Endoscopic Probe Disinfection Market
A comprehensive overview of the endoscopic probe disinfection market is delivered in this report, featuring market size insights, revenue projections, competitive assessment, demand evaluation, growth propellers, restraining factors, Type trends, End Use trends, supply chain analysis, and strategic opportunities ahead.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Endoscopic Probe Disinfection Market Size, Market Forecast and Outlook By FMI
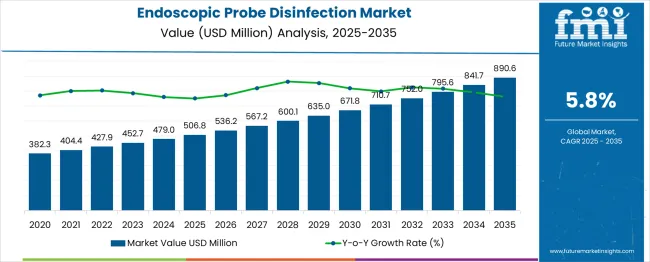
The endoscopic probe disinfection market was valued at USD 506.80 million in 2025, projected to reach USD 536.19 million in 2026, and is forecast to expand to USD 942.28 million by 2036 at a 5.8% CAGR. As per FMI, rising ultrasound-guided interventional procedure volumes, tightening high-level disinfection mandates for semi-critical probes, and growing awareness of infection transmission risk from inadequately reprocessed transesophageal and intracavitary probes are sustaining demand for automated probe disinfection systems and validated consumable chemistries. Infection control guidelines from the CDC, AIUM, and European Society of Radiology now classify all intracavitary ultrasound probes as semi-critical devices requiring high-level disinfection between patient uses.
Summary of Endoscopic Probe Disinfection Market
- Market snapshot
- The endoscopic probe disinfection market is valued at USD 506.80 million in 2025 and is projected to reach USD 942.28 million by 2036.
- The industry is expected to grow at a 5.8% CAGR from 2026 to 2036, creating an incremental opportunity of USD 406.09 million.
- The market is a regulated infection control category where probe classification as semi-critical devices, high-level disinfection mandates, and procedural throughput requirements define procurement patterns.
- Demand and growth drivers
- Demand is sustained by infection control guidelines classifying intracavitary ultrasound probes as semi-critical devices requiring high-level disinfection between uses.
- Automated UV-C and hydrogen peroxide vapour systems are gaining adoption, delivering validated disinfection within sub-ten-minute cycles that fit ASC scheduling constraints.
- Rising ultrasound-guided interventional procedure volumes across cardiology, obstetrics, and interventional radiology sustain consumable chemistry demand.
- Among key countries, China leads at 7.8% CAGR, followed by India at 7.3%, Germany at 6.7%, France at 6.1%, the UK at 5.5%, the USA at 4.9%, and Brazil at 4.4%.
- Product and segment view
- The endoscopic probe disinfection market covers disinfection instruments (automated UV-C, hydrogen peroxide vapour, and chemical immersion systems) and consumables (disinfectant chemistries, wipes, and sheaths) for ultrasound probe reprocessing.
- Probe types include convex, linear, and transesophageal ultrasound probes requiring different disinfection protocols based on patient contact classification.
- Endoscopic Probes (Ultrasound) leads by Type with 60.3% share in 2026 as probe-specific disinfection requirements drive the majority of equipment and consumable procurement.
- Ambulatory Surgical Centers leads by End Use with 44.2% share in 2026 as high-volume ultrasound-guided procedures in compressed schedules drive automated disinfection adoption.
- Scope includes probe disinfection instruments and consumables; excludes ultrasound probes themselves, general endoscope reprocessing systems, and surface disinfection products.
- Geography and competitive outlook
- China and India are the fastest-growing markets due to diagnostic imaging infrastructure expansion. The USA and Germany anchor mature, guideline-compliance-driven demand.
- Competition is shaped by validated disinfection cycle time, kill-rate documentation, and compatibility across ultrasound probe OEM platforms.
