Corneal Ulcer Treatment Market Size, Market Forecast and Outlook By FMI
Summary of the Corneal Ulcer Treatment Market
- Demand and Growth Drivers
- Emergency eye care volume stays firm since microbial keratitis can progress quickly and ophthalmologists start treatment before culture confirmation in severe cases.
- Contact lens wear keeps a steady treated pool as bacterial keratitis keeps affecting lens wearers and non-lens wearers in routine practice.
- Fungal ulcers sustain a larger treatment burden in tropical settings due to delayed presentation often requiring longer anti-fungal therapy and closer follow up.
- Product and Segment View
- Drug therapy is projected to lead treatment type demand as most ulcer episodes begin with intensive topical anti-infective prescribing.
- Eye drops are expected to lead dosage form demand since corneal exposure and dose frequency align well with topical schedules.
- Bacterial ulcers are expected to lead ulcer type demand due to broad spectrum topical antibiotics remaining central in empiric care.
- Geography and Competitive Outlook
- India and China lead the country growth set as high microbial keratitis burden and expanding ophthalmic access keep treated volume on an upward path.
- United States holds a large revenue base due to branded ophthalmic products and compounded rescue therapy influencing treatment spend per episode.
- Companies such as Dompé and Santen together with Bausch & Lomb and Harrow contribute through anti-infective depth corneal healing support and eye care channel reach.
- Analyst Opinion
- FMI observes that "Corneal ulcer treatment demand is aligned to speed of diagnosis and confidence in first-line prescribing. Clinicians move quickly since a few missed hours can worsen scarring risk and increase the chance of surgical rescue. Suppliers with dependable topical anti-infective access and a stronger corneal healing portfolio hold a better position in referral-driven ophthalmology care. Growth through 2036 is likely to come from deeper fungal care coverage together with branded therapy for persistent epithelial defects and neurotrophic ulcer cases."
- Corneal Ulcer Treatment Market Value Analysis
- Acute care prescribing keeps annual treatment demand stable across hospitals and ophthalmology clinics as corneal infection can threaten vision within a short treatment window.
- Revenue holds strongest in branded and compounded ophthalmic lines that support intensive dosing schedules during the early treatment phase.
- Revenue for the sector rises in persistent epithelial defect care since biologic and nerve growth factor therapies command a higher spend per treated case.
- Hospital pharmacy access shapes realized sales as emergency dispensing shortens treatment delay in severe bacterial and fungal presentations.
Corneal Ulcer Treatment Market Definition
The corneal ulcer treatment sector covers prescription drugs and supportive ophthalmic therapies used to manage infectious ulcers and severe non-infectious epithelial defects that present with stromal loss or a persistent corneal surface break.
Corneal Ulcer Treatment Market Inclusions
The study includes global sizing and forecast from 2026 to 2036 for ulcer type treatment type drug class dosage form end user distribution channel and geography. Country growth patterns and competitive positioning are included together with a scope table and segment taxonomy.
Corneal Ulcer Treatment Market Exclusions
Refractive surgery devices diagnostic imaging systems contact lens hardware and general dry eye products stay outside this study unless they are prescribed directly for active corneal ulcer management or epithelial healing support.
Corneal Ulcer Treatment Market Research Methodology
- Primary Research: Involved review of current product labels and official company pages from ophthalmic therapy suppliers with active commercial presence in corneal care.
- Desk Research: Combined public clinical references treatment guidance government disease burden information and peer-reviewed studies on microbial keratitis and ulcer management.
- Market Sizing and Forecasting: Aligned treated episode burden with therapy mix by ulcer type and weighted spend by treatment setting and dosage intensity.
- Data Validation: Cross-checked segment leadership country outlook and company positioning against current labels official updates and current eye care practice references.
Why is the Corneal Ulcer Treatment Market Growing?
- Urgent eye care demand sustains a steady prescribing activity as microbial keratitis can scar the cornea quickly after delayed presentation.
- Topical anti-infective therapy remains important as empiric treatment starts before culture results in a large share of acute episodes.
- Neurotrophic ulcer therapy adds value due to persistent epithelial defects require longer treatment duration and specialist follow-up.
Hospital ophthalmology teams sustain demand for keratitis treatment as first-line care starts as soon as stromal infiltration and epithelial loss are confirmed. Broad-spectrum drops maintain the largest prescribing share as clinicians require fast corneal surface coverage during the first few treatment days. This prescribing pattern suggests positive outlook for ophthalmic eye drop sector with ulcer care across emergency rooms and referral clinics.
