FHIR Prior Authorization Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
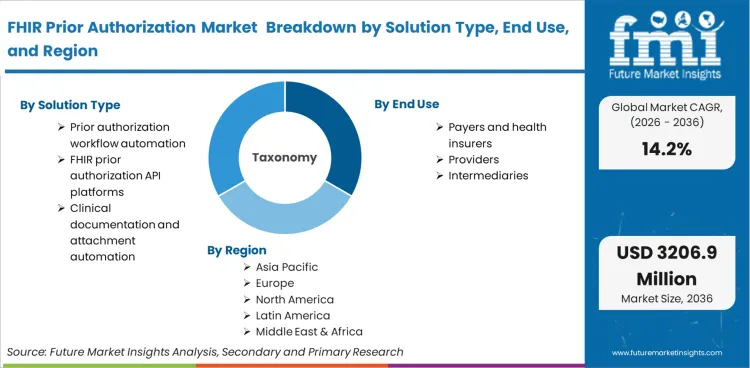
The FHIR Prior Authorization Market is segmented by Solution Type (FHIR prior authorization API platforms, Prior authorization workflow automation, Clinical documentation and attachment automation), End User (Payers and health insurers, Providers, Intermediaries), Deployment Model (Cloud and SaaS, On-premise, Hybrid) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
FHIR Prior Authorization Market Forecast and Outlook By FMI
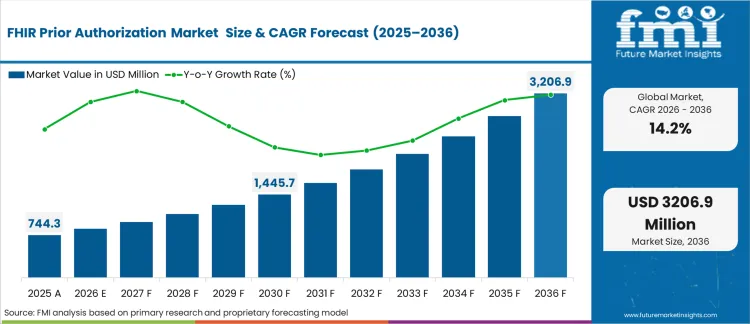
In 2025, the FHIR prior authorization market was valued at USD 745 million. Based on Future Market Insights' analysis, demand for FHIR prior authorization solutions is estimated to grow to USD 850 million in 2026 and USD 3,200 million by 2036. FMI projects a CAGR of 14.2% during the forecast period.
Absolute dollar growth of USD 2,350 million over 2026 to 2036 reflects a shift from fax-led utilization management toward API-mediated exchange of authorization status, clinical attachments, and decision responses. Growth is steady because payers and large provider groups keep funding administrative automation, yet timelines stretch where legacy claims stacks, prior authorization portals, and EHR workflows still need integration and governance.
Industry experts note that administrative burdens in prior authorization processes can be reduced through automation, allowing clinicians to focus more on patient care.
Summary of FHIR Prior Authorization Market
- Market Definition:
- FHIR prior authorization solutions enable standards-based submission, attachment exchange, status tracking, and determinations between payers, providers, and intermediaries using HL7 FHIR APIs.
- Demand Drivers in the Market:
- CMS interoperability and prior authorization requirements push impacted payers to implement FHIR-based API capabilities and response workflows.
- Payers and large provider groups fund automation to cut turnaround time and reduce manual follow-up tied to missing documentation and status uncertainty.
- National digital health programs lower integration friction by promoting standardized exchange rails for claims and clinical data.
- Key Segments Analyzed in the FMI Report:
- Solution type: Prior authorization workflow automation leads with 46% share in 2026 because it governs routing, exceptions, and operational throughput.
- End user: Payers and health insurers lead with 52% share in 2026 because decision systems, policy logic, and compliance accountability sit with plans.
- Deployment model: Cloud and SaaS leads with 62% share in 2026 as API traffic, audit logs, and partner onboarding require scalable operations.
- Analyst Opinion at FMI:
- Analysts emphasize that executives should align standards with workflow ROI and prioritize automation to reduce rework.