- Key players include STERIS plc, Olympus Corporation, GE Healthcare, Philips Healthcare, Chison Medical Imaging, CS Medical LLC, CIVCO Medical Solutions, Tristel Plc, Germitec, and Nanosonics.
- Analyst opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare observe that: 'Based on FMI's report, endoscopic probe disinfection is moving from manual wipe-based protocols toward automated systems that deliver documented, reproducible high-level disinfection with audit-trail evidence. Facilities relying on manual protocols face increasing accreditation risk as inspectors demand cycle-level disinfection documentation. Equipment suppliers without validated compatibility across GE, Philips, and Siemens probe platforms face limited formulary inclusion. The consumable replacement cycle creates a predictable recurring revenue stream that incentivises system placement at subsidised capital costs.'
- Strategic implications / Executive takeaways
- Imaging department directors must evaluate automated probe disinfection systems on validated cycle time and audit-trail documentation capabilities to meet accreditation inspection requirements.
- Infection control officers should standardise on high-level disinfection protocols for all intracavitary ultrasound probes, replacing manual wipe-only practices with documented automated systems.
- Procurement teams must verify probe disinfection system compatibility across the facility's installed ultrasound probe OEM platforms before capital equipment selection.
Endoscopic Probe Disinfection Market key takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 536.19 million |
| Industry Value (2036) | USD 942.28 million |
| CAGR (2026 to 2036) | 5.8% |
Source: Future Market Insights, 2026
FMI opines that the ambulatory surgical centre segment commands the largest end-use share because these facilities perform high volumes of ultrasound-guided interventional procedures in compressed turnaround schedules where manual disinfection protocols create throughput bottlenecks. Automated UV-C and hydrogen peroxide vapour disinfection systems are gaining adoption because they deliver validated kill rates within cycle times under ten minutes, enabling probe reuse between sequential patients without disrupting procedure scheduling. The consumables segment sustains recurring revenue through validated disinfectant wipes, sheaths, and chemistry refills that must be replaced per manufacturer-specified cycle counts.
China leads with a 7.8% CAGR through 2036 as ultrasound-guided interventional procedure volumes expand across hospital and imaging centre networks. India follows at 7.3%, driven by growing diagnostic imaging capacity and NABH infection control compliance. Germany registers a 6.7% pace as EU MDR probe reprocessing validation mandates tighten. France expands at 6.1% on the back of HAS accreditation ultrasound hygiene requirements. The United Kingdom maintains a 5.5% trajectory driven by NHS decontamination standards for intracavitary probes. The United States tracks at 4.9%, reflecting mature CDC guideline compliance with incremental growth from automated disinfection system adoption. Brazil records a 4.4% expansion driven by diagnostic imaging infrastructure growth.
Endoscopic Probe Disinfection Market definition
Endoscopic probe disinfection encompasses the instruments and consumable chemistries used to perform high-level disinfection of ultrasound probes (convex, linear, transesophageal) between patient uses. The category covers automated UV-C disinfection systems, hydrogen peroxide vapour units, chemical immersion devices, and validated disinfectant consumables serving hospitals, ASCs, diagnostic laboratories, and imaging centres.
Endoscopic Probe Disinfection Market inclusions
Market scope includes automated and manual probe disinfection instruments and consumable chemistries. The report covers global and regional market sizes for 2026 to 2036 with segment breakdowns by type and end user.
Endoscopic Probe Disinfection Market exclusions
The scope excludes ultrasound probes themselves, general endoscope reprocessing systems for flexible endoscopes, and surface disinfection products not validated for probe-level disinfection.
Endoscopic Probe Disinfection Market research methodology
- Primary research: Analysts engaged with imaging department managers, infection control officers, and ASC operations directors to map probe disinfection procurement triggers and system selection criteria.
- Desk research: Data collection aggregated CDC ultrasound probe disinfection guidelines, AIUM infection control recommendations, and EU MDR probe reprocessing validation requirements.
