Customized Premix Market
Customized Premix Market Analysis- Size, Share, and Forecast 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Customized Premix Market Forecast and Outlook 2026 to 2036
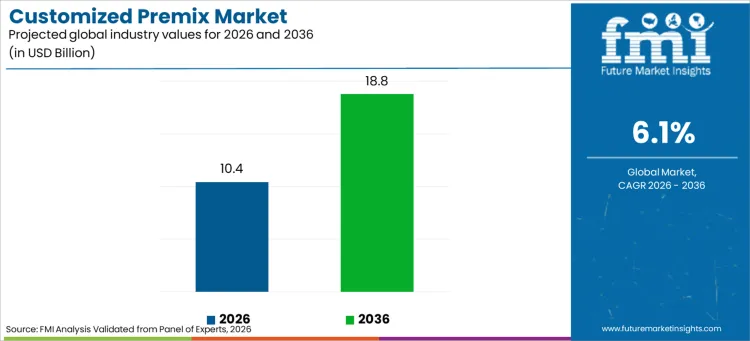
The customized premix market is projected to expand from USD 10.40 billion in 2026 to USD 18.79 billion by 2036, registering a 6.1% CAGR. Formulation programs that require repeatable nutrient delivery, stable batch outcomes, and predictable sourcing through long procurement cycles are reinforcing growth. Customers are tightening acceptance thresholds around blend uniformity, mixing reliability, and specification discipline, which raises the value of suppliers that can deliver consistent output across repeated replenishment.
Buying behavior is also shifting toward system-style procurement, where premix decisions are coordinated across product pipelines rather than made SKU by SKU. This approach is supporting broader adoption of premix solutions that align with structured fortification roadmaps and standardized ingredient strategies used across customized premixes and adjacent food premix portfolios.
Qualification cycles remain deliberate because any change in ingredient type, blending pathway, or form can trigger revalidation across product performance checks and quality documentation routines. Once a blend is embedded into a finished product program, switching suppliers becomes less frequent due to requalification cost and operational disruption risk.
Quick Stats for Customized Premix Market
- Customized premix market value in 2026: USD 10.40 billion
- Customized premix forecast value in 2036: USD 18.79 billion
- Forecast CAGR from 2026 to 2036: 6.1%
- Leading product type share: Premix Blends 71.7%
- Leading form share: Powder 67.7%
- Leading ingredient type share: Vitamin Premix 45.5%
- Leading function share: Bone Health 29.3%
- Leading application share: Pharma OTC Drugs 39.4%
- Fast-growth countries: China, India
- Key players: Archer-Daniels-Midland Company; Barentz International B.V.; BASF SE; Budenheim GmbH; Coalescence LLC; Farbest Brands; Fenchem Biotek Ltd.
Customized Premix Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 10.40 billion |
| Market Forecast Value (2036) | USD 18.79 billion |
| Forecast CAGR (2026 to 2036) | 6.1% |
Why is Customized Premix Demand Being Shaped by Formulation Control and Batch Repeatability?
Customized premix demand is expanding because buyers are increasing reliance on blends that reduce formulation variability across high-volume production. Product owners and quality teams are aligning premix selection with repeatable mixing outcomes, consistency across lot cycles, and documented compliance support that fits audit-ready workflows. This is especially relevant where finished products require stable nutrient targets and strict label discipline across long shelf-life programs.
Vitamin premix leadership at 45.5% reflects how micronutrient strategies remain central to many fortification and supplementation roadmaps. Demand also supports mineral premix, nucleotide premix, amino acids premix, enzymes, and other ingredient types where stakeholders manage differentiated outcomes across specific use cases. These demand themes often run parallel to broader investment in vitamin premix and mineral premixes, where quality discipline and repeat performance remain core purchase drivers.
Customers are also increasing preference for suppliers that can support technical conversations around blending tolerance, stability across storage conditions, and predictable lead times. This improves supplier retention once qualification is completed.
Segmental Analysis
Why do Premix Blends dominate the product type mix?