Fungal care adds another growth layer in tropical practice since deeper infiltrates and late presentation often lengthen therapy and follow-up burden. Commercial expansion in fungal keratitis treatment and ocular drug delivery systems supports this longer-contact therapy use as ophthalmologists look for steadier corneal contact and better adherence under intensive dosing schedules.
Market Segmentation Analysis
- Drug therapy is expected to account for 69.0% share in 2026 as most ulcer cases start with topical anti-infective treatment before referral escalation.
- Eye drops are projected to contribute 74.0% share in 2026 due to frequent corneal dosing being easier to manage through topical dispensing.
- Bacterial ulcers are anticipated to represent 41.0% share in 2026 since empiric antibiotic coverage remains the common opening step in acute care.
- Hospitals are likely to secure 46.0% share in 2026 as severe ulcer cases often enter care through emergency and inpatient linked ophthalmology units.
Segment structure in this sector tracks ulcer type treatment intensity drug class dosage form care setting and dispensing route as prescribing patterns shift with severity and organism profile. Referral ophthalmologists give more weight to early anti-infective access in bacterial care while fungal and neurotrophic cases lift demand for longer treatment support. Channel mix adds another layer because hospital pharmacies supply acute therapy quickly while retail and specialty outlets support refill cycles after discharge.
Corneal Ulcer Treatment Market Analysis by Ulcer Type
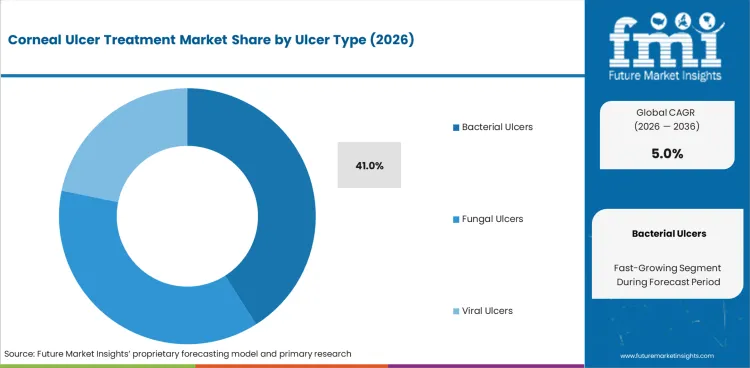
- Bacterial ulcers are expected to account for 41.0% share of ulcer type demand in 2026 since empiric antibacterial therapy is the first response in a large share of acute presentations.
- Broad-spectrum topical coverage keeps this group ahead as clinicians must act quickly before culture results arrive in emergency practice.
Insights into the Fungal Ulcers Segment
- Fungal ulcers are likely to advance at a quicker pace as tropical disease burden and longer treatment duration lift value per managed case.
- Ophthalmologists keep close watch on these cases since deeper stromal involvement raises scarring risk and extends follow up time.
Corneal Ulcer Treatment Market Analysis by Treatment Type
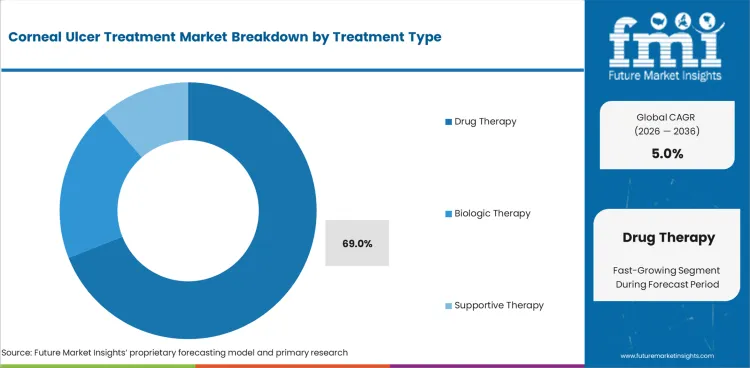
- Drug therapy is projected to contribute 69.0% share in 2026 as anti-infective drops and adjunct medicines lead first-line care.
- Referral clinics favor this path since treatment can start on the same day without waiting for surgical scheduling.
Insights into the Biologic Therapy Segment
- Biologic therapy is likely to rise faster due to persistent epithelial defects and neurotrophic ulcers requiring stronger surface-healing support.
- Adoption of serum-based and nerve growth factor-aligned care is rising as specialists try to close stubborn defects before perforation risk rises.
