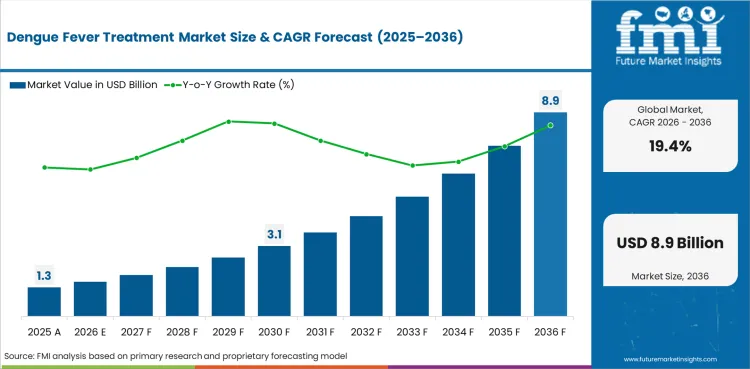
The dengue fever treatment market covers pharmaceutical and vaccine products used in the prevention and management of dengue virus infection, including vaccines, antiviral agents, supportive care therapeutics, and fluid management products distributed through hospital, public health, and pharmacy channels.
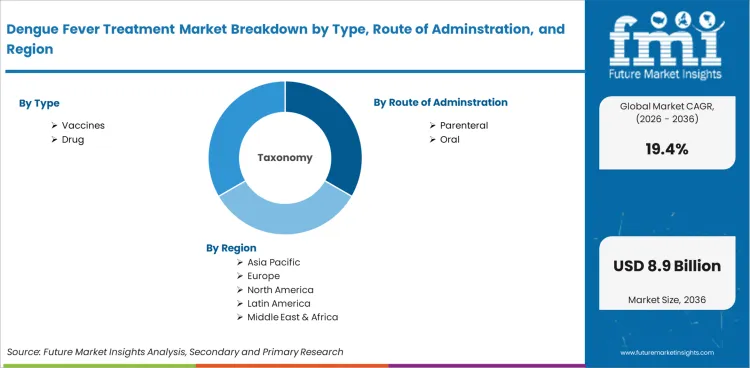
Market scope covers all commercially available dengue fever treatments categorized by type (vaccines, antiviral agents, supportive care, fluid management, others), route of administration (parenteral, oral, intravenous), end user (hospitals, public health centres, clinics, others), and distribution channel (hospital pharmacies, retail pharmacies, public health supply chains). Revenue coverage spans 2026 to 2036.
The scope does not include mosquito control products, diagnostic testing kits, or general antipyretic medications not specifically indicated for dengue management.
Demand for dengue fever treatment reflects the expanding global burden of dengue infection and the development of vaccine-based prevention that is transforming the market from purely supportive care toward integrated prevention and treatment. An estimated 100 to 400 million dengue infections occur annually, with approximately 500,000 cases progressing to severe dengue requiring hospitalization.
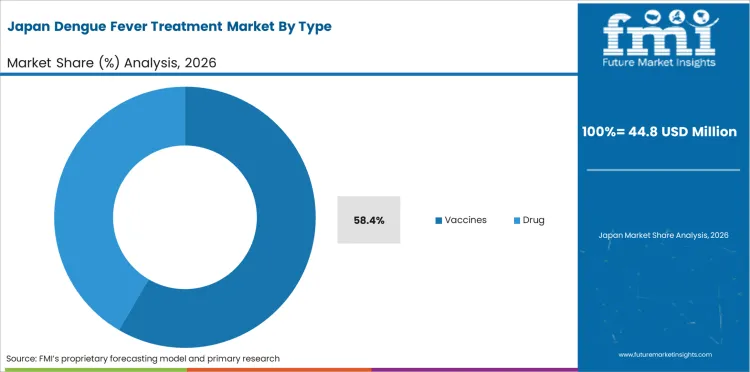
Adoption of vaccine products leads at 58.4% of type demand, driven by the approval and deployment of Dengvaxia (Sanofi) and Qdenga/TAK-003 (Takeda) in endemic countries. Vaccine procurement through government immunization programs represents the primary demand mechanism, with WHO prequalification supporting inclusion in national vaccination schedules.
