, converted through credibility-led retail environments, and concentrated among demographic cohorts with higher regimen intensity and premium skincare engagement.
Why do Anti-Aging Creams Hold the Largest Product-Type Share in 2026?
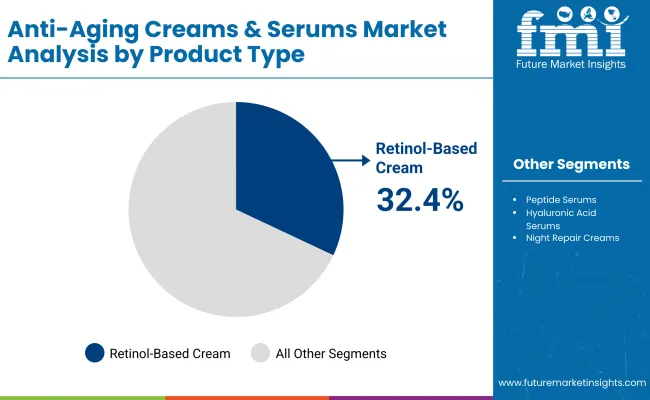
Anti-aging creams lead with a 61.3% share in 2026 because the category’s unit economics are built around habitual, daily-use formats that scale through broad retail coverage and predictable replenishment cycles. The structural signal is how large consumer and skincare groups keep allocating innovation and marketing weight to regimen-based skincare platforms that can be repeated across markets and price tiers. Beiersdorf’s reporting shows anti-aging innovation being delivered into a mass brand format through NIVEA Q10 Dual Action Serum, indicating that creams and adjacent leave-on formats remain the primary vehicle for translating R&D into high-volume consumption. This format dominance is reinforced by the need to manage active delivery and tolerance in everyday routines, which keeps creams central when brands must balance performance with compliance. In parallel, L’Oréal’s disclosed scale in dermatology-linked skincare engagement indicates that the industry is building growth through credibility systems that support routine adherence, not one-off purchases.
Why do Women Represent the Dominant End-User Segment in 2026?
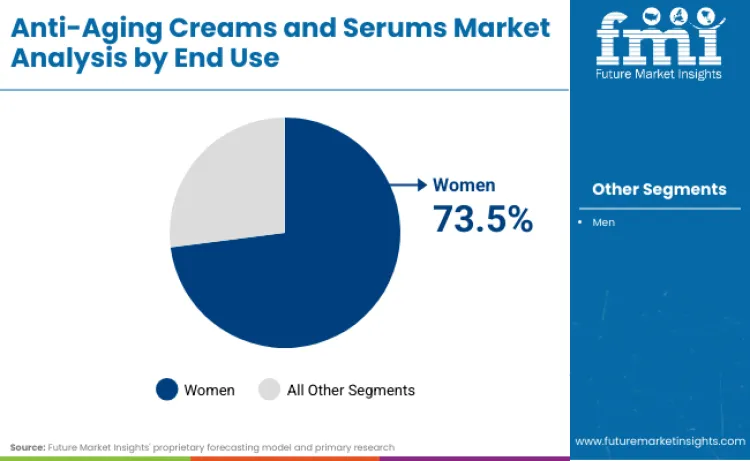
Women lead with a 73.5% share in 2026 because the category is still anchored in higher routine intensity, higher product repertoire per user, and more frequent engagement with premium retail touchpoints that reinforce anti-aging regimen adherence. The structural signals sit in how companies design and scale anti-aging innovation and compliance messaging around the highest-penetration cohort, particularly as regulatory changes alter retinoid concentrations and warnings. The women-led demand base also aligns with the dominant conversion lane. Specialty stores, the largest channel in 2026, are built around assisted regimen formation and repeat replenishment, and this reinforces share concentration in the cohort with the highest willingness to sustain multi-step routines. At the same time, the industry’s push toward science-backed skincare platforms, evidenced by R&D-led launches in mass and prestige portfolios, increases the depth of regimen architectures that historically over-index in women’s purchasing patterns.
What are the Key Trends and Restraints Shaping the Anti-Aging Creams and Serums Market?
A defining trend is the regulatory tightening around high-impact actives, which is reshaping formulation roadmaps and forcing companies to translate efficacy into compliant concentration and labelling systems. The trend reinforces scale advantages because large operators can absorb testing, documentation, and multi-country rollout complexity while maintaining claims discipline and retail readiness.