Corneal Ulcer Treatment Market Analysis by Drug Class
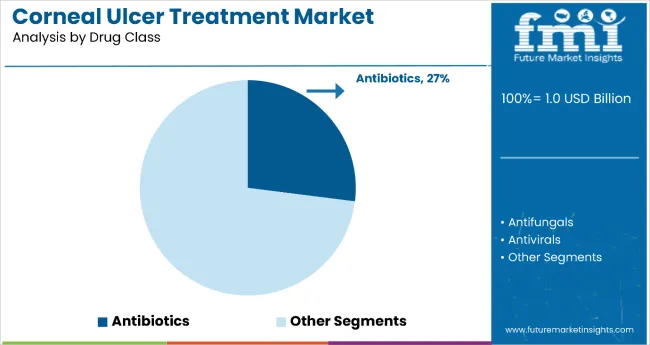
- Antibiotics are expected to hold 36.0% share of drug class demand in 2026 since suspected bacterial disease sustains steady use of fluoroquinolone and fortified drops.
- Empiric prescribing supports this class as bacterial ulcers account for a large part of urgent corneal infection workload.
Insights into the Antifungals Segment
- Antifungals are likely to post faster gains as natamycin-based care and specialist escalation are used longer in filamentous disease.
- Treatment burden rises in this antifungal sector since delayed fungal diagnosis often lengthens dosing frequency and clinic review cadence.
Corneal Ulcer Treatment Market Analysis by Dosage Form
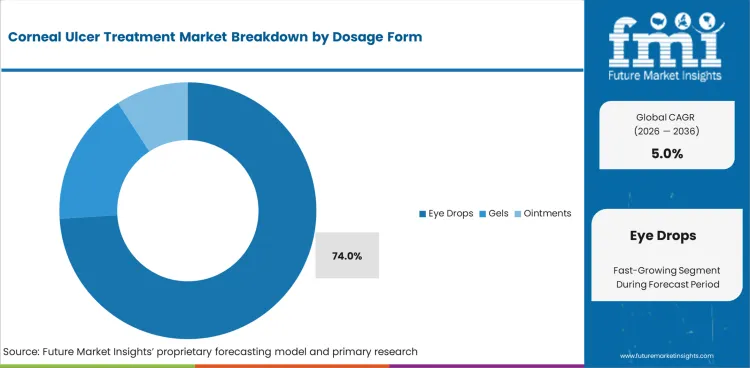
- Eye drops are set to make up 74.0% share in 2026 due to topical delivery being the standard route in most ulcer pathways.
- Frequent instillation contributes to the rise in dosage form as corneal drug exposure requires close dose spacing during the first treatment phase.
Insights into the Gels Segment
- Gels are likely to record faster gains as overnight surface protection and adherence support help in persistent epithelial defect care.
- Specialists use these formats to reduce surface dryness and extend contact time in selected recovery periods.
Corneal Ulcer Treatment Market Analysis by End User
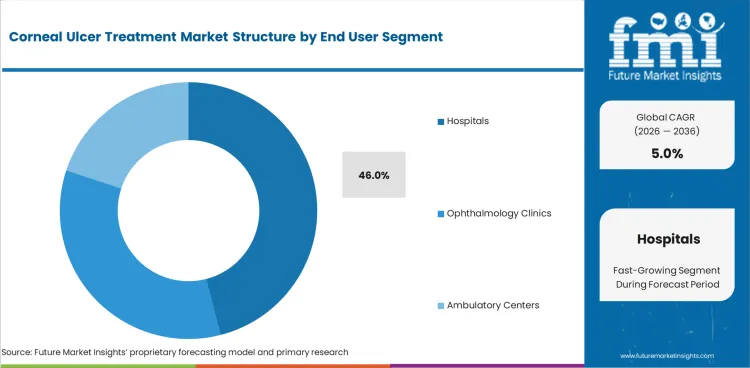
- Hospitals are projected to secure 46.0% share in 2026 due to advanced ulcer cases often entering care through emergency ophthalmology services.
- Severe pain hypopyon and perforation risk keep hospital based treatment channels ahead in high-acuity disease.
Insights into the Ophthalmology Clinics Segment
- Ophthalmology clinics are projected to expand at a rapid pace as follow up titration and culture guided therapy integrate into outpatient specialist care after initial stabilization.
- Clinic-based review remains steady as healing progress and scarring risk need repeated slit lamp assessment over several visits.
Corneal Ulcer Treatment Market Analysis by Distribution Channel
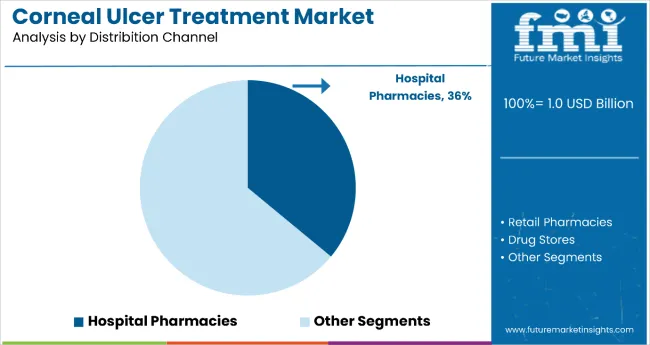
- Hospital pharmacies are expected to account for 43.0% share of distribution channel demand in 2026 since urgent dispensing shortens treatment delay in acute care.