- Market sizing and forecasting: Baseline values derive from a bottom-up aggregation of probe disinfection equipment installations and consumable consumption per ultrasound procedure, applying procedure volume growth curves.
- Data validation and update cycle: Projections are cross-validated against published infection control equipment company revenue disclosures and imaging facility survey data.
Why is the Endoscopic Probe Disinfection Market Growing?
Market expansion is being supported by the increasing focuses on infection control and patient safety and the corresponding demand for effective disinfection solutions that can eliminate pathogens from endoscopic equipment and prevent healthcare-associated infections. Modern healthcare systems are increasingly focused on comprehensive infection prevention strategies that require validated and reliable disinfection procedures for all medical devices that come into contact with patients. Endoscopic probe disinfection's proven effectiveness in reducing infection risks while maintaining equipment functionality makes it essential technology for safe endoscopic procedures and patient care.
The growing adoption of minimally invasive procedures and endoscopic diagnostics is driving demand for endoscopic probe disinfection solutions that can support high-volume procedure environments while ensuring consistent disinfection quality. Healthcare provider preference for disinfection systems that combine efficacy with workflow efficiency is creating opportunities for automated and point-of-care disinfection technologies. The rising influence of regulatory requirements and accreditation standards is also contributing to increased adoption of validated disinfection protocols and equipment that can demonstrate compliance with infection control guidelines.
Segmental Analysis
The market is segmented by type, end use, and region. By type, the market is divided into endoscopic probes [ultrasound (EUS)] including convex probes, linear probes, and transesophageal (TEE) probes, as well as products including instruments and consumables. Based on end use, the market is categorized into ambulatory surgical centers (ASCs), hospitals, diagnostic laboratories and imaging centers, and others. Regionally, the market is divided into North America, Europe, East Asia, South Asia & Pacific, Latin America, and Middle East & Africa.
By Type, Endoscopic Probes [Ultrasound (EUS)] Segment Accounts for 60.3% Market Share
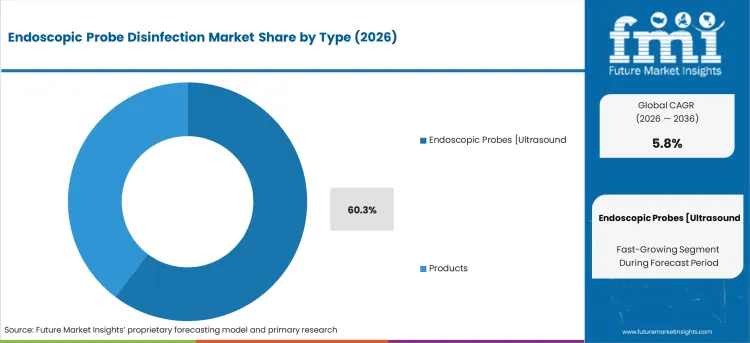
The endoscopic probes [ultrasound (EUS)] segment is projected to account for 60.3% of the endoscopic probe disinfection market in 2025, reaffirming its position as the dominant probe category. Healthcare facilities increasingly utilize ultrasound endoscopic probes for their versatility in diagnostic and therapeutic procedures across multiple medical specialties including gastroenterology, cardiology, and pulmonology. EUS probes' frequent patient contact and complex design features directly address critical disinfection requirements for ensuring patient safety and preventing cross-contamination between procedures.
This probe type forms the foundation of endoscopic disinfection protocols, as it represents the most commonly used and infection-prone equipment requiring specialized disinfection procedures and validation. Healthcare facility investments in probe-specific disinfection systems and ongoing compliance with infection control guidelines continue to strengthen adoption of EUS probe disinfection solutions. With healthcare providers prioritizing patient safety and regulatory compliance, EUS probe disinfection aligns with both clinical requirements and risk management objectives, making it the central component of endoscopic infection prevention strategies.