- Strategic Implications/Executive Takeaways:
- Set a single attachment policy and exception path across lines of business so automation does not move work into manual queues.
- Fund provider onboarding and testing capacity early, since partner readiness often sets the go-live ceiling.
- Treat auditability, consent, and access controls as product requirements because authorization artifacts become regulated exchange records.
- Methodology:
- Forecasts blend policy dates, standards maturity, and payer-provider implementation patterns to size adoption curves.
- Country rates reflect documented program milestones and local payer structures rather than generic digitization assumptions.
- Validation checks use public specifications, government releases, and documented deployments before finalizing outputs.
India leads the growth set at 17.0% CAGR as national claims and health data rails mature under ABDM-linked gateways. The United States follows at 15.5% CAGR on compliance-driven API buildouts tied to payer workflows. The United Kingdom posts 14.8% CAGR as NHS referral digitization expands via FHIR APIs. Germany grows at 14.0% CAGR as telematics and ePA-linked specifications normalize FHIR-based exchange. Canada rises at 13.6% CAGR where provincial eReferral and pan-Canadian FHIR implementation workstreams reduce integration friction.
| Metric | Value |
|---|---|
| Market Size (2026) | USD 850.0 Million |
| Forecast Value (2036) | USD 3,200.0 Million |
| CAGR (2026-2036) | 14.2% |
FHIR Prior Authorization Market Definition
FHIR prior authorization solutions are software and services that exchange authorization requests, supporting clinical details, and payer decisions using HL7 FHIR-based APIs. Buyers use them to reduce manual phone and fax steps, standardize attachments, and track status updates inside payer and provider workflows. The market covers API platforms, workflow engines, and automation that connect health plans, providers, and intermediaries to submit requests, receive determinations, and reconcile what was authorized with what was delivered.
FHIR Prior Authorization Market Inclusions
This report covers global and regional market sizing for 2025, 2026, and 2036 with a 2026 to 2036 forecast. It provides segmentation by solution type, end user, and deployment model, along with regional assessment and country-level growth rates. Analysis includes policy and standards alignment for FHIR-based prior authorization exchange, implementation considerations across payer-provider connectivity, and adoption patterns across mature and emerging health IT environments.
FHIR Prior Authorization Market Exclusions
The scope excludes non-FHIR prior authorization tools that rely only on proprietary portals, standalone fax management, or manual utilization management services without API exchange. It also excludes claims adjudication platforms, core EHR licensing, and broader revenue cycle management suites unless the offering directly supports FHIR prior authorization workflows or clinical attachment automation as a primary function.
FHIR Prior Authorization Market Research Methodology
- Primary Research: FMI conducted interviews with payer IT leaders, provider revenue cycle directors, health information network operators, and implementation partners involved in electronic prior authorization rollouts tied to API exchange.
- Desk Research: Secondary inputs included CMS rulemaking materials, HL7 Da Vinci implementation guidance, NHS developer specifications, government digital health releases, and public technical documentation from standards communities and public agencies.
- Market-Sizing and Forecasting: A hybrid approach combined bottom-up provider and payer adoption curves with top-down administrative spend and regulatory compliance timelines to model addressable workflow volumes.
- Data Validation and Update Cycle: Forecast outputs were cross-checked against policy implementation dates, published specification updates, and documented deployment milestones, then refreshed on a rolling cycle when major standards or policy changes occur.
Segmental Analysis
FHIR Prior Authorization Market Analysis by Solution Type

Based on FMI's FHIR prior authorization market report, prior authorization workflow automation is estimated to hold 46% share in 2026. Workflow automation dominates because it is the layer that converts standards into operational throughput, routing requests, tracking statuses, and managing exception handling across payers and providers.
- Compliance Integration: CMS released the Interoperability and Prior Authorization Final Rule in January 2024, establishing requirements that push payers toward API-based prior authorization exchange, which increases demand for workflow orchestration around those APIs [2].
- Operational proof points include case studies that demonstrate time savings when clinical intelligence is integrated into authorization workflows, supporting investment cases.