Pricing reflects the contrast between cost-accessible generic supportive care medications and premium-priced vaccine products. Vaccine pricing is negotiated through government procurement mechanisms, with tiered pricing frameworks providing lower costs for endemic developing countries while maintaining commercial pricing in travel medicine and private healthcare markets.
The dengue fever treatment market is segmented by type, route of adminstration, end-user, and distribution channel.
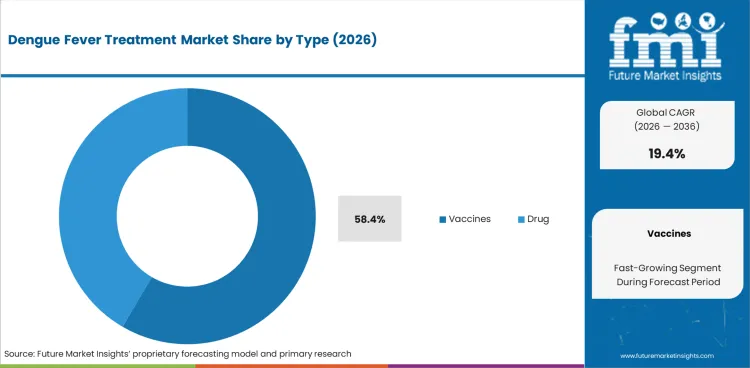
Vaccines lead at 58.4% driven by the growing regulatory approval and public health deployment of dengue vaccines. Dengvaxia and TAK-003 represent approved products with distinct target populations, creating complementary procurement demand across seropositive screening-based and broader population-based vaccination strategies.
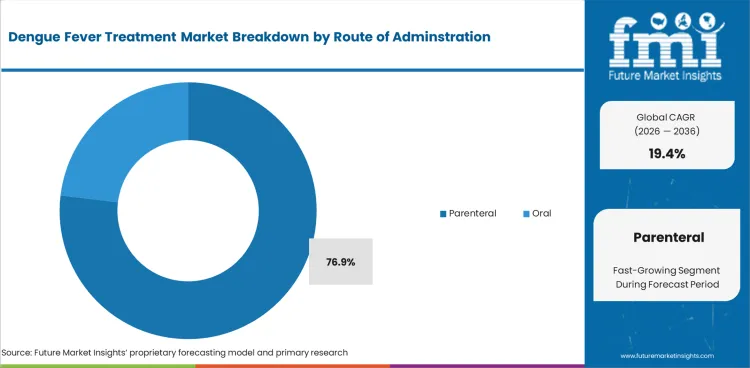
Parenteral administration represents 76.9% of route demand, reflecting the injectable nature of dengue vaccines and the intravenous fluid and medication requirements for hospitalized severe dengue patients.
The dengue fever treatment market is shaped by regulatory, technology, and procurement dynamics.
Demand is shaped by expanding regulatory approvals for dengue vaccines across endemic countries. WHO prequalification and national immunization program inclusion create structured government procurement demand.
Growth reflects increasing dengue infection rates driven by climate change expanding mosquito habitat, urbanization concentrating population density, and international travel spreading virus serotypes to new regions.
Adoption is supported by sustained government and international organization investment in dengue prevention and treatment programs across endemic regions in Southeast Asia, Latin America, and South Asia.
In the absence of dengue-specific antiviral therapy, supportive care including fluid management, antipyretics, and monitoring remains the treatment foundation for clinical dengue cases, sustaining pharmaceutical demand proportional to case volumes.
.webp)
| Country | CAGR |
|---|---|
| South Korea | 19.8% |
| USA | 19.7% |
| Japan | 19.6% |
| EU | 19.4% |
| UK | 18.9% |
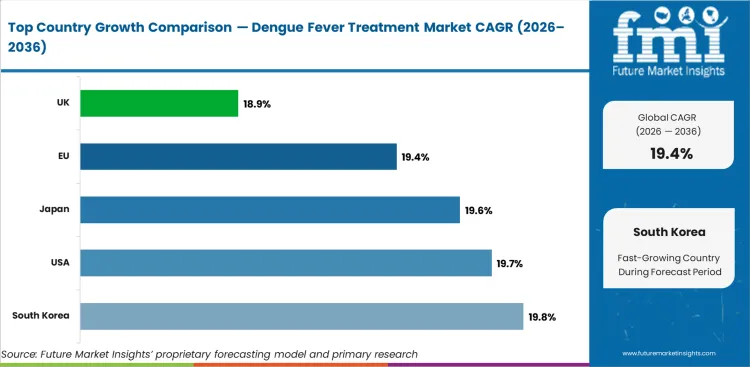
The global dengue fever treatment market is expected to grow at 19.4% per year from 2026 to 2036.