By End Use, Ambulatory Surgical Centers (ASCs) Segment Accounts for 44.2% Market Share
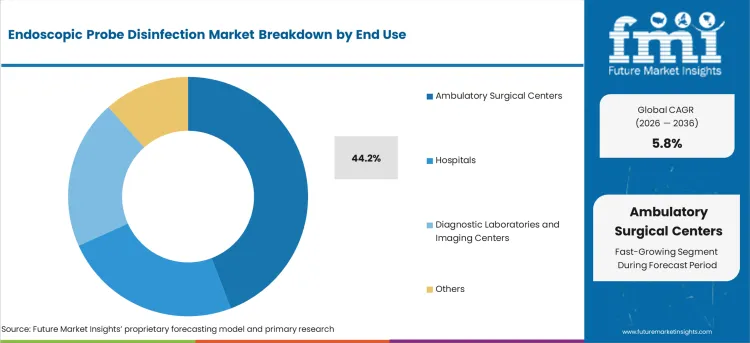
Ambulatory surgical centers are projected to represent 44.2% of endoscopic probe disinfection demand in 2025, underscoring their critical role as primary venues for minimally invasive procedures requiring endoscopic equipment. ASCs prefer comprehensive disinfection solutions for their ability to support high-volume procedure schedules while maintaining strict infection control standards and operational efficiency. Positioned as essential infrastructure for outpatient endoscopic procedures, ASCs require reliable and rapid disinfection capabilities that can support continuous workflow demands.
The segment is supported by increasing shift toward outpatient procedures and growing focuses on cost-effective healthcare delivery that requires efficient infection control processes. ASCs must maintain accreditation standards and regulatory compliance that mandate validated disinfection procedures for all patient-contact medical equipment. As healthcare continues to move toward ambulatory settings, ASCs will continue to dominate the market while supporting accessible and safe endoscopic care delivery.
What are the Drivers, Restraints, and Key Trends of the Endoscopic Probe Disinfection Market?
The endoscopic probe disinfection market is advancing steadily due to increasing healthcare-associated infection concerns and growing regulatory requirements for medical device reprocessing and infection control. The market faces challenges including high costs of automated disinfection systems, complexity of disinfection validation procedures, and workflow disruption during disinfection processes. Innovation in rapid disinfection technologies and point-of-care solutions continue to influence product development and market expansion patterns.
Expansion of Automated and Point-of-Care Disinfection Systems
The growing adoption of automated disinfection systems is enabling healthcare facilities to standardize disinfection procedures while reducing human error and improving workflow efficiency. Automated systems provide consistent disinfection cycles with documentation and validation capabilities that support regulatory compliance and quality assurance. Healthcare providers are increasingly recognizing the operational and safety advantages of automated disinfection for high-volume endoscopic procedures.
Integration of Smart Monitoring and Validation Technologies
Modern disinfection system manufacturers are incorporating smart monitoring capabilities, data logging, and validation systems that provide real-time verification of disinfection efficacy and compliance documentation. These technologies improve safety assurance while providing automated record-keeping that supports accreditation and regulatory requirements. Advanced monitoring also enables predictive maintenance and quality control that enhances system reliability and performance consistency.