A core restraint is rising compliance load for cosmetics operations, which increases time-to-market friction and raises the cost of maintaining broad SKU architectures in anti-aging. In the United States, FDA guidance on MoCRA-mandated facility registration and product listing operationalises statutory requirements and standardises the information brands must submit, increasing governance expectations across the supply chain. The compliance overhead affects smaller brands disproportionately and encourages portfolio consolidation, delayed launches, and higher reliance on established manufacturing systems that can sustain documentation quality without disrupting replenishment.
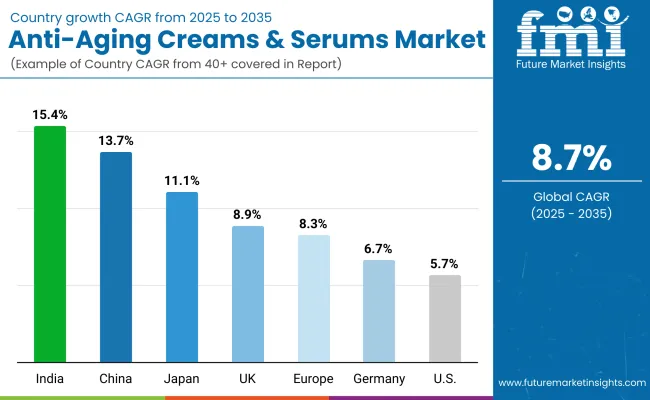
Anti-Aging Creams and Serums Market Analysis by Key Countries
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 9.1% |
| India | 8.5% |
| Germany | 7.8% |
| Brazil | 7.1% |
| USA | 6.4% |
| UK | 5.7% |
| Japan | 5.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
Why is China the Fastest-Growing Market for Anti-Aging Across 2026 to 2036?
China grows at a 9.1% CAGR because its regulatory architecture is tightening around cosmetics supervision and ingredient governance, which favours scaled, compliant anti-aging portfolios and accelerates the shift toward evidence-backed skincare. The NMPA’s implementing provisions for supervision and administration establish NMPA responsibility for cosmetics oversight nationwide and formalise enforcement structures that push brands toward disciplined documentation and compliance. This governance environment changes competitive behaviour: companies prioritise robust regulatory affairs capability, faster compliant reformulation, and clearer claims substantiation, which benefits leaders with deep China operating systems and multi-channel execution. For anti-aging, this supports faster scaling of retinoid-adjacent and advanced active platforms that can withstand scrutiny, while weaker compliance operators face slower approvals and higher relabel risk.
What Drives India’s 2026 to 2036 Growth Trajectory in Anti-Aging?
India expands at an 8.5% CAGR because import and market access for cosmetics are governed through formal registration and regulatory processes that are progressively standardising supply, raising quality baselines, and enabling broader branded portfolio penetration. CDSCO states that no cosmetic shall be imported unless the product is registered under the Cosmetics Rules, 2020 by the Central Licensing Authority, which increases compliance gating and shifts volume toward brands that can execute documentation and variant-level registration. This mechanism supports anti-aging growth by reducing grey-market variability, improving consumer trust in efficacy-led products, and encouraging global players to expand compliant anti-aging assortments through organised retail and specialty formats in major cities. The tightening of import discipline also promotes more consistent availability of regulated actives-led products, reinforcing regimen adoption and repeat purchase across the decade.
How does Germany’s Regulatory Perimeter Translate into Strong Anti-Aging Expansion?
Germany grows at a 7.8% CAGR because EU-wide cosmetics governance and ingredient restrictions are directly shaping anti-aging formulation and labelling decisions, accelerating compliant portfolio refresh cycles across the market. Regulation (EC) No 1223/2009 provides the EU’s harmonised cosmetics framework, which strengthens in-market control and safety assessment requirements across member states, including Germany. The 2024 amendment that restricts Vitamin A compounds in cosmetics forces retinoid-heavy anti-aging portfolios to reformulate within specified thresholds and adopt mandated warnings, creating a multi-year replacement cycle across SKUs and retail sets. This drives both innovation cadence and procurement preference for suppliers with robust compliance systems, sustaining market expansion through regulated product refresh and higher-value, evidence-backed anti-aging routines.
Why does Brazil Maintain High Growth in Anti-Aging Despite Operating Complexity?