- Standards Alignment: The HL7 Da Vinci Prior Authorization Support (PAS) Implementation Guide provides a common FHIR exchange pattern for request and response flows, lowering custom interface burden for platform vendors [3].
FHIR Prior Authorization Market Analysis by End User
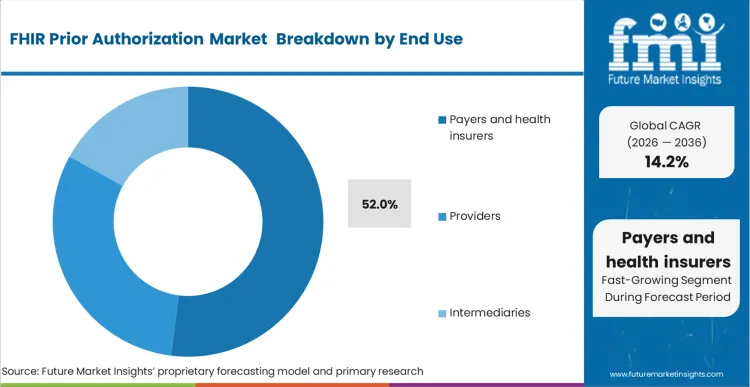
Based on FMI's FHIR prior authorization market report, adoption by payers and health insurers is estimated to hold 52% share in 2026. Payers lead because they control authorization policy, decision rules, and the core systems that issue determinations, so most platform spend and compliance accountability sits inside health plans and pharmacy benefit managers.
- Policy-Driven Spend: CMS requirements detailed in the January 2024 final rule extend patient access to authorization information and set API expectations, which pushes payer budgets toward standards-based interfaces and monitoring [2].
- Network Leverage: Health information networks and intermediaries that connect many EHRs and plans can scale onboarding, which strengthens payer preference for connected hubs rather than one-off interfaces.
- Provider Pull-Through: Where referral and consult workflows shift to structured exchange, providers demand tighter integration of authorization status into scheduling and care coordination queues.
FHIR Prior Authorization Market Analysis by Deployment Model
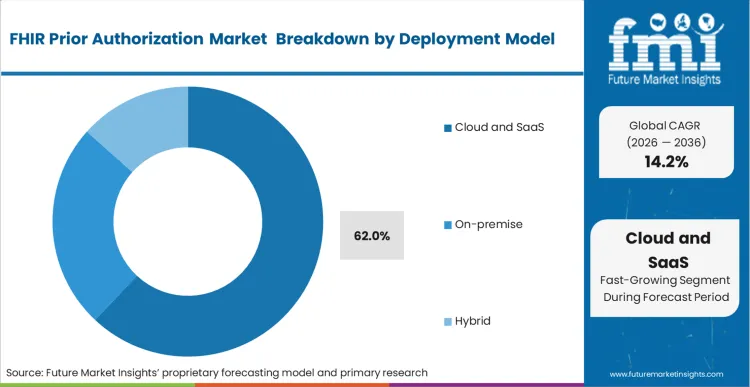
Based on FMI's FHIR prior authorization market report, Cloud and SaaS deployment is estimated to hold 62% share in 2026. Cloud-led delivery dominates because transaction volumes fluctuate, API uptime must be measured continuously, and cross-entity connectivity requires scalable security, logging, and routing services.
- API Operations: Standards-based exchange introduces traffic management, authentication, and audit requirements that are easier to operate with cloud-native monitoring and scaling patterns supported by FHIR implementation guides [3].
- Jurisdiction Rollouts: Public sector eReferral and interoperability programs publish evolving implementation guidance, which suits SaaS update cycles over on-premise release cadence.
- Cross-Network Onboarding: Cloud deployment supports faster partner testing cycles across multiple payers and provider groups, reducing time-to-connect versus local installs.