Analysis of Endoscopic Probe Disinfection Market by Key Country
.webp)
| Country | CAGR (2026-2036) |
| China | 7.8% |
| India | 7.3% |
| Germany | 6.7% |
| France | 6.1% |
| UK | 5.5% |
| USA | 4.9% |
| Brazil | 4.4% |
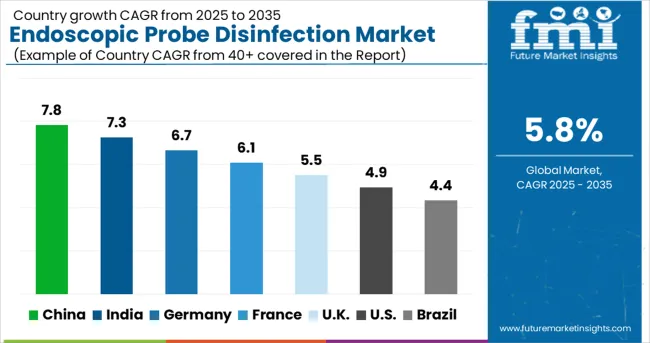
The global market is expected to grow at a CAGR of 5.8% between 2026 and 2036, driven by rising demand for infection control and stringent regulatory standards in healthcare facilities. China leads with 7.8% growth, supported by increasing adoption of advanced disinfection technologies and expanding hospital infrastructure. India follows at 7.3%, reflecting growing awareness of endoscope hygiene and rising healthcare investments. Germany records 6.7%, driven by strict compliance with sterilization protocols and advanced clinical practices. France is projected at 6.1%, supported by adoption of automated disinfection systems and infection prevention initiatives. The UK grows at 5.5% with enhanced focus on patient safety, while the USA expands at 4.9%, influenced by regulatory mandates and increasing use of high-level disinfection solutions. Brazil shows 4.4%, reflecting gradual market penetration and growing healthcare standards.
The report covers an in-depth analysis of 40+ countries, top-performing countries are highlighted below.
Market Potential of Endoscopic Probe Disinfection in China
China is expected to achieve a CAGR of 7.8% from 2026 to 2036, driven by rapid growth of hospitals and specialized diagnostic centers. Increased awareness of hospital-acquired infections and government infection control policies have accelerated adoption of automated probe disinfection systems. Local manufacturers have expanded production capacity, while multinational firms have partnered with distributors and hospital networks to enhance market penetration. The growing number of endoscopic procedures, particularly in gastroenterology and bronchoscopy, has sustained demand. Technological innovation, including automated and compact disinfection systems, has been a key driver. China’s market is poised for strong growth as healthcare infrastructure modernization and regulatory compliance initiatives continue across urban centers.
In-depth Analysis of Endoscopic Probe Disinfection Market in India
India is forecasted to grow at a CAGR of 7.3% from 2026 to 2036, driven by rising endoscopic procedure volumes and enhanced infection control awareness. Hospitals and diagnostic centers have increasingly adopted automated high-level disinfection systems to comply with clinical hygiene standards. Public health initiatives promoting patient safety and infection prevention have encouraged adoption, while private hospitals have implemented advanced systems to reduce liability and improve outcomes. Device manufacturers have conducted training programs for staff to ensure correct usage and compliance. With the growing number of gastroenterology and bronchoscopy procedures in metropolitan centers, India’s market is expected to expand steadily over the forecast period.
Growth Outlook on Endoscopic Probe Disinfection Market in Germany
Germany is projected to grow at a CAGR of 6.7% between 2026 and 2036, supported by strict infection control regulations and high hospital standards. Endoscopic probe disinfection systems have been widely deployed in gastroenterology, pulmonology, and urology units to reduce hospital-acquired infections. Hospitals have adopted automated high-level disinfection devices with integrated monitoring for regulatory compliance. Device manufacturers, including Getinge and Olympus, have invested in training programs and service contracts to enhance adoption. Germany’s aging population and high procedural volumes for endoscopic examinations have contributed to market stability, while continuous technological upgrades in disinfection equipment have improved operational efficiency and safety standards.
Demand Outlook on Endoscopic Probe Disinfection Market in France
France is expected to grow at a CAGR of 6.1% between 2026 and 2036, driven by hospital initiatives to prevent hospital-acquired infections and ensure patient safety. Endoscopic probe disinfection systems have been widely adopted in gastroenterology, pulmonology, and ENT units. Integration of automated high-level disinfection with monitoring systems has been emphasized to meet regulatory and accreditation standards. Device providers have focused on expanding product offerings and conducting staff training programs. Government infection control policies and public awareness campaigns have facilitated adoption, particularly in urban hospitals and private clinics. Ongoing technological advancements and rising endoscopic procedure volumes are expected to sustain market growth throughout the forecast period.