Brazil advances at a 7.1% CAGR because regulatory regularisation processes for personal hygiene products, cosmetics and perfumes are formalised through electronic procedures and GMP expectations, pushing the market toward compliant branded portfolios that can scale. ANVISA’s RDC 752/2022 sets the basis for definition, classification, and regularisation procedures and establishes electronic regularisation through ANVISA’s portal, which creates a structured pathway for product notification or registration. ANVISA also points to specific GMP frameworks for cosmetics and fragrances through RDC 48/2013, reinforcing that scaled operators with quality systems are structurally advantaged. For anti-aging, this regulatory architecture supports growth through improved product reliability and claims discipline, while raising barriers for informal or weakly documented offerings. The result is expansion driven by formalisation and portfolio scaling by compliant players.
What Makes the United States a Steady Growth Market for Anti-Aging Through 2036?
The United States grows at a 6.4% CAGR because MoCRA-driven operational requirements are raising compliance discipline across cosmetics portfolios, favouring large brands with strong manufacturing and documentation systems and sustaining confidence in actives-led anti-aging. FDA guidance on registration and listing of cosmetic product facilities and products details statutory requirements and submission expectations, creating a governance layer that pushes brands to invest in compliant operations and traceable product architectures. This increases the value of established quality systems and reduces the ability of marginal operators to scale quickly without compliance infrastructure, supporting steady market expansion anchored in trusted brands and regulated portfolio refresh.
Why is the UK’s Growth Moderate but Durable Through the Forecast Period?
The UK expands at a 5.7% CAGR because post-Brexit cosmetics placement requires formal notification through the UK’s Submit Cosmetic Product Notifications service for products made available in Great Britain, reinforcing compliance-led market participation and stabilising brand execution. GOV.UK guidance states that cosmetic products must be notified using the Submit service before being placed on the GB market. This creates a structured compliance baseline that supports credible anti-aging portfolios and strengthens retailer confidence in documentation, while also adding administrative friction that moderates acceleration relative to faster-growth markets. Growth remains durable as brands optimise SKUs, claims, and distribution under a stable notification regime.
What Underpins Japan’s Anti-Aging Growth While Keeping It Below China and India?
Japan grows at a 5.1% CAGR because quasi-drug style regulatory pathways for efficacy-leaning cosmetic claims create higher documentation and submission expectations, shaping portfolio choices and moderating speed of rollout while supporting trusted, high-value anti-aging platforms. PMDA documentation on manufacturing and marketing procedures indicates that application documents for quasi-drugs are submitted to PMDA and sit under formal accreditation structures, reflecting a more stringent environment for efficacy-positioned products. This raises the bar for claims and supports brand trust, but it also increases time and cost for launches, keeping growth steady rather than explosive across 2026 to 2036.
How is Competition Structured in the Global Anti-Aging Creams and Serums Market?
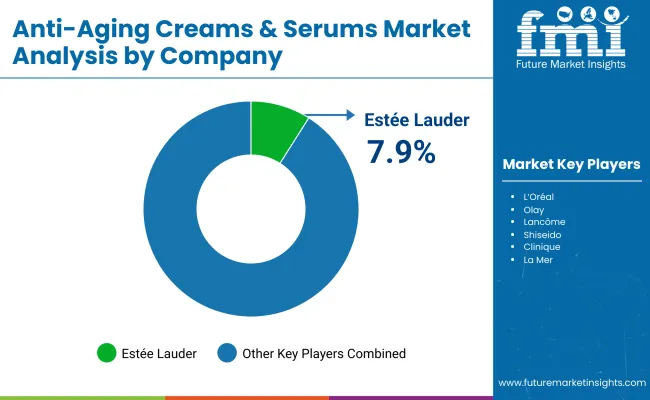
Competition is led by multi-category beauty and FMCG groups with scale in R&D, compliance, and omnichannel execution. Scope includes topical anti-aging creams and serums positioned for wrinkle reduction, firming, hydration-linked ageing support, and tone correction within cosmetics regulations. Scope excludes injectables, medical devices, and prescription therapeutics. L’Oréal is the largest global player in this competitive set by portfolio breadth and disclosed investment in beauty R&I and digital commerce, with its annual reporting citing 28.2% of sales in e-commerce and a scaled science platform supporting skincare innovation at global level. In North America, Estée Lauder remains structurally important in prestige skincare, and its fiscal 2024 results disclosure shows skincare performance sensitivity to China and travel retail dynamics, which shapes regional emphasis and channel strategy. In Asia, Japan remains distinct: local leaders operate under quasi-drug style procedural expectations that can advantage domestic incumbents on trust and compliance, meaning global leadership does not automatically translate into Japanese leadership.
