Cold-Plasma Sterilised Films Market
Cold-Plasma Sterilised Films Market Size and Share Forecast Outlook 2026 to 2036
Cold-Plasma Sterilised Films Market Forecast and Outlook 2026 to 2036
The Cold-Plasma Sterilised Films market is anticipated to grow from USD 557 million in 2026 to USD 1,476 million by 2036, reflecting a CAGR of 10.2%. The expansion is driven by rising demand for sterilised films in medical, pharmaceutical, and aseptic food packaging, where cold-plasma technology ensures surface decontamination without affecting material integrity. Adoption is particularly strong in sterile lidding, form-fill-seal, and rollstock films, enabling safer packaging and extended shelf life for sensitive products.
Medical and pharmaceutical applications lead the market, accounting for the largest share, followed by aseptic food packaging and hospital-use films. Innovations in inline plasma treatment, high-speed plasma chambers, and cost-efficient units support scalability and process efficiency. Manufacturers are emphasizing compatibility with PE, PET, PA, and multilayer films while ensuring regulatory compliance. Increasing global exports of sterile medical disposables further bolster demand, positioning cold-plasma sterilised films as a critical component in hygienic and high-performance packaging solutions.
Quick Stats of the Cold-Plasma Sterilised Films Market
- Cold-Plasma Sterilised Films Market Value (2026): USD 557 million
- Cold-Plasma Sterilised Films Forecast Value (2036): USD 1,476 million
- Cold-Plasma Sterilised Films Market Forecast CAGR (2026 to 2036): 10.2%
- Leading Packaging Format in the Market: Sterile Lidding & Pouch Films (45%)
- Key Growth Regions in the Market: India, China, USA, Brazil, Germany
- Top Players in the Market: Amcor, Mondi, Wanhua, UFlex, Toray, DuPont, Local converters
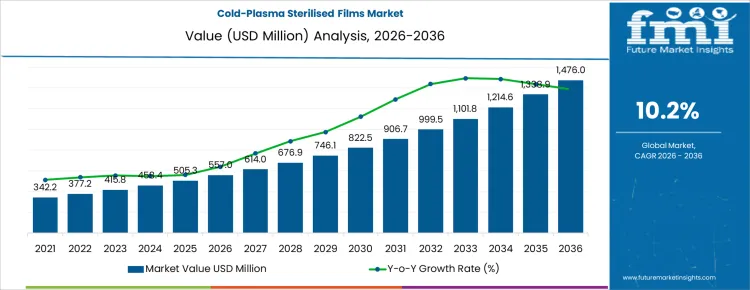
What is the Growth Forecast for the Cold-Plasma Sterilised Films Market through 2036?
From 2026 to 2031, the cold-plasma sterilised films market grows from USD 557 million to approximately USD 795 million, representing the early adoption phase. Annual value additions increase from USD 42 million in 2026 to USD 75 million in 2031. Growth is driven by adoption in food and beverage packaging, pharmaceuticals, and medical device applications, where sterilisation, extended shelf life, and microbial control are critical. Uptake is concentrated in North America and Europe, accounting for roughly 65% of the market, with pilot projects validating sterilisation efficacy, material compatibility, and process integration for thermoforming and flexible films.
Between 2031 and 2036, the market expands from roughly USD 795 million to USD 1,476 million, illustrating the late adoption phase characterized by broader commercialization and structural deployment. Annual increments rise from USD 75 million to USD 156 million, driven by multi-region adoption, repeat procurement, and portfolio-wide integration in industrial and consumer packaging lines. Growth is reinforced by regulatory support for safe food packaging, rising demand for pathogen-free surfaces, and adoption in medical and pharmaceutical packaging. Early-stage growth relied on selective high-value pilots, while later expansion is fueled by full-scale integration and normalized use of cold-plasma sterilisation across global packaging production.
Cold Plasma Sterilised Films Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 557 million |
| Forecast Value (2036) | USD 1,476 million |
| Forecast CAGR (2026–2036) | 10.2% |
Why Are Cold Plasma Sterilised Films Reframing Sterility at the Material Level?