Sales Analysis of Endoscopic Probe Disinfection Market in the United Kingdom
The United Kingdom is projected to grow at a CAGR of 5.5% between 2026 and 2036, with market growth driven by hospital infection control initiatives and rising endoscopic procedures. NHS hospitals and private clinics have deployed automated high-level disinfection systems for gastrointestinal and pulmonary endoscopies. Compliance with UK Department of Health standards and regulatory guidelines has shaped adoption strategies. Major manufacturers have provided training and service support to ensure correct system usage and adherence to protocols. While market growth is moderate due to centralized procurement and budget constraints, increasing endoscopic procedural volumes and focus on patient safety are expected to sustain adoption over the forecast period.
Growth Assessment of Endoscopic Probe Disinfection Market in the United States
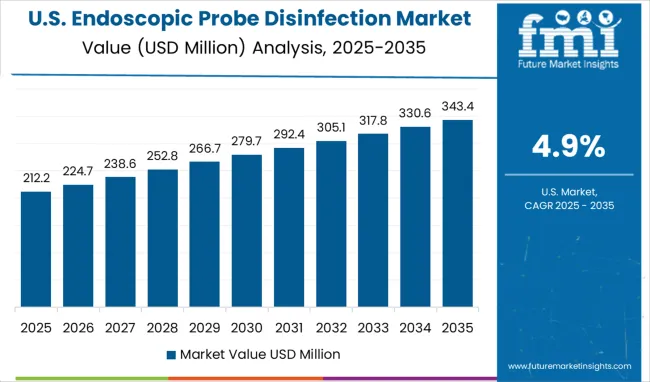
The market in the United States is projected to grow at a CAGR of 4.9% between 2026 and 2036, driven by increasing hospital-acquired infection awareness and rising endoscopic procedures. Hospitals and outpatient centers have been adopting automated high-level disinfection systems to ensure compliance with infection control standards. Regulatory oversight by the FDA and CDC guidelines has influenced both product design and adoption rates. Major players such as Cantel Medical and Getinge have expanded their market presence through product innovation and training programs for clinical staff. Increasing procedural volumes in gastroenterology, pulmonology, and urology have created steady demand for probe disinfection solutions. The market growth remains moderate due to high equipment costs and the need for staff training.
Opportunity Analysis of Endoscopic Probe Disinfection Market in Brazil
Brazil is expected to grow at a CAGR of 4.4% between 2026 and 2036, with adoption concentrated in urban tertiary hospitals and private clinics. Hospitals have increasingly implemented automated probe disinfection systems to reduce hospital-acquired infections. Market growth is influenced by public awareness campaigns and partnerships with multinational device providers offering training and service support. Adoption is moderated by limited reimbursement frameworks and budget constraints in public hospitals. Private healthcare providers have been more proactive in implementing automated systems, particularly in gastroenterology and pulmonology units. Technological upgrades and increasing procedural volumes are expected to gradually support market expansion in major cities.
Europe Market Split by Country
The market in Europe demonstrates mature development across major economies with Germany showing strong presence through its advanced healthcare system and focuses on infection control excellence, supported by healthcare facilities leveraging clinical expertise to implement comprehensive endoscopic disinfection protocols that emphasize patient safety, regulatory compliance, and workflow efficiency across hospital and outpatient settings.
France represents a significant market driven by its healthcare system excellence and sophisticated understanding of infection prevention and medical device safety, with healthcare providers focusing on comprehensive disinfection solutions that combine French medical expertise with advanced disinfection technologies for enhanced patient safety and clinical excellence in endoscopic procedures.
The UK exhibits considerable growth through its focuses on National Health Service infection control standards and patient safety excellence, with strong adoption of endoscopic probe disinfection systems across NHS hospitals, private healthcare facilities, and specialized endoscopy centers. Germany and France show expanding interest in automated disinfection applications, particularly in high-volume endoscopy centers and ambulatory surgical facilities. BENELUX countries contribute through their focus on healthcare innovation and advanced medical technology adoption, while Eastern Europe and Nordic regions display growing potential driven by increasing healthcare investment and expanding endoscopic procedure capabilities.