Recent developments:
- In April 2024, Beiersdorf announced a patented anti-glycation active after ten years of research and 1,700 ingredients tested, targeting sugar-induced collagen damage to reduce wrinkle formation and address glycation-driven premature skin aging, validated through extensive scientific studies globally applied.
- In December 2024, the USA Food and Drug Administration issued final guidance outlining recommendations for cosmetic facility registration and product listing, clarifying statutory requirements, responsible parties, submission content, timelines, and procedures under FDA-2023-D-1716, while emphasizing the nonbinding, advisory nature of guidance documents.
Key Players
- L’Oréal
- The Estée Lauder Companies
- Procter & Gamble
- Unilever
- Beiersdorf
- Shiseido Company
- Johnson & Johnson
- Amorepacific Corporation
- Coty Inc.
- Kao Corporation
Market Definition
The Anti-Aging Creams and Serums market covers topical cosmetic formulations sold as creams and serums positioned for visible ageing concerns including wrinkles, loss of firmness, dryness-linked ageing appearance, and tone unevenness, and marketed under cosmetic regulatory frameworks. It includes products using recognised cosmetic actives such as retinoids, hyaluronic acid, peptides, antioxidants, and botanical extracts, sold through retail and direct channels. It excludes injectables, prescription drugs, energy-based devices, and surgical interventions. The market is governed by cosmetics safety and labelling rules, including EU framework requirements and ingredient restrictions that directly shape retinoid portfolio design.
Market Inclusion
Included revenues cover branded anti-aging creams and serums sold through specialty stores, supermarkets and hypermarkets, pharmacies and drug stores, online retail, and DTC channels. Included also covers products reformulated or relabelled to comply with updated ingredient governance, such as EU retinoid limits and warning requirements, and compliance systems that standardise product placement such as FDA registration and listing guidance linked to MoCRA. Included end users cover women and men purchasing topical anti-aging cosmetic products for routine use.
Market Exclusion
Excluded revenues cover aesthetic injectables, prescription retinoids and other regulated medicines, dermatology procedures performed as medical services, and devices used for resurfacing or tightening. Also excluded are bulk active ingredients sold as intermediates without finished consumer packaging, and contract manufacturing revenue not attributable to branded finished goods sell-through. Market participation requirements and compliance overhead that do not directly relate to cosmetics placement and labelling are excluded unless they directly gate product availability, such as national cosmetics import registration rules and cosmetics notification mandates in specific jurisdictions.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type Segments | Anti-Aging Creams; Anti-Aging Serums |
| Ingredient Type Segments | Retinoids; Hyaluronic Acid; Peptides; Antioxidants; Botanical Extracts |
| Distribution Channels | Specialty Stores; Supermarkets & Hypermarkets; Pharmacies & Drug Stores; Online Retail; Direct-to-Consumer |
| End User Segments | Women; Men |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia & Pacific, East Asia, Middle East & Africa |
| Key Countries | China, India, Germany, Brazil, United States, United Kingdom, Japan |
| Key Companies Profiled | L’Oréal; The Estée Lauder Companies; Procter & Gamble; Unilever; Beiersdorf; Shiseido Company; Johnson & Johnson; Amorepacific Corporation; Coty Inc.; Kao Corporation |
| Additional Attributes | Dollar sales measured for topical anti-aging creams and serums by product format, active ingredient platform, retail and digital distribution mix, end-user regimen intensity, regulatory compliance impact (retinoid concentration limits, labelling requirements, MoCRA-linked registration), portfolio reformulation cycles, and country-level governance effects on product availability |
Anti-Aging Creams and Serums Market by Key Segments
Product Type:
- Anti-Aging Creams
- Anti-Aging Serums
Ingredient Type:
- Retinoids
- Hyaluronic Acid
- Peptides
- Antioxidants
- Botanical Extracts
Distribution Channel:
- Specialty Stores
- Supermarkets and Hypermarkets
- Pharmacies and Drug Stores
- Online Retail
End User:
- Women
- Men
Bibliography
- Beiersdorf. (2024, April 22). Pioneering anti-aging innovation with a new active ingredient targeting sugar-induced skin aging.