FHIR Prior Authorization Market Drivers, Restraints, and Opportunities
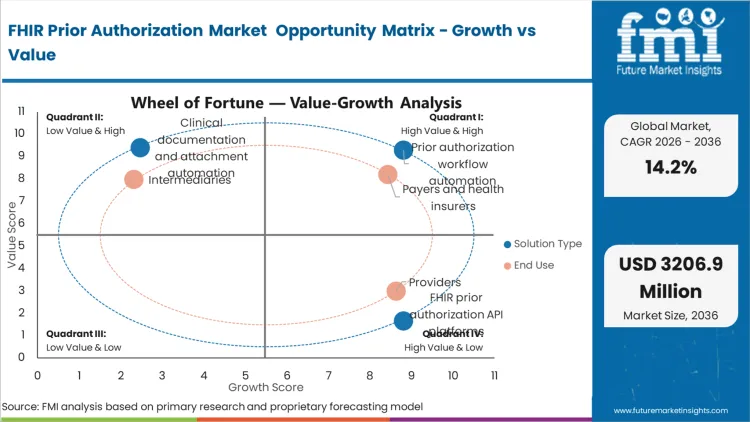
Future Market Insights analysis finds that prior authorization remains a high-friction administrative process because policy rules, clinical documentation, and payer decisioning sit in different systems. The market exists at its current size because payers and providers have long paid for portals, call centers, and manual staff time to move authorizations through. FHIR-based exchange reframes that spend into software and connectivity budgets that can be measured by turnaround time, denial reasons, and staff hours avoided.
FMI analysts observe a transition from document-centric submission toward structured exchange where attachments, status changes, and determinations can be routed and audited in near real time. Demand rises fastest where policy creates deadlines and where digital health infrastructure reduces integration effort. Demand is slower where provider workflows are fragmented across multiple EHR instances, or where payer utilization management stacks require major re-platforming before APIs can be exposed at scale.
- Regulatory Timelines: CMS prior authorization interoperability requirements finalized January 2024 push health plans toward standardized APIs and structured status exchange, tightening investment cycles through 2027 milestones [2].
- National Digital Health Rails: ABDM-linked claims exchange and interoperability initiatives reduce manual claim and authorization data entry, which raises the value of standardized attachments and audit trails. [8]
- Referral Digitization Pull: NHS FHIR referral APIs expand structured referral exchange, which increases pressure to align pre-approval and authorization checks with the same integration patterns.
Regional Analysis
North America, Europe, and Asia Pacific are assessed as the core regions for adoption, with country-level growth reflecting regulatory mandates, national digital health infrastructure, and payer-provider connectivity models. The full report includes market attractiveness analysis by region and country.
.webp)
| Country | CAGR (2026-2036) |
|---|---|
| India | 17.0% |
| United States | 15.5% |
| United Kingdom | 14.8% |
| Germany | 14.0% |
| Canada | 13.6% |
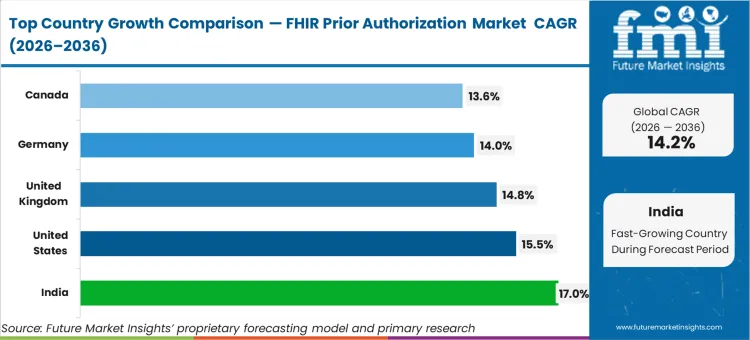
Source: Future Market Insights (FMI) analysis, based on a proprietary forecasting model and primary research
North America FHIR Prior Authorization Market Analysis
North America acts as the compliance-led adoption engine, where payer mandates and large integrated delivery networks set the pace for API exchange and workflow automation. Surescripts and payer connectivity platforms influence scaling by embedding authorization exchange into e-prescribing and EHR workflows, while Epic and Oracle Health shape provider-side integration patterns through platform capabilities and implementation services.