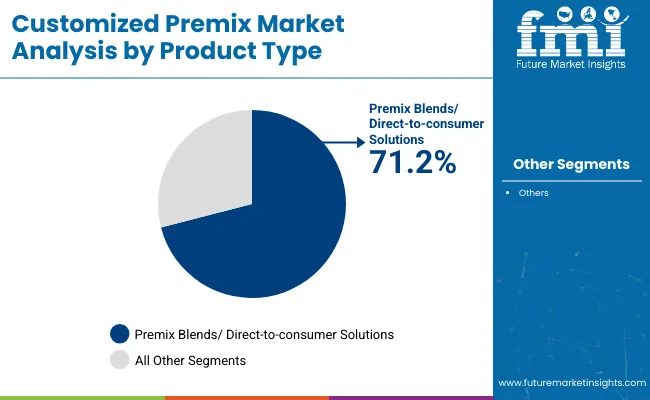
Premix blends account for a 71.7% share, reflecting demand for ready-to-use solutions that simplify formulation workflows. Buyers often select premix blends to reduce process steps, standardize nutrient delivery, and maintain consistency across multiple production locations. This structure supports repeat procurement once performance is validated in commercial runs.
Drum-to-hopper formulations remain relevant where customers prioritize direct handling compatibility within specific plant workflows. Adoption is strengthened when process routines are designed around controlled loading and efficient plant throughput, a pattern also visible across drum-to-hopper blends premix programs.
Why does Powder lead the form mix?
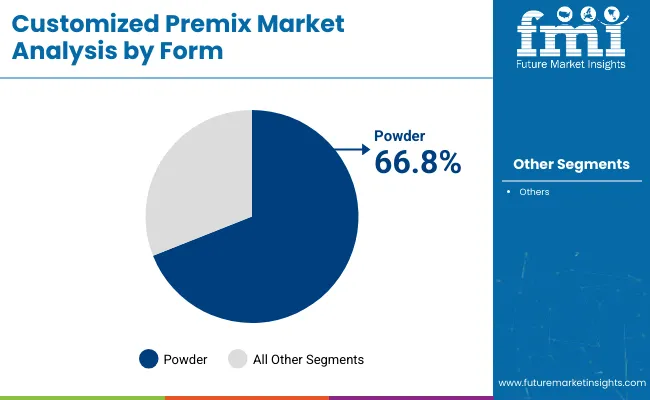
Powder holds a 67.7% share, supported by storage practicality and operational compatibility across high-volume blending environments. Powder formats also fit standardized dosing routines used by many food, supplement, and OTC product manufacturers, which makes them easier to deploy across recurring production schedules.
Liquid remains relevant where rapid incorporation and streamlined handling matter, particularly in programs designed around specific mixing systems and controlled viscosity requirements.
Why does Vitamin Premix remain the leading ingredient type?
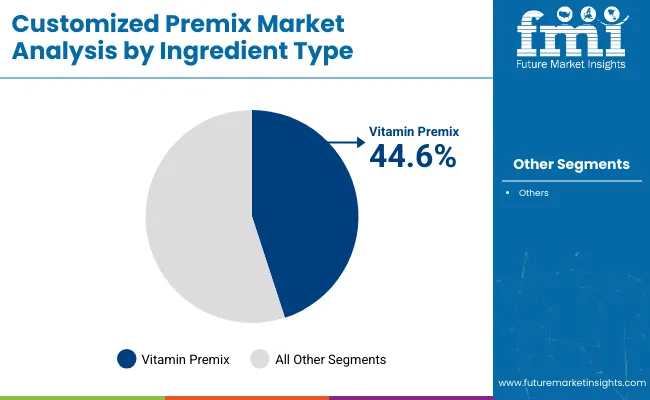
Vitamin premix leads with a 45.5% share, reflecting broad relevance across supplementation and fortified product pipelines. Buyers often treat vitamin blends as core inputs that require consistent output across lots, as variability can disrupt finished product compliance and internal quality checks.
Other ingredient types such as mineral premix, nucleotide premix, amino acids premix, enzymes, and other ingredients support diversification across multiple use pathways, which strengthens overall demand resilience.
Why does Bone Health lead by function?