- Distribution channel demand is influenced by severe ulcer cases often requiring initial doses before discharge or transfer to specialist follow-up.
Insights into the Specialty Pharmacies Segment
- Specialty pharmacies are likely to rise faster as biologic therapy and hard-to-source ophthalmic products require closer fulfillment support.
- Access programs and cold chain handling support uptake in ulcer cases that move beyond common anti-infective dispensing.
Corneal Ulcer Treatment Market Drivers Restraints and Opportunities
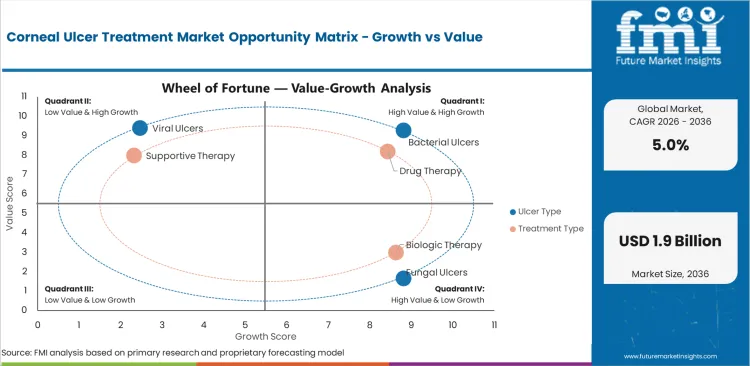
- Urgent microbial keratitis treatment supports repeat ophthalmic prescribing through hospital ophthalmology units and referral clinic pathways.
- Culture delay and poor early adherence can slow treatment success since corneal infection can deepen before targeted therapy is established.
- Biologic surface healing support opens commercial room for suppliers that can serve persistent epithelial defects through specialist eye care channels.
Corneal ulcer treatment demand expands through acute anti-infective therapy and specialist follow-up as vision-loss risk pushes clinicians toward fast intervention. Hospitals and ophthalmology clinics keep therapy volume consistent as bacterial ulcers and fungal disease and neurotrophic surface defects each require repeated review and dose adjustment. Growth faces limits from delayed presentation along with poor culture access, and the need for very frequent dosing during early care.
Rapid First Line Therapy Supports Demand
Empiric therapy keeps the largest revenue base as ophthalmologists must protect the cornea before culture confirmation is available. Topical anti-infective prescribing is the first choice in most care paths. This keeps antibacterial and antifungal drops demand firm across emergency and referral settings. Brand presence is important here since formulation tolerance and supply continuity influence which products reach first-line use.
Delayed Diagnosis Slows Outcomes
Late presentation reduces treatment efficiency as stromal damage can progress before the correct pathogen is identified. Fungal disease and acanthamoeba cases are more exposed to this problem since early signs can look similar to bacterial infection. This delay raises the chance of longer therapy and corneal scarring and referral to surgical rescue.
Healing Focused Therapy Opens Upside
Persistent epithelial defects create a stronger value pocket as specialists need products that support re-epithelialization after infection control is achieved. Therapeutic contact lenses along with advanced biologic surface care can expand treatment depth in referral practice. Suppliers that connect corneal healing support with specialist distribution can capture this higher spending segment.
Regional Analysis
.webp)
| Country |
CAGR 2026 to 2036 |
| India |
6.6% |
| China |
6.1% |
| Brazil |
5.9% |
| Mexico |
5.4% |
| Saudi Arabia |
5.3% |
| Germany |
4.8% |
| United States |
4.3% |
Source: Future Market Insights analysis based on current clinical burden references and covered country narratives in 2026.
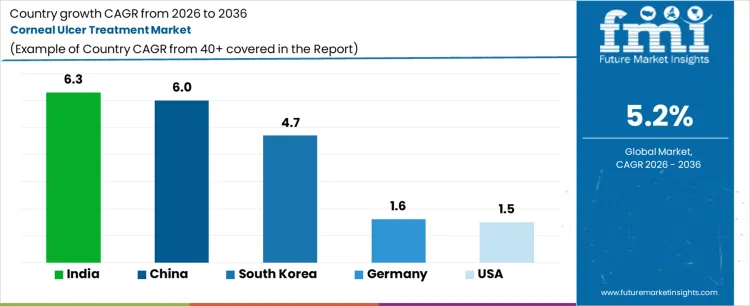
Analysis of Corneal Ulcer Treatment Market by Key Countries
- India is projected to post the fastest pace at 6.6% CAGR through 2036 since fungal keratitis burden and expanding ophthalmology access keep treated case volume high.