- United States: In the United States, FHIR prior authorization solutions are projected to expand at a 15.5% CAGR through 2036. Growth is anchored in the CMS Interoperability and Prior Authorization Final Rule released January 17, 2024, which requires impacted payers to improve prior authorization data sharing through FHIR-based APIs and related interoperability provisions [2]. Evidence of operational ROI also supports budget conversion from manual staffing to automation. In October 2024, Surescripts published results from a collaboration reported in a KLAS case study that quantified approval-time reductions and higher rates of automated approvals when clinical intelligence is integrated into electronic prior authorization workflows [1].
- Canada: Canada is expected to record a 13.6% CAGR for FHIR prior authorization solutions through 2036. Growth is driven by provincial eReferral and interoperability programs that build reusable patterns for service request exchange, which lowers the effort required to extend similar patterns to authorization and pre-approval workflows. [6]
FMI's analysis of FHIR prior authorization market in North America consists of country-wise assessment that includes the United States and Canada. Readers can find payer compliance timelines, provider onboarding levers, and deployment-model preferences tied to API operations at scale.
Europe FHIR Prior Authorization Market Analysis
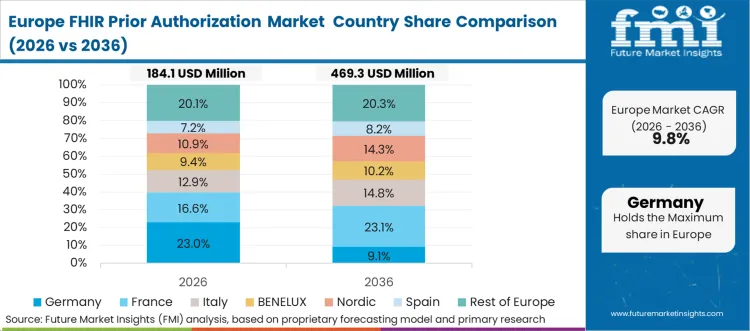
Europe acts as the standards-and-infrastructure adoption engine, where national health systems and regulated digital health programs set technical patterns for structured exchange. NHS ecosystem APIs influence how referral and pre-approval steps are digitized in the United Kingdom, while Germany's telematics infrastructure and ePA-linked specifications shape how FHIR implementation guidance is published and adopted across vendors.
- United Kingdom: The United Kingdom is projected to grow at 14.8% CAGR for FHIR prior authorization solutions through 2036. Growth follows the operational need to digitize referral and care coordination workflows and reduce manual transfer of clinical context between primary and secondary care. NHS Digital maintains an e-Referral Service (e-RS) that supports paperless referral creation and exchange, which reinforces FHIR as an accepted integration route for high-volume workflow messaging in England. [4] Adoption is paced by NHS change control, supplier assurance, and the need to align provider systems with a national service.
- Germany: Demand for FHIR prior authorization solutions in Germany is projected to rise at 14.0% CAGR through 2036. Growth is tied to the country's telematics infrastructure approach, where specifications and implementation guides are published to standardize exchange across approved systems. gematik maintains specifications for national digital health infrastructure that reference FHIR-based resources, indicating institutional backing for FHIR artifacts within Germany's healthcare system. [10] For prior authorization, the structural driver is alignment between payer policy workflows and standardized exchange patterns that can be audited and integrated with national infrastructure components.
FMI's analysis of the FHIR prior authorization market in Europe consists of country-wise assessments that include the United Kingdom and Germany. Readers can find country-level adoption drivers, public specification signals, and integration constraints linked to governance and certification.