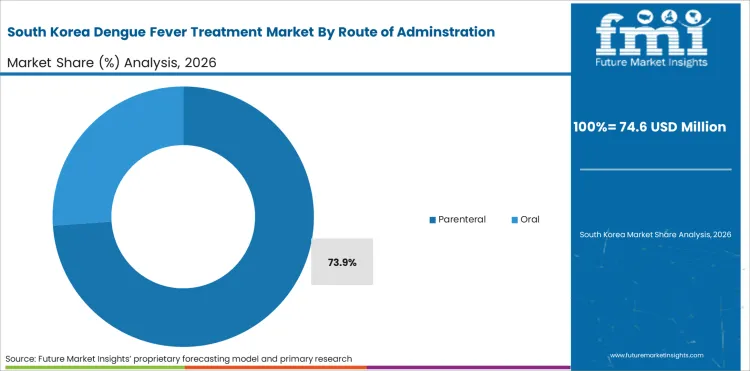
South Korea is expected to grow at 19.8% through 2036, driven by travel medicine vaccine demand, growing awareness of dengue risk for international travelers, and investment in infectious disease preparedness.
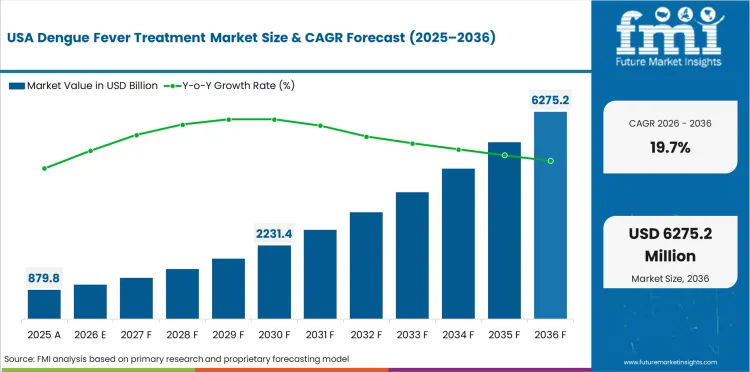
The USA is expected to grow at 19.7% through 2036, supported by travel medicine vaccine demand, FDA-approved dengue vaccines, and growing domestic dengue risk in southern states.
Japan is expected to grow at 19.3% through 2036, supported by travel vaccination programs, tropical disease research investment, and growing dengue awareness following domestic transmission events.
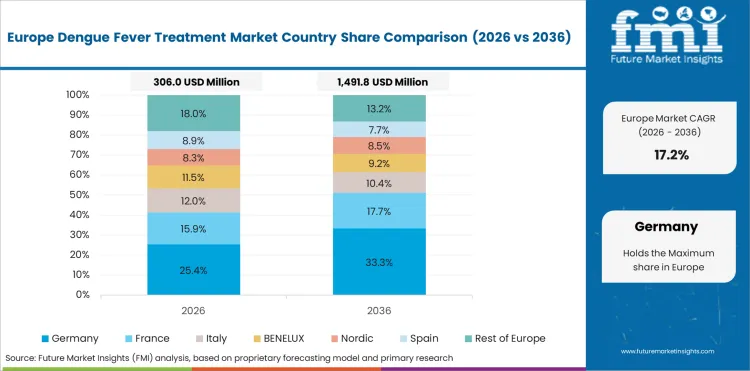
The EU is expected to grow at 19.5% through 2036, driven by travel medicine demand, EMA-approved vaccines, and public health coordination for imported dengue case management.
The UK is expected to grow at 19.1% through 2036, driven by travel health clinic vaccination demand, NHS tropical disease management, and public health preparedness for imported cases.