Competitive Landscape of Endoscopic Probe Disinfection Market
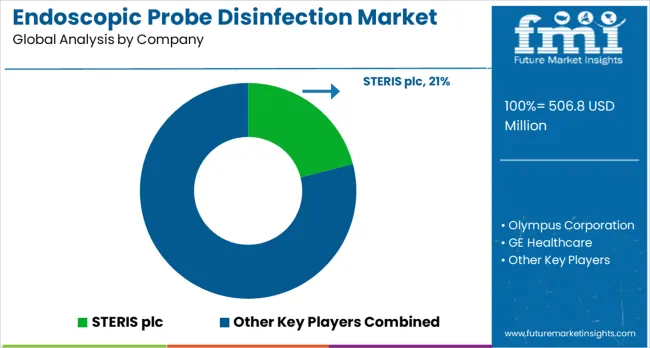
The market has been shaped by global medical technology leaders and specialized hygiene solution providers, competing on effectiveness, speed, and safety. STERIS plc, Olympus Corporation, GE Healthcare, and Philips Healthcare have focused on automated disinfection systems that deliver consistent results while minimizing manual handling. Strategies include integrated cleaning protocols, rapid cycle times, and compatibility with a wide range of endoscopic equipment. Chison Medical Imaging and CS Medical LLC have targeted smaller clinical setups with compact, easy-to-use disinfection solutions.
Regional and niche players such as CIVCO Medical Solutions, Tristel Plc, Germitec, and Nanosonics have differentiated through advanced chemical formulations, portable systems, and user-friendly devices. Product brochures highlight high-level disinfection, device compatibility, and reduced turnaround time for clinical procedures. Systems are designed to improve workflow efficiency, reduce contamination risks, and ensure compliance with infection control standards. STERIS and Olympus promote large-scale automated units, while Tristel and Nanosonics focus on compact, environmentally friendly technologies for smaller clinics and endoscopy suites.
Competition in the market is defined by disinfection efficacy, operational efficiency, and device compatibility. Philips and GE Healthcare provide integrated solutions that connect to hospital management systems, while CIVCO Medical Solutions and Germitec highlight customizable and portable devices for rapid deployment. Brochures communicate safety performance, validated cleaning protocols, and workflow benefits, positioning each company as a reliable partner for infection prevention in endoscopic procedures.
Key Players in the Endoscopic Probe Disinfection Market
- STERIS plc
- Olympus Corporation
- GE Healthcare
- Philips Healthcare
- Chison Medical Imaging
- CS Medical LLC
- CIVCO Medical Solutions
- Tristel Plc
- Germitec
- Nanosonics
Scope of the report
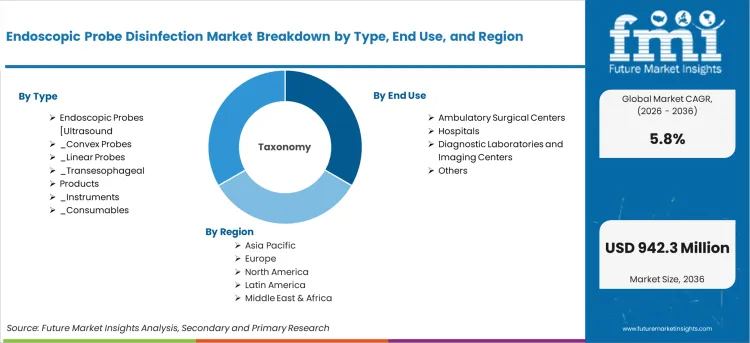
| Metric | Value |
|---|---|
| Quantitative Units | USD 536.19 million to USD 942.28 million, at a CAGR of 5.8% |
| Market Definition | Endoscopic probe disinfection covers instruments and consumables for high-level disinfection of ultrasound probes between patient uses in clinical settings. |
| Type Segmentation | Endoscopic Probes (Ultrasound) (Convex, Linear, Transesophageal), Products (Instruments, Consumables) |
| End Use Segmentation | Ambulatory Surgical Centers, Hospitals, Diagnostic Laboratories and Imaging Centers, Others |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, India, Germany, France, UK, USA, Brazil, and 40 plus countries |
| Key Companies Profiled | STERIS plc, Olympus Corporation, GE Healthcare, Philips Healthcare, Chison Medical Imaging, CS Medical LLC, CIVCO Medical Solutions, Tristel Plc, Germitec, Nanosonics |
| Forecast Period | 2026 to 2036 |
| Approach | Bottom-up methodology from probe disinfection installations and consumable consumption per procedure. |
Endoscopic Probe Disinfection Market by Segments
Type:
- Endoscopic Probes [Ultrasound (EUS)]
- Convex Probes
- Linear Probes