Cold plasma sterilised films gained attention when manufacturers encountered limits in treating finished packages rather than materials themselves. Films exposed to heat or chemical sterilants warped, lost seal integrity, or required long aeration times. In response, plasma treatment was applied directly to film surfaces to neutralise microorganisms without bulk heating. This allowed sterility to be addressed upstream, before forming or sealing. Early demand was narrow and technical, concentrated in medical packaging lines where product sensitivity left no alternative. Adoption followed manufacturing constraints, not a general shift in sterilisation philosophy, and remained confined to high-control environments.
Future demand reflects a deeper change in how sterility is specified. Film sterility is increasingly treated as an intrinsic property rather than a post-process condition. Cold plasma enables inline treatment with short cycle times and minimal residue, aligning with automated, high-throughput production. Designers now select films based on compatibility with plasma exposure and downstream sealing behaviour. Growth follows specification driven manufacturing, where sterility, surface energy, and process stability are linked. Cold plasma moves from workaround to baseline capability.
What Factors Are Affecting the Demand for Cold Plasma Sterilised Films Market in Terms of End Use Application and Packaging Format and Type?
Demand for cold plasma sterilised films is segmented by end use application and packaging format and type across sterile and aseptic packaging environments. Medical and pharma packaging accounts for about 40% of total demand, making it the leading end use segment. Aseptic food packaging, medical disposables, pharma exports, hospital packaging, and advanced medical packs represent additional applications without disclosed share splits. These segments differ in sterility assurance requirements, regulatory oversight, and material compatibility.
Packaging format and type influence sterilisation effectiveness and barrier performance. Sterile lidding and pouch films account for about 45% of demand, followed by form fill seal films, rollstock films, sachet and pouch films, and other formats. Together, these segments explain demand formation shaped by sterilisation validation needs and material integrity rather than overall film consumption volumes.
What Drives Demand by End Use Application in the Cold Plasma Sterilised Films Market?
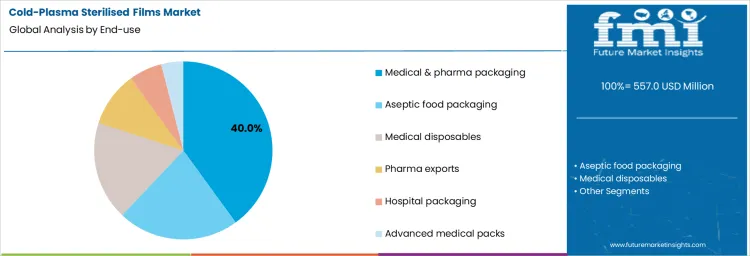
Medical and pharma packaging leads demand with a 40% share because cold plasma sterilisation provides surface level microbial reduction without high thermal exposure. These applications require validated sterility while preserving polymer properties. Aseptic food packaging adopts cold plasma films where heat sensitive products require non thermal treatment. Medical disposables apply sterilised films to maintain hygiene during storage and transport. Pharma export packaging relies on consistent sterility across long transit times. Hospital packaging adopts selectively based on infection control protocols. Advanced medical packs apply cold plasma films where device sensitivity limits alternative sterilisation methods. End use demand therefore reflects sterility assurance requirements rather than packaging turnover rates.
Application driven demand remains stable because sterilisation methods are qualified during packaging design. Medical and pharma uses embed cold plasma films into approved packaging systems. Food applications scale cautiously based on regulatory acceptance. Disposable and export uses follow compliance driven adoption. These patterns limit short term volatility. End use segmentation highlights reliance on non thermal sterilisation where material sensitivity restricts conventional methods.
How Does Packaging Format and Type Shape Demand for Cold Plasma Sterilised Films Market?
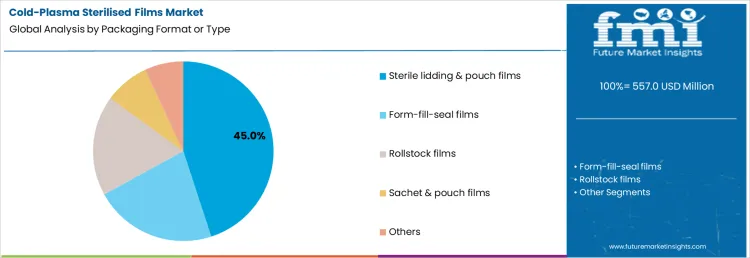
Sterile lidding and pouch films account for about 45% of total demand, making them the leading packaging format. Their dominance reflects widespread use in sealed medical trays and device packs. Form fill seal films are adopted in aseptic food and pharma packaging requiring inline sterilisation compatibility. Rollstock films support high volume disposable packaging. Sachet and pouch films serve pharma exports and unit dose packaging. Other formats address niche hospital and specialty applications. Packaging format selection depends on seal integrity, sterility maintenance, and process compatibility. Demand follows sterilisation reliability and barrier performance rather than innovation in film structure.