- Beiersdorf. (2025). Beiersdorf annual report 2024.
- Central Drugs Standard Control Organization. (n.d.). Cosmetics: Import registration and requirements under Cosmetics Rules, 2020. Ministry of Health and Family Welfare, Government of India.
- European Union. (2009). Regulation (EC) No 1223/2009 on cosmetic products.
- European Union. (2024). Commission Regulation (EU) 2024/996 amending Regulation (EC) No 1223/2009 regarding the use of Vitamin A and other substances.
- UK Government. (2020). Submit a cosmetic product notification.
- USA Food and Drug Administration. (2024). Guidance for industry: Registration and listing of cosmetic product facilities and products.
Frequently Asked Questions
How do EU retinoid limits change anti-aging portfolio strategy over 2026 to 2036?
They force compliant reformulation cycles and warning-led labelling, concentrating share in brands that can execute regulated retinoid architectures.
Why do creams remain the dominant product type in 2026 despite serum premiumisation?
Creams sit at the centre of daily routines and scale best through mass reach formats, sustaining the 61.3% share in 2026.
Which channel’s economics best explain specialty store leadership in 2026?
Assisted selling and regimen-building mechanics drive conversion for actives-led anti-aging, supporting the 34.9% specialty share in 2026.
What is the most material compliance restraint in the United States for anti-aging brands?
MoCRA-linked FDA registration and listing expectations raise operational and documentation load for cosmetics portfolios.
Why does China lead the country growth set through 2036 in this dataset?
Tighter national cosmetics supervision favours compliant scaled portfolios and accelerates formal, evidence-backed anti-aging adoption, supporting a 9.1% CAGR.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports and Integrated Reports
- Cosmetics Regulatory Publications
- Corporate Websites and Product Documentation
- Investor Presentations and Earnings Communications
- Ingredient Governance and Safety Frameworks
- Scientific and Dermatology Literature
- Trade Journals and Beauty Industry Magazines
- Press Releases and Verified Media Coverage
- Government Publications and Regulatory Databases
- FMI Internal Knowledge Base
- Subscription-based Industry Databases
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Validation Approach
- Why Primary Evidence is Used
- Field Techniques
- Brand and Portfolio Leader Interviews
- Regulatory and Compliance Expert Consultations
- Dermatology and Professional Skincare Interviews
- Retail and Channel Partner Consultations
- R&D and Formulation Expert Discussions
- Stakeholder Universe Engaged
- C-suite Executives
- Brand and Category Heads
- Regulatory and Quality Leaders
- R&D and Formulation Scientists
- Retail and Digital Commerce Executives
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Skincare Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Year-on-Year Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Year-on-Year Growth Trend Analysis By Product Type
- Absolute $ Opportunity Analysis By Product Type
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Year-on-Year Growth Trend Analysis By Ingredient Type
- Absolute $ Opportunity Analysis By Ingredient Type
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Year-on-Year Growth Trend Analysis By Distribution Channel
- Absolute $ Opportunity Analysis By Distribution Channel
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Year-on-Year Growth Trend Analysis By End User
- Absolute $ Opportunity Analysis By End User
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- Key Countries Market Analysis
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Product Type
- By Ingredient Type
- By Distribution Channel
- By End User
- By Region
- Competition Analysis
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Anti-Aging Creams and Serums Market Key Takeaways
- Global Anti-Aging Creams and Serums Market Size, 2021 to 2036
- Anti-Aging Creams and Serums Market Size by Product Type
- Anti-Aging Creams and Serums Market Size by Ingredient Type
- Anti-Aging Creams and Serums Market Size by Distribution Channel
- Anti-Aging Creams and Serums Market Size by End User
- Anti-Aging Creams and Serums Market Size by Region
- Anti-Aging Creams and Serums Market CAGR by Country
- Scope of Report
List of Figures
- Anti-Aging Creams and Serums Market Growth Trajectory, 2026 to 2036
- Ingredient Governance and Compliance Framework
- Product Type Share Analysis
- Ingredient Type Share Distribution
- Distribution Channel Mix Analysis
- End User Demand Concentration
- Regional Market Attractiveness Analysis
- Competitive Landscape Overview
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