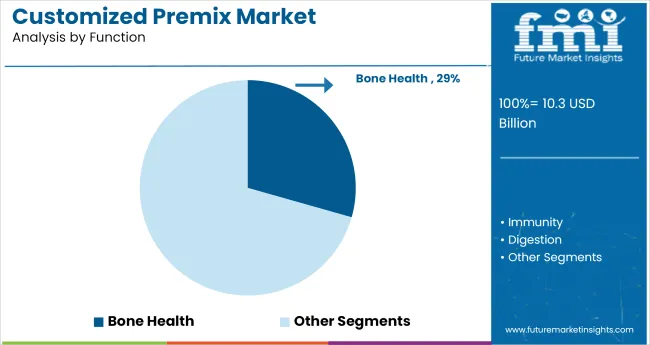
Bone health accounts for a 29.3% share, indicating sustained focus on function-led positioning in premix selection. Function mapping often determines which ingredient set is approved, how stability is validated, and what documentation is retained for internal control.
Immunity, digestion, energy, heart health, weight management, and other functional targets remain active, supporting broader portfolio segmentation and specialized blend development across customer pipelines.
Why do Pharma OTC Drugs lead application demand?
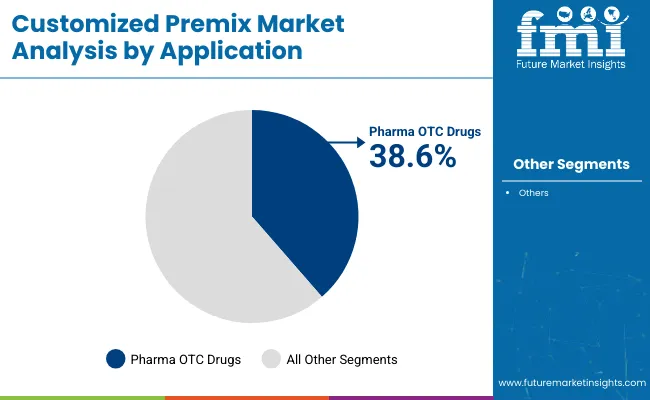
Pharma OTC drugs hold a 39.4% share, supported by structured procurement environments where documentation readiness and consistency influence supplier acceptance. This application also reinforces long-cycle supplier relationships because switching costs rise when requalification and stability documentation must be repeated.
Dietary supplements remain a major demand base, supported by repeat ordering cycles and portfolio expansion across multiple product lines. Food sector and pet food applications add diversification, improving demand stability across different purchasing rhythms.
What is sustaining growth across vitamin-led blend development?
Repeat formulation programs that prioritize consistent nutrient delivery and stable quality outcomes support growth. Vitamin premix leadership reflects how buyers treat micronutrient blends as long-cycle inputs, with supplier selection driven by repeatability, specification control, and predictable replenishment. These procurement patterns often align with standardized use of premixes across broader complete premixes and food premix portfolios.
How are function-led programs influencing portfolio design?
Function-led demand is shaping how premix portfolios are organized. Bone health leadership indicates how customers anchor product plans around defined outcomes and then select blends that fit target positioning and internal performance checks. This encourages suppliers to build modular capabilities that can support multiple functional directions without compromising stability and blend uniformity.
What limits speed of supplier switching in high-compliance applications?
Supplier switching is slowed by qualification steps tied to mixing uniformity, stability testing, and documentation review. In pharma OTC settings, acceptance routines are structured, which increases time-to-approval for new suppliers. These barriers slow early conversion yet support retention once suppliers pass validation checkpoints.
Analysis of the Customized Premix Market by Key Countries
Country-level momentum differs based on scale of manufacturing output, pace of product innovation, and how quickly demand programs expand across supplements, food sector applications, and OTC pipelines.
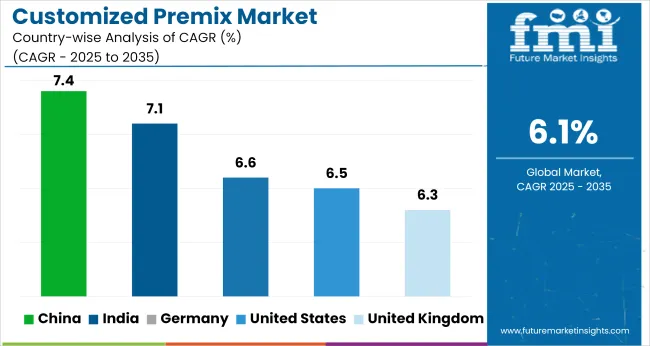
| Country | CAGR (2026-2036) |
|---|---|
| USA | 6.6% |
| UK | 6.4% |
| Germany | 6.7% |
| India | 7.2% |
| China | 7.5% |
Why does China lead growth within this country set?