Sterile lidding formats maintain leadership due to established medical packaging standards. FFS films scale where continuous processing is required. Rollstock formats remain relevant for disposables. Sachets apply selectively based on dosing needs. Producers avoid altering formats once sterilisation validation is complete. This stabilizes format driven demand. Packaging format and type therefore reinforces concentration in proven sterile packaging structures.
How Are Cold Plasma Sterilised Films Being Applied Across Healthcare and Food Packaging?
Use appears in applications requiring microbial decontamination without thermal or chemical exposure. Medical device packaging, sterile pharmaceutical pouches, and lab consumables incorporate cold-plasma sterilised films to maintain sterility while preserving material properties. Food packaging for perishable items such as ready-to-eat meals, fresh produce, and dairy products uses these films to extend shelf life and prevent pathogen growth. These applications reflect operational and safety priorities rather than decorative or aesthetic purposes, with adoption driven by sterility assurance, material integrity, and regulatory compliance.
What Operational and Material Conditions Support Adoption of Cold Plasma Sterilised Films?
Selection aligns with polymer films compatible with plasma exposure, maintaining barrier properties, mechanical strength, and optical clarity. Processing lines require controlled plasma generation, exposure time, and gas composition to achieve effective sterilisation without surface degradation. Manufacturers rely on validated protocols to meet ISO or FDA sterilisation standards while preserving flexibility and heat-sealability. These conditions emerge from operational reliability, sterility assurance, and material performance priorities in structured packaging workflows.
Which Practical Considerations Limit Wider Use of Cold Plasma Sterilised Films?
High capital investment in plasma equipment and operational expertise can restrict adoption to high-value or regulated products. Effectiveness depends on film thickness, composition, and surface topology. Integration into existing packaging lines may require redesign of sealing, handling, or transport systems. Validation and regulatory approval for medical or food contact use adds time and cost. These factors lead to selective deployment where sterility assurance, product protection, and regulatory compliance justify incremental investment and operational complexity.
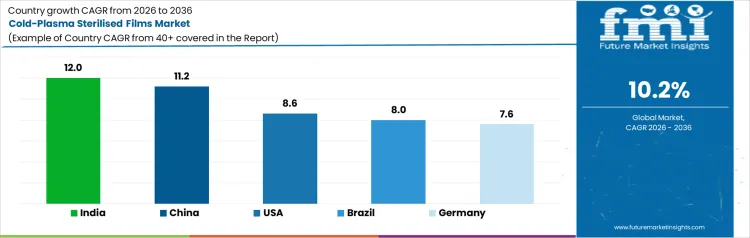
What is the Demand for Cold Plasma Sterilised Films by Country?
| Country | CAGR (%) |
|---|---|
| USA | 8.6% |
| Germany | 7.6% |
| China | 11.2% |
| India | 12.0% |
| Brazil | 8.0% |
The demand for cold plasma sterilised films varies across countries, driven by food safety requirements, medical packaging, and adoption of advanced sterilisation technologies. India leads with a 12.0% CAGR, supported by growing food and pharmaceutical industries, increasing demand for hygienic packaging, and adoption of plasma sterilisation processes. China follows at 11.2%, driven by large scale food production, medical packaging needs, and regulatory compliance for sterilised packaging. The USA grows at 8.6%, reflecting adoption in healthcare, pharmaceuticals, and food sectors. Brazil records 8.0%, shaped by packaging modernization and hygiene standards. Germany posts 7.6%, supported by established packaging industries and strict sterilisation regulations.
How Is Cold-Plasma Sterilization Driving Growth in Films in the United States?