Asia Pacific FHIR Prior Authorization Market Analysis
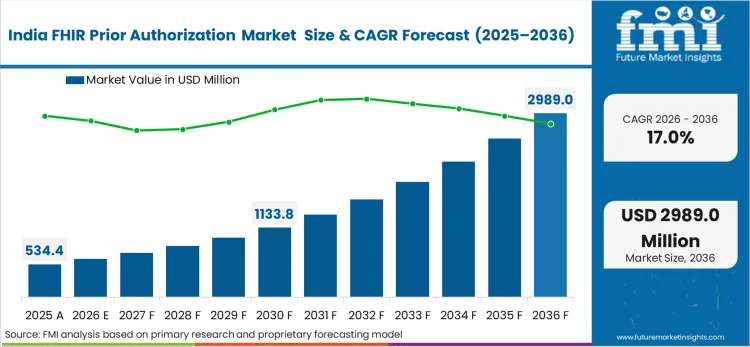
Asia Pacific acts as the digital-rails adoption engine, where national platforms can compress integration effort once identity, consent, and claims exchange gateways mature. India is the lead growth market because ABDM-linked infrastructure and claims exchange programs create a path for structured, machine-readable workflows that can expand beyond pilots when payer and provider connectivity improves.
- India: India is anticipated to grow at 17.0% CAGR for FHIR prior authorization solutions through 2036. Growth is anchored in ABDM-aligned programs that aim to standardize health data and claims exchange, which can reduce manual steps that currently dominate insurer-provider interactions. India's growth rate is supported by the broader ABDM ecosystem push, which positions ABHA identity and digital health exchange as foundational elements for interoperable health workflows. [9]
FMI's analysis of FHIR prior authorization market in Asia Pacific consists of country-wise assessment that includes India. Readers can find program-driven adoption signals, infrastructure constraints by provider tier, and deployment preferences tied to scaled connectivity.
Competitive Aligners for Market Players
Competition in FHIR prior authorization is shaped by connectivity density and workflow depth rather than pure feature checklists. The market is moderately concentrated around a set of networks and intermediaries that already sit in payer-provider transaction paths, alongside large EHR and health IT platforms that can embed prior authorization APIs into clinical workflows. Differentiation comes from partner reach, attachment handling, audit logging, and the ability to support multiple implementation guides across use cases.
Structural advantages accrue to firms with existing network effects in e-prescribing, eligibility, claims, and payer-provider messaging, since onboarding is often the limiting step. Those players can spread certification, security, and integration costs across a wider base of participants. EHR and core health IT platform vendors hold leverage because they control the user interface where authorizations are initiated and tracked, which can reduce workflow fragmentation and raise switching costs for providers.
Buyer behavior reflects risk management and operational continuity. Large payers avoid dependency on a single vendor by using multi-vendor strategies, separating API gateways from workflow engines, and maintaining governance over decision rules and policy content. Providers push vendors to embed status and attachment prompts into existing intake and scheduling queues to avoid "yet another portal," which increases pressure on vendors to integrate deeply with EHR workflows while still supporting payer compliance specifications.
Key Players in the FHIR Prior Authorization Market
- Surescripts
- CoverMyMeds (McKesson)
- DrFirst
- CenterX
- Availity
- Optum Rx (Optum)
- athenahealth
- Epic Systems
- Oracle Health (Cerner)
- Veradigm (Allscripts)
Scope of the Report
| Item | Value |
|---|---|
| Quantitative Units | USD 850.0 million (2026) to USD 3,200.0 million (2036), at a CAGR of 14.2% |
| Market Definition | The FHIR prior authorization market comprises software platforms and services that enable electronic prior authorization request, clinical attachment exchange, status tracking, and determinations using HL7 FHIR-based APIs across payer-provider workflows. |
| Solution Type Segmentation | FHIR prior authorization API platforms, Prior authorization workflow automation, Clinical documentation and attachment automation |
| End User Segmentation | Payers and health insurers, Providers, Intermediaries |
| Deployment Model Segmentation | Cloud and SaaS, On-premise, Hybrid |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | India, United States, United Kingdom, Germany, Canada |
| Key Companies Profiled | Surescripts, CoverMyMeds (McKesson), DrFirst, CenterX, Availity, Optum Rx (Optum), athenahealth, Epic Systems, Oracle Health (Cerner), Veradigm (Allscripts) |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling anchored on policy and standards timelines, validated through primary interviews and triangulated with public specifications and official releases. |
FHIR Prior Authorization Market Analysis by Segments
By Solution Type:
- FHIR prior authorization API platforms
- Prior authorization workflow automation
- Clinical documentation and attachment automation
By End User:
- Payers and health insurers
- Providers
- Intermediaries
By Deployment Model:
- Cloud and SaaS
- On-premise
- Hybrid
By Region:
- North America
- Europe
- Asia Pacific
Bibliography
- [1] Surescripts. (2024). KLAS Reports Significant Time Saved with Surescripts Medication Prior Authorization Innovations, Leveraging Clinical Intelligence, Collaboration. Company Press Release, October 16, 2024.