China is projected to expand at a 7.5% CAGR, supported by accelerating adoption across food sector production and supplement pipelines where scale-up favors standardized premix inputs. Growth benefits from wider use of nutrient blends that can be deployed across large manufacturing networks with consistent outcomes.
Why is India sustaining strong expansion through diversified application pull?
India is expected to grow at a 7.2% CAGR, supported by broadening demand across dietary supplements and food sector programs where buyers are increasing focus on repeatable blend performance and stable supply continuity. Adoption also strengthens where suppliers support flexible blend designs aligned with evolving product portfolios.
Why does Germany reflect steady growth anchored in validation discipline?
Germany is forecast to advance at a 6.7% CAGR, shaped by procurement routines that emphasize documentation readiness and supplier verification. Growth tends to be supported by structured scale-up after performance and consistency benchmarks are confirmed.
Why does the USA remain a stable growth anchor?
The USA is projected to grow at a 6.6% CAGR, supported by recurring demand from pharma OTC drugs and dietary supplements where procurement systems favor validated suppliers. Demand benefits from long-cycle ordering patterns once blends are embedded into finished product programs.
Why does the UK show consistent growth through structured purchasing cycles?
The UK is expected to progress at a 6.4% CAGR, supported by steady demand programs that prioritize predictable quality outcomes and supplier reliability. Growth remains linked to controlled adoption across supplements and OTC-linked production, where qualification steps reinforce stable reordering.
Competitive Landscape
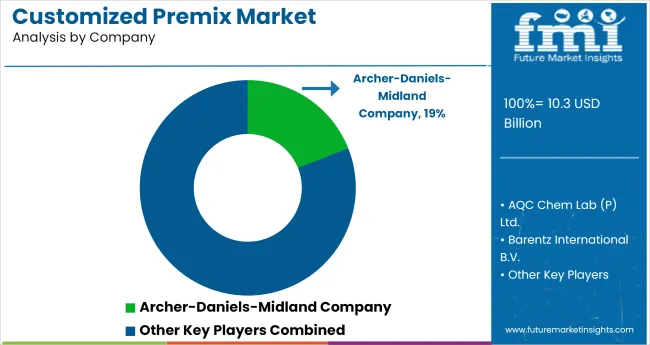
Competition is shaped by formulation capability, batch consistency, and the ability to deliver documentation that supports regulated and high-visibility applications. Suppliers differentiate through blending discipline, repeatable output across lots, and technical support that helps customers maintain stable outcomes across changing production schedules.
Portfolio depth remains an advantage. Vendors that can support vitamin premix leadership while maintaining flexibility for mineral, amino acids, nucleotide, enzyme, and specialty ingredient blends are better positioned to secure long-term supply relationships. Strong positioning is also supported by capability across both premix blends and drum-to-hopper pathways, which improves fit across different plant operating models.
Key Players
- Archer-Daniels-Midland Company
- Barentz International B.V.
- BASF SE
- Budenheim GmbH
- Coalescence LLC
- Farbest Brands
- Fenchem Biotek Ltd.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Billion |
| Product Type | Premix Blends; Drum-to-Hopper Formulations |
| Form | Powder; Liquid |
| Ingredient Type | Vitamin Premix; Mineral Premix; Nucleotide Premix; Amino Acids Premix; Enzymes; Other Ingredients |
| Function | Bone Health; Immunity; Digestion; Energy; Heart Health; Weight Management; Others |
| Application | Pharma OTC Drugs; Dietary Supplements; Food Sector; Pet Food |
| Key Countries | USA; UK; Germany; India; China |
| Key Companies Profiled | Archer-Daniels-Midland Company; Barentz International B.V.; BASF SE; Budenheim GmbH; Coalescence LLC; Farbest Brands; Fenchem Biotek Ltd. |
Customized Premix Market Segmentation
By Product Type
- Premix Blends
- Drum-to-Hopper Formulations
By Form
- Powder
- Liquid
By Ingredient Type
- Vitamin Premix
- Mineral Premix
- Nucleotide Premix
- Amino Acids Premix
- Enzymes
- Other Ingredients
By Function
- Bone Health
- Immunity
- Digestion
- Energy
- Heart Health
- Weight Management
- Others
By Application
- Pharma OTC Drugs
- Dietary Supplements
- Food Sector
- Pet Food
By Region
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East and Africa
Bibliography (APA Style)
- European Food Safety Authority. (2025). Overview on tolerable upper intake levels as derived by the Scientific Committee on Food and the EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA) (Version 11, August 2025). EFSA.