In the United States, revenue from the Cold-Plasma Sterilised Films Market is expanding at a CAGR of 8.6% through 2036, driven by adoption of cold-plasma technology to sterilize and decontaminate films used in food packaging, medical devices, and industrial applications. Manufacturers are integrating cold-plasma sterilization to enhance microbial safety, extend shelf life, and maintain film integrity without heat or chemicals. Demand is concentrated in food and beverage packaging, medical packaging, and industrial protective films. Domestic suppliers provide high-performance, processing-compatible films suitable for extrusion, lamination, and thermoforming processes. Regulatory compliance, recurring packaging production, and hygiene standards are sustaining predictable procurement nationwide.
- Food and beverage packaging, medical packaging, and industrial films drive adoption
- Cold-plasma sterilization improves microbial safety and extends shelf life
- Domestic suppliers provide processing-compatible, high-performance films
- Recurring packaging production sustains predictable procurement
How Are Sterilization Standards Supporting Growth in Germany?
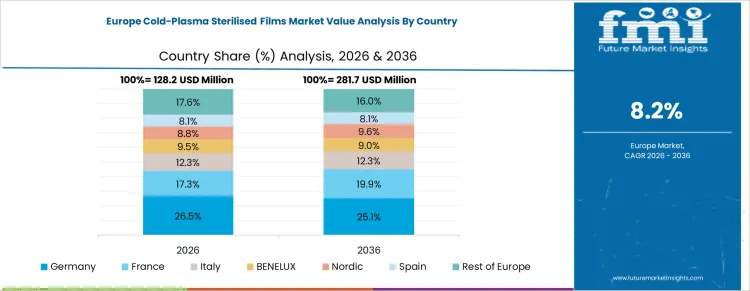
Germany continues to record steady growth in the Cold-Plasma Sterilised Films Market at a CAGR of 7.6% through 2036, supported by stringent hygiene, sterilization, and packaging quality standards. Manufacturers are adopting cold-plasma technology to sterilize films used in food, medical, and industrial applications while preserving material properties. Demand is concentrated in food and beverage packaging, medical device films, and industrial protective films. Domestic suppliers provide high-performance, processing-compatible films suitable for extrusion, lamination, and thermoforming processes. Compliance with sterilization regulations and recurring production cycles is sustaining measured adoption nationwide.
- Food and beverage packaging, medical devices, and industrial films anchor demand
- Cold-plasma technology sterilizes films while preserving material integrity
- Domestic suppliers provide processing-compatible, high-performance films
- Recurring production cycles sustain steady adoption
How Is Industrial and Food Packaging Expansion Accelerating Adoption in China?
In China, revenue from the Cold-Plasma Sterilised Films Market is growing at a CAGR of 11.2% through 2036, driven by rapid adoption of sterilized films in food packaging, medical devices, and industrial applications. Manufacturers are integrating cold-plasma sterilization to improve microbial control, maintain film properties, and extend shelf life. Demand is strong across food and beverage packaging, medical device films, and industrial protective films. Domestic suppliers are scaling production of high-performance, processing-compatible films suitable for extrusion, lamination, and thermoforming. Expanding packaging operations and recurring sterilized film production are sustaining rapid adoption nationwide.
- Food and beverage packaging, medical devices, and industrial films drive adoption
- Cold-plasma sterilization ensures microbial control and extends shelf life
- Domestic suppliers scale processing-compatible, high-performance films
- Recurring sterilized film production sustains consistent adoption
How Is Food Safety and Industrial Application Supporting Growth in India?
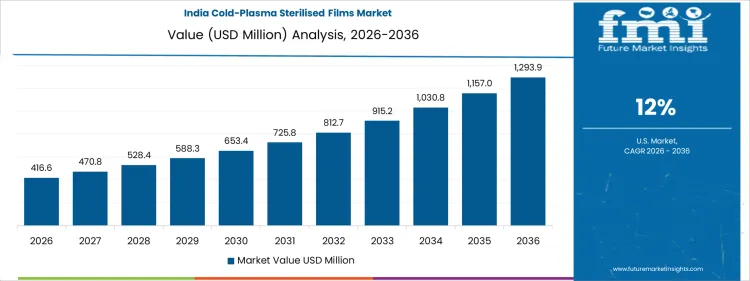
In India, revenue from the Cold-Plasma Sterilised Films Market is expanding at a CAGR of 12% through 2036, supported by increasing use of sterilized films in food packaging, medical devices, and industrial protective applications. Manufacturers are adopting cold-plasma sterilization to reduce microbial contamination, preserve film integrity, and extend product shelf life. Demand is concentrated in food and beverage packaging, medical packaging, and industrial protective films. Domestic suppliers provide high-performance, processing-compatible films suitable for extrusion, lamination, and thermoforming processes. Growth in food safety standards, packaging operations, and recurring sterilized film production are sustaining robust adoption nationwide.