- China is expected to follow at 6.1% CAGR through 2036 due to large urban hospital systems and high outpatient eye care traffic influencing faster diagnosis and treatment conversion.
- Brazil at 5.9% CAGR and Mexico at 5.4% CAGR are set to maintain strong growth through 2036 as referral eye care access expands in large metropolitan corridors.
Country growth differs with microbial burden specialist access treatment spend and the speed at which patients enter ophthalmology care. India and China rise faster as treated volume is larger and fungal disease contributes more value intensive care episodes. The United States expands at a steadier pace as the therapy base is larger and branded ophthalmic spending is already well established.
Demand Outlook for Corneal Ulcer Treatment Market in India
Demand for the sector in India is projected to record a 6.6% CAGR through 2036 since fungal keratitis volume is high and tertiary eye hospitals handle a large referral load from delayed rural presentation. Large ophthalmic networks keep treatment output consistent influencing a faster use of anti-fungal therapy and specialist follow-up. Referral patterns in India lift value as late-stage cases often need longer care before the corneal surface closes.
- Tropical disease burden sustains a steady anti-fungal use in tertiary eye centers as fungal corneal ulcers need longer treatment and closer follow up.
- Late presentation increases treatment duration in severe referrals as patients often reach specialist hospitals after the infection has advanced.
- Specialist hospital networks support faster treatment conversion after diagnosis since corneal clinics manage complex ulcer cases with structured follow up.
Demand Outlook for Corneal Ulcer Treatment Market in China
The sector in China is expected to rise at a 6.1% CAGR through 2036 since high patient volumes in urban eye hospitals support faster diagnosis and broader treatment capture for microbial keratitis. Hospital-based care demand remains firm in large cities influencing topical anti-infective dispensing together with close follow up in specialist clinics. Revenue growth in China is supported by larger outpatient flow and rising use of advanced ophthalmic therapies in referral practice.
- Urban eye hospitals process large volumes of acute corneal infection cases since major cities have dense outpatient ophthalmology networks.
- Hospital pharmacies support fast access to first line topical therapy due to many diagnosed patients receiving treatment inside large eye care facilities.
- Referral care in major cities lifts use of higher-value ophthalmic products as specialist clinicians manage difficult ulcers through repeat visits.
Opportunity Analysis of Corneal Ulcer Treatment Market in Brazil
The market adoption in Brazil is likely to grow at a 5.9% CAGR through 2036 as public and private referral eye care channels are expanding corneal infection treatment access in major states. Follow up intensity supports revenue growth since specialists manage fungal disease and persistent epithelial defects through repeated outpatient review. Growth in Brazil is aligned to wider ophthalmic service access and stronger dispensing depth in urban centers.
- Metropolitan eye care access supports faster treatment initiation after diagnosis since large state centers concentrate specialist ophthalmology services.
- Repeat specialist review raises demand for refill therapy due to fungal disease and slow epithelial healing often requiring longer care.
- Private ophthalmology channels add value in higher spend treatment episodes as insured patients can access specialist corneal care more quickly.
Sales Analysis of Corneal Ulcer Treatment Market in Mexico
Mexico is expected to rise at a 5.4% CAGR through 2036 since referral access in large cities is improving and ophthalmology services in hospitals are capturing more acute corneal cases. Bacterial ulcer treatment holds the largest revenue share and retail refill activity influences continuity after hospital discharge. Mexico benefits from stronger dispensing routes in urban regions with growing specialist eye care availability.
- Urban referral hospitals capture higher acuity corneal infections since large cities concentrate more on emergency eye care and specialist follow-up.
- Retail refill channels support continued dosing after early emergency treatment as patients often require topical therapy after discharge.
- Specialist access in large cities improves treatment continuity as ophthalmologists can monitor healing and adjust therapy during follow up.
Sales Analysis of Corneal Ulcer Treatment Market in Saudi Arabia
Saudi Arabia is projected to expand at a 5.3% CAGR through 2036 due to tertiary hospital investment and wider ophthalmology coverage supporting earlier specialist management of severe corneal disease. Hospital pharmacies are significant channels in this country as they keep acute care dispensing closely aligned to inpatient linked eye services. Growth in Saudi Arabia is supported by expanding referral capacity rather than by a very large disease pool.
- Tertiary hospitals hold a strong role in acute corneal care pathways since severe eye infections are usually managed inside specialist facilities.