- European Food Safety Authority. (2024). Guidance on the assessment of the efficacy of feed additives (EFSA Journal, 22). EFSA.
- USA Food and Drug Administration. (2024, April 24). Current good manufacturing practices (CGMPs) for food and dietary supplements. FDA. USA Food and Drug Administration. (2025, November 21). Current good manufacturing practice (CGMP) regulations. FDA.
- National Institutes of Health, Office of Dietary Supplements. (2024, July 17). Multivitamin/mineral supplements: Fact sheet for health professionals. NIH.
- The Food Supplements and Food for Specific Groups (Vitamins and Minerals) (Amendment etc.) Regulations 2023. (2023). UK Statutory Instruments 2023 No. 28. UK Government.
Frequently Asked Questions
What is the projected customized premix market size for 2026?
The customized premix market is expected to total USD 10.40 billion in 2026.
What value is expected for customized premix in 2036?
In 2036, demand for customized premix is forecast to reach USD 18.79 billion.
At what rate will the customized premix market progress from 2026 to 2036?
Customized premix demand is expected to grow at a 6.1% CAGR during 2026 to 2036.
Which form leads demand and what share does it hold?
Powder leads by form with a 67.7% share.
Which ingredient type leads demand and what share does it hold?
Vitamin premix leads by ingredient type with a 45.5% share.
Which function leads demand and what share does it hold?
Bone health leads by function with a 29.3% share.
Which application leads demand and what share does it hold?
Pharma OTC drugs lead by application with a 39.4% share.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Premix Blends
- Drum-to-Hopper Formulations
- Premix Blends
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Form
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Form, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Form, 2026 to 2036
- Powder
- Liquid
- Powder
- Y to o to Y Growth Trend Analysis By Form, 2021 to 2025
- Absolute $ Opportunity Analysis By Form, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Ingredient Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Ingredient Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Ingredient Type, 2026 to 2036
- Vitamin Premix
- Mineral Premix
- Nucleotide Premix
- Amino Acids Premix
- Enzymes
- Other Ingredients
- Vitamin Premix
- Y to o to Y Growth Trend Analysis By Ingredient Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Ingredient Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Function
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Function, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Function, 2026 to 2036
- Bone Health
- Immunity
- Digestion
- Energy
- Heart Health
- Weight Management
- Others
- Bone Health
- Y to o to Y Growth Trend Analysis By Function, 2021 to 2025
- Absolute $ Opportunity Analysis By Function, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Pharma OTC Drugs
- Dietary Supplements
- Food Sector
- Pet Food
- Pharma OTC Drugs
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Form
- By Ingredient Type
- By Function
- By Application
- Competition Analysis
- Competition Deep Dive
- Archer-Daniels-Midland Company
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Barentz International B.V.
- BASF SE
- Budenheim GmbH
- Coalescence LLC
- Farbest Brands
- Fenchem Biotek Ltd.
- Archer-Daniels-Midland Company
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Form, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Ingredient Type, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Function, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Form
- Figure 9: Global Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Ingredient Type
- Figure 12: Global Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Function
- Figure 15: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Application
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Form
- Figure 35: North America Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Ingredient Type
- Figure 38: North America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Function
- Figure 41: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Application
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Form
- Figure 51: Latin America Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Ingredient Type
- Figure 54: Latin America Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Function
- Figure 57: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Application
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Form
- Figure 67: Western Europe Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Ingredient Type
- Figure 70: Western Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Function
- Figure 73: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Application
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Form
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Ingredient Type
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Function
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Application
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Form
- Figure 99: East Asia Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Ingredient Type
- Figure 102: East Asia Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Function
- Figure 105: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Application
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Form
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Ingredient Type
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Function
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Form, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Form, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Form
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Ingredient Type, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Ingredient Type, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Ingredient Type
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Function, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Function, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Function
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