- Food and beverage packaging, medical packaging, and industrial protective films drive adoption
- Cold-plasma sterilization reduces contamination and preserves film integrity
- Domestic suppliers provide high-performance, processing-compatible films
- Recurring production cycles sustain robust adoption
How Is Packaging and Industrial Film Expansion Supporting Adoption in Brazil?
Brazil is seeing strong growth in the Cold-Plasma Sterilised Films Market at a CAGR of 8% through 2036, supported by increasing adoption of sterilized films in food, medical, and industrial applications. Manufacturers are integrating cold-plasma sterilization to improve microbial safety, maintain film properties, and extend product shelf life. Demand is concentrated in food and beverage packaging, medical device films, and industrial protective films. Domestic suppliers are expanding production of high-performance, processing-compatible films suitable for extrusion, lamination, and thermoforming. Growth in packaging infrastructure and recurring sterilized film production are sustaining robust procurement nationwide.
- Food and beverage packaging, medical devices, and industrial protective films drive adoption
- Cold-plasma sterilization improves microbial safety and maintains film integrity
- Domestic suppliers expand processing-compatible, high-performance films
- Recurring production cycles sustain robust procurement
Which Companies Compete in The Cold Plasma Sterilised Films Market When Sterilisation Equipment Dictates Material Choice?
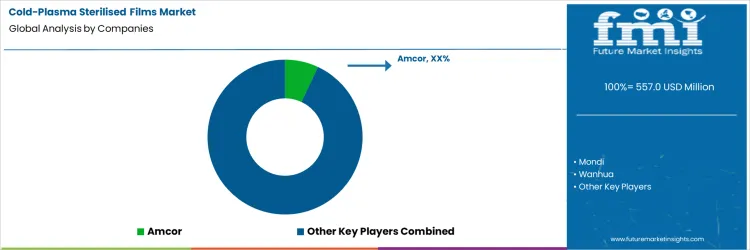
Cold plasma sterilisation shifts control away from film suppliers toward sterilisation system constraints, forcing material selection to start with surface chemistry tolerance rather than packaging design. Film failure modes such as surface embrittlement, loss of sealability, or uncontrolled oxidation determine acceptance. Toray and DuPont enter the value chain through polymer films and surface materials that maintain molecular stability after repeated plasma exposure. Amcor and Mondi are evaluated not as generic flexible packaging suppliers, but on whether specific film structures survive plasma treatment without disrupting downstream forming, sealing, or print adhesion. In this market, qualification often begins inside pilot sterilisation chambers, with materials rejected early if surface response is inconsistent across treatment cycles.
Competitive outcomes are decided during conversion, not resin specification. UFlex and regional converters determine commercial viability by running plasma treated films at production speeds, where yield loss and line stoppages carry more weight than laboratory barrier data. Wanhua participates upstream through polymer supply aligned with plasma compatible formulations rather than finished films. Local converters frequently act as gatekeepers, choosing which films progress from trial to scaled use based on equipment fit and operational stability. Market structure remains fragmented because sterilisation parameters differ widely across facilities, limiting standardisation. Competitive positioning depends on how predictably materials behave under real plasma exposure, how easily converters adapt them to existing lines, and how quickly failures can be diagnosed and corrected.