- Transesophageal (TEE) Probes
- Products
- Instruments
- Consumables
End Use:
- Ambulatory Surgical Centers (ASCs)
- Hospitals
- Diagnostic Laboratories and Imaging Centers
- Others
Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia & Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia & Pacific
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- 1. Centers for Disease Control and Prevention. (2024). Guideline for disinfection and sterilization in healthcare facilities: Ultrasound probe update. CDC.
- 2. American Institute of Ultrasound in Medicine. (2024). AIUM guidelines for cleaning and preparing external and internal ultrasound probes. AIUM.
- 3. European Commission. (2024). EU MDR 2017/745: Reprocessing requirements for semi-critical ultrasound devices. European Commission.
- 4. World Health Organization. (2024). Decontamination and reprocessing of medical devices. WHO.
- 5. Australasian College for Infection Prevention and Control. (2024). Ultrasound transducer reprocessing guidelines. ACIPC.
- 6. Organisation for Economic Co-operation and Development. (2024). Health at a Glance 2024: Diagnostic imaging procedure volumes. OECD.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for endoscopic probe disinfection in 2026?
Demand for endoscopic probe disinfection in the global market is estimated to be valued at USD 536.19 million in 2026.
What will be the market size by 2036?
Market size for endoscopic probe disinfection is projected to reach USD 942.28 million by 2036.
What is the expected demand growth between 2026 and 2036?
Demand is expected to grow at a CAGR of 5.8% between 2026 and 2036.
Which Type leads in 2026?
Endoscopic Probes (Ultrasound) accounts for 60.3% in 2026 as probe-specific disinfection requirements drive the majority of procurement.
Which End Use leads in 2026?
Ambulatory Surgical Centers accounts for 44.2% in 2026 as high-volume procedures in compressed schedules drive automated disinfection adoption.
What is driving demand in China?
Expanding ultrasound-guided interventional procedure volumes across hospital and imaging centre networks sustain disinfection equipment and consumable demand.
What is included in the scope?
The scope covers probe disinfection instruments and consumables; excludes probes themselves, general endoscope reprocessing, and surface disinfection products.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2026 to 2036
- Endoscopic Probes [Ultrasound
- Convex Probes
- Linear Probes
- Transesophageal
- Products
- Instruments
- Consumables
- Endoscopic Probes [Ultrasound
- Y to o to Y Growth Trend Analysis By Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Ambulatory Surgical Centers
- Hospitals
- Diagnostic Laboratories and Imaging Centers
- Others
- Ambulatory Surgical Centers
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Type
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By End Use
- Competition Analysis
- Competition Deep Dive
- STERIS plc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Olympus Corporation
- GE Healthcare
- Philips Healthcare
- Chison Medical Imaging
- CS Medical LLC
- CIVCO Medical Solutions
- Tristel Plc
- Germitec
- Nanosonics
- STERIS plc
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Type
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