- Hospital pharmacy access shortens treatment delay in severe presentations as therapy can begin soon after ophthalmology review.
- Referral expansion supports higher treatment conversion in specialist centers since more patients are opting for an early structured corneal care treatment.
Demand Outlook for Corneal Ulcer Treatment Market in Germany
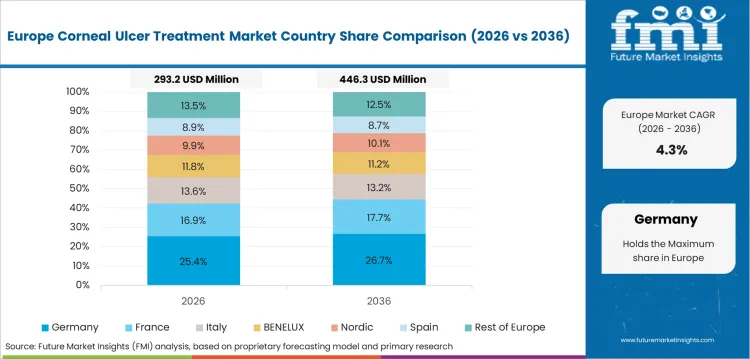
The sector in Germany is expected to record a 4.8% CAGR through 2036 since specialist ophthalmology capacity is strong with a more mature base and treatment capture being already well developed. Revenue growth comes from corneal healing support and longer managed care in persistent epithelial defects. Germany benefits from organized referral pathways and dependable hospital access across advanced eye care centers.
- Mature specialist networks support stable treatment conversion after diagnosis since ophthalmology access is well developed across hospital systems.
- Advanced corneal care raises value per persistent epithelial defect case due to difficult healing episodes requiring repeat treatment and monitoring.
- A larger established base sustains a measured future expansion measured as diagnosis and treatment capture are already well developed.
Demand Outlook for Corneal Ulcer Treatment Market in the United States
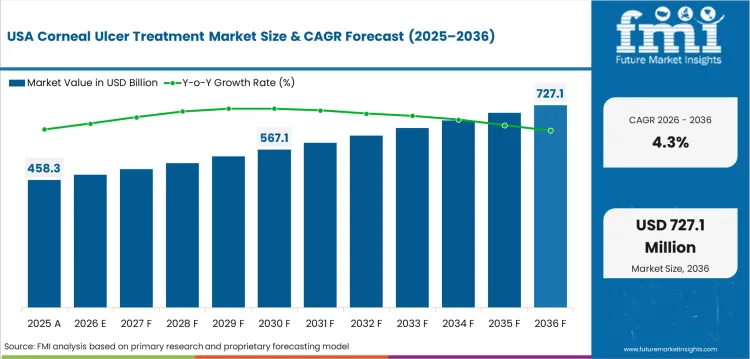
Demand in the United States is projected to grow at a 4.3% CAGR through 2036 as the therapy base is large and branded ophthalmic spending already lifts current treatment value. Clinical guidance and compounding support sustain established anti-infective care across hospitals and corneal specialist clinics. Growth in the United States is steadier as penetration is high and future gains come more from product mix than from untreated volume capture.
- Branded ophthalmic therapy lifts spend per treated episode as advanced products and specialist prescribing influence the treatment mix.
- Compounded fortified drops support severe bacterial ulcer management since corneal specialists use tailored therapy for difficult infections.
- A mature base sustains a steady future expansion as treatment access is already high across hospitals and corneal specialist clinics.
Competitive Landscape and Strategic Positioning
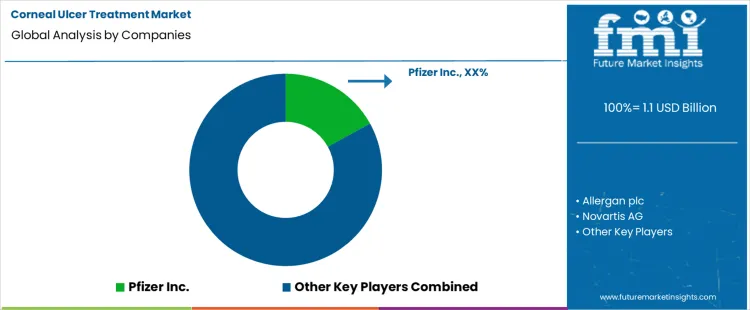
- Competition is moderately concentrated across ophthalmic suppliers with anti-infective depth along with corneal healing support and established eye care channel access.
- Dompé holds a strong position through Oxervate and specialist corneal disease focus that supports higher value epithelial healing care.