Key Players in the Cold Plasma Sterilised Films Market
- Amcor
- Mondi
- Wanhua
- UFlex
- Local converters
- Toray
- DuPont
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| End-use / Application | Medical & pharma packaging, Aseptic food packaging, Medical disposables, Pharma exports, Hospital packaging, Advanced medical packs |
| Packaging Format / Type | Sterile lidding & pouch films, Form-fill-seal films, Rollstock films, Sachet & pouch films, Others |
| Material | PE, PET, PA films, Multilayer barrier films, PET / PE blends, PE films, Others |
| Technology | Atmospheric cold-plasma surface sterilisation, Inline plasma treatment, High-speed plasma chambers, Cost-optimised plasma units, Others |
| Region | Asia Pacific (China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Rest of Asia Pacific), Europe (Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, Rest of Europe), North America (United States, Canada, Mexico), Latin America (Brazil, Chile, Rest of Latin America), Middle East & Africa (KSA, Other GCC Countries, Turkey, South Africa, Other African Union, Rest of Middle East & Africa) |
| Key Companies Profiled | Amcor, Mondi, Wanhua, UFlex, Local converters, Toray, DuPont |
| Additional Attributes | Dollar by sales by end-use application, Dollar by sales by packaging format, Dollar by sales by material, Dollar by sales by technology adoption, Dollar by sales by region, Sterilisation performance and validation, Process integration and throughput, Film compatibility with plasma treatment, Material integrity and barrier properties, Adoption in regulated food and medical packaging |
Cold Plasma Sterilised Films Market Segmentation
By End-use / Application:
- Medical & pharma packaging
- Aseptic food packaging
- Medical disposables
- Pharma exports
- Hospital packaging
- Advanced medical packs
By Packaging Format / Type:
- Sterile lidding & pouch films
- Form-fill-seal films
- Rollstock films
- Sachet & pouch films
- Others
By Material:
- PE, PET, PA films
- Multilayer barrier films
- PET / PE blends
- PE films
- Others
By Technology:
- Atmospheric cold-plasma surface sterilisation
- Inline plasma treatment
- High-speed plasma chambers
- Cost-optimised plasma units
- Others
By Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the cold-plasma sterilised films market in 2026?
The global cold-plasma sterilised films market is estimated to be valued at USD 557.0 million in 2026.
What will be the size of cold-plasma sterilised films market in 2036?
The market size for the cold-plasma sterilised films market is projected to reach USD 1,476.0 million by 2036.
How much will be the cold-plasma sterilised films market growth between 2026 and 2036?
The cold-plasma sterilised films market is expected to grow at a 10.2% CAGR between 2026 and 2036.
What are the key product types in the cold-plasma sterilised films market?
The key product types in cold-plasma sterilised films market are medical & pharma packaging, aseptic food packaging, medical disposables, pharma exports, hospital packaging and advanced medical packs.
Which packaging format or type segment to contribute significant share in the cold-plasma sterilised films market in 2026?
In terms of packaging format or type, sterile lidding & pouch films segment to command 45.0% share in the cold-plasma sterilised films market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use , 2026 to 2036
- Medical & pharma packaging
- Aseptic food packaging
- Medical disposables
- Pharma exports
- Hospital packaging
- Advanced medical packs
- Y to o to Y Growth Trend Analysis By End-use , 2021 to 2025
- Absolute $ Opportunity Analysis By End-use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging Format or Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging Format or Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging Format or Type, 2026 to 2036
- Sterile lidding & pouch films
- Form-fill-seal films
- Rollstock films
- Sachet & pouch films
- Others
- Y to o to Y Growth Trend Analysis By Packaging Format or Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging Format or Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material, 2026 to 2036
- PE, PET, PA films
- Multilayer barrier films
- PET / PE blends
- PE films
- Others
- Y to o to Y Growth Trend Analysis By Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Material, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Atmospheric cold-plasma surface sterilisation
- Inline plasma treatment
- High-speed plasma chambers
- Cost-optimised plasma units
- Others
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-use
- By Packaging Format or Type
- By Material
- By Technology
- Competition Analysis
- Competition Deep Dive
- Amcor
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Mondi
- Wanhua
- UFlex
- Local converters
- Toray
- DuPont
- Amcor
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Packaging Format or Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End-use
- Figure 6: Global Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Packaging Format or Type
- Figure 9: Global Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Material
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End-use
- Figure 29: North America Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Packaging Format or Type
- Figure 32: North America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Material
- Figure 35: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Technology
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End-use
- Figure 42: Latin America Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Packaging Format or Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Material
- Figure 48: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Technology
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by End-use
- Figure 55: Western Europe Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Packaging Format or Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Material
- Figure 61: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Technology
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by End-use
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Packaging Format or Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Material
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by End-use
- Figure 81: East Asia Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Packaging Format or Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Material
- Figure 87: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Technology
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by End-use
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Packaging Format or Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by End-use
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Packaging Format or Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging Format or Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Packaging Format or Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