- [2] Centers for Medicare & Medicaid Services (CMS). (2024). CMS Interoperability and Prior Authorization Final Rule (CMS-0057-F). Federal Register, February 8, 2024.
- [3] HL7 International. (2024). Da Vinci Prior Authorization Support (PAS) Implementation Guide. HL7 FHIR Implementation Guide, Version 2.1.0.
- [4] NHS Digital. (2024). e-Referral Service (e-RS) FHIR API. NHS Developer Documentation.
- [5] eHealth Ontario. (2024). Ontario eReferral - eConsult HL7 FHIR Implementation Guide. Provincial Digital Health Specification.
- [6] Canada Health Infoway. (2024). eReferral/eConsult Pan-Canadian Interoperability Specification. Canada Health Infoway Featured Initiatives.
- [7] Canada Health Infoway. (2024). FHIR Implementations Community. InfoCentral Community Portal.
- [8] Ministry of Health and Family Welfare, Government of India. (2024). Update on National Health Claims Exchange. Press Release, July 26, 2024.
- [9] National Health Authority, Government of India. (2024). Ayushman Bharat Digital Mission (ABDM). ABDM Official Portal.
- [10] gematik GmbH. (2024). Implementation Guide ePA MHD Service. ePA FHIR Implementation Guide, Version 1.1.0.
This bibliography is provided for reader reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
This Report Addresses
- Market sizing and 10-year forecast from 2026 to 2036 aligned to FMI master numbers and segment shares.
- Opportunity mapping by solution type, end user, and deployment model with leading sub-segment share benchmarks.
- Country-level CAGR analysis for priority markets to support go-to-market sequencing and partner onboarding plans.
- Competitive assessment covering network effects, integration depth, and buyer leverage in payer-provider connectivity.
- Standards and policy tracking tied to FHIR implementation guidance, API requirements, and program-led adoption signals.
- Deployment model analysis focused on cloud operations, audit logging, uptime monitoring, and scalable onboarding.
- Regional narratives linking adoption to payer structures, national digital health programs, and public specifications.
- Deliverables packaged for strategy and implementation teams across PDF narrative, Excel tables, and presentation-ready charts.
Frequently Asked Questions
FHIR Prior Authorization Market
It reaches USD 850.0 million in 2026, reflecting payer-led compliance programs and provider onboarding for API-based authorization exchange.
What will the market size be by 2036?
FMI projects USD 3,200.0 million by 2036 as workflow automation scales, attachments become standardized, and cloud operations support higher transaction volumes.
What CAGR is expected for 2026 to 2036?
The market is projected to expand at 14.2% CAGR from 2026 to 2036, driven by interoperability mandates and measurable administrative cycle-time reduction.
How much absolute dollar growth occurs over the forecast period?
Absolute growth totals USD 2,350.0 million from 2026 to 2036 as manual authorization steps shift into API-mediated workflow orchestration and monitoring.
Which solution type segment leads the market in 2026?
Prior authorization workflow automation leads with 46% share in 2026 because it routes requests, manages exceptions, and delivers the fastest operational ROI.
Which end user segment holds the largest share in 2026?
Payers and health insurers hold 52% share in 2026 since they fund compliance delivery, own decision systems, and manage network connectivity at scale.
Which deployment model is most adopted in 2026?
Cloud and SaaS leads with 62% share in 2026 because uptime monitoring, scaling, and cross-entity onboarding are easier to operate centrally.
Why is India the fastest-growing market in this set?