- Santen, Bausch & Lomb, Harrow and Alcon compete through prescription ophthalmology portfolios, hospital access and corneal care relationships.
Competition in corneal ulcer treatment centers on speed of ophthalmology access and confidence in topical prescribing under urgent care conditions. Suppliers with dependable anti-infective depth hold an advantage in bacterial and fungal ulcer management as clinicians need immediate access to a familiar product line. Harrow contributes through NATACYN distribution and ophthalmic access programs while Viatris and Alcon benefit from established anti-infective channel relationships across eye care settings.
Corneal healing care further drives the competition as persistent epithelial defects and neurotrophic ulcer cases are influenced by referral driven therapy selection. Dompé stands out through Oxervate and its specialist corneal disease positioning. Santen and Bausch & Lomb are expanding this field through corneal health work and targeted development activity that can deepen their role beyond standard anti-infective use.
Route to clinic is important in this sector since hospital pharmacies and referral networks influence which products reach first-line use and which are reserved for rescue care. Eye infection treatment and ophthalmic drug packaging sectors reflect how adjacent ophthalmic supply base supports brand reach in ulcer care. Companies that combine ophthalmology sales access with specialist education can defend pricing better in higher-acuity segments.
Competitive Benchmarking: Corneal Ulcer Treatment Market
| Company |
Anti Infective Depth |
Corneal Healing Portfolio |
Ophthalmology Access |
Geographic Reach |
| Dompé |
Medium |
High |
Strong |
High |
| Santen Pharmaceutical |
Medium |
Medium |
Strong |
High |
| Bausch & Lomb |
Medium |
Medium |
Strong |
High |
| Harrow |
High |
Low |
Strong |
Medium |
| Alcon |
Low |
Low |
Strong |
High |
| Viatris |
Medium |
Low |
Moderate |
High |
Source: Future Market Insights competitive analysis based on current product labels company updates and covered country narratives in 2026.
Key Developments in Corneal Ulcer Treatment Market
- In July 2024, Harrow’s NATACYN label was revised in the United States and NATACYN remained indicated for fungal blepharitis, conjunctivitis, and keratitis.
- In July 2024, Bausch & Lomb acquired Trukera Medical to expand its USA surgical presence and strengthen its dry eye diagnostic position.
- In September 2025, Harrow launched Harrow Access for All to expand pricing access across its ophthalmic portfolio.
- In December 2025, Santen and SERI announced SONIC 2.0 to advance disease-modifying therapies for major eye diseases including ocular surface diseases.
Key Players in the Corneal Ulcer Treatment Market
Major Global Players
- Dompé
- Santen Pharmaceutical
- Bausch & Lomb
- Alcon
- AbbVie
Regional Participants
Report Scope and Coverage
| Item |
Value |
| Market value in 2025 |
USD 1.0 billion |
| Market value in 2026 |
USD 1.1 billion |
| Market value in 2036 |
USD 1.8 billion |
| CAGR from 2026 to 2036 |
5.0% |
| Quantitative units |
USD billion |
| Market definition |
Therapies used to manage infectious corneal ulcers and severe epithelial defect driven ulceration |
| By ulcer type |
Bacterial Ulcers, Fungal Ulcers, Viral Ulcers, Acanthamoeba Ulcers, Neurotrophic Ulcers, Sterile Ulcers |
| By treatment type |
Drug Therapy, Biologic Therapy, Supportive Therapy, Compounded Therapy, Adjunct Procedures |
| By drug class |
Antibiotics, Antifungals, Antivirals, Antiparasitics, Nerve Growth Factor, Artificial Tears |
| By dosage form |
Eye Drops, Gels, Ointments, Suspension, Biologic Solution |
| By end user |
Hospitals, Ophthalmology Clinics, Ambulatory Centers |
| By distribution channel |
Hospital Pharmacies, Retail Pharmacies, Specialty Pharmacies, Online Pharmacies |
| Regions covered |
North America, Latin America, Europe, East Asia, South Asia, Middle East and Africa |
| Countries covered |
India, China, Brazil, Mexico, Saudi Arabia, Germany, United States |
| Key companies profiled |
Dompé, Santen Pharmaceutical, Bausch + Lomb, Harrow, Alcon, Viatris, AbbVie |
| Forecast period |
2026 to 2036 |
| Approach |
FMI public treatment model aligned with clinical burden references, labels and company updates |
Corneal Ulcer Treatment Market Breakdown by Ulcer Type, Treatment Type, Drug Class, Dosage Form, End User, Distribution Channel, and Region
Corneal Ulcer Treatment Market Segmented by Ulcer Type
- Bacterial Ulcers
- Fungal Ulcers
- Viral Ulcers
- Acanthamoeba Ulcers
- Neurotrophic Ulcers
- Sterile Ulcers
Corneal Ulcer Treatment Market Segmented by Treatment Type
- Drug Therapy
- Biologic Therapy
- Supportive Therapy
- Compounded Therapy
- Adjunct Procedures
Corneal Ulcer Treatment Market Segmented by Drug Class
- Antibiotics
- Antifungals
- Antivirals
- Antiparasitics
- Nerve Growth Factor
- Artificial Tears
Corneal Ulcer Treatment Market Segmented by Dosage Form
- Eye Drops
- Gels
- Ointments
- Suspension
- Biologic Solution
Corneal Ulcer Treatment Market Segmented by End User
- Hospitals
- Ophthalmology Clinics
- Ambulatory Centers
Corneal Ulcer Treatment Market Segmented by Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Specialty Pharmacies
- Online Pharmacies
Corneal Ulcer Treatment Market by Region
- North America
- Latin America
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- East Asia
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
Research Sources and Bibliography
- USA National Library of Medicine. (2024). NATACYN suspension label. DailyMed.