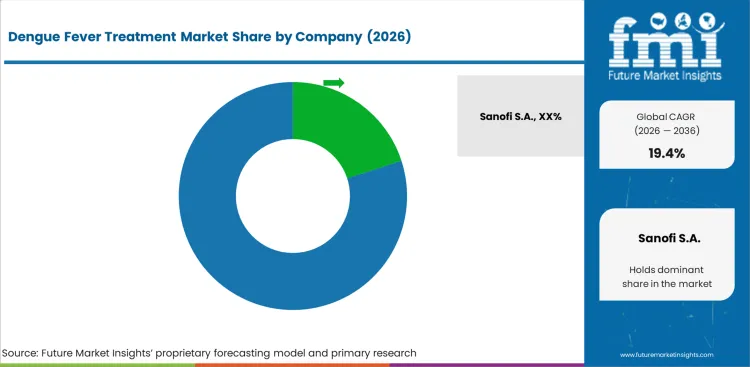
Sanofi S.A. leads through Dengvaxia, the first approved dengue vaccine, with established government procurement contracts in endemic countries and travel medicine distribution in non-endemic markets.
Takeda Pharmaceutical Company maintains a strong position through Qdenga (TAK-003), a dengue vaccine approved without prior serostatus screening requirement, expanding the eligible vaccination population.
Johnson & Johnson (Janssen) and GlaxoSmithKline contribute through dengue vaccine pipeline development and tropical disease research programs.
Entry barriers include the complex clinical trial requirements for dengue vaccine development, government procurement relationship establishment, and the WHO prequalification process required for inclusion in public health vaccination programs.
Key global companies leading the dengue fever treatment market include:
| Company | Vaccine Portfolio | Government Procurement | Manufacturing Scale | Geographic Reach |
|---|---|---|---|---|
| Sanofi S.A. | High | Strong | High | Global |
| Takeda Pharmaceutical | High | Strong | High | Global |
| Johnson & Johnson (Janssen) | Medium | Moderate | High | Global |
| GlaxoSmithKline plc | Medium | Moderate | High | Global |
| Merck & Co., Inc. | Low | Moderate | High | Global |
| Valneva | Low | Low | Medium | Europe |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Dengue Fever Treatment Market
Major Global Players
Emerging Players/Startups
| Parameter | Details |
|---|---|
| Quantitative Units | USD 1.51 billion to USD 8.89 billion, at a CAGR of 19.4% |
| Market Definition | The dengue fever treatment market covers pharmaceutical and vaccine products used in the prevention and management of dengue virus infection, including vaccines, antiviral agents, supportive care therapeutics, and fluid management products distributed through hospital, public health, and pharmacy channels. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, France, Germany, Italy, South Korea, Japan, China, India, 30 plus countries |
| Key Companies Profiled | Sanofi S.A., Takeda Pharmaceutical Company, Johnson & Johnson (Janssen Pharmaceuticals), GlaxoSmithKline plc (GSK), Merck & Co., Inc., Roche Diagnostics, Abbott Laboratories, Serum Institute of India, Eli Lilly and Company, BioNTech SE |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with publication dates, URLs, and supporting data for all cited works.
What is the global market demand for Dengue Fever Treatment in 2026?
In 2026, the global market is expected to be worth USD 1.51 billion.
How big will the Dengue Fever Treatment Market be in 2036?
By 2036, the market is expected to be worth USD 8.89 billion.
How much is demand expected to grow between 2026 and 2036?
Between 2026 and 2036, demand is expected to grow at a CAGR of 19.4%.
Which Type segment is expected to lead in 2026?
Vaccines is expected to account for 58.4% of the segment in 2026.
What is causing demand to rise in South Korea?
South Korea is expected to grow at 19.8% through 2036.
What is causing demand to rise in USA?
USA is expected to grow at 19.7% through 2036.
What does this report mean by Dengue Fever Treatment Market definition?
The dengue fever treatment market covers pharmaceutical and vaccine products used in the prevention and management of dengue virus infection, including vaccines, antiviral agents, supportive care ther.
How does FMI make the forecast?
Forecasting uses a hybrid bottom-up and top-down methodology starting with verified transaction data, projecting adoption velocity across segments and regions.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.