India posts 17.0% CAGR as ABDM-linked digital rails and claims exchange gateways reduce manual data entry and support standardized authorization artifacts.
What drives growth in the United States market?
The United States grows at 15.5% CAGR as payer compliance with CMS API requirements pushes new builds, while providers integrate status tracking into workflows.
What keeps the United Kingdom near the global growth rate?
The United Kingdom grows at 14.8% CAGR because national referral digitization supports structured exchange, but rollout pace is shaped by NHS governance cycles.
Why does Germany track close to the global CAGR?
Germany's 14.0% CAGR reflects institutional FHIR specification signals and telematics infrastructure alignment, balanced by certification cycles and multi-stakeholder governance.
What explains Canada's growth profile versus the United States?
Canada grows at 13.6% CAGR as provincial eReferral programs standardize workflows, yet payer models vary by province, which slows national-scale uniform adoption.
How do buyers measure ROI for workflow automation platforms?
Buyers track approval turnaround time, staff hours avoided, denial rework reduction, and fewer abandoned treatments where authorization delays previously disrupted care initiation.
What is the main restraint slowing adoption in mature payer markets?
Legacy utilization management systems and fragmented provider workflows extend integration timelines, raising exception queues and testing effort even when standards are clear.
Which use cases benefit most from clinical attachment automation?
Imaging, specialty drugs, and procedure authorizations benefit most, since structured attachment prompts reduce missing information, cutting denials tied to incomplete documentation.
How do intermediaries fit into the market structure?
Intermediaries provide hubs for routing, identity, and monitoring across many payers and providers, reducing point-to-point interfaces and speeding partner onboarding cycles.
What role do HL7 Da Vinci guides play in implementation?
Da Vinci implementation guides standardize FHIR request and response patterns, lowering custom interface design effort and improving interoperability testing across vendors.
What deployment risks should buyers plan for with cloud models?
Cloud deployment requires strong audit logging, security controls, traffic management, and failover planning, since authorization exchange becomes a workflow dependency for care.
How should payers sequence implementation to reduce disruption?
Start with high-volume authorizations, align attachment policies, then onboard major provider groups through staged pilots, keeping manual fallback paths for exception handling.
What should executives look for when selecting vendors in this market?
Executives should prioritize connectivity reach, proven onboarding capacity, attachment and status depth, auditability, and integration into EHR workflows without duplicative portals.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Solution Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Solution Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Solution Type , 2026 to 2036
- Prior authorization workflow automation
- FHIR prior authorization API platforms
- Clinical documentation and attachment automation
- Prior authorization workflow automation
- Y-o-Y Growth Trend Analysis By Solution Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Solution Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Payers and health insurers
- Providers
- Intermediaries
- Payers and health insurers
- Y-o-Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Model, 2026 to 2036
- Cloud and SaaS
- On-premise
- Hybrid
- Cloud and SaaS
- Y-o-Y Growth Trend Analysis By Deployment Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Solution Type
- By End Use
- By Deployment Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Solution Type
- By End Use
- By Deployment Model
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Solution Type
- By End Use
- By Deployment Model
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Solution Type
- By End Use
- By Deployment Model
- Competition Analysis
- Competition Deep Dive
- Surescripts
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CoverMyMeds (McKesson)
- DrFirst
- CenterX
- Availity
- Optum Rx (Optum)
- athenahealth
- Epic Systems
- Oracle Health (Cerner)
- Veradigm (Allscripts)
- Surescripts
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Solution Type , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Deployment Model, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Solution Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Deployment Model
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Solution Type
- Figure 26: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End Use
- Figure 29: North America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Deployment Model
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Solution Type
- Figure 36: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by End Use
- Figure 39: Latin America Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Deployment Model
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Solution Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by End Use
- Figure 49: Western Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Deployment Model
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Solution Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Deployment Model
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Solution Type
- Figure 66: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by End Use
- Figure 69: East Asia Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Deployment Model
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Solution Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Deployment Model
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Solution Type , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Solution Type , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Solution Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Deployment Model, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Model, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Deployment Model
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