- National Center for Biotechnology Information. (2025). Corneal ulcer. StatPearls.
- Centers for Disease Control and Prevention. (2025, May 27). What causes contact lens-related bacterial keratitis?
- European Medicines Agency. (2024). Oxervate: EPAR product information.
- Harrow. (2025, September 25). Harrow launches Harrow Access for All.
- Bausch + Lomb. (2024, July 23). Bausch + Lomb acquires diagnostic company Trukera Medical.
- Santen Pharmaceutical Co., Ltd. (2025, December 3). Singapore Eye Research Institute and Santen announce SONIC 2.0 collaboration.
- American Academy of Ophthalmology. (2024). Bacterial keratitis preferred practice pattern. Ophthalmology.
- Shi, Q., Li, Y., et al. (2024). Direct cost analysis of microbial keratitis in North China. Tropical Medicine and Infectious Disease.
The bibliography is provided for reader reference and direct source checking.
This Report Answers
- How large the corneal ulcer treatment sector is in 2026 and how far it can rise by 2036?
- Which ulcer type holds the lead and which treatment format leads in 2026?
- Why do eye drops and drug therapy hold the clearest lead in the current treatment structure?
- Why do India and China lead the published country outlook through 2036?
- How hospital pharmacies shape early treatment access in severe ulcer care?
- Which supplier capabilities help ophthalmic companies defend position in anti-infective and corneal healing care?
- What clinical burden factors and treatment pathways support the forecast methodology used in this study?
Frequently Asked Questions
How large is the corneal ulcer treatment market in 2026?
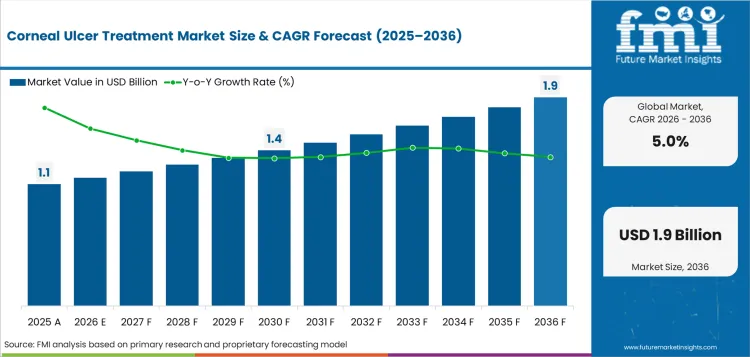
The corneal ulcer treatment sector is expected to cross USD 1.1 billion in 2026 based on modeled treated episode burden and therapy mix.
What will the corneal ulcer treatment market be worth by 2036?
Industry value is expected to reach USD 1.8 billion by 2036 as specialist corneal care and longer healing support lift treatment spend.
What CAGR is projected for the corneal ulcer treatment market from 2026 to 2036?
The corneal ulcer treatment sector is projected to expand at a 5.0% CAGR from 2026 to 2036 under a measured ophthalmic care outlook.
Which treatment type leads the corneal ulcer treatment market?
Drug therapy leads treatment type demand with a projected 69.0% share in 2026 as first line care starts with topical anti-infective prescribing.
Which dosage form leads the corneal ulcer treatment market?
Eye drops lead dosage form demand with an expected 74.0% share in 2026 since most ulcer pathways use frequent topical dosing schedules.
Which country is projected to grow fastest in the corneal ulcer treatment market?
India leads the country outlook with an expected 6.6% CAGR through 2036 as fungal disease burden and referral eye care volume stay high.








.webp)